Optimization of Goat Milk with ACE Inhibitory Peptides Fermented by Lactobacillus bulgaricus LB6 Using Response Surface Methodology
Abstract
:1. Introduction
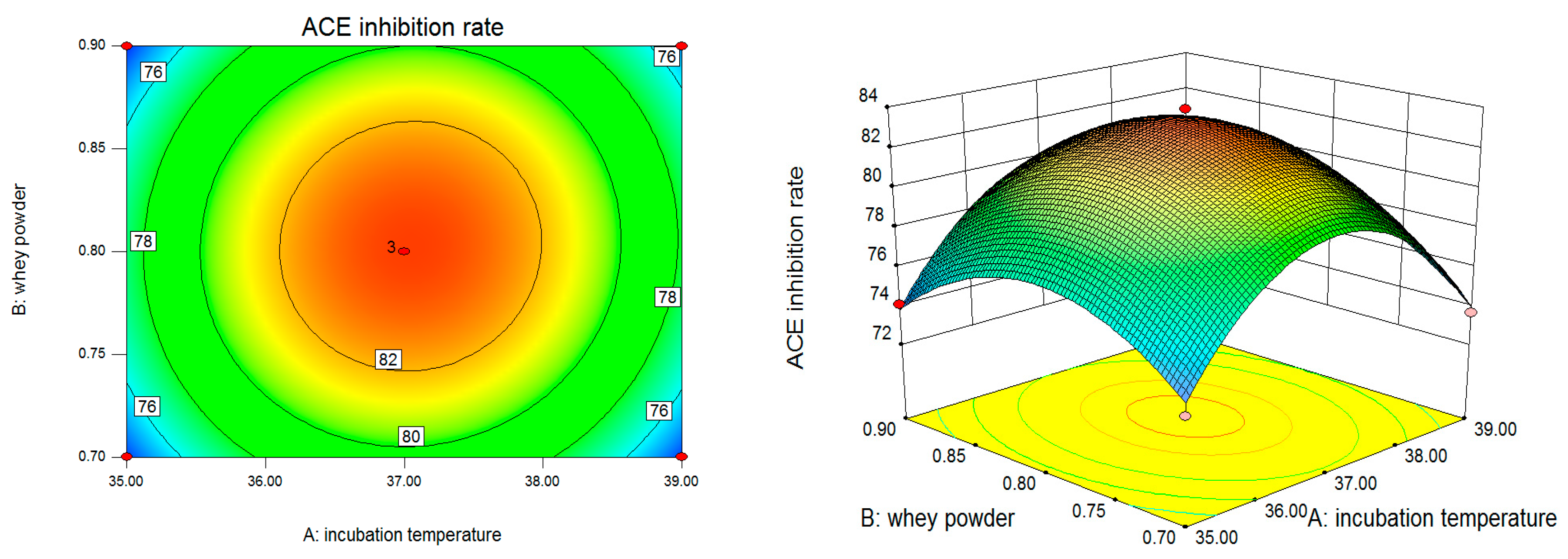
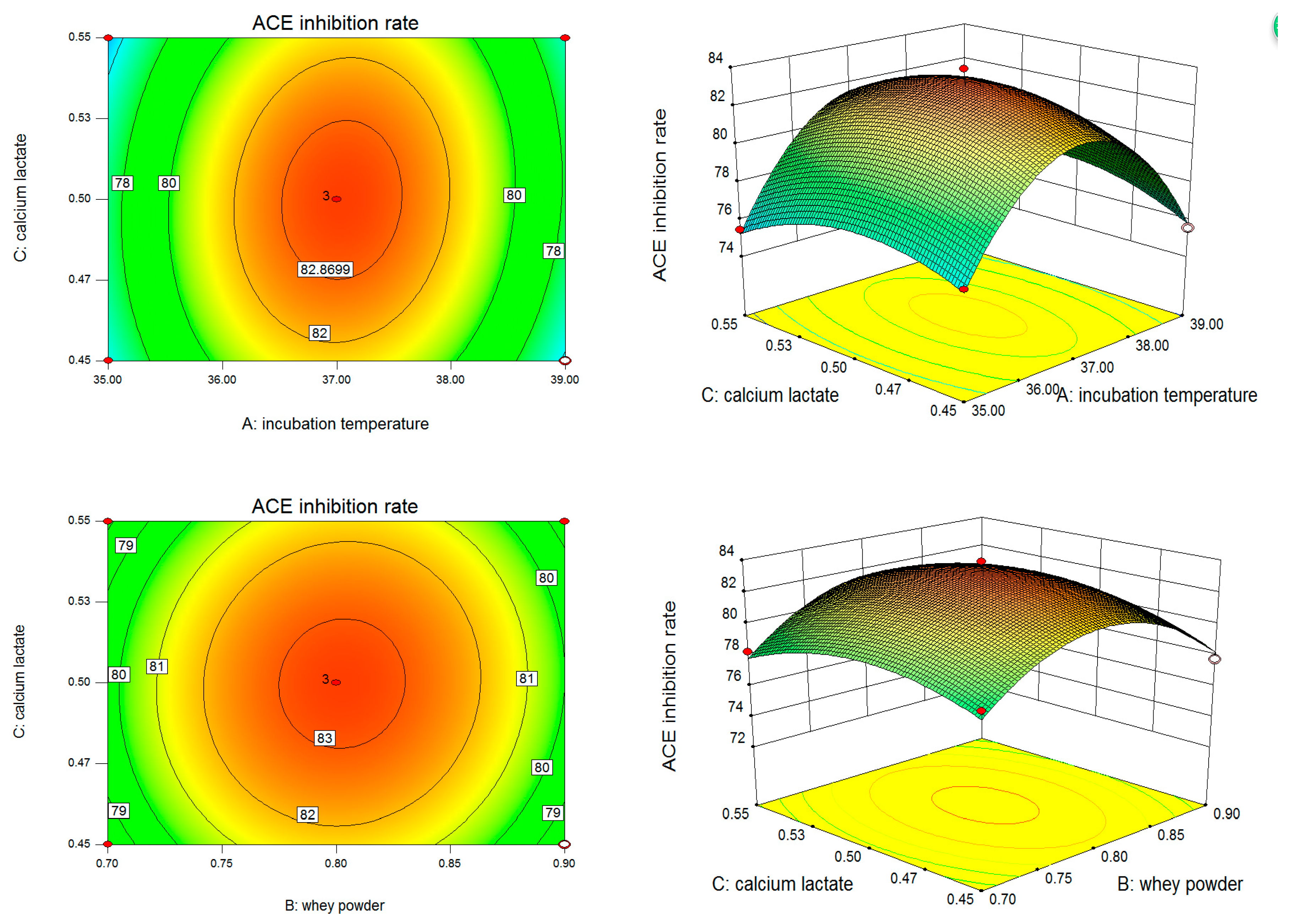
2. Results
2.1. The Experimental Design and Results of Box–Behnken
2.2. Regression Analysis
2.3. Variance Analysis
3. Discussion
4. Materials and Methods
4.1. Strain
4.2. Preparation of Fermented Goat Milk
4.3. Determination of ACE Inhibitory Activity
4.4. Calculation of the ACE Inhibition Rate
4.5. Measurement of Viable Counts
4.6. Optimization of Fermentation Conditions by RSM
4.7. Validation of the Model
4.8. Statistical Analysis of the Data
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Macmahon, S.; Peto, R.; Collins, R.; Godwin, J.; Cutler, J.; Sorlie, P.; Abbott, R.; Neaton, J.; Dyer, A.; Stamler, J. Blood pressure, stroke and coronary heart disease. Lancet 1990, 335, 827–838. [Google Scholar] [CrossRef]
- Duprez, D.; Helshoecht, P.V.; Eynde, W.V.; Leeman, M. Prevalance of hypertension in the adult population of Belgium: Report of a worksite study, Attention Hypertension. J. Hum. Hypertens. 2002, 16, 47–52. [Google Scholar] [CrossRef] [PubMed]
- James, P.A.; Oparil, S.; Carter, B.L.; Cushman, W.C.; Dennisonhimmelfarb, C.; Handler, J.; Lackland, D.T.; LeFevre, M.L.; MacKenzie, T.D.; Ogedegbe, O.; et al. 2014 evidence-based guideline for the management of high blood pressure in adults. JAMA 2014, 311, 507–520. [Google Scholar] [CrossRef] [PubMed]
- Grandi, A.M.; Gaudio, G.; Fachinetti, A.; Bianchi, L.; Nardo, B.; Zanzi, P.; Ceriani, L.; Guasti, L.; Venco, A. Hyperinsulinemia, family history of hypertension, and essential hypertension. Am. J. Hypertens. 1996, 9, 732–738. [Google Scholar] [CrossRef]
- Cushman, D.W.; Cheung, H.S. Spectrophotometric assay and properties of the angiotensin-converting-enzyme of rabbit lung. Biochem. Pharmacol. 1971, 20, 1637–1638. [Google Scholar] [CrossRef]
- Skeggs, L.T.; Kahn, J.R.; Shumway, N.P. The preparation and function of the hypertension-converting enzyme. J. Exp. Med. 1956, 103, 295–299. [Google Scholar] [CrossRef] [PubMed]
