Anti-Proliferative Activity of Triterpenoids and Sterols Isolated from Alstonia scholaris against Non-Small-Cell Lung Carcinoma Cells
Abstract
:1. Introduction
2. Results
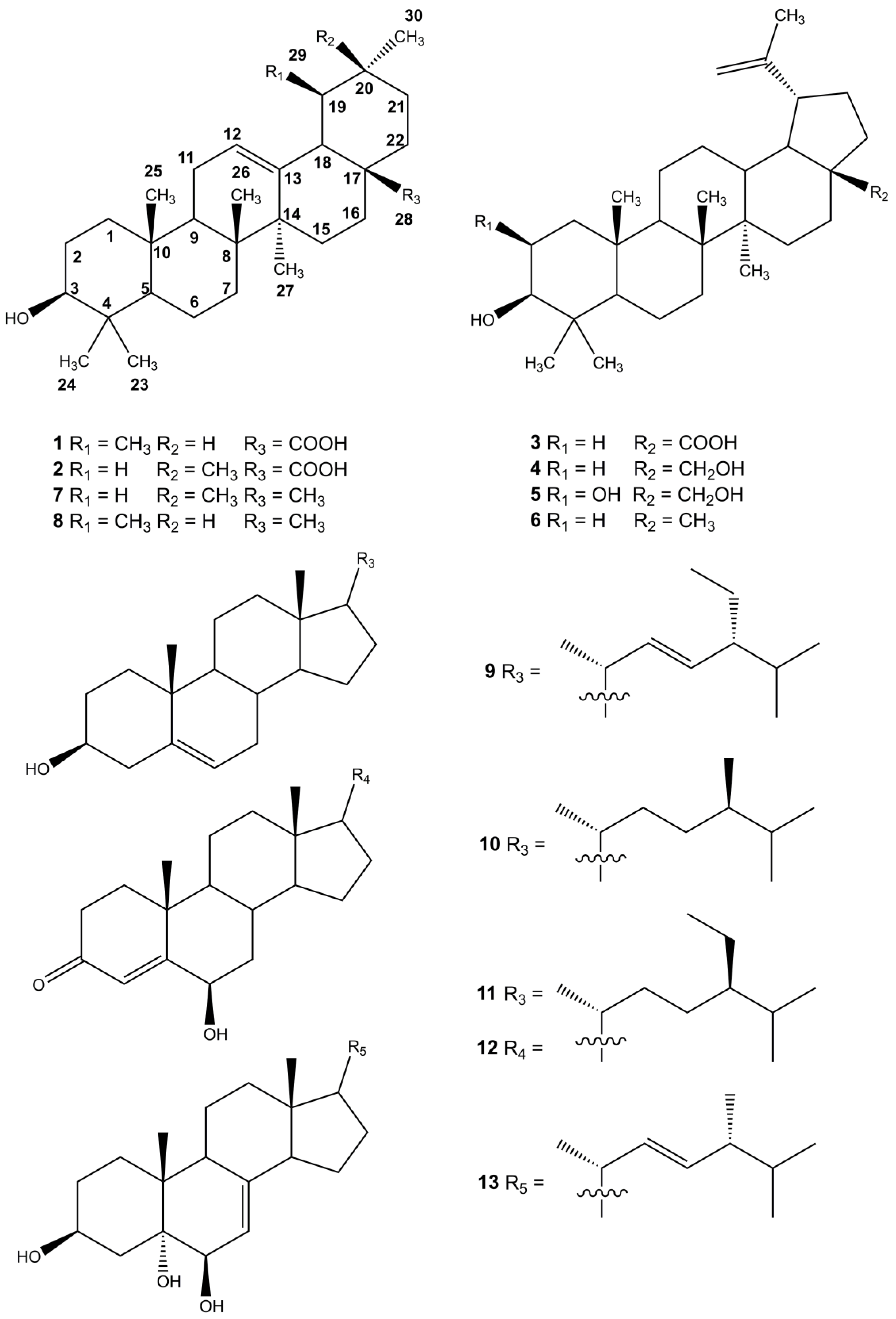
2.1. Isolation and Identification of Triterpenoids from A. scholaris
2.2. Antiproliferation Activity of Triterpenoids and Steriols against NSCLC
2.3. The Inhibitory Concentrations (IC50) of Triterpenoids and Steriols on NSCLC
3. Discussion
4. Materials and Methods
4.1. General Procedures
4.2. Plant Materials
4.3. Isolation and Identification of Triterpenoids and Sterols
4.4. Anti-Proliferative Activity
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| AMPK | AMP-activated protein kinase |
| COSY | Correlation spectroscopy |
| DEPT | Distortionless Enhancement by Polarization Transfer |
| DMEM | Dulbecco’s Modified Eagle Medium |
| HMBC | Heteronuclear Multiple Bond Correlation |
| HMQC | Heteronuclear Multiple-Quantum Correlation |
| mTOR | Mammalian target of rapamycin |
| MTT | 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| MRSA | Methicillin-resistant Staphylococcus aureus |
| NMR | Nuclear Magnetic Resonance |
| NSCLC | Non-Small Cell Lung Cancer |
| SCLC | Small Cell Lung Cancer |
| TLC | Thin layer chromatography |
References
- Khanal, N.; Ganti, A.K. Emerging targeted therapies in non-small cell lung cancer. Expert Rev. Anticancer Ther. 2016, 16, 177–187. [Google Scholar] [CrossRef] [PubMed]
- Zhao, T.; Pan, H.; Feng, Y.; Li, H.; Zhao, Y. Petroleum ether extract of Chenopodium album L. prevents cell growth and induces apoptosis of human lung cancer cells. Exp. Ther. Med. 2016, 12, 3301–3307. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Liu, C.Y.; Fan, P.W.; Hsieh, C.H.; Lin, H.Y.; Lee, M.C.; Fang, K. The aqueous extract of Brucea javanica suppresses cell growth and alleviates tumorigenesis of human lung cancer cells by targeting mutated epidermal growth factor receptor. Drug Des. Dev. Ther. 2016, 10, 3599–3609. [Google Scholar] [CrossRef] [PubMed]
- Tang, Z.H.; Chen, X.; Wang, Z.Y.; Chai, K.; Wang, Y.F.; Xu, X.H.; Wang, X.W.; Lu, J.H.; Wang, Y.T.; Chen, X.P.; et al. Induction of C/EBP homologous protein-mediated apoptosis and autophagy by licochalcone A in non-small cell lung cancer cells. Sci. Rep. 2016, 6, 26241. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.R.; Li, S.; Ho, C.T.; Chang, Y.H.; Tan, K.T.; Chung, T.W.; Wang, B.Y.; Chen, Y.K.; Lin, C.C. Tangeretin derivative, 5-acetyloxy-6,7,8,4′-tetramethoxyflavone induces G2/M arrest, apoptosis and autophagy in human non-small cell lung cancer cells in vitro and in vivo. Cancer Biol. Ther. 2016, 17, 48–64. [Google Scholar] [CrossRef] [PubMed]
