Phospholipids of Animal and Marine Origin: Structure, Function, and Anti-Inflammatory Properties
Abstract
1. Introduction
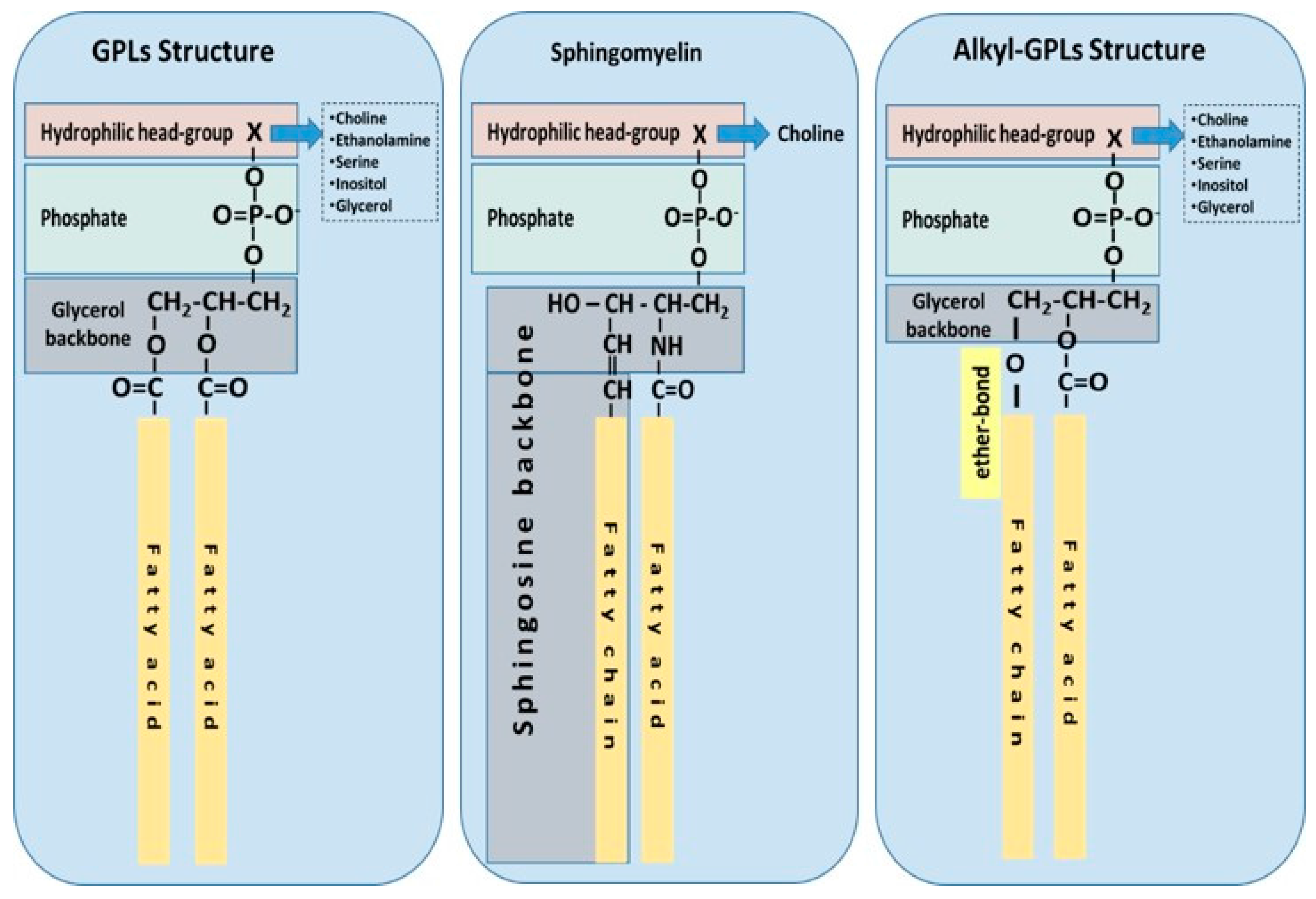
1.1. Phospholipid Classes and Biological Functions
1.2. Glycerophospholipid and Sphingophospholipid Biosynthesis
1.3. Inflammation and Lipid Inflammatory Mediators
1.4. Dietary Phospholipids: Digestion and Absorption
2. Phospholipids of Animal Origin
2.1. Meat Phospholipids
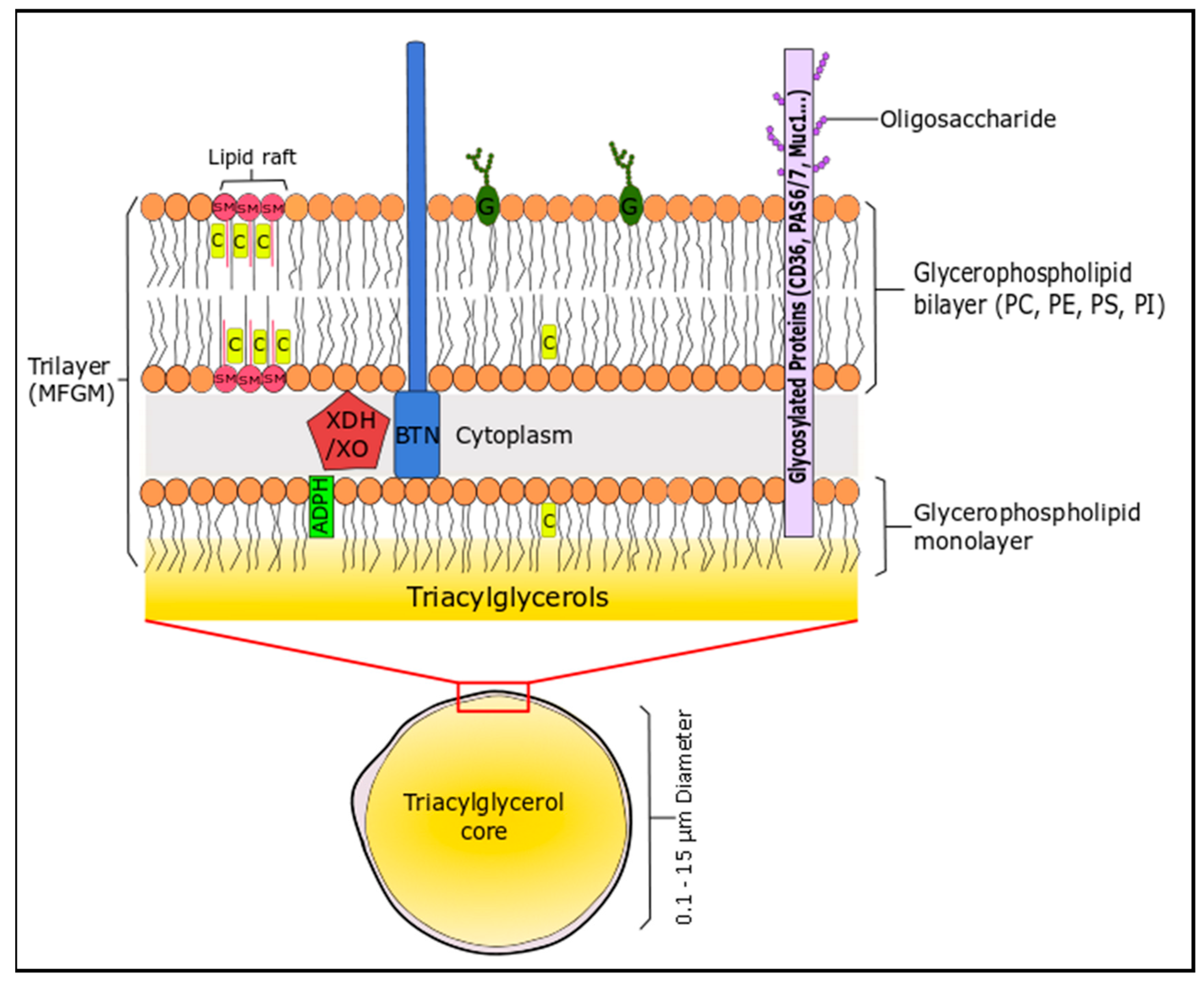
2.2. Milk and Dairy Phospholipids
2.3. Egg Phospholipids
3. Marine Origin
3.1. Sources of Marine Phospholipids
3.2. Oxidation of Marine Phospholipids—Pro-Inflammatory Mediators
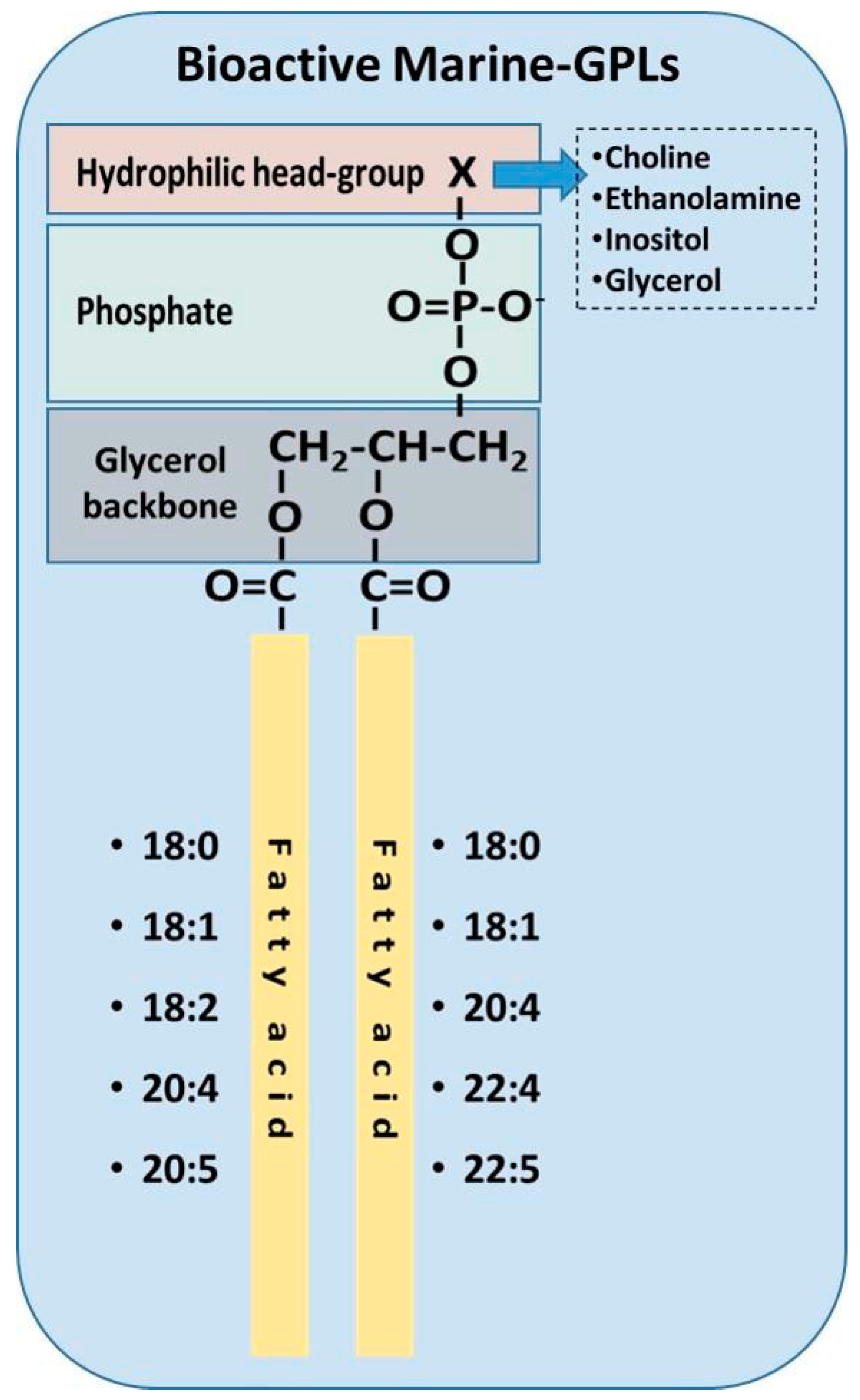
3.3. Bioavailability and Biofunctionality of Marine Phospholipids
3.4. Marine Phospholipids and Inflammation: The Missing Link
4. Conclusions and Future Perspectives
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Castro-Gómez, P.; Garcia-Serrano, A.; Visioli, F.; Fontecha, J. Relevance of dietary glycerophospholipids and sphingolipids to human health. Prostaglandins Leukot. Essent. Fatty Acids 2015, 101, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Contarini, G.; Povolo, M. Phospholipids in milk fat: Composition, biological and technological significance, and analytical strategies. Int. J. Mol. Sci. 2013, 14, 2808–2831. [Google Scholar] [CrossRef] [PubMed]
- Rombaut, R.; Dewettinck, K. Properties, analysis and purification of milk polar lipids. Int. Dairy J. 2006, 16, 1362–1373. [Google Scholar] [CrossRef]
- Caforio, A.; Driessen, A.J.M. Archaeal phospholipids: Structural properties and biosynthesis. Biochim. Biophys. Acta. 2017, 1862, 1325–1339. [Google Scholar] [CrossRef] [PubMed]
- Demopoulos, C.; Pinckard, R.; Hanahan, D.J. Platelet-activating factor. Evidence for 1-o-alkyl-2-acetyl-sn-glyceryl-3-phosphorylcholine as the active component (a new class of lipid chemical mediators). J. Biol. Chem. 1979, 254, 9355–9358. [Google Scholar] [PubMed]
- Megson, I.L.; Whitfield, P.D.; Zabetakis, I. Lipids and cardiovascular disease: Where does dietary intervention sit alongside statin therapy? Food Funct. 2016, 7, 2603–2614. [Google Scholar] [CrossRef] [PubMed]
- Tsoupras, A.B.; Iatrou, C.; Frangia, C.; Demopoulos, C.A. The implication of platelet activating factor in cancer growth and metastasis: Potent beneficial role of paf-inhibitors and antioxidants. Infect. Disord.-Drug Targets (Former. Curr. Drug Targets-Infect. Disord.) 2009, 9, 390–399. [Google Scholar] [CrossRef]
- Küllenberg, D.; Taylor, L.A.; Schneider, M.; Massing, U. Health effects of dietary phospholipids. Lipids Health Dis. 2012, 11, 1. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K. Omega-3 phospholipids. In Polar Lipids: Biology, Chemistry, and Technology; Ahmad, M.U., Xu, X., Eds.; AOCS Press: Urbana, IL, USA, 2015; pp. 463–493. [Google Scholar]
- Mollinedo, F.; Gajate, C. Fas/cd95 death receptor and lipid rafts: New targets for apoptosis-directed cancer therapy. Drug Resist. Updat. 2006, 9, 51–73. [Google Scholar] [CrossRef] [PubMed]
- Tessaro, F.H.; Ayala, T.S.; Martins, J.O. Lipid mediators are critical in resolving inflammation: A review of the emerging roles of eicosanoids in diabetes mellitus. BioMed Res. Int. 2015, 2015, 568408. [Google Scholar] [CrossRef] [PubMed]
- Gundermann, K.-J.; Gundermann, S.; Drozdzik, M.; Prasad, V.M. Essential phospholipids in fatty liver: A scientific update. Clin. Exp. Gastroenterol. 2016, 9, 105. [Google Scholar] [PubMed]
- Choy, C.H.; Han, B.-K.; Botelho, R.J. Phosphoinositide diversity, distribution, and effector function: Stepping out of the box. Bioessays 2017. [Google Scholar] [CrossRef] [PubMed]
- Mejia, E.M.; Hatch, G.M. Mitochondrial phospholipids: Role in mitochondrial function. J. Bioenerg. Biomembr. 2016, 48, 99–112. [Google Scholar] [CrossRef] [PubMed]
- Vance, J.E. Phospholipid synthesis and transport in mammalian cells. Traffic 2015, 16, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Henneberry, A.L.; Wright, M.M.; McMaster, C.R. The major sites of cellular phospholipid synthesis and molecular determinants of fatty acid and lipid head group specificity. Mol. Biol. Cell 2002, 13, 3148–3161. [Google Scholar] [CrossRef] [PubMed]
