Application of an Ultrafine Shearing Method for the Extraction of C-Phycocyanin from Spirulina platensis
Abstract
:1. Introduction
2. Results and Discussion
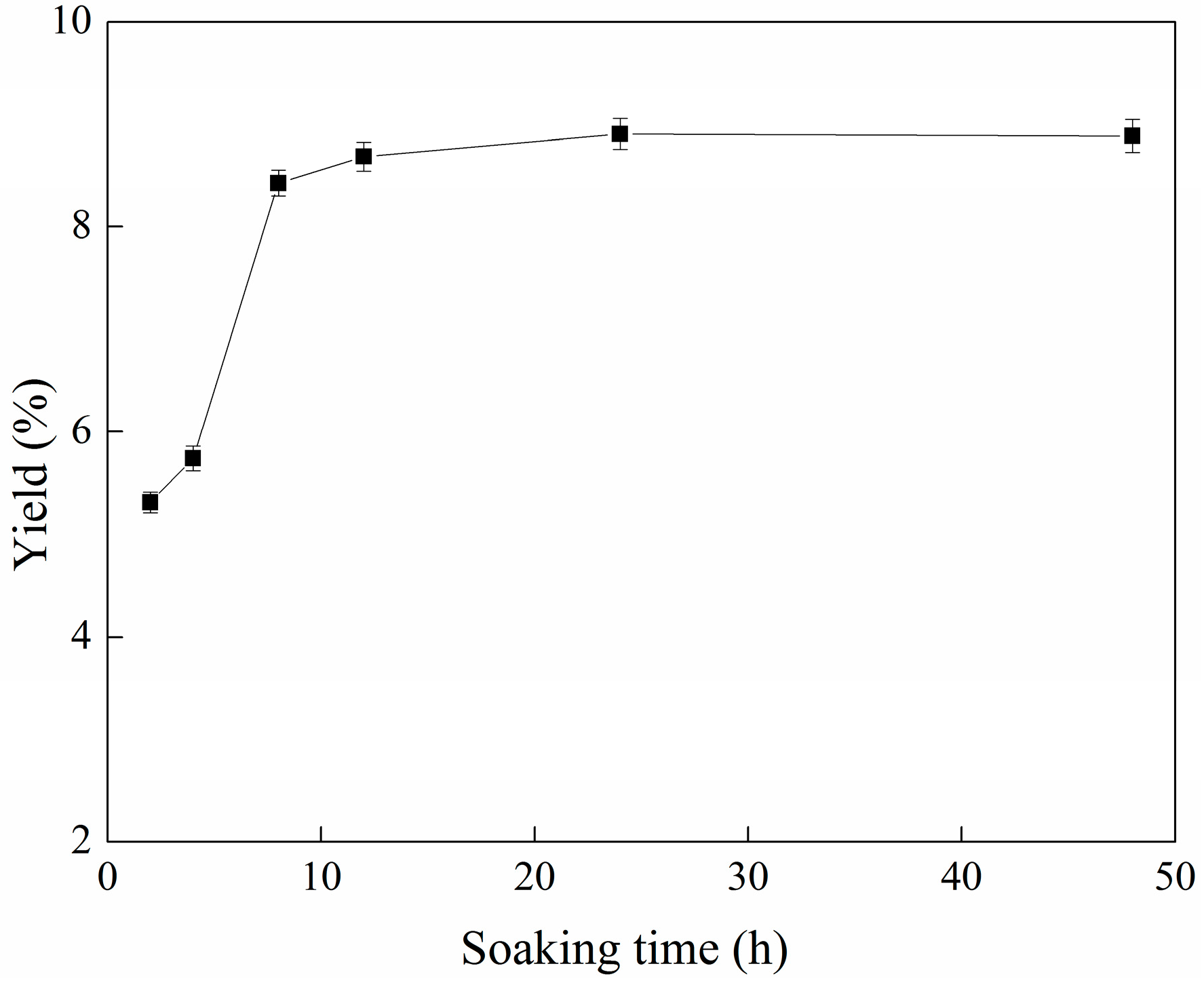
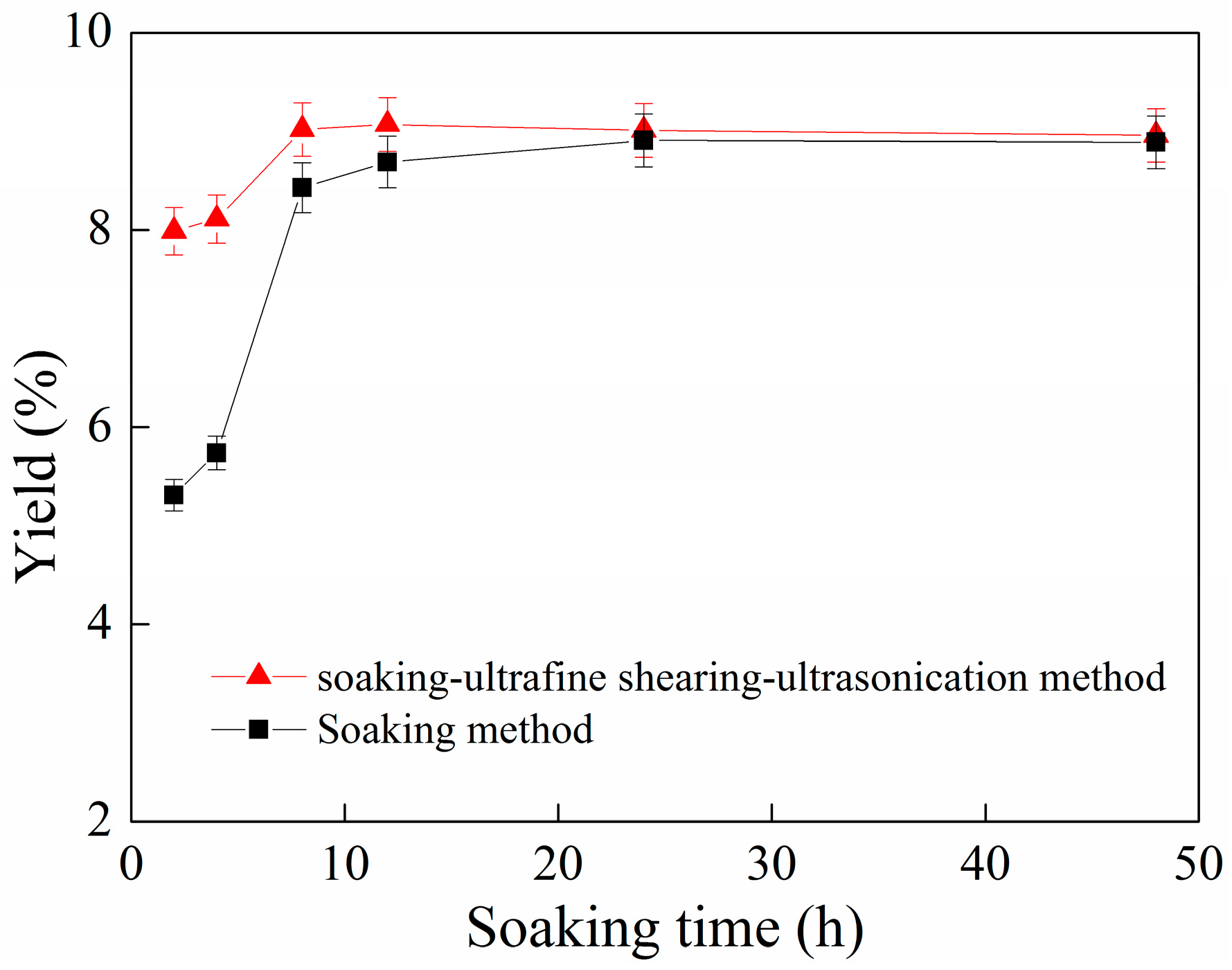
2.1. Effect of Soaking Time on the Yield of C-PC
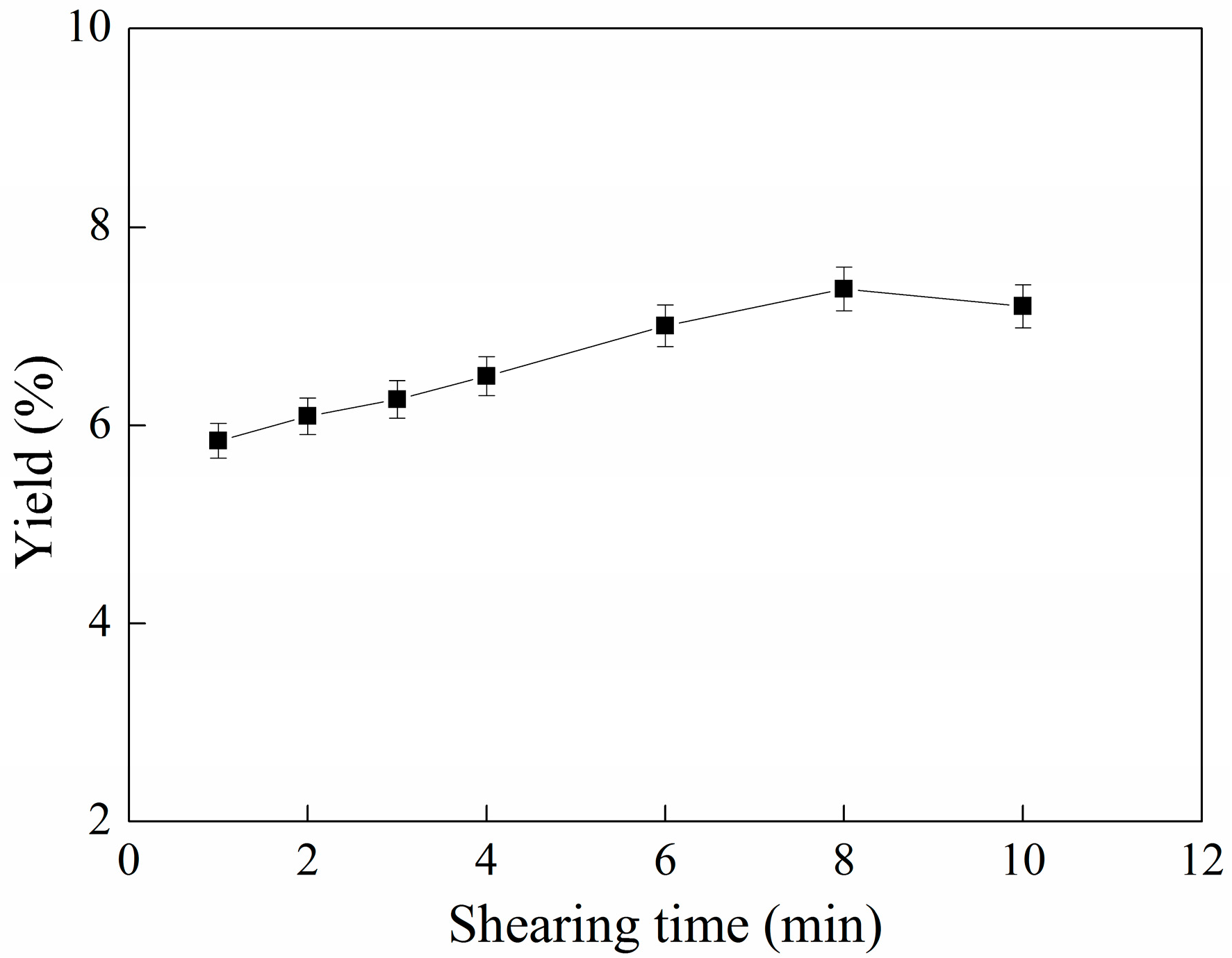
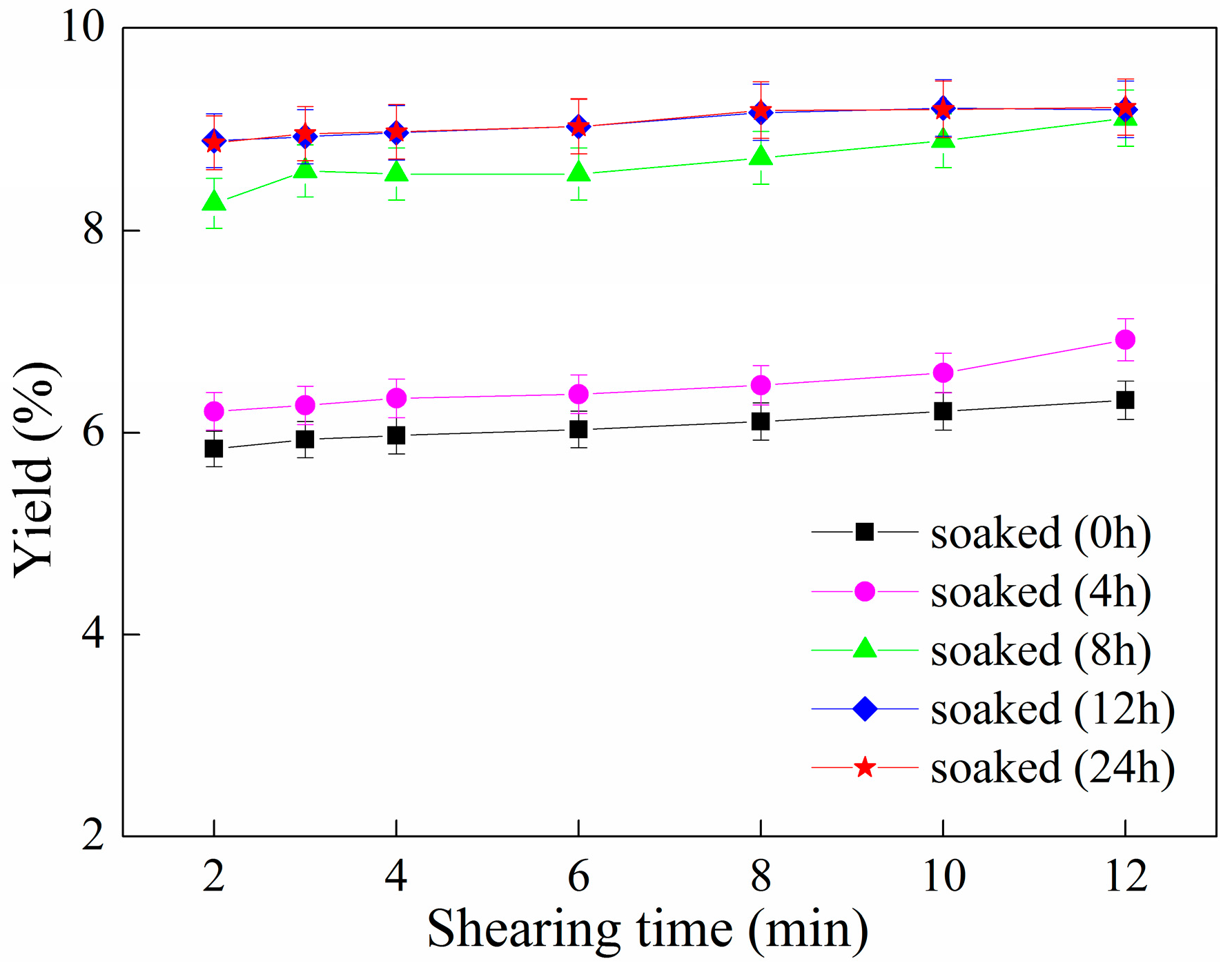
2.2. Effect of Ultrafine Shearing Time on the Yield of C-PC
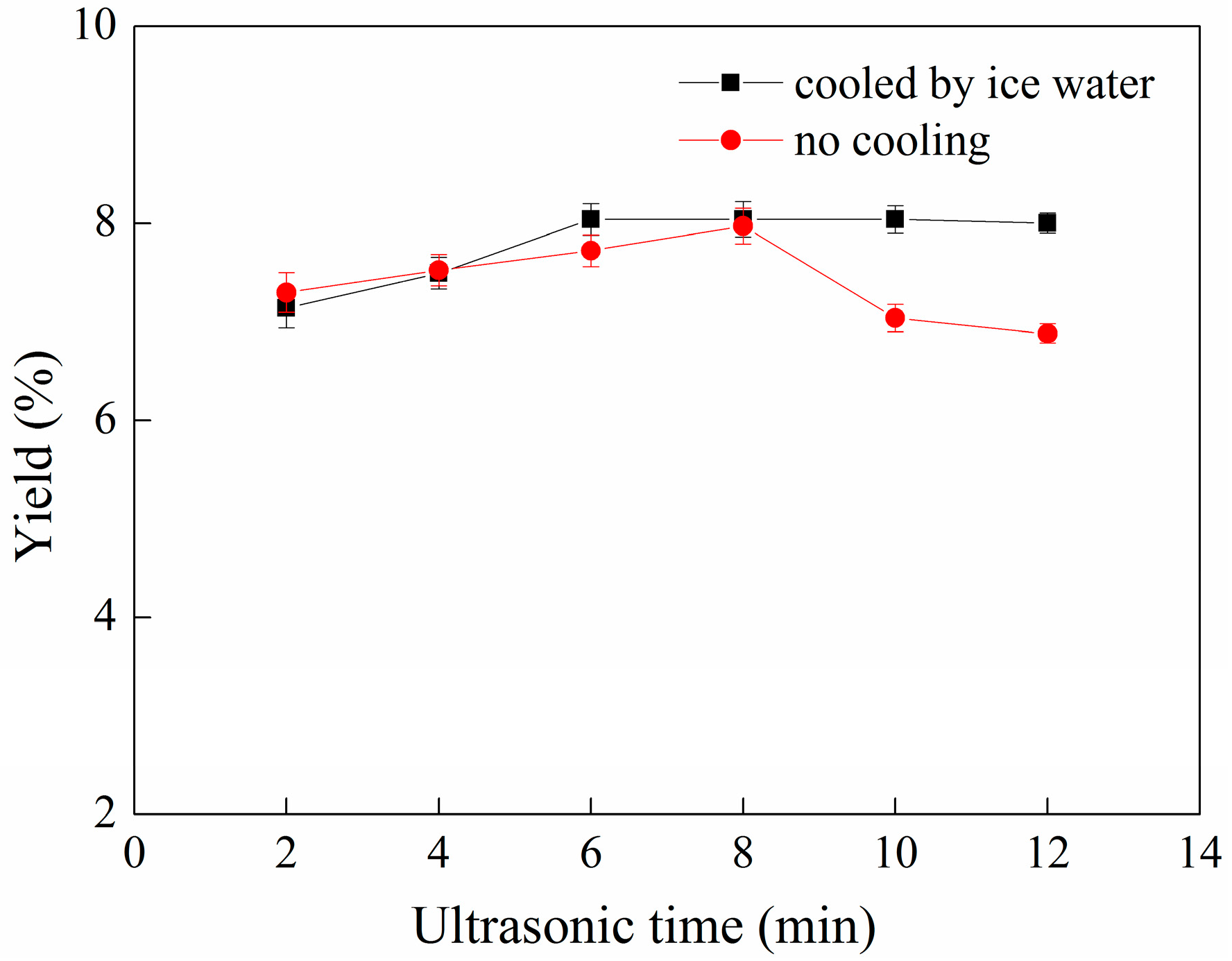
2.3. Effect of Ultrasonication Time on the Yield of C-PC
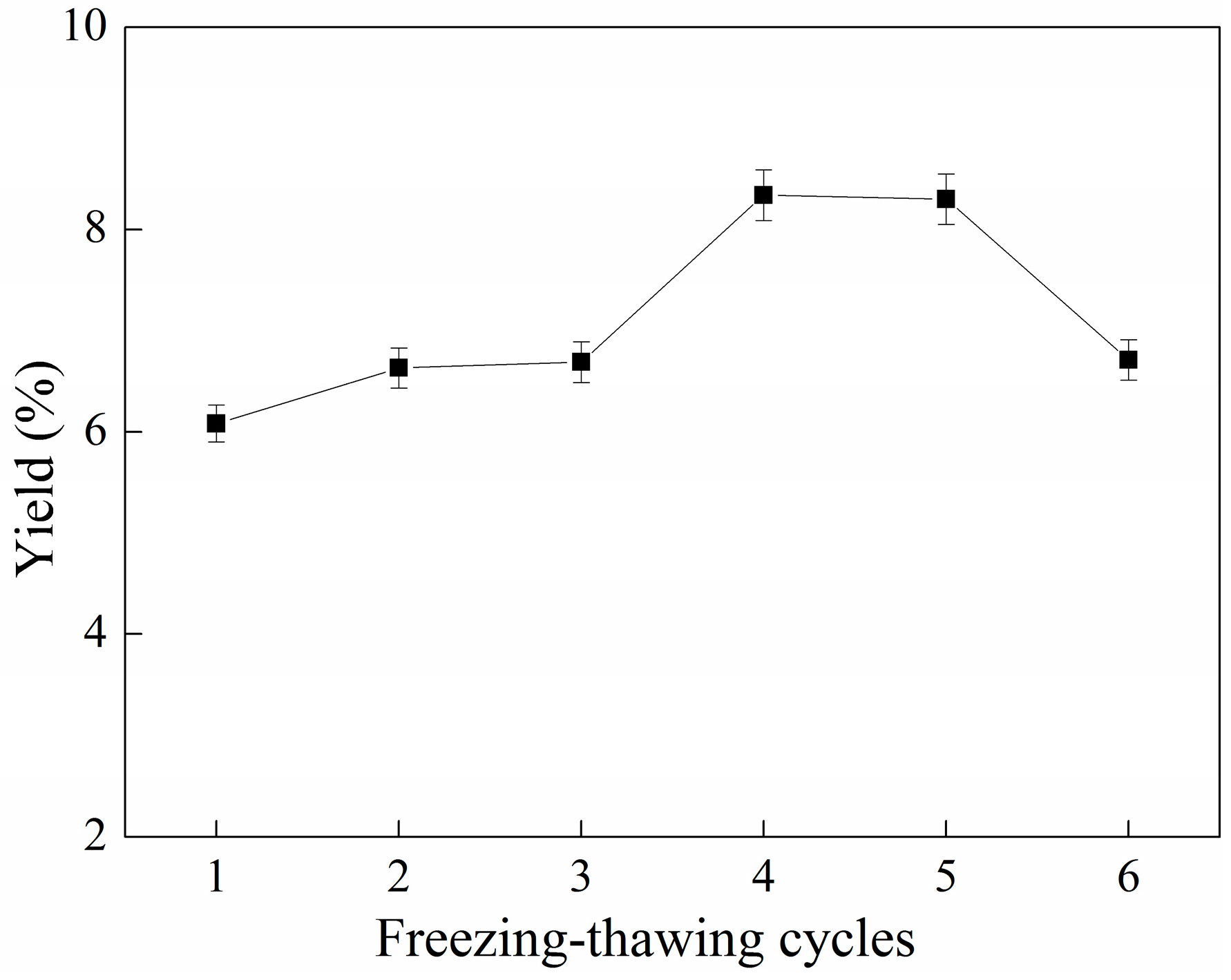
2.4. Effect of Cycles of Freezing-Thawing on the Yield of C-PC
2.5. Effect of Soaking Time and Ultrafine Shearing Time on the Yield of C-PC
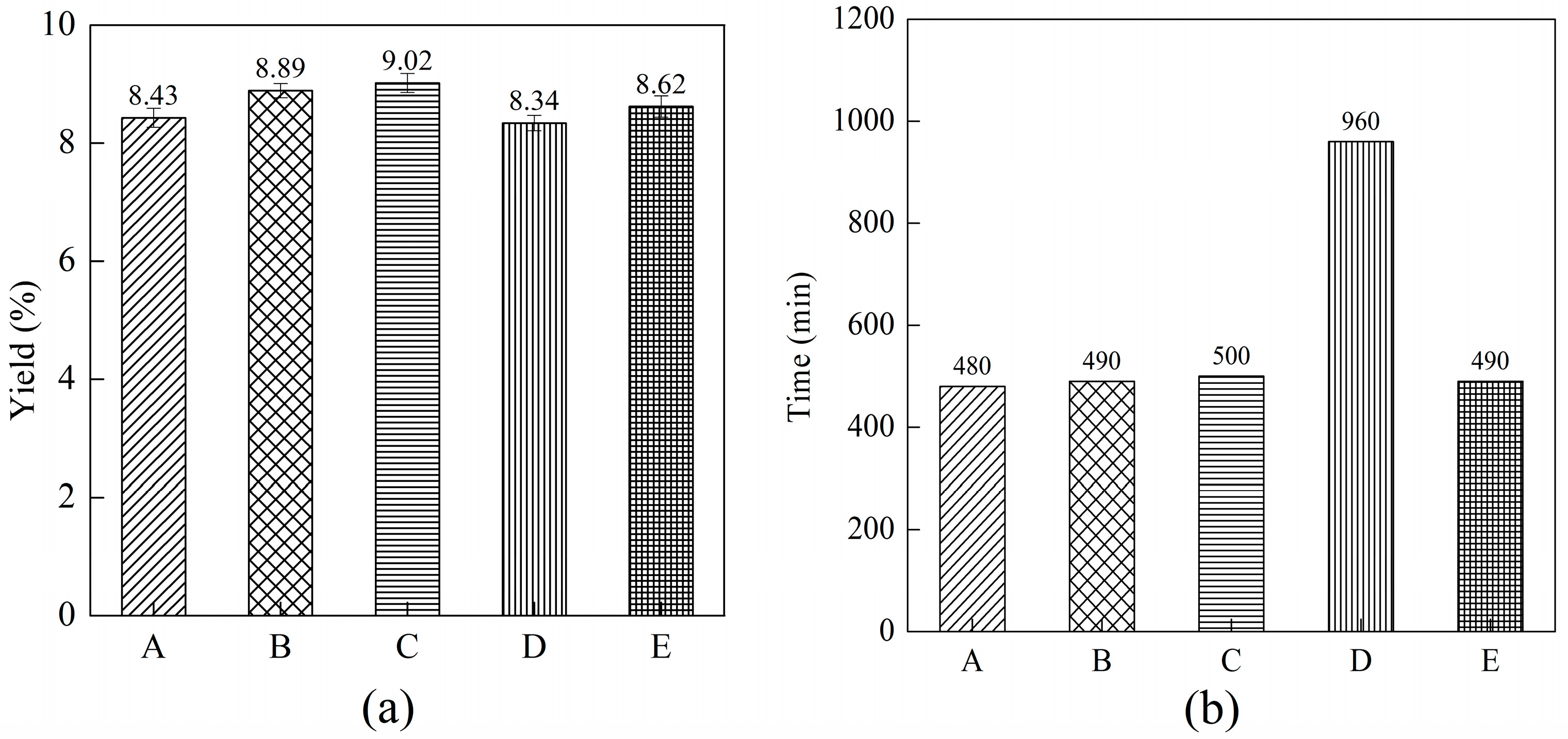
2.6. The Combined Effect of the Soaking Method, Ultrafine Shearing Method and Ultrasonication Method on the Yield of C-PC
2.7. The Combined Effect of the Soaking Method, Ultrafine Shearing Method and Ultrasonication Method on the Yield of C-PC
3. Materials and Methods
3.1. Materials
