One-Step Partially Purified Lipases (ScLipA and ScLipB) from Schizophyllum commune UTARA1 Obtained via Solid State Fermentation and Their Applications
Abstract
:1. Introduction
2. Results and Discussion
2.1. Optimization of Fermentation Parameters
2.2. Partial Purification of Crude Enzyme Extract
2.3. Lipase Stability and Deactivation
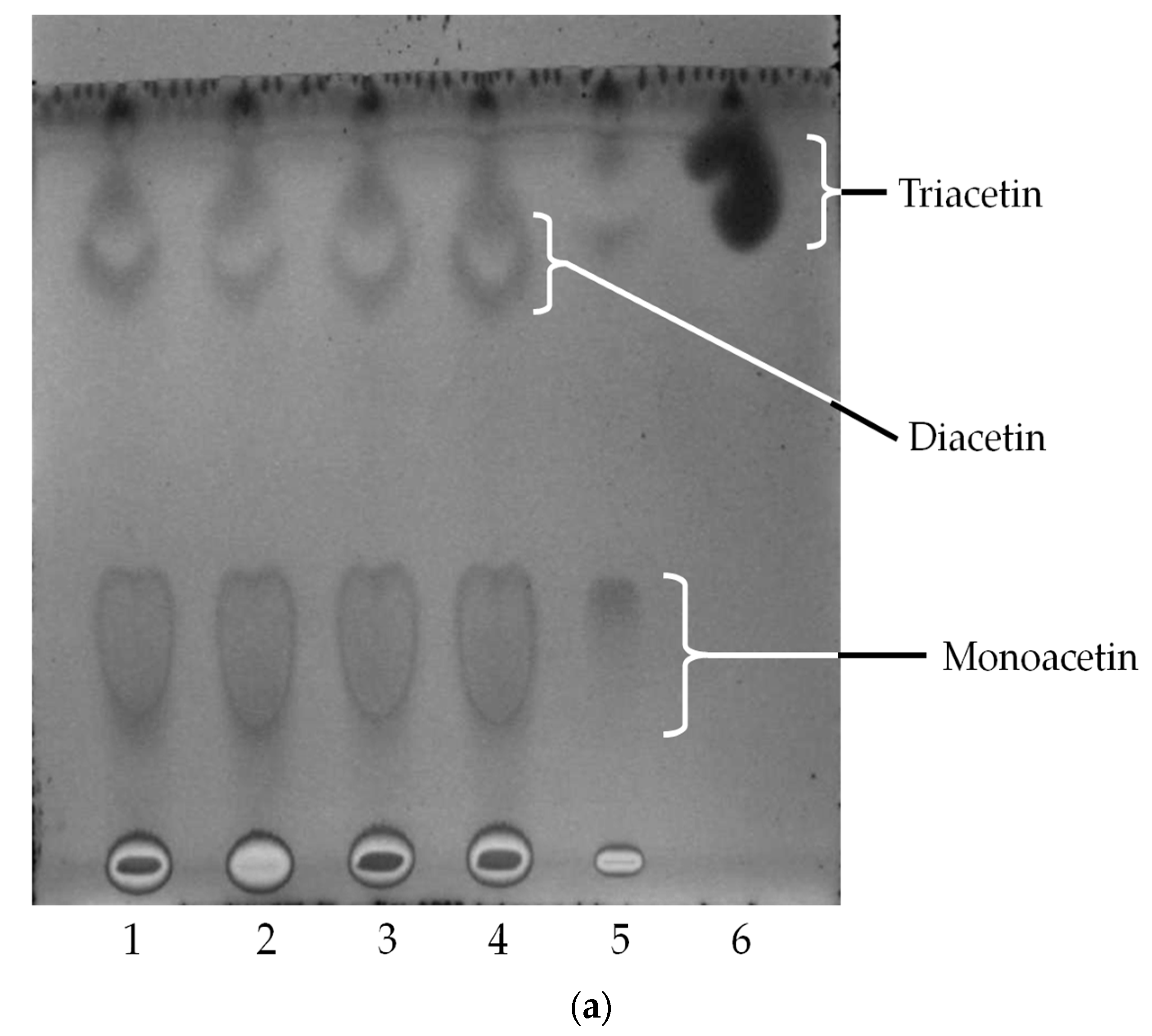
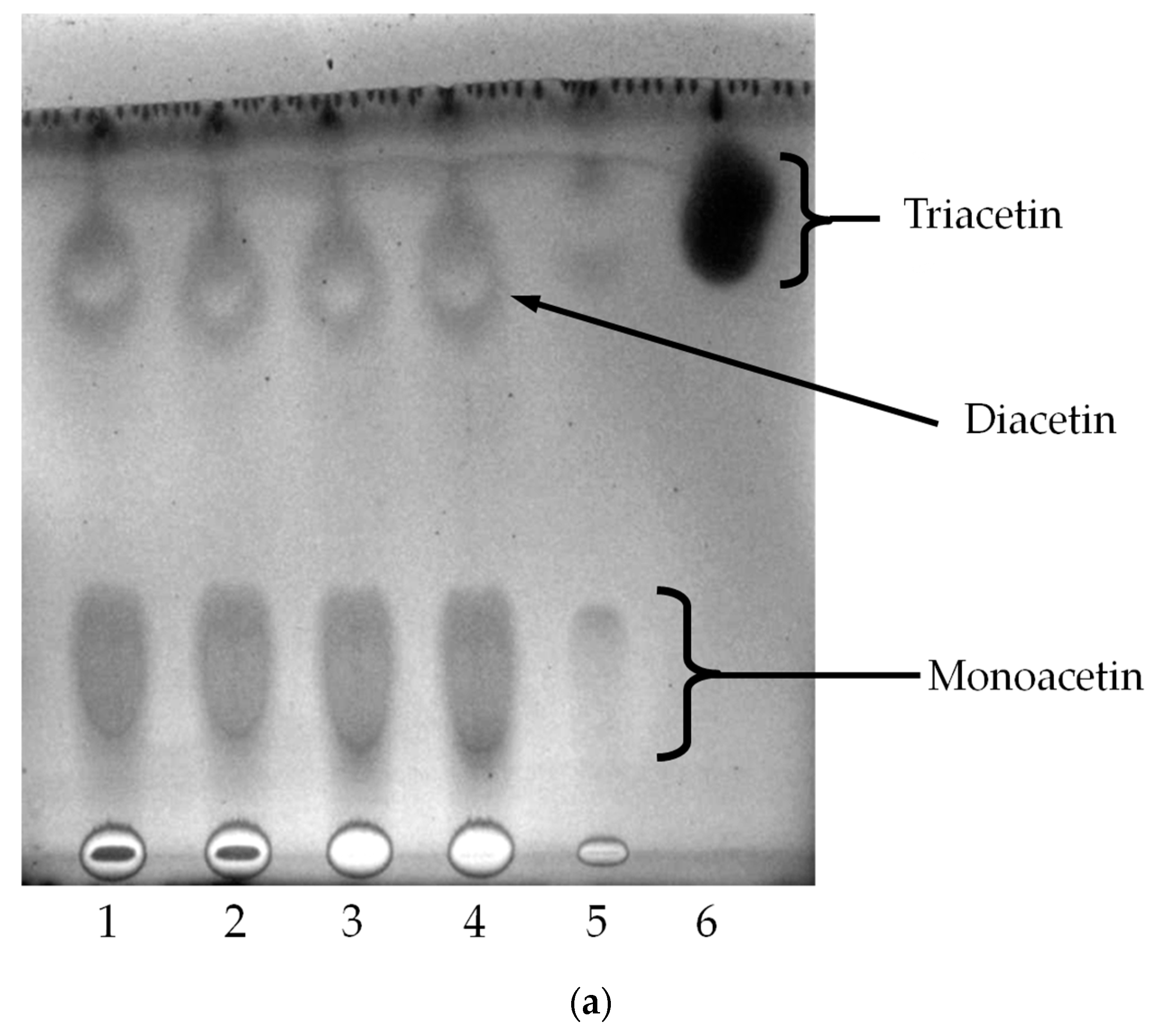
2.4. Oil Hydrolysis and Glycerol Esterification
3. Materials and Methods
3.1. Fungal Cultivation and Solid State Fermentation
3.2. Optimization of Fermentation Parameters
3.3. Crude Enzyme Extraction and Lipase Assay
3.4. Partial Purification of Crude Enzyme Extract
3.5. Lipase Stability and Deactivation
3.6. Crude Fish Oil Extraction
3.7. Oil Hydrolysis and Glycerol Esterification
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mirfat, A.H.S.; Noorlidah, A.; Vikineswary, S. Antimicrobial activities of split gill mushroom Schizophyllum commune Fr. Am. J. Res. Commun. 2014, 2, 113–124. [Google Scholar]
- Teoh, Y.P.; Mashitah, M.D. Optimization of parameters for mycelia growth by Schizophyllum commune and a kinetic model study of its growth morphology. J. Appl. Sci. 2012, 12, 1100–1105. [Google Scholar] [CrossRef]