- Vance, J.E.; Vance, D.E. Biochemistry of Lipids, Lipoproteins and Membranes, 5th ed.; Elsevier: Oxford, UK, 2008. [Google Scholar]
- Vance, J.E.; Tasseva, G. Formation and function of phosphatidylserine and phosphatidylethanolamine in mammalian cells. Biochim. Biophys. Acta 2013, 1831, 543–554. [Google Scholar] [CrossRef] [PubMed]
- Gardocki, M.E.; Jani, N.; Lopes, J.M. Phosphatidylinositol biosynthesis: Biochemistry and regulation. Biochim. Biophys. Acta 2005, 1735, 89–100. [Google Scholar] [CrossRef] [PubMed]
- Maceyka, M.; Spiegel, S. Sphingolipid metabolites in inflammatory disease. Nature 2014, 510, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Zemski Berry, K.A.; Murphy, R.C. Free radical oxidation of plasmalogen glycerophosphocholine containing esterified docosahexaenoic acid: Structure determination by mass spectrometry. Antioxid. Redox Signal. 2005, 7, 157–169. [Google Scholar] [CrossRef] [PubMed]
- Braverman, N.E.; Moser, A.B. Functions of plasmalogen lipids in health and disease. Biochim. Biophys. Acta 2012, 1822, 1442–1452. [Google Scholar] [CrossRef] [PubMed]
- Lordan, R.; Zabetakis, I. Invited review: The anti-inflammatory properties of dairy lipids. J. Dairy Sci. 2017, 100, 4197–4212. [Google Scholar] [CrossRef] [PubMed]
- O’Keefe, J.H.; Gheewala, N.M.; O’Keefe, J.O. Dietary strategies for improving post-prandial glucose, lipids, inflammation, and cardiovascular health. J. Am. Coll. Cardiol. 2008, 51, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Lecomte, M.; Bourlieu, C.; Meugnier, E.; Penhoat, A.; Cheillan, D.; Pineau, G.; Loizon, E.; Trauchessec, M.; Claude, M.; Ménard, O. Milk polar lipids affect in vitro digestive lipolysis and postprandial lipid metabolism in mice. J. Nutr. 2015, 145, 1770–1777. [Google Scholar] [CrossRef] [PubMed]
- O’Keefe, J.H.; Bell, D.S.H. Postprandial hyperglycemia/hyperlipidemia (postprandial dysmetabolism) is a cardiovascular risk factor. Am. J. Cardiol. 2007, 100, 899–904. [Google Scholar] [CrossRef] [PubMed]
- Medzhitov, R. Origin and physiological roles of inflammation. Nature 2008, 454, 428. [Google Scholar] [CrossRef] [PubMed]
- Demopoulos, C.A.; Karantonis, H.C.; Antonopoulou, S. Platelet activating factor—A molecular link between atherosclerosis theories. Eur. J. Lipid Sci. Technol. 2003, 105, 705–716. [Google Scholar] [CrossRef]
- Libby, P.; Ridker, P.M.; Maseri, A. Inflammation and atherosclerosis. Circulation 2002, 105, 1135–1143. [Google Scholar] [CrossRef] [PubMed]
- Kotas, M.E.; Medzhitov, R. Homeostasis, inflammation, and disease susceptibility. Cell 2015, 160, 816–827. [Google Scholar] [CrossRef] [PubMed]
- Ross, R. Atherosclerosis—An inflammatory disease. N. Engl. J. Med. 1999, 340, 115–126. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, A.S.; Obin, M.S. Obesity and the role of adipose tissue in inflammation and metabolism. Am. J. Clin. Nutr. 2006, 83, 461S–465S. [Google Scholar] [PubMed]
- Reuter, S.; Gupta, S.C.; Chaturvedi, M.M.; Aggarwal, B.B. Oxidative stress, inflammation, and cancer: How are they linked? Free Radic. Biol. Med. 2010, 49, 1603–1616. [Google Scholar] [CrossRef] [PubMed]
- Van Eldik, L.J.; Carrillo, M.C.; Cole, P.E.; Feuerbach, D.; Greenberg, B.D.; Hendrix, J.A.; Kennedy, M.; Kozauer, N.; Margolin, R.A.; Molinuevo, J.L.; et al. The roles of inflammation and immune mechanisms in alzheimer’s disease. Alzheimer’s Dement. Transl. Res. Clin. Interv. 2016, 2, 99–109. [Google Scholar] [CrossRef] [PubMed]
- DeBoer, M.D. Obesity, systemic inflammation, and increased risk for cardiovascular disease and diabetes among adolescents: A need for screening tools to target interventions. Nutrition 2013, 29, 379–386. [Google Scholar] [CrossRef] [PubMed]
- Garcia, C.; Feve, B.; Ferré, P.; Halimi, S.; Baizri, H.; Bordier, L.; Guiu, G.; Dupuy, O.; Bauduceau, B.; Mayaudon, H. Diabetes and inflammation: Fundamental aspects and clinical implications. Diabetes Metab. 2010, 36, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Liao, K.P. Cardiovascular disease in patients with rheumatoid arthritis. Trends. Cardiovasc. Med. 2017, 27, 136–140. [Google Scholar] [CrossRef] [PubMed]