- Ondetti, M.A.; Rubin, B.; Cushman, D.W. Design of specific inhibitors of angiotensin-converting enzyme: New class of orally active antihypertensive agents. Science 1977, 196, 441–444. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y.; Masuda, O.; Takano, T. Decrease of tissue angiotensin I-converting enzyme activity upon feeding sour milk in spontaneous hypertensive rats. Biosci. Biotechnol. Biochem. 1996, 60, 488–489. [Google Scholar] [CrossRef] [PubMed]
- Pihlanto-Leppala, A. Bioactive peptides derived from bovine whey proteins: Opioid and ACE-inhibitory peptides. Trends Food Sci. Technol. 2001, 11, 347–356. [Google Scholar] [CrossRef]
- Leclerc, P.L.; Gauthier, S.F.; Bachelardb, H.; Santure, M.; Roy, D. Antihypertensive activity of casein-enriched milk fermented by Lactobacillus helveticus. Int. Dairy J. 2002, 12, 995–1004. [Google Scholar] [CrossRef]
- Tenenbaum, A.; Grossman, E.; Shemesh, J.; Fisman, E.Z.; Nosrati, I.; Motro, M. Intermediate but not low doses of aspirin can suppress angiotensin-converting enzyme inhibitor-induced cough. Am. J. Hypertens. 2000, 13, 776–782. [Google Scholar] [CrossRef]
- Abassi, Z.; Winaver, J.; Feuerstein, G.Z. The biochemical pharmacology of renin inhibitors: Implications for translational medicine in hypertension, diabetic nephropathy and heart failure: Expectations and reality. Biochem. Pharmacol. 2009, 78, 933–940. [Google Scholar] [CrossRef] [PubMed]
- Ondetti, M.A.; Cushman, D.W. Enzymes of the renin-angiotensin system and their inhibitors. Annu. Rev. Biochem. 1982, 51, 283–308. [Google Scholar] [CrossRef] [PubMed]
- Maruyama, S.; Mitachi, H.; Awaya, J.; Kurono, M.; Tomizuka, N.; Suzuki, H. Angiotensin-I-converting enzyme inhibitory activity of the C-terminal hexapeptide of αs1-casein. Biosci. Biotechnol. Biochem. 2006, 51, 2557–2561. [Google Scholar] [CrossRef]
- Hernandezledesma, B.; Recio, I.; Ramos, M.; Amigo, L. Preparation of ovine and caprine β-lactoglobulin hydrolysates with ACE-inhibitory activity. Identification of active peptides from caprine β-lactoglobulin hydrolysed with thermolysin. Int. Dairy J. 2002, 12, 805–812. [Google Scholar] [CrossRef]
- Belewu, M.A.; Aiyegbusi, O.F. Comparison of the mineral content and apparent biological value of milk from human, cow and goat. J. Food Technol. Afr. 2002, 7, 9–11. [Google Scholar] [CrossRef]
- Park, Y.W.; Juárez, M.; Ramos, M.; Haenlein, G.F.W. Physico-chemical characteristics of goat and sheep milk. Small Rumin. Res. 2007, 68, 88–113. [Google Scholar] [CrossRef]