- Singh, M.K.; Singh, J.; Kumar, M.; Thakur, I.S. Novel lipase from basidiomycetes Schizophyllum commune ISTL04, produced by solid state fermentation of Leucaena leucocephala seeds. J. Mol. Catal. B Enzym. 2014, 110, 92–99. [Google Scholar] [CrossRef]
- Bornscheur, U.; Stamatis, H.; Xenakis, A.; Yamane, T.; Kolisis, F.N. A comparison of different strategies for lipase-catalyzed synthesis of partial glycerides. Biotechnol. Lett. 1994, 16, 697–702. [Google Scholar] [CrossRef]
- Yvergnaux, F. Lipases: Particularly effective biocatalysts for cosmetic active ingredients. OCL 2017, 24, D408. [Google Scholar] [CrossRef]
- Nascimento, V.L.V.D.; Bermúdez, V.M.S.; Oliveira, A.L.L.D.; Kleinberg, M.N.; Ribeiro, R.D.T.M.; Abreu, R.F.A.D.; Carioca, J.O.B. Characterization of a hydrolyzed oil obtained from fish waste for nutraceutical application. Food Sci. Technol. 2015, 35, 321–325. [Google Scholar] [CrossRef]
- Kumar, P.K.P.; Krishna, A.G.G. Physiochemical characteristics of commercial coconut oils produced in India. Grasas Aceites 2015, 66, e062. [Google Scholar] [CrossRef]
- Júnior, S.J.H.; Ract, J.N.R.; Gioielli, L.A.; Vitolo, M. Conversion of triolein into mono- and diacylglycerols by immobilized lipase. Arab. J. Sci. Eng. 2017, 580, 1–9. [Google Scholar] [CrossRef]