- Tsoupras, A.B.; Antonopoulou, S.; Baltas, G.; Samiotaki, M.; Panayotou, G.; Kotsifaki, H.; Mantzavinos, Z.; Demopoulos, C.A. Isolation and identification of hydroxyl–platelet-activating factor from natural sources. Life Sci. 2006, 79, 1796–1803. [Google Scholar] [CrossRef] [PubMed]
- Antonopoulou, S.; Tsoupras, A.; Baltas, G.; Kotsifaki, H.; Mantzavinos, Z.; Demopoulos, C.A. Hydroxyl-platelet-activating factor exists in blood of healthy volunteers and periodontal patients. Mediat. Inflamm. 2003, 12, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, M.A.; Satoh, H.; Favelyukis, S.; Babendure, J.L.; Imamura, T.; Sbodio, J.I.; Zalevsky, J.; Dahiyat, B.I.; Chi, N.-W.; Olefsky, J.M. Jnk and tumor necrosis factor-α mediate free fatty acid-induced insulin resistance in 3t3-l1 adipocytes. J. Biol. Chem. 2005, 280, 35361–35371. [Google Scholar] [CrossRef] [PubMed]
- Tsoupras, A.B.; Chini, M.; Mangafas, N.; Tsogas, N.; Stamatakis, G.; Tsantila, N.; Fragopoulou, E.; Antonopoulou, S.; Gargalianos, P.; Demopoulos, C.A.; et al. Platelet-activating factor and its basic metabolic enzymes in blood of naive hiv-infected patients. Angiology 2012, 63, 343–352. [Google Scholar] [CrossRef] [PubMed]
- Meirow, Y.; Baniyash, M. Immune biomarkers for chronic inflammation related complications in non-cancerous and cancerous diseases. Cancer Immunol. Immunother. 2017, 66, 1089–1101. [Google Scholar] [CrossRef] [PubMed]
- Burri, L.; Hoem, N.; Banni, S.; Berge, K. Marine omega-3 phospholipids: Metabolism and biological activities. Int. J. Mol. Sci. 2012, 13, 15401–15419. [Google Scholar] [CrossRef] [PubMed]
- Zabetakis, I. Food security and cardioprotection: The polar lipid link. J. Food Sci. 2013, 78, 1101–1104. [Google Scholar] [CrossRef] [PubMed]
- Dennis, E.A.; Norris, P.C. Eicosanoid storm in infection and inflammation. Nat. Rev. Immunol. 2015, 15, 511–523. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Marine omega-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance. Biochim. Biophys. Acta 2015, 1851, 469–484. [Google Scholar] [CrossRef] [PubMed]
- Ardies, C.M. Diet, Excercise and Chronic Disease: The Biological Basis of Prevention; CRC Press: Boca Raton, FL, USA, 2014. [Google Scholar]
- Simopoulos, A.P. The importance of the omega-6/omega-3 fatty acid ratio in cardiovascular disease and other chronic diseases. Exp. Biol. Med. 2008, 233, 674–688. [Google Scholar] [CrossRef] [PubMed]
- De Caterina, R. N-3 fatty acids in cardiovascular disease. N. Engl. J. Med. 2011, 364, 2439–2450. [Google Scholar] [CrossRef] [PubMed]
- Murphy, K.J.; Meyer, B.J.; Mori, T.A.; Burke, V.; Mansour, J.; Patch, C.S.; Tapsell, L.C.; Noakes, M.; Clifton, P.A.; Barden, A.; et al. Impact of foods enriched with n-3 long-chain polyunsaturated fatty acids on erythrocyte n-3 levels and cardiovascular risk factors. Br. J. Nutr. 2007, 97, 749–757. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A. An increase in the omega-6/omega-3 fatty acid ratio increases the risk for obesity. Nutrients 2016, 8, 128. [Google Scholar] [CrossRef] [PubMed]
- Rizos, E.C.; Ntzani, E.E.; Bika, E.; Kostapanos, M.S.; Elisaf, M.S. Association between omega-3 fatty acid supplementation and risk of major cardiovascular disease events: A systematic review and meta-analysis. JAMA 2012, 308, 1024–1033. [Google Scholar] [CrossRef] [PubMed]
- Enns, J.E.; Yeganeh, A.; Zarychanski, R.; Abou-Setta, A.M.; Friesen, C.; Zahradka, P.; Taylor, C.G. The impact of omega-3 polyunsaturated fatty acid supplementation on the incidence of cardiovascular events and complications in peripheral arterial disease: A systematic review and meta-analysis. BMC Cardiovasc. Disord. 2014, 14, 70. [Google Scholar] [CrossRef] [PubMed]
- Kwak, S.; Myung, S.; Lee, Y.; Seo, H.; Korean Meta-Analysis Study Group. Efficacy of omega-3 fatty acid supplements (eicosapentaenoic acid and docosahexaenoic acid) in the secondary prevention of cardiovascular disease: A meta-analysis of randomized, double-blind, placebo-controlled trials. Arch. Intern. Med. 2012, 172, 686–694. [Google Scholar] [PubMed]