- Lópezaliaga, I.; Díazcastro, J.; Ma, J.M.A.; Barrionuevo, M.; Campos, M.S. A review of the nutritional and health aspects of goat milk in cases of intestinal resection. Dairy Sci. Technol. 2010, 90, 611–622. [Google Scholar] [CrossRef]
- Haenlein, G.F.W. Goat milk in human nutrition. Small Rumin. Res. 2004, 51, 155–163. [Google Scholar] [CrossRef]
- Kumar, R.; Chaudhary, K.; Sharma, M.; Nagpal, G.; Chauhan, J.S.; Singh, S.; Gautam, A.; Raghava, G.P.S. AHTPDB: A comprehensive platform for analysis and presentation of antihypertensive peptides. Nucleic Acids Res. 2015, 43, 956–962. [Google Scholar] [CrossRef] [PubMed]
- Parmar, H.; Hati, S.; Sakure, A. In vitro and in silico analysis of novel ACE-inhibitory bioactive peptides derived from fermented goat milk. Int. J. Pept. Res. Ther. 2017, 3, 1–13. [Google Scholar] [CrossRef]
- Quirós, A.; Hernández-Ledesma, B.; Ramos, M.; Amigo, L.; Recio, I. Angiotensin-converting enzyme inhibitory activity of peptides derived from caprine kefir. J. Dairy Sci. 2005, 88, 3480–3487. [Google Scholar] [CrossRef]
- Chen, H.; Ji, Z.; Shu, G.W.; Xing, H.N. Effect of probiotic lactobacillus strains on angiotensin i converting enzyme inhibitory activity from fermented goat milk. Adv. Mater. Res. 2012, 531, 442–445. [Google Scholar] [CrossRef]
- Shu, G.W.; Yang, H.; Chen, H.; Ji, Z.; Xing, H. Effect of carbon source and salts on angiotensin-I-converting enzyme (ACE) inhibitory activity in fermented goat milk by Lactobacillus bulgaricus LB6. J. Pure Appl. Microbiol. 2013, 7, 301–308. [Google Scholar]
- Shu, G.W.; Yang, H.; Chen, H.; Ji, Z.; Xing, H. Effect of organic nitrogen source on angiotensin-I-converting enzyme (ACE) inhibitory peptides fermented by Lactobacillus bulgaricus LB6 from goat milk. Adv. J. Food Sci. Technol. 2014, 6, 221–227. [Google Scholar] [CrossRef]
- Ewe, J.A.; Wanabdullah, W.N.; Alias, A.K.; Bhat, R.; Liong, M.T. ACE inhibitory activity and bioconversion of isoflavones by lactobacillus in soymilk supplemented with B-vitamins. Br. Food J. 2011, 113, 1127–1146. [Google Scholar] [CrossRef]
- Yeo, S.K.; Liong, M.T. Angiotensin-I-converting enzyme inhibitory activity and bioconversion of isoflavones by probiotics in soymilk supplemented with prebiotics. Int. J. Food Sci. Nutr. 2010, 61, 161–181. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Z.M.; Gang, W.U.; Huo, G.C.; Tian, B. Study on external conditions of angiotensin converting enzyme inhibitory peptide derived from fermented milk. Sci. Technol. Food Ind. 2011, 32, 106–108. [Google Scholar]