3.2. Methods
3.2.1. Concentration and Yield of C-PC
3.2.2. Soaking
3.2.3. Ultrasonication
3.2.4. Freezing-Thawing
3.2.5. Soaking-Ultrafine Shearing
3.2.6. Soaking-Ultrafine Shearing-Ultrasonication
3.2.7. Scanning Electron Micrograph (SEM) Analysis
3.2.8. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Gupta, M.; Dwivedi, U.N.; Khandelwal, S. C-Phycocyanin: An effective protective agent against thymic atrophy by tributyltin. Toxicol. Lett. 2011, 204, 2–11. [Google Scholar] [CrossRef] [PubMed]
- Setyoningrum, T.M.; Nur, M.M.A. Optimization of C-phycocyanin production from S. platensis cultivated on mixotrophic condition by using response surface methodology. Biocatal. Agric. Biotechnol. 2015, 4, 603–607. [Google Scholar] [CrossRef]
- Ismaiel, M.M.S.; El-Ayouty, Y.M.; Piercey-Normore, M. Role of pH on antioxidants production by Spirulina (Arthrospira) platensis. Braz. J. Microbiol. 2016, 47, 298–304. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.K.; Shrivastav, A.; Mishra, S. Effect of preservatives for food grade C-PC from Spirulina platensis. Process Biochem. 2008, 43, 339–345. [Google Scholar] [CrossRef]