- Walz, C.P.; Barry, A.R.; Koshman, S.L. Omega-3 polyunsaturated fatty acid supplementation in the prevention of cardiovascular disease. Can. Pharm. J. 2016, 149, 166–173. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, R.; Stevens, S.; Gorman, D.; Pan, A.; Warnakula, S.; Chowdhury, S.; Ward, H.; Johnson, L.; Crowe, F.; Hu, F.B. Association between fish consumption, long chain omega 3 fatty acids, and risk of cerebrovascular disease: Systematic review and meta-analysis. BMJ 2012, 345, e6698. [Google Scholar] [CrossRef] [PubMed]
- Hishikawa, D.; Valentine, W.J.; Iizuka-Hishikawa, Y.; Shindou, H.; Shimizu, T. Metabolism and functions of docosahexaenoic acid-containing membrane glycerophospholipids. FEBS. Lett. 2017, 591, 2730–2744. [Google Scholar] [CrossRef] [PubMed]
- Murru, E.; Banni, S.; Carta, G. Nutritional properties of dietary omega-3-enriched phospholipids. BioMed Res. Int. 2013, 2013, 965417. [Google Scholar] [CrossRef] [PubMed]
- Murray, M.; Hraiki, A.; Bebawy, M.; Pazderka, C.; Rawling, T. Anti-tumor activities of lipids and lipid analogues and their development as potential anticancer drugs. Pharmacol. Ther. 2015, 150, 109–128. [Google Scholar] [CrossRef] [PubMed]
- Wallner, S.; Schmitz, G. Plasmalogens the neglected regulatory and scavenging lipid species. Chem. Phys. Lipids 2011, 164, 573–589. [Google Scholar] [CrossRef] [PubMed]
- Bochkov, V.; Gesslbauer, B.; Mauerhofer, C.; Philippova, M.; Erne, P.; Oskolkova, O.V. Pleiotropic effects of oxidized phospholipids. Free Radic. Biol. Med. 2017, 111, 6–24. [Google Scholar] [CrossRef] [PubMed]
- Reis, A. Oxidative phospholipidomics in health and disease: Achievements, challenges and hopes. Free Radic. Biol. Med. 2017, 111, 25–37. [Google Scholar] [CrossRef] [PubMed]
- Venable, M.E.; Zimmerman, G.A.; McIntyre, T.M.; Prescott, S.M. Platelet-activating factor: A phospholipid autacoid with diverse actions. J. Lipid Res. 1993, 34, 691–702. [Google Scholar] [PubMed]
- Triggiani, M.; Schleimer, R.; Warner, J.; Chilton, F. Differential synthesis of 1-acyl-2-acetyl-sn-glycero-3-phosphocholine and platelet-activating factor by human inflammatory cells. J. Immunol. 1991, 147, 660–666. [Google Scholar] [PubMed]
- Francescangeli, E.; Freysz, L.; Goracci, G. Paf-synthesizing enzymes in neural cells during differentiation and in gerbil brain during ischemia. In Platelet-Activating Factor and Related Lipid Mediators 2; Snyder, F., Ed.; Springer: Berlin, Germany, 1996; pp. 21–27. [Google Scholar]
- Palur Ramakrishnan, A.V.K.; Varghese, T.P.; Vanapalli, S.; Nair, N.K.; Mingate, M.D. Platelet activating factor: A potential biomarker in acute coronary syndrome? Cardiovasc. Ther. 2017, 35, 64–70. [Google Scholar] [CrossRef] [PubMed]
- Castro Faria Neto, H.C.; Stafforini, D.M.; Prescott, S.M.; Zimmerman, G.A. Regulating inflammation through the anti-inflammatory enzyme platelet-activating factor-acetylhydrolase. Mem. Inst. Oswaldo Cruz 2005, 100, 83–91. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Melnikova, V.; Bar-Eli, M. Inflammation and melanoma growth and metastasis: The role of platelet-activating factor (paf) and its receptor. Cancer Metastasis Rev. 2007, 26, 359. [Google Scholar] [CrossRef] [PubMed]
- Reznichenko, A.; Korstanje, R. The role of platelet-activating factor in mesangial pathophysiology. Am. J. Pathol. 2015, 185, 888–896. [Google Scholar] [CrossRef] [PubMed]
- Feige, E.; Mendel, I.; George, J.; Yacov, N.; Harats, D. Modified phospholipids as anti-inflammatory compounds. Curr. Opin. Lipidol. 2010, 21, 525–529. [Google Scholar] [CrossRef] [PubMed]
- Papakonstantinou, V.D.; Lagopati, N.; Tsilibary, E.C.; Demopoulos, C.A.; Philippopoulos, A.I. A review on platelet activating factor inhibitors: Could a new class of potent metal-based anti-inflammatory drugs induce anticancer properties? Bioinorg. Chem. Appl. 2017, 2017, 6947034. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Singh, I.N.; Mondal, S.C.; Singh, L.; Garg, V.K. Platelet-activating factor (paf)-antagonists of natural origin. Fitoterapia 2013, 84, 180–201. [Google Scholar] [CrossRef] [PubMed]