- Rosu, R.; Yasui, M.; Iwasaki, Y.; Yamane, T. Enzymatic synthesis of symmetrical 1,3-diacylglycerols by direct esterification of glycerol in solvent-free system. J. Am. Oil Chem. Soc. 1999, 76, 839–843. [Google Scholar] [CrossRef]
- Bansal, K.; McCrady, J.; Hansen, A.; Bhalerao, K. Thin layer chromatography and image analysis to detect glycerol in biodiesel. Fuel 2008, 87, 3369–3372. [Google Scholar] [CrossRef]
- Moquin, P.H.L.; Temelli, F.; King, J.W. Kinetic modeling of glycerolysis reaction for soybean oils in supercritical carbon dioxide media. J. Am. Oil Chem. Soc. 2005, 82, 613–617. [Google Scholar] [CrossRef]
- Bayoumi, R.A.; El-Louboudey, S.S.; Sidkey, N.M.; Abd-El-Rahman, M.A. Production, purification and characterization of thermoalkalophilic lipase for application in bio-detergent industry. J. Appl. Sci. Res. 2007, 3, 1752–1765. [Google Scholar]
- Orsavova, J.; Misurcova, L.; Ambrozova, J.V.; Vicha, R.; Mlcek, J. Fatty acids composition of vegetable oils and its contribution to dietary energy intake and dependence of cardiovascular mortality on dietary intake of fatty acids. Int. J. Mol. Sci. 2015, 16, 12871–12890. [Google Scholar] [CrossRef] [PubMed]