- Wang, L.F.; Jin, J.; Yuan, J.; Rong, H.E.; Xingrong, J.U. Optimization of liquid-state fermentation conditions for preparing rapeseed angiotensin-I-converting enzyme inhibitory peptides. Food Sci. 2010, 31, 226–231. [Google Scholar]
- Gonzalezgonzalez, C.R.; Tuohy, K.M.; Jauregi, P. Production of angiotensin-I-converting enzyme (ACE) inhibitory activity in milk fermented with probiotic strains: Effects of calcium, pH and peptides on the ACE-inhibitory activity. Int. Dairy J. 2011, 21, 615–622. [Google Scholar] [CrossRef]
- Li, Y.; Sadiq, F.A.; Liu, T.J.; Chen, J.C.; He, G.Q. Purification and identification of novel peptides with inhibitory effect against angiotensin-I-converting enzyme and optimization of process conditions in milk fermented with the yeast kluyveromyces marxianus. J. Funct. Foods 2015, 16, 278–288. [Google Scholar] [CrossRef]
- Pan, D.D.; Guo, Y.X. Optimization of sour milk fermentation for the production of ACE inhibitory peptides and purification of a novel peptide from whey protein hydrolysate. Int. Dairy J. 2010, 20, 472–479. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |
| Factors | Coded Levels | ||
|---|---|---|---|
| −1 | 0 | 1 | |
| Temperature (°C) | 35 | 37 | 39 |
| Whey powder (%) | 0.7 | 0.8 | 0.9 |
| Calcium lactate (%) | 0.45 | 0.50 | 0.55 |
| Runs | A | B | C | R1 (%) | R2 [lg(cfu·mL−1)] | ||||
|---|---|---|---|---|---|---|---|---|---|
| Actual Value | Predicted Value | Residual | Actual Value | Predicted Value | Residual | ||||
| 1 | −1 | 1 | 0 | 74.08 | 73.81 | 0.27 | 7.7 | 7.67 | 0.031 |
| 2 | 0 | 1 | −1 | 77.77 | 78.21 | −0.44 | 7.82 | 7.83 | −0.015 |
| 3 | −1 | 0 | 1 | 75.45 | 75.23 | 0.22 | 7.71 | 7.72 | −0.014 |
| 4 | 0 | 1 | 1 | 78.04 | 78.53 | −0.49 | 7.8 | 7.82 | −0.018 |
| 5 | 0 | 0 | 0 | 83.19 | 83.26 | −0.07 | 8.05 | 8.04 | 0.013 |
| 6 | 0 | −1 | −1 | 78.72 | 78.23 | 0.49 | 7.9 | 7.88 | 0.017 |
| 7 | 0 | −1 | 1 | 78.25 | 77.81 | 0.44 | 7.82 | 7.81 | 0.015 |
| 8 | 1 | 0 | −1 | 75.56 | 75.78 | −0.22 | 7.8 | 7.79 | 0.014 |
| 9 | 1 | 1 | 0 | 75.49 | 74.83 | 0.66 | 7.67 | 7.67 | 0.00125 |
| 10 | 0 | 0 | 0 | 82.67 | 83.26 | −0.59 | 8.03 | 8.04 | 0.00667 |
| 11 | 1 | −1 | 0 | 73.68 | 73.96 | −0.28 | 7.67 | 7.70 | −0.031 |