- Fragopoulou, E.; Nomikos, T.; Tsantila, N.; Mitropoulou, A.; Zabetakis, I.; Demopoulos, C.A. Biological activity of total lipids from red and white wine/must. J. Agric. Food Chem. 2001, 49, 5186–5193. [Google Scholar] [CrossRef] [PubMed]
- Xanthopoulou, M.N.; Asimakopoulos, D.; Antonopoulou, S.; Demopoulos, C.A.; Fragopoulou, E. Effect of robola and cabernet sauvignon extracts on platelet activating factor enzymes activity on u937 cells. Food Chem. 2014, 165, 50–59. [Google Scholar] [CrossRef] [PubMed]
- Xanthopoulou, M.N.; Kalathara, K.; Melachroinou, S.; Arampatzi-Menenakou, K.; Antonopoulou, S.; Yannakoulia, M.; Fragopoulou, E. Wine consumption reduced postprandial platelet sensitivity against platelet activating factor in healthy men. Eur. J. Nutr. 2016, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Argyrou, C.; Vlachogianni, I.; Stamatakis, G.; Demopoulos, C.A.; Antonopoulou, S.; Fragopoulou, E. Postprandial effects of wine consumption on platelet activating factor metabolic enzymes. Prostaglandins Other Lipid Mediat. 2017, 130, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Fragopoulou, E.; Nomikos, T.; Antonopoulou, S.; Mitsopoulou, C.A.; Demopoulos, C.A. Separation of biologically active lipids from red wine. J. Agric. Food. Chem. 2000, 48, 1234–1238. [Google Scholar] [CrossRef] [PubMed]
- Fragopoulou, E.; Antonopoulou, S.; Tsoupras, A.; Tsantila, N.; Grypioti, A.; Gribilas, G.; Gritzapi, H.; Konsta, E.; Skandalou, E.; Papadopoulou, A. Antiatherogenic properties of Red/White Wine, Musts, Grape-Skins, and Yeast. In Proceedings of the 45th International Conference on the Bioscience of Lipids, Ioannina, Greece, 25–29 May 2004; Elsevier: Ioannina, Greece; p. 66. [Google Scholar]
- Sioriki, E.; Nasopoulou, C.; Demopoulos, C.A.; Zabetakis, I. Comparison of sensory and cardioprotective properties of olive-pomace enriched and conventional gilthead sea bream (Sparus aurata): The effect of grilling. J. Aquat. Food Prod. Technol. 2015, 24, 782–795. [Google Scholar] [CrossRef]
- Sioriki, E.; Smith, T.K.; Demopoulos, C.A.; Zabetakis, I. Structure and cardioprotective activities of polar lipids of olive pomace, olive pomace-enriched fish feed and olive pomace fed gilthead sea bream (Sparus aurata). Food Res. Int. 2016, 83, 143–151. [Google Scholar] [CrossRef]
- Nasopoulou, C.; Karantonis, H.C.; Perrea, D.N.; Theocharis, S.E.; Iliopoulos, D.G.; Demopoulos, C.A.; Zabetakis, I. In vivo anti-atherogenic properties of cultured gilthead sea bream (Sparus aurata) polar lipid extracts in hypercholesterolaemic rabbits. Food Chem. 2010, 120, 831–836. [Google Scholar] [CrossRef]
- Nasopoulou, C.; Nomikos, T.; Demopoulos, C.; Zabetakis, I. Comparison of antiatherogenic properties of lipids obtained from wild and cultured sea bass (Dicentrarchus labrax) and gilthead sea bream (Sparus aurata). Food Chem. 2007, 100, 560–567. [Google Scholar] [CrossRef]
- Nasopoulou, C.; Tsoupras, A.B.; Karantonis, H.C.; Demopoulos, C.A.; Zabetakis, I. Fish polar lipids retard atherosclerosis in rabbits by down-regulating paf biosynthesis and up-regulating paf catabolism. Lipids Health Dis. 2011, 10, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Rementzis, J.; Antonopoulou, S.; Argyropoulos, D.; Demopoulos, C.A. Biologically active lipids from s. Scombrus. In Platelet-Activating Factor and Related Lipid Mediators 2: Roles in Health and Disease; Nigam, S., Kunkel, G., Prescott, S.M., Eds.; Springer: Boston, MA, USA, 1996; pp. 65–72. [Google Scholar]
- Nasopoulou, C.; Smith, T.; Detopoulou, M.; Tsikrika, C.; Papaharisis, L.; Barkas, D.; Zabetakis, I. Structural elucidation of olive pomace fed sea bass (Dicentrarchus labrax) polar lipids with cardioprotective activities. Food Chem. 2014, 145, 1097–1105. [Google Scholar] [CrossRef] [PubMed]