- Baliga, M.S. Review of the phytochemical, pharmacological and toxicological properties of Alstonia scholaris Linn. R. Br (Saptaparna). Chin. J. Integr. Med. 2012, 18, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Khyade, M.S.; Kasote, D.M.; Vaikos, N.P. Alstonia scholaris (L) R. Br. and Alstonia macrophylla Wall. ex G. Don: A comparative review on traditional uses, phytochemistry and pharmacology. J. Ethnopharmacol. 2014, 153, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Abe, F.; Chen, R.F.; Yamauchi, T.; Marubayashi, N.; Ueda, I. Studies on the Constituents of Alstonia-Scholaris 1. Alschomine and Isoalschomine, New Alkaloids from the Leaves of Alstonia-Scholaris. Chem. Pharm. Bull. 1989, 37, 887–890. [Google Scholar] [CrossRef]
- Attaurrahman; Alvi, K.A. Indole Alkaloids from Alstonia-Scholaris. Phytochemistry 1987, 26, 2139–2142. [Google Scholar]
- Atta-ur-Rahman, A.; Asif, M.; Ghazala, M.; Fatima, J.; Alvi, K.A. Scholaricine, an Alkaloid from Alstonia scholaris. Phytochemistry 1985, 24, 2771–2773. [Google Scholar] [CrossRef]
- Cai, X.H.; Du, Z.Z.; Luo, X.D. Unique monoterpenoid indole alkaloids from Alstonia scholaris. Org. Lett. 2007, 9, 1817–1820. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.H.; Shang, J.H.; Feng, T.; Luo, X.D. Novel Alkaloids from Alstonia scholaris. Z. Naturforsch. B 2010, 65, 1164–1168. [Google Scholar] [CrossRef]
- Cai, X.H.; Tan, Q.G.; Liu, Y.P.; Feng, T.; Du, Z.Z.; Li, W.Q.; Luo, X.D. A cage-monoterpene indole alkaloid from Alstonia scholaris. Org. Lett. 2008, 10, 577–580. [Google Scholar] [CrossRef] [PubMed]
- El-Askary, H.I.; El-Olemy, M.M.; Salama, M.M.; Sleem, A.A.; Amer, M.H. Bioguided isolation of pentacyclic triterpenes from the leaves of Alstonia scholaris (Linn.) R. Br. growing in Egypt. Nat. Prod. Res. 2012, 26, 1755–1758. [Google Scholar] [CrossRef] [PubMed]
- Shang, J.H.; Cai, X.H.; Feng, T.; Zhao, Y.L.; Wang, J.K.; Zhang, L.Y.; Yan, M.; Luo, X.D. Pharmacological evaluation of Alstonia scholaris: Anti-inflammatory and analgesic effects. J. Ethnopharmacol. 2010, 129, 174–181. [Google Scholar] [CrossRef] [PubMed]