- Elsayed, H.H.; Elrahman, M.K.A.; Emara, A.H.; El-Hafez, A.A. Compare effects of fatty acid composition (olive, coconut oil and butter) on adipose liver tissue, and serum lipid profile in albino rats. IOSR-JBB 2015, 1, 28–38. [Google Scholar]
- Singh, J.; Singh, M.K.; Kumar, M.; Thakur, I.S. Immobilized lipase from Schizophyllum commune ISTL04 for the production of fatty acids methyl esters from cyanobacterial oil. Bioresour. Technol. 2015, 188, 214–218. [Google Scholar] [CrossRef] [PubMed]
- Yan, Q.J.; Duan, X.J.; Liu, Y.; Jiang, Z.Q.; Yang, S.Q. Expression and characterization of a novel 1,3-regioselective cold-adapted lipase from Rhizomucor endophyticus suitable for biodiesel synthesis. Biotechnol. Biofuels 2016, 9, 86. [Google Scholar] [CrossRef] [PubMed]
- Lotrakul, P.; Dharmsthiti, S. Purification and characterization of lipase from Aeromonas sobria LP004. J. Biotechnol. 1997, 54, 113–120. [Google Scholar] [CrossRef]
- Tong, X.X.; Busk, P.K.; Lange, L. Characterization of a new sn-1,3-regioselective triacylglycerol lipase from Malbranchea cinnamomea. Biotechnol. Appl. Biochem. 2015, 63, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Langone, M.A.P.; Sant’Anna, G.L., Jr. Enzymatic synthesis of medium-chain triglycerides in a solvent-free system. Appl. Biochem. Biotechnol. 1999, 77–79, 759–770. [Google Scholar] [CrossRef]