- Antelo, F.S.; Costa, J.A.V.; Kalil, S.J. Thermal degradation kinetics of the phycocyanin from Spirulina platensis. Biochem. Eng. J. 2008, 41, 43–47. [Google Scholar] [CrossRef]
- Chaiklahan, R.; Chirasuwan, N.; Loha, V.; Tia, S.; Bunnag, B. Separation and purification of phycocyanin from Spirulina sp. using a membrane process. Bioresour. Technol. 2011, 102, 7159–7164. [Google Scholar] [CrossRef] [PubMed]
- Patil, G.; Raghavarao, K.S.M.S. Aqueous two phase extraction for purification of C-phycocyanin. Biochem. Eng. J. 2007, 34, 156–164. [Google Scholar] [CrossRef]
- Martínez, J.M.; Luengo, E.; Saldaña, G.; Álvarez, I.; Raso, J. C-phycocyanin extraction assisted by pulsed electric field from Artrosphira platensis. Food Res. Int. 2017, 99, 1042–1047. [Google Scholar] [CrossRef] [PubMed]
- Niu, J.F.; Wang, G.C.; Lin, X.Z.; Zhou, B.C. Large-scale recovery of C-phycocyanin from Spirulina platensis using expanded bed adsorption chromatography. J. Chromatogr. B 2007, 850, 267–276. [Google Scholar] [CrossRef] [PubMed]