- Shang, J.H.; Cai, X.H.; Zhao, Y.L.; Feng, T.; Luo, X.D. Pharmacological evaluation of Alstonia scholaris: Anti-tussive, anti-asthmatic and expectorant activities. J. Ethnopharmacol. 2010, 129, 293–298. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.-M.; Chen, H.-T.; Wu, Z.-Y.; Jhan, Y.-L.; Shyu, C.-L.; Chou, C.-H. Antibacterial and synergistic activity of pentacyclic triterpenoids isolated from Alstonia scholaris. Molecules 2016, 21, 139. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.M.; Jhan, Y.L.; Tsai, S.J.; Chou, C.H. The Pleiotropic Antibacterial Mechanisms of Ursolic Acid against Methicillin-Resistant Staphylococcus aureus (MRSA). Molecules 2016, 21. [Google Scholar] [CrossRef] [PubMed]
- Seebacher, W.; Simic, N.; Weis, R.; Saf, R.; Kunert, O. Complete assignments of 1H- and 13C-NMR resonances of oleanolic acid, 18 α-oleanolic acid, ursolic acid and their 11-oxo derivatives. Magn. Reson. Chem. 2003, 41, 636–638. [Google Scholar] [CrossRef]
- Ikuta, A.; Itokawa, H. Triterpenoids of Paeonia Japonica Callus-Tissue. Phytochemistry 1988, 27, 2813–2815. [Google Scholar] [CrossRef]
- Prachayasittikul, S.; Saraban, P.; Cherdtrakulkiat, R.; Ruchirawat, S.; Prachayasittikul, V. New bioactive triterpenoids and antimalarial activity of Diospyros rubra Lec. EXCLI J. 2010, 9, 1–10. [Google Scholar]
- Okoye, N.N.; Ajaghaku, D.L.; Okeke, H.N.; Ilodigwe, E.E.; Nworu, C.S.; Okoye, F.B. β-Amyrin and α-amyrin acetate isolated from the stem bark of Alstonia boonei display profound anti-inflammatory activity. Pharm. Biol. 1478, 52, 1478–1486. [Google Scholar] [CrossRef] [PubMed]
- Soldi, C.; Pizzolatti, M.G.; Luiz, A.P.; Marcon, R.; Meotti, F.C.; Mioto, L.A.; Santos, A.R. Synthetic derivatives of the α- and β-amyrin triterpenes and their antinociceptive properties. Bioorg. Med. Chem. 2008, 16, 3377–3386. [Google Scholar] [CrossRef] [PubMed]
- Marsan, M.P.; Warnock, W.; Muller, I.; Nakatani, Y.; Ourisson, G.; Milon, A. Synthesis of Deuterium-Labeled Plant Sterols and Analysis of Their Side-Chain Mobility by Solid State Deuterium NMR. J. Organ. Chem. 1996, 61, 4252–4257. [Google Scholar] [CrossRef]
- Morikawa, T.; Mizutani, M.; Aoki, N.; Watanabe, B.; Saga, H.; Saito, S.; Oikawa, A.; Suzuki, H.; Sakurai, N.; Shibata, D.; et al. Cytochrome P450 CYP710A encodes the sterol C-22 desaturase in Arabidopsis and tomato. Plant Cell 2006, 18, 1008–1022. [Google Scholar] [CrossRef] [PubMed]