- Liao, X.Y.; Zhu, Y.L.; Wang, S.G.; Li, Y.W. Producing triacetylglycerol with glycerol by two steps: Esterification and acetylation. Fuel Process. Technol. 2009, 90, 988–993. [Google Scholar] [CrossRef]
- Saxena, R.K.; Isar, J.; Saran, S.; Kaushik, R.; Davidson, W.S. Efficient microwave-assisted hydrolysis of triolein and synthesis of bioester, bio-surfactant and glycerides using Aspergillus carneus lipase. Curr. Sci. 2005, 89, 1000–1003. [Google Scholar]
- Sharma, K.; Rathore, M. Comparative evaluation of specific activity of crude and partially purified lipase preparation from lipolytic bacteria. IJPSR 2010, 1, 31–38. [Google Scholar]
- Kam, Y.C.; Hii, S.L.; Sim, C.Y.Y.; Ong, L.G.A. Schizophyllum commune lipase production on pretreated sugarcane bagasse and its effectiveness. Int. J. Polym. Sci. 2016, 2016, 2918202. [Google Scholar] [CrossRef]
- Bondioli, P.; Bella, L.D. An alternative spectrophotometric method for determination of free glycerol in biodiesel. EJLST 2005, 107, 153–157. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |
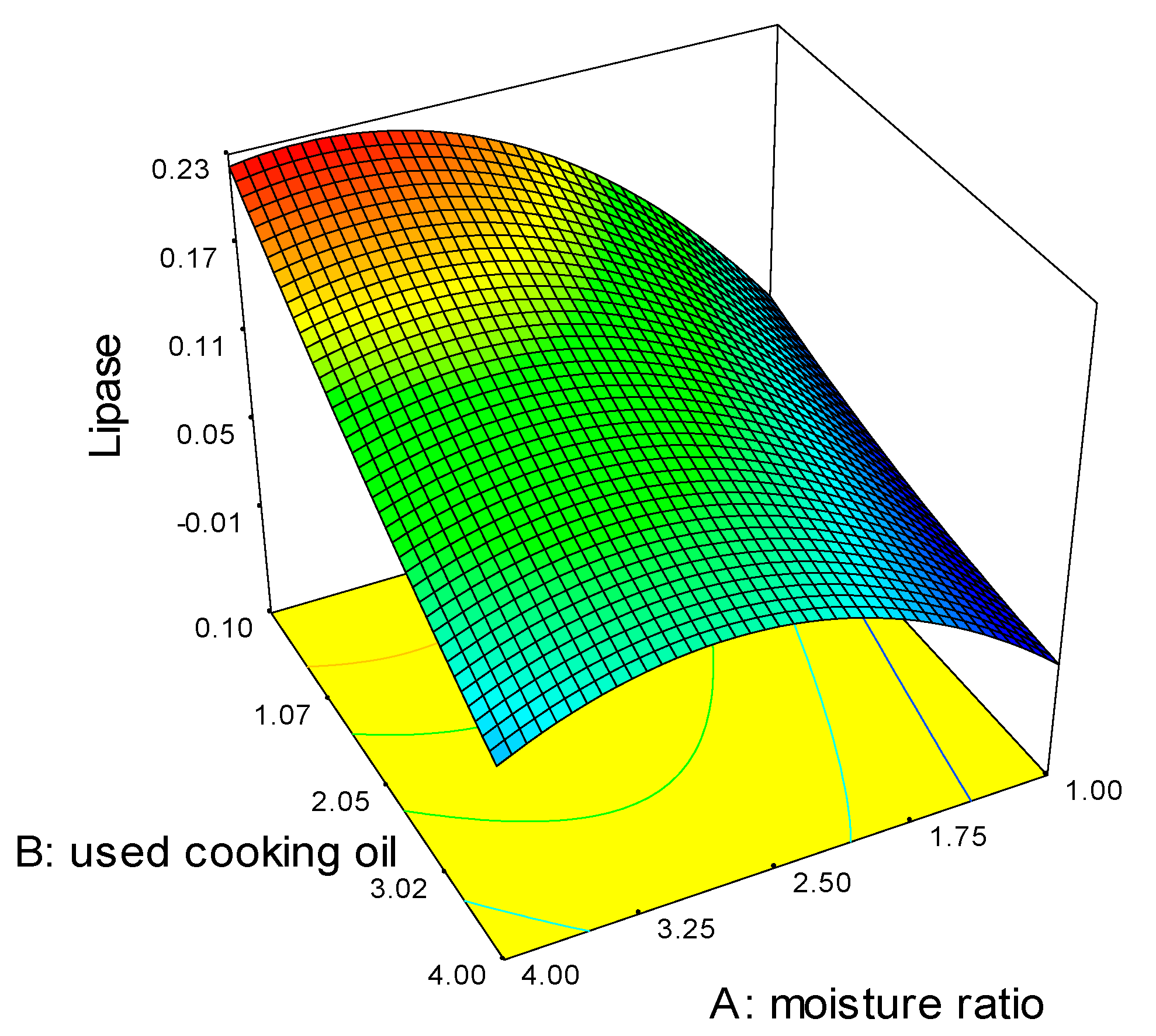
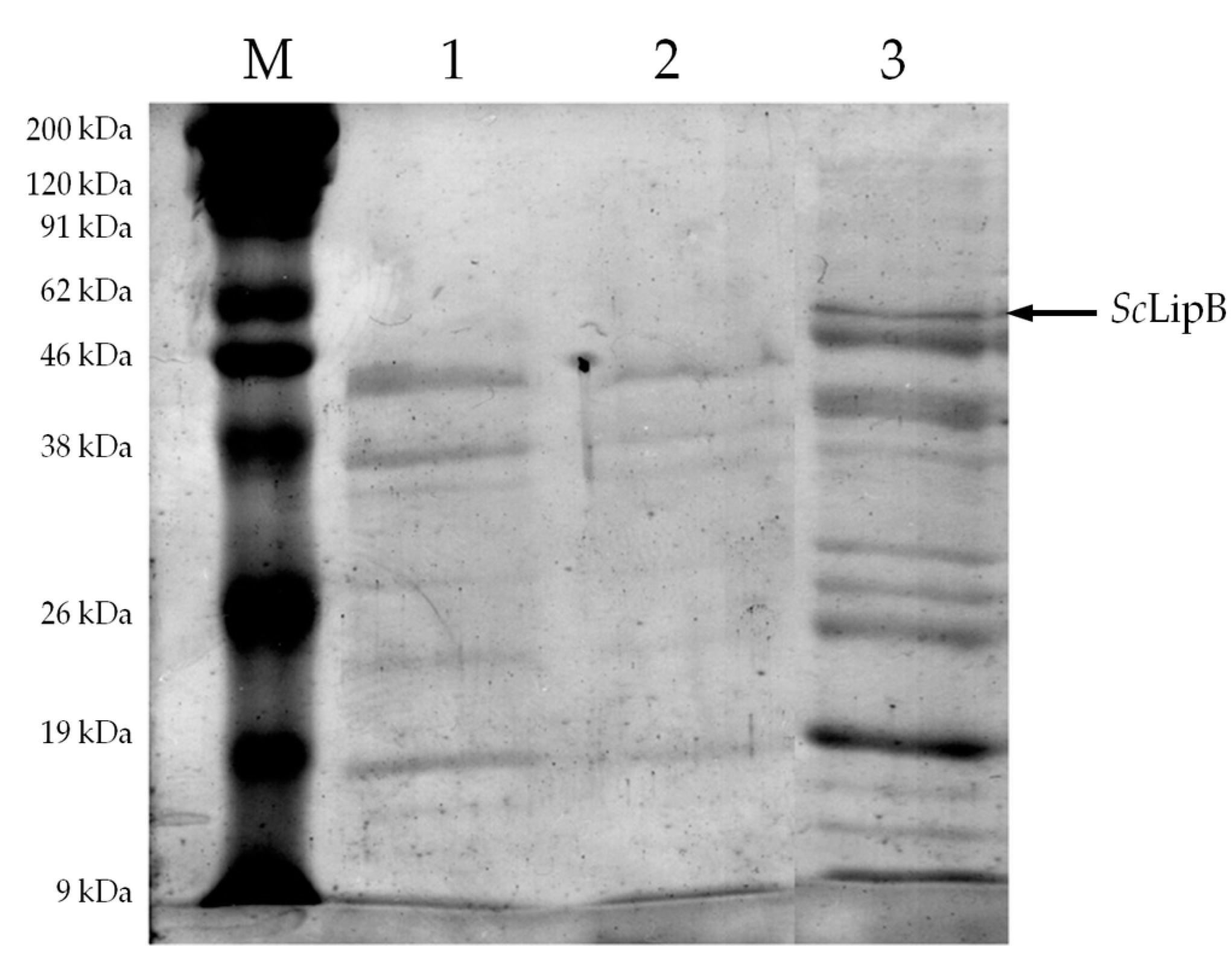
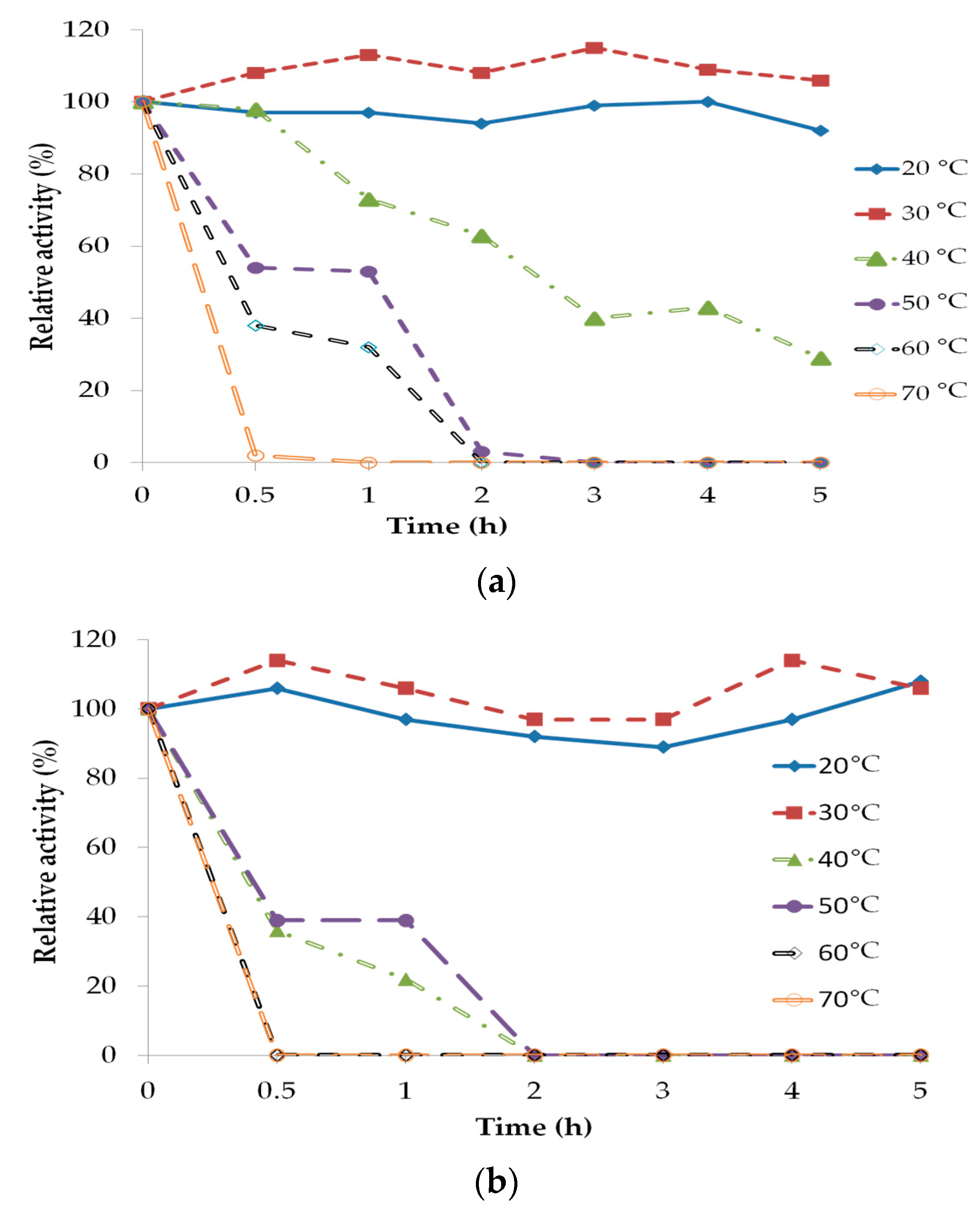
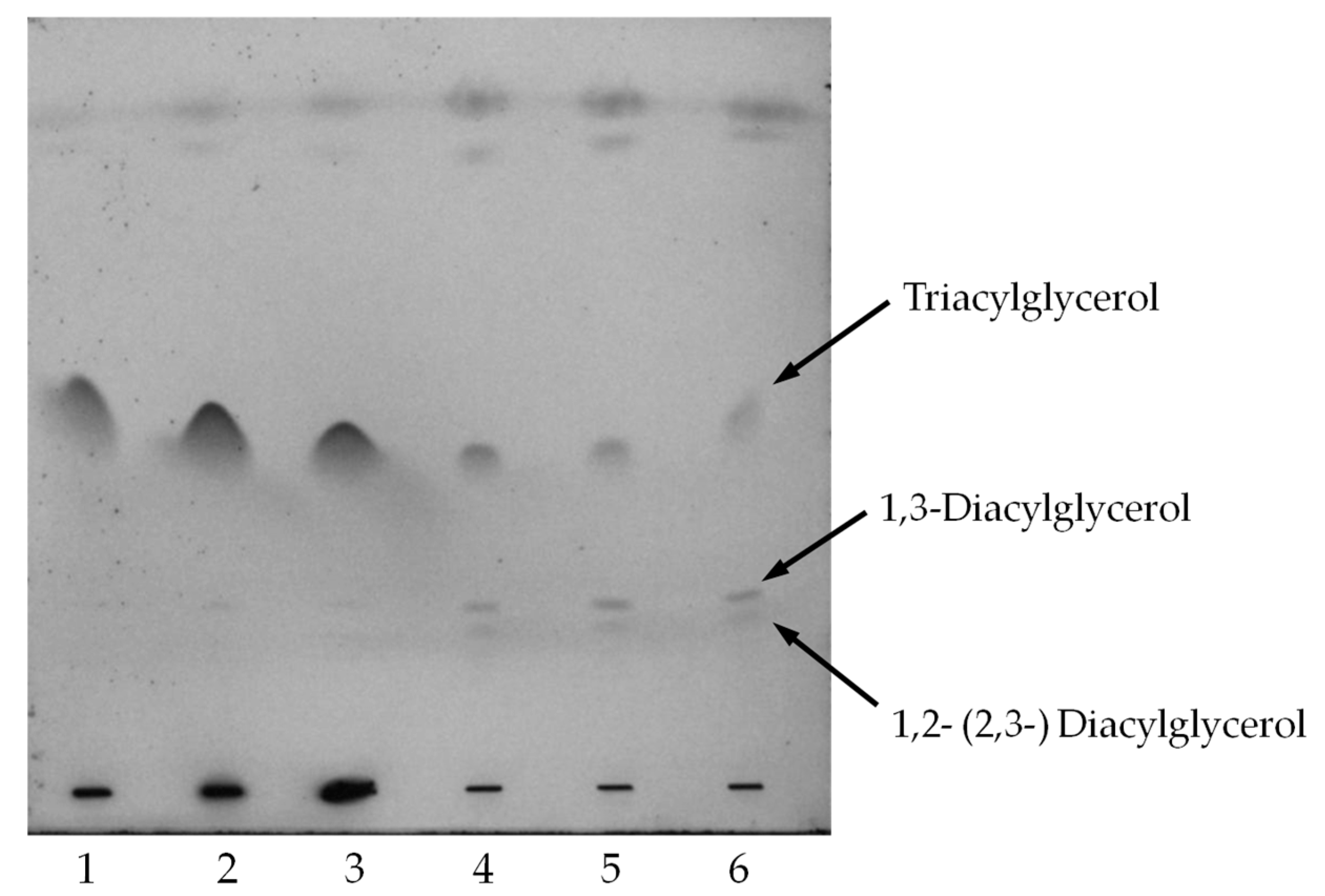


| Source | Sum of Squares | Degrees of Freedom | Mean Square | F Value | p-Value Prob. > F |
|---|---|---|---|---|---|
| Model | 0.078 | 7 | 0.011 | 4.22 | 0.0014 |
| A 1 | 9.663 × 10−3 | 1 | 9.663 × 10−3 | 3.64 | 0.0637 |
| B 2 | 0.029 | 1 | 0.029 | 10.77 | 0.0021 |
| C 3 | 4.760 × 10−3 | 1 | 4.760 × 10−3 | 1.80 | 0.1875 |
| H 4 | 0.018 | 1 | 0.018 | 6.89 | 0.0122 |
| AB | 7.701 × 10−3 | 1 | 7.701 × 10−3 | 2.91 | 0.0959 |
| AC | 4.641 × 10−3 | 1 | 4.641 × 10−3 | 1.75 | 0.1930 |
| AH | 4.681 × 10−3 | 1 | 4.681 × 10−3 | 1.77 | 0.1912 |
| Lack of fit | 0.030 | 8 | 3.772 × 10−3 | 1.59 | 0.1658 |
| Source | Sum of Squares | Degree of Freedom | Mean Square | F-Value | p-Value Prob. > F |
|---|---|---|---|---|---|
| Model | 0.130 | 3 | 0.044 | 14.24 | <0.0001 |
| A 1 | 0.052 | 1 | 0.052 | 16.72 | 0.0005 |
| B 2 | 0.059 | 1 | 0.059 | 19.20 | 0.0002 |
| AB | 0.013 | 1 | 0.013 | 4.27 | 0.0503 |
| Lack of fit | 0.027 | 5 | 5.404 × 10−3 | 2.22 | 0.0973 |
| Step | Volume (mL) | Total Protein (mg) | Total Activity (U·L−1) | Specific Activity (U·mg−1) | Purification (Fold) | Yield (%) |