- Panayiotou, A.; Samartzis, D.; Nomikos, T.; Fragopoulou, E.; Karantonis, H.C.; Demopoulos, C.A.; Zabetakis, I. Lipid fractions with aggregatory and antiaggregatory activity toward platelets in fresh and fried cod (Gadus morhua): Correlation with platelet-activating factor and atherogenesis. J. Agric. Food Chem. 2000, 48, 6372–6379. [Google Scholar] [CrossRef] [PubMed]
- Tsoupras, A.B.; Fragopoulou, E.; Nomikos, T.; Iatrou, C.; Antonopoulou, S.; Demopoulos, C.A. Characterization of the de novo biosynthetic enzyme of platelet activating factor, ddt-insensitive cholinephosphotransferase, of human mesangial cells. Mediat. Inflamm. 2007, 2007, 27683. [Google Scholar] [CrossRef] [PubMed]
- Karantonis, H.C.; Antonopoulou, S.; Demopoulos, C.A. Antithrombotic lipid minor constituents from vegetable oils. Comparison between olive oils and others. J. Agric. Food Chem. 2002, 50, 1150–1160. [Google Scholar] [CrossRef] [PubMed]
- Karantonis, H.C.; Antonopoulou, S.; Perrea, D.N.; Sokolis, D.P.; Theocharis, S.E.; Kavantzas, N.; Iliopoulos, D.G.; Demopoulos, C.A. In vivo antiatherogenic properties of olive oil and its constituent lipid classes in hyperlipidemic rabbits. Nutr. Metab. Cardiovasc. Dis. 2006, 16, 174–185. [Google Scholar] [CrossRef] [PubMed]
- Tsantila, N.; Karantonis, H.C.; Perrea, D.N.; Theocharis, S.E.; Iliopoulos, D.G.; Antonopoulou, S.; Demopoulos, C.A. Antithrombotic and antiatherosclerotic properties of olive oil and olive pomace polar extracts in rabbits. Mediat. Inflamm. 2007, 2007, 36204. [Google Scholar] [CrossRef] [PubMed]
- Tsoupras, A.; Fragopoulou, E.; Iatrou, C.; Demopoulos, C. In vitro protective effects of olive pomace polar lipids towards platelet activating factor metabolism in human renal cells. Curr. Top. Nutraceutical Res. 2011, 9, 105. [Google Scholar]
- Nasopoulou, C.; Gogaki, V.; Panagopoulou, E.; Demopoulos, C.; Zabetakis, I. Hen egg yolk lipid fractions with antiatherogenic properties. J. Anim. Sci. 2013, 84, 264–271. [Google Scholar] [CrossRef] [PubMed]
- Tsorotioti, S.E.; Nasopoulou, C.; Detopoulou, M.; Sioriki, E.; Demopoulos, C.A.; Zabetakis, I. In vitro anti-atherogenic properties of traditional greek cheese lipid fractions. Dairy Sci. Technol. 2014, 94, 269–281. [Google Scholar] [CrossRef]
- Poutzalis, S.; Anastasiadou, A.; Nasopoulou, C.; Megalemou, K.; Sioriki, E.; Zabetakis, I. Evaluation of the in vitro anti-atherogenic activities of goat milk and goat dairy products. Dairy Sci. Technol. 2016, 96, 317–327. [Google Scholar] [CrossRef]
- Megalemou, K.; Sioriki, E.; Lordan, R.; Dermiki, M.; Nasopoulou, C.; Zabetakis, I. Evaluation of sensory and in vitro anti-thrombotic properties of traditional greek yogurts derived from different types of milk. Heliyon 2017, 3, e00227. [Google Scholar] [CrossRef] [PubMed]
- Thomson, A.B.R.; Keelan, M.; Garg, M.L.; Clandinin, M.T. Intestinal aspects of lipid absorption: In review. Can. J. Physiol. Pharmacol. 1989, 67, 179–191. [Google Scholar] [CrossRef] [PubMed]
- Ramırez, M.; Amate, L.; Gil, A. Absorption and distribution of dietary fatty acids from different sources. Early Hum. Dev. 2001, 65, S95–S101. [Google Scholar] [CrossRef]
- Zierenberg, O.; Grundy, S. Intestinal absorption of polyenephosphatidylcholine in man. J. Lipid Res. 1982, 23, 1136–1142. [Google Scholar] [PubMed]
- Tall, A.R.; Blum, C.B.; Grundy, S.M. Incorporation of radioactive phospholipid into subclasses of high-density lipoproteins. Am. J. Physiol. Endocrinol. Metab. 1983, 244, E513–E516. [Google Scholar]
- Carey, M.C.; Small, D.M.; Bliss, C.M. Lipid digestion and absorption. Annu. Rev. Physiol. 1983, 45, 651–677. [Google Scholar] [CrossRef] [PubMed]
- Hussain, M.M. Intestinal lipid absorption and lipoprotein formation. Curr. Opin. Lipidol. 2014, 25, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Ohlsson, L.; Hertervig, E.; Jönsson, B.A.; Duan, R.-D.; Nyberg, L.; Svernlöv, R.; Nilsson, Å. Sphingolipids in human ileostomy content after meals containing milk sphingomyelin. Am. J. Clin. Nutr. 2010, 91, 672–678. [Google Scholar] [CrossRef] [PubMed]
- Amate, L.; Gil, A.; Ramírez, M. Feeding infant piglets formula with long-chain polyunsaturated fatty acids as triacylglycerols or phospholipids influences the distribution of these fatty acids in plasma lipoprotein fractions. J. Nutr. 2001, 131, 1250–1255. [Google Scholar] [PubMed]