- Gomes, A.; Saha, A.; Chatterjee, I.; Chakravarty, A.K. Viper and cobra venom neutralization by β-sitosterol and stigmasterol isolated from the root extract of Pluchea indica Less. (Asteraceae). Phytomedicine 2007, 14, 637–643. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.T.; Shi, Y.P. Steroids from Saussurea Ussuriensis. Die Pharm. Int. J. Pharm. Sci. 2005, 60, 464–467. [Google Scholar] [CrossRef]
- Jinming, G.; Lin, H.; Jikai, L. A novel sterol from Chinese truffles Tuber indicum. Steroids 2001, 66, 771–775. [Google Scholar] [CrossRef]
- Hao, J.; Zhang, X.; Zhang, P.; Liu, J.; Zhang, L.; Sun, H. Efficient access to isomeric 2,3-dihydroxy lupanes: First synthesis of alphitolic acid. Tetrahedron 2009, 65, 7975–7984. [Google Scholar] [CrossRef]
- Jager, S.; Trojan, H.; Kopp, T.; Laszczyk, M.N.; Scheffler, A. Pentacyclic triterpene distribution in various plants—Rich sources for a new group of multi-potent plant extracts. Molecules 2009, 14, 2016–2031. [Google Scholar] [CrossRef] [PubMed]
- Feng, L.; Chen, Y.; Yuan, L.; Liu, X.; Gu, J.F.; Zhang, M.H.; Wang, Y. A combination of alkaloids and triterpenes of Alstonia scholaris (Linn.) R. Br. leaves enhances immunomodulatory activity in C57BL/6 mice and induces apoptosis in the A549 cell line. Molecules 2013, 18, 13920–13939. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.M.; Chen, H.T.; Li, T.C.; Weng, J.H.; Jhan, Y.L.; Lin, S.X.; Chou, C.H. The role of pentacyclic triterpenoids in the allelopathic effects of Alstonia scholaris. J. Chem. Ecol. 2014, 40, 90–98. [Google Scholar] [CrossRef] [PubMed]
- Laszczyk, M.N. Pentacyclic triterpenes of the lupane, oleanane and ursane group as tools in cancer therapy. Planta Med. 2009, 75, 1549–1560. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Q.Y.; Li, P.P.; Jin, F.S.; Yao, C.; Zhang, G.H.; Zang, T.; Ai, X. Ursolic acid induces ER stress response to activate ASK1-JNK signaling and induce apoptosis in human bladder cancer T24 cells. Cell. Signal. 2013, 25, 206–213. [Google Scholar] [CrossRef] [PubMed]
- Wozniak, L.; Skapska, S.; Marszalek, K. Ursolic Acid—A Pentacyclic Triterpenoid with a Wide Spectrum of Pharmacological Activities. Molecules 2015, 20, 20614–20641. [Google Scholar] [CrossRef] [PubMed]
- De Angel, R.E.; Smith, S.M.; Glickman, R.D.; Perkins, S.N.; Hursting, S.D. Antitumor effects of ursolic acid in a mouse model of postmenopausal breast cancer. Nutr. Cancer 2010, 62, 1074–1086. [Google Scholar] [CrossRef] [PubMed]