| 12 | −1 | 0 | −1 | 76.31 | 76.15 | 0.16 | 7.83 | 7.85 | −0.016 |
| 13 | 0 | 0 | 0 | 83.92 | 83.26 | 0.66 | 8.03 | 8.04 | 0.00667 |
| 14 | −1 | −1 | 0 | 73.32 | 73.98 | −0.66 | 7.67 | 7.67 | 0.00125 |
| 15 | 1 | 0 | 1 | 76.44 | 76.60 | −0.16 | 7.83 | 7.81 | 0.016 |
| Source | ACE Inhibition Rate | Viable Counts of LB6 | |||||
|---|---|---|---|---|---|---|---|
| DF | MS | F | Pr > F | MS | F | Pr > F | |
| Model | 9 | 17.58 | 31.10 | 0.0007 *** | 0.0260 | 31.58 | 0.0007 *** |
| A | 1 | 0.51 | 0.89 | 0.3880 | 0.0005 | 0.54 | 0.4967 |
| B | 1 | 0.25 | 0.44 | 0.5366 | 0.0006 | 0.73 | 0.4317 |
| C | 1 | 0.004 | 0.007 | 0.9358 | 0.0045 | 5.38 | 0.0680 |
| AB | 1 | 0.28 | 0.49 | 0.5161 | 0.0002 | 0.27 | 0.6265 |
| AC | 1 | 0.76 | 1.34 | 0.2995 | 0.0056 | 6.71 | 0.0488 * |
| BC | 1 | 0.14 | 0.24 | 0.6435 | 0.0009 | 1.07 | 0.3476 |
| A2 | 1 | 119.39 | 211.20 | <0.0001 *** | 0.1500 | 177.64 | <0.0001 *** |
| B2 | 1 | 43.47 | 76.90 | 0.0003 *** | 0.0930 | 110.41 | 0.0001 *** |
| C2 | 1 | 9.86 | 17.43 | 0.0087 ** | 0.0069 | 8.27 | 0.0348 * |
| Residual | 5 | 0.57 | 0.0008 | ||||
| Lack of fit | 3 | 0.68 | 1.72 | 0.3878 | 0.0013 | 9.81 | 0.0939 |
| Pure error | 2 | 0.39 | 0.0001 | ||||
| Cor Total | 14 | ||||||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shu, G.; Shi, X.; Chen, H.; Ji, Z.; Meng, J. Optimization of Goat Milk with ACE Inhibitory Peptides Fermented by Lactobacillus bulgaricus LB6 Using Response Surface Methodology. Molecules 2017, 22, 2001. https://doi.org/10.3390/molecules22112001
Shu G, Shi X, Chen H, Ji Z, Meng J. Optimization of Goat Milk with ACE Inhibitory Peptides Fermented by Lactobacillus bulgaricus LB6 Using Response Surface Methodology. Molecules. 2017; 22(11):2001. https://doi.org/10.3390/molecules22112001
Chicago/Turabian StyleShu, Guowei, Xiaoyu Shi, He Chen, Zhe Ji, and Jiangpeng Meng. 2017. "Optimization of Goat Milk with ACE Inhibitory Peptides Fermented by Lactobacillus bulgaricus LB6 Using Response Surface Methodology" Molecules 22, no. 11: 2001. https://doi.org/10.3390/molecules22112001
APA StyleShu, G., Shi, X., Chen, H., Ji, Z., & Meng, J. (2017). Optimization of Goat Milk with ACE Inhibitory Peptides Fermented by Lactobacillus bulgaricus LB6 Using Response Surface Methodology. Molecules, 22(11), 2001. https://doi.org/10.3390/molecules22112001