|---|---|---|---|---|---|---|
| Crude | 600.0 | 159.18 | 14.22 | 0.089 | 1.00 | 100.00 |
| 30% | 87.0 | 16.23 | 5.10 | 0.314 | 3.53 | 35.86 |
| 40% | 28.9 | 3.76 | 0.88 | 0.234 | 2.63 | 6.19 |
| 50% | 152.0 | 18.16 | 1.14 | 0.063 | 0.71 | 8.02 |
| 60% | 108.0 | 11.21 | 0.81 | 0.072 | 0.81 | 5.70 |
| 70% | 5.0 | 2.48 | 0.18 | 0.073 | 0.82 | 1.27 |
| 80% | 7.5 | 9.20 | 0.17 | 0.018 | 0.20 | 1.20 |
| 90% | 5.5 | 3.68 | 0.00 | 0.000 | 0.00 | 0.00 |
| Leftover | 500 | 73.88 | 0.00 | 0.000 | 0.00 | 0.00 |
| Partial Purified Lipases | Substrates | Glycerol Production (μmol/mgprotein/day) |
|---|---|---|
| ScLipA | Crude fish oil | 2.489 ± 0.03 |
| Coconut oil | 0.405 ± 0.01 | |
| Butter | 0.236 ± 0.02 | |
| ScLipB | Crude fish oil | 0.460 ± 0.03 |
| Coconut oil | nd 1 | |
| Butter | nd 1 |
| Variable | Level | |
|---|---|---|
| −1 | +1 | |
| Inoculum density (number of mycelia discs) | 2 | 10 |
| Moisture ratio (mL per g of SB) | 1:1 | 1:4 |
| Urea (%) | 0.1 | 1.5 |
| Temperature (°C) | 28 | 35 |
| SB solids (g) | 2 | 6 |
| Glucose (%) | 0 | 1 |
| SB particle size (mm) | 0.5–0.85 | 1.5–2.0 |
| UCO ratio (mL per g of SB) | 1:0.1 | 1:4 |
| Variable | Level | ||
|---|---|---|---|