- Shao, J.W.; Dai, Y.C.; Xue, J.P.; Wang, J.C.; Lin, F.P.; Guo, Y.H. In vitro and in vivo anticancer activity evaluation of ursolic acid derivatives. Eur. J. Med. Chem. 2011, 46, 2652–2661. [Google Scholar] [CrossRef] [PubMed]
- Way, T.D.; Tsai, S.J.; Wang, C.M.; Ho, C.T.; Chou, C.H. Chemical Constituents of Rhododendron formosanum Show Pronounced Growth Inhibitory Effect on Non-Small-Cell Lung Carcinoma Cells. J. Agric. Food Chem. 2014, 62, 875–884. [Google Scholar] [CrossRef] [PubMed]
- Eiznhamer, D.A.; Xu, Z.Q. Betulinic acid: A promising anticancer candidate. IDrugs 2004, 7, 359–373. [Google Scholar] [PubMed]
- Mullauer, F.B.; Kessler, J.H.; Medema, J.P. Betulinic acid, a natural compound with potent anticancer effects. Anti-Cancer Drugs 2010, 21, 215–227. [Google Scholar] [CrossRef] [PubMed]
- Shin, Y.G.; Cho, K.H.; Chung, S.M.; Graham, J.; Das Gupta, T.K.; Pezzuto, J.M. Determination of betulinic acid in mouse blood, tumor and tissue homogenates by liquid chromatography-electrospray mass spectrometry. J. Chromatogr. B Biomed. Sci. Appl. 1999, 732, 331–336. [Google Scholar] [CrossRef]
- Udeani, G.O.; Zhao, G.M.; Geun Shin, Y.; Cooke, B.P.; Graham, J.; Beecher, C.W.; Kinghorn, A.D.; Pezzuto, J.M. Pharmacokinetics and tissue distribution of betulinic acid in CD-1 mice. Biopharm. Drug Dispos. 1999, 20, 379–383. [Google Scholar] [CrossRef]
- Fulda, S. Betulinic Acid for cancer treatment and prevention. Int. J. Mol. Sci. 2008, 9, 1096–1107. [Google Scholar] [CrossRef] [PubMed]
- Zuco, V.; Supino, R.; Righetti, S.C.; Cleris, L.; Marchesi, E.; Gambacorti-Passerini, C.; Formelli, F. Selective cytotoxicity of betulinic acid on tumor cell lines, but not on normal cells. Cancer Lett. 2002, 175, 17–25. [Google Scholar] [CrossRef]
- Ali-Seyed, M.; Jantan, I.; Vijayaraghavan, K.; Bukhari, S.N. Betulinic Acid: Recent Advances in Chemical Modifications, Effective Delivery, and Molecular Mechanisms of a Promising Anticancer Therapy. Chem. Biol. Drug Des. 2016, 87, 517–536. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds 1–13 are not available from the authors. |


| Compound | 5 | 2β,3β,28-lup-20(29)-ene-triol | ||
|---|---|---|---|---|
| Position | 1H | 13C | 1H | 13C |
| 1 | 2.15, 1.09 | 44.4 | 44.5 | |
| 2 | 4.09 (dd, J = 3.6, 6.6) | 71.1 | 4.08 (brs) | 71.2 |
| 3 | 3.19 (d, J = 4.2) | 78.4 | 3.19 (brs) | 78.5 |
| 4 | 38.1 | 38.2 | ||