- Pohndorf, R.S.; Camara, Á.S.; Larrosa, A.P.Q.; Pinheiro, C.P.; Strieder, M.M.; Pinto, L.A.A. Production of lipids from microalgae Spirulina sp.: Influence of drying, cell disruption and extraction methods. Biomass Bioenergy 2016, 93, 25–32. [Google Scholar] [CrossRef]
- Passos, F.; Uggetti, E.; Carrère, H.; Ferrer, I. Pretreatment of microalgae to improve biogas production: A review. Bioresour. Technol. 2014, 172, 403–412. [Google Scholar] [CrossRef] [PubMed]
- Martelli, G.; Folli, C.; Visai, L.; Daglia, M.; Ferrari, D. Thermal stability improvement of blue colorant C-Phycocyanin from Spirulina platensis for food industry applications. Process Biochem. 2014, 49, 154–159. [Google Scholar] [CrossRef]
- Tan, C.H.; Show, P.L.; Chang, J.S.; Ling, T.C.; Lan, J.C.W. Novel approaches of producing bioenergies from microalgae: A recent review. Biotechnol. Adv. 2015, 33, 1219–1227. [Google Scholar] [CrossRef] [PubMed]
- Brennan, L.; Owende, P. Biofuels from microalgae—A review of technologies for production, processing, and extractions of biofuels and co-products. Renew. Sustain. Energy Rev. 2010, 14, 557–577. [Google Scholar] [CrossRef]
- Preece, K.E.; Hooshyar, N.; Krijgsman, A.J.; Fryer, P.J.; Zuidam, N.J. Intensification of protein extraction from soybean processing materials using hydrodynamic cavitation. Innov. Food Sci. Emerg. Technol. 2017, 41, 47–55. [Google Scholar] [CrossRef]
- Gerde, J.A.; Wang, T.; Yao, L.; Jung, S.; Johnson, L.A.; Lamsal, B. Optimizing protein isolation from defatted and non-defatted Nannochloropsis microalgae biomass. Algal Res. 2013, 2, 145–153. [Google Scholar] [CrossRef]
- Biller, P.; Friedman, C.; Ross, A.B. Hydrothermal microwave processing of microalgae as a pre-treatment and extraction technique for bio-fuels and bio-products. Bioresour. Technol. 2013, 136, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Safi, C.; Ursu, A.V.; Laroche, C.; Zebib, B.; Merah, O.; Pontalier, P.Y.; Vaca-Garcia, C. Aqueous extraction of proteins from microalgae: Effect of different cell disruption methods. Algal Res. 2014, 3, 61–65. [Google Scholar] [CrossRef]