- Buang, Y.; Wang, Y.-M.; Cha, J.-Y.; Nagao, K.; Yanagita, T. Dietary phosphatidylcholine alleviates fatty liver induced by orotic acid. Nutrition 2005, 21, 867–873. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, P.; Szabó, A.; Erős, G.; Gurabi, D.; Horváth, G.; Németh, I.; Ghyczy, M.; Boros, M. Anti-inflammatory effects of phosphatidylcholine in neutrophil leukocyte-dependent acute arthritis in rats. Eur. J. Pharmacol. 2009, 622, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.S.; Nam, Y.; Chung, Y.H.; Kim, H.R.; Park, E.S.; Chung, S.J.; Kim, J.H.; Sohn, U.D.; Kim, H.-C.; Oh, K.W.; et al. Beneficial effects of phosphatidylcholine on high-fat diet-induced obesity, hyperlipidemia and fatty liver in mice. Life Sci. 2014, 118, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Tőkés, T.; Tuboly, E.; Varga, G.; Major, L.; Ghyczy, M.; Kaszaki, J.; Boros, M. Protective effects of l-alpha-glycerylphosphorylcholine on ischaemia–reperfusion-induced inflammatory reactions. Eur. J. Nutr. 2015, 54, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Abbasi, A.; Dallinga-Thie, G.M.; Dullaart, R.P.F. Phospholipid transfer protein activity and incident type 2 diabetes mellitus. Clin. Chim. Acta 2015, 439, 38–41. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, B.C.; Andrews, V.G. Influence of dietary egg and soybean phospholipids and triacylglycerols on human serum lipoproteins. Lipids 1993, 28, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Scholey, A.B.; Camfield, D.A.; Hughes, M.E.; Woods, W.; K Stough, C.K.; White, D.J.; Gondalia, S.V.; Frederiksen, P.D. A randomized controlled trial investigating the neurocognitive effects of lacprodan® pl-20, a phospholipid-rich milk protein concentrate, in elderly participants with age-associated memory impairment: The phospholipid intervention for cognitive ageing reversal (plicar): Study protocol for a randomized controlled trial. Trials 2013, 14, 404. [Google Scholar] [PubMed]
- Dasgupta, S.; Bhattacharyya, D.K. Dietary effect of eicosapentaenoic acid (epa) containing soyphospholipid. J. Oleo Sci 2007, 56, 563–568. [Google Scholar] [CrossRef] [PubMed]
- Shirouchi, B.; Nagao, K.; Inoue, N.; Ohkubo, T.; Hibino, H.; Yanagita, T. Effect of dietary omega 3 phosphatidylcholine on obesity-related disorders in obese otsuka long-evans tokushima fatty rats. J. Agric. Food Chem. 2007, 55, 7170–7176. [Google Scholar] [CrossRef] [PubMed]
- Cohn, J.; Kamili, A.; Wat, E.; Chung, R.W.; Tandy, S. Dietary phospholipids and intestinal cholesterol absorption. Nutrients 2010, 2, 116–127. [Google Scholar] [CrossRef] [PubMed]
- Blesso, C. Egg phospholipids and cardiovascular health. Nutrients 2015, 7, 2731–2747. [Google Scholar] [CrossRef] [PubMed]
- Tellis, C.C.; Tselepis, A. Pathophysiological role and clinical significance of lipoprotein-associated phospholipase a2 (lp-pla2) bound to ldl and hdl. Curr. Pharm. Des. 2014, 20, 6256–6269. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, M.; Otvos, J.; Nikolic, D.; Montalto, G.; Toth, P.; Banach, M. Subfractions and subpopulations of hdl: An update. Curr. Med. Chem. 2014, 21, 2881–2891. [Google Scholar] [CrossRef] [PubMed]
- Marathe, G.K.; Pandit, C.; Lakshmikanth, C.L.; Chaithra, V.H.; Jacob, S.P.; D’Souza, C.J.M. To hydrolyze or not to hydrolyze: The dilemma of platelet-activating factor acetylhydrolase. J. Lipid Res. 2014, 55, 1847–1854. [Google Scholar] [CrossRef] [PubMed]
- Givens, D. Saturated fats, dairy foods and health: A curious paradox? Nutr. Bull. 2017, 42, 274–282. [Google Scholar] [CrossRef]
- Givens, D.; Livingstone, K.; Pickering, J.; Fekete, Á.; Dougkas, A.; Elwood, P. Milk: White elixir or white poison? An examination of the associations between dairy consumption and disease in human subjects. Anim. Front. 2014, 4, 8–15. [Google Scholar] [CrossRef]
- Fernandez, L.M. Eggs and health special issue. Nutrients 2016, 8, 784. [Google Scholar] [CrossRef] [PubMed]
- De Smet, S.; Vossen, E. Meat: The balance between nutrition and health. A review. Meat Sci. 2016, 120, 145–156. [Google Scholar] [CrossRef] [PubMed]