| Moisture ratio (mL per g of SB) | 1:1 | 1:2 | 1:4 |
| UCO ratio (mL per g of SB) | 1:0.1 | 1:1 | 1:4 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kam, Y.C.; Woo, K.K.; Ong, L.G.A. One-Step Partially Purified Lipases (ScLipA and ScLipB) from Schizophyllum commune UTARA1 Obtained via Solid State Fermentation and Their Applications. Molecules 2017, 22, 2106. https://doi.org/10.3390/molecules22122106
Kam YC, Woo KK, Ong LGA. One-Step Partially Purified Lipases (ScLipA and ScLipB) from Schizophyllum commune UTARA1 Obtained via Solid State Fermentation and Their Applications. Molecules. 2017; 22(12):2106. https://doi.org/10.3390/molecules22122106
Chicago/Turabian StyleKam, Yew Chee, Kwan Kit Woo, and Lisa Gaik Ai Ong. 2017. "One-Step Partially Purified Lipases (ScLipA and ScLipB) from Schizophyllum commune UTARA1 Obtained via Solid State Fermentation and Their Applications" Molecules 22, no. 12: 2106. https://doi.org/10.3390/molecules22122106
APA StyleKam, Y. C., Woo, K. K., & Ong, L. G. A. (2017). One-Step Partially Purified Lipases (ScLipA and ScLipB) from Schizophyllum commune UTARA1 Obtained via Solid State Fermentation and Their Applications. Molecules, 22(12), 2106. https://doi.org/10.3390/molecules22122106