- Safi, C.; Frances, C.; Ursu, A.V.; Laroche, C.; Pouzet, C.; Vaca-Garcia, C.; Pontalier, P.Y. Understanding the effect of cell disruption methods on the diffusion of Chlorella vulgaris proteins and pigments in the aqueous phase. Algal Res. 2015, 8, 61–68. [Google Scholar] [CrossRef]
- Chen, F.; Zhang, X.; Zhang, Q.; Du, X.; Yang, L.; Zu, Y.; Yang, F. Simultaneous synergistic microwave–ultrasonic extraction and hydrolysis for preparation of trans-resveratrol in tree peony seed oil-extracted residues using imidazolium-based ionic liquid. Ind. Crop. Prod. 2016, 94, 266–280. [Google Scholar] [CrossRef]
- Mittal, R.; Tavanandi, H.A.; Mantri, V.A.; Raghavarao, K.S.M.S. Ultrasound assisted methods for enhanced extraction of phycobiliproteins from marine macro-algae, Gelidium pusillum (Rhodophyta). Ultrason. Sonochem. 2017, 38, 92–103. [Google Scholar] [CrossRef] [PubMed]
- Chemat, F.; Rombaut, N.; Meullemiestre, A.; Turk, M.; Perino, S.; Fabiano-Tixier, A.; Abert-Vian, M. Review of green food processing techniques. Preservation, transformation, and extraction. Innov. Food Sci. Emerg. Technol. 2017, 41, 357–377. [Google Scholar] [CrossRef]
- Show, K.Y.; Lee, D.J.; Tay, J.H.; Lee, T.M.; Chang, J.S. Microalgal drying and cell disruption—Recent advances. Bioresour. Technol. 2015, 184, 258–266. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Yuan, W. Microalgal cell disruption in a high-power ultrasonic flow system. Bioresour. Technol. 2015, 193, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Feng, Y.; Lun, J. Aqueous two-phase countercurrent distribution for the separation of C-phycocyanin and allophycocyanin from Spirulina platensis. Food Bioprod. Process. 2012, 90, 111–117. [Google Scholar] [CrossRef]
- Wang, F.; Liu, Y.H.; Ma, Y.; Cui, Z.G.; Shao, L.L. Application of TMA-PEG to promote C-phycocyanin extraction from S. platensis in the PEG ATPS. Process Biochem. 2017, 52, 283–294. [Google Scholar] [CrossRef]
- Bennett, A.; Bogorad, L. Complementary chromatic adaptation in a filamentous blue-green alga. J. Cell Biol. 1973, 58, 419–435. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples are available from the authors. |

© 2017 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, J. Application of an Ultrafine Shearing Method for the Extraction of C-Phycocyanin from Spirulina platensis. Molecules 2017, 22, 2023. https://doi.org/10.3390/molecules22112023
Yu J. Application of an Ultrafine Shearing Method for the Extraction of C-Phycocyanin from Spirulina platensis. Molecules. 2017; 22(11):2023. https://doi.org/10.3390/molecules22112023
Chicago/Turabian StyleYu, Jianfeng. 2017. "Application of an Ultrafine Shearing Method for the Extraction of C-Phycocyanin from Spirulina platensis" Molecules 22, no. 11: 2023. https://doi.org/10.3390/molecules22112023
APA StyleYu, J. (2017). Application of an Ultrafine Shearing Method for the Extraction of C-Phycocyanin from Spirulina platensis. Molecules, 22(11), 2023. https://doi.org/10.3390/molecules22112023