| 5 | 0.77 (d, J = 9.5) | 55.2 | 55.3 | |
| 6 | 1.56 | 18.2 | 18.1 | |
| 7 | 1.41 | 34.1 | 34.2 | |
| 8 | 41 | 41.1 | ||
| 9 | 1.24 | 50.8 | 50.9 | |
| 10 | 36.8 | 36.9 | ||
| 11 | 1.45 | 20.9 | 21.0 | |
| 12 | 1.65, 1.05 | 25.2 | 25.3 | |
| 13 | 1.65 | 37.2 | 37.3 | |
| 14 | 42.8 | 42.9 | ||
| 15 | 1.71, 1.05 | 26.9 | 27.0 | |
| 16 | 1.93, 1.21 | 29.1 | 29.2 | |
| 17 | 47.7 | 47.8 | ||
| 18 | 1.58 | 48.7 | 48.8 | |
| 19 | 2.39 m | 47.7 | 47.8 | |
| 20 | 150.4 | 150.4 | ||
| 21 | 29.7 | 29.8 | ||
| 22 | 1.86, 1.04 | 33.9 | 34.0 | |
| 23 | 0.99 s | 29.5 | 0.99 s | 29.6 |
| 24 | 0.98 s | 17.1 | 0.98 s | 17.1 |
| 25 | 1.14 s | 17.0 | 1.14 s | 17.1 |
| 26 | 1.04 s | 15.9 | 1.04 s | 16.0 |
| 27 | 0.97 s | 14.7 | 0.97 s | 14.7 |
| 28 | 3.80, 3.33 (d, J = 10.8) | 60.5 | 3.80, 3.33 (d, J = 10.8) | 60.6 |
| 29 | 4.69, 4.59 | 109.6 | 4.69, 4.58 | 109.7 |
| 30 | 1.68 s | 19.2 | 1.68 s | 19.1 |
| Compound | NSCLC (A549 Cell Line) | |
|---|---|---|
| IC50 (μM) | S.E. + | |
| Ursolic acid (1) | 39.8 | 0.09 |
| Oleanolic acid (2) | >400 | - |
| Betulinic acid (3) | 40.1 | 0.51 |
| Betulin (4) | 240.5 | 4.04 |
| 2β,3β,28-lup-20(29)-ene-triol (5) | 172.6 | 0.44 |
| Lupeol (6) | >400 | - |
| β-amyrin (7) | >400 | - |
| α-amyrin (8) | >400 | - |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, C.-M.; Tsai, S.-J.; Jhan, Y.-L.; Yeh, K.-L.; Chou, C.-H. Anti-Proliferative Activity of Triterpenoids and Sterols Isolated from Alstonia scholaris against Non-Small-Cell Lung Carcinoma Cells. Molecules 2017, 22, 2119. https://doi.org/10.3390/molecules22122119
Wang C-M, Tsai S-J, Jhan Y-L, Yeh K-L, Chou C-H. Anti-Proliferative Activity of Triterpenoids and Sterols Isolated from Alstonia scholaris against Non-Small-Cell Lung Carcinoma Cells. Molecules. 2017; 22(12):2119. https://doi.org/10.3390/molecules22122119
Chicago/Turabian StyleWang, Chao-Min, Shang-Jie Tsai, Yun-Lian Jhan, Kuei-Lin Yeh, and Chang-Hung Chou. 2017. "Anti-Proliferative Activity of Triterpenoids and Sterols Isolated from Alstonia scholaris against Non-Small-Cell Lung Carcinoma Cells" Molecules 22, no. 12: 2119. https://doi.org/10.3390/molecules22122119
APA StyleWang, C.-M., Tsai, S.-J., Jhan, Y.-L., Yeh, K.-L., & Chou, C.-H. (2017). Anti-Proliferative Activity of Triterpenoids and Sterols Isolated from Alstonia scholaris against Non-Small-Cell Lung Carcinoma Cells. Molecules, 22(12), 2119. https://doi.org/10.3390/molecules22122119