- McNeill, S.H. Inclusion of red meat in healthful dietary patterns. Meat Sci. 2014, 98, 452–460. [Google Scholar] [CrossRef] [PubMed]
- Andersen, C.J. Bioactive egg components and inflammation. Nutrients 2015, 7, 7889–7913. [Google Scholar] [CrossRef] [PubMed]
- Weihrauch, J.L.; Son, Y.-S. Phospholipid content of foods. J. Am. Oil Chem. Soc. 1983, 60, 1971–1978. [Google Scholar] [CrossRef]
- Lewis, E.D.; Zhao, Y.-Y.; Richard, C.; Bruce, H.L.; Jacobs, R.L.; Field, C.J.; Curtis, J.M. Measurement of the abundance of choline and the distribution of choline-containing moieties in meat. Int. J. Food Sci. Nutr. 2015, 66, 743–748. [Google Scholar] [CrossRef] [PubMed]
- Kunsman, J.E.; Field, R.A. The lipid content of mechanically deboned red meats. J. Food Sci. 1976, 41, 1439–1441. [Google Scholar] [CrossRef]
- Gray, G.M.; Macfarlane, M.G. Composition of phospholipids of rabbit, pigeon and trout muscle and various pig tissues. Biochem. J. 1961, 81, 480. [Google Scholar] [CrossRef] [PubMed]
- Kuchmak, M.; Dugan, L.R. Phospholipids of pork muscle tissues. J. Am. Oil Chem. Soc. 1963, 40, 734–736. [Google Scholar] [CrossRef]
- Acosta, S.O.; Marion, W.W.; Forsythe, R. Total lipids and phospholipids in turkey tissues. Poult. Sci. 1966, 45, 169–184. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Liu, F.; Zhu, Y.; Xu, W. Changes of phospholipids in duck muscle by different heating methods. J. Food Process. Technol. 2011, 2. [Google Scholar] [CrossRef]
- Zancada, L.; Pérez-Díez, F.; Sánchez-Juanes, F.; Alonso, J.; García-Pardo, L.; Hueso, P. Phospholipid classes and fatty acid composition of ewe’s and goat’s milk. Grasas y Aceites 2013, 64, 304–310. [Google Scholar]
- Thomas, A.J.; Patton, S. Phospholipids of fish gills. Lipids 1972, 7, 76–78. [Google Scholar] [CrossRef] [PubMed]
- Ackman, R.G. Marine Biogenic Lipids, Fats and Oils; CRC Press: Boca Raton, FL, USA, 1989; Volume 2. [Google Scholar]
- Subra-Paternault, P.; ThongDeng, H.; Grélard, A.; Cansell, M. Extraction of phospholipids from scallop by-product using supercritical co2/alcohol mixtures. LWT-Food Sci. Technol. 2015, 60, 990–998. [Google Scholar] [CrossRef]
- Ishii, K.; Okajima, H.; Okada, Y.; Watanabe, H. Studies on furan fatty acids of salmon roe phospholipids. J. Biochem. 1988, 103, 836–839. [Google Scholar] [CrossRef] [PubMed]
- Nemova, N.N.; Murzina, S.A.; Nefedova, Z.A.; Veselov, A.E. Features in the lipid status of two generations of fingerlings (0+) of atlantic salmon (Salmo salar L.) inhabiting the arenga river (kola peninsula). Int. J. Mol. Sci. 2015, 16, 17535–17545. [Google Scholar] [CrossRef] [PubMed]
- Benedito-Palos, L.; Navarro, J.; Kaushik, S.; Pérez-Sánchez, J. Tissue-specific robustness of fatty acid signatures in cultured gilthead sea bream (Sparus aurata L.) fed practical diets with a combined high replacement of fish meal and fish oil. J. Anim. Sci. 2010, 88, 1759–1770. [Google Scholar] [CrossRef] [PubMed]
- Benedito-Palos, L.; Calduch-Giner, J.A.; Ballester-Lozano, G.F.; Pérez-Sánchez, J. Effect of ration size on fillet fatty acid composition, phospholipid allostasis and mrna expression patterns of lipid regulatory genes in gilthead sea bream (Sparus aurata). Br. J. Nutr. 2012, 109, 1175–1187. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cordier, M.; Brichon, G.; Weber, J.-M.; Zwingelstein, G. Changes in the fatty acid composition of phospholipids in tissues of farmed sea bass (Dicentrarchus labrax) during an annual cycle. Roles of environmental temperature and salinity. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2002, 133, 281–288. [Google Scholar] [CrossRef]
- Gallagher, M.L.; Paramore, L.; Alves, D.; Rulifson, R.A. Comparison of phospholipid and fatty acid composition of wild and cultured striped bass eggs. J. Fish Biol. 1998, 52, 1218–1228. [Google Scholar] [CrossRef]
- Adeyeye, E. Fatty acids, sterols and phospholipids levels in the muscle of acanthurus montoviaeand lutjanus goreensisfish. La Rivista Italiana Delle Sostanze Grasse 2015, 92, 123–138. [Google Scholar]
- Wu, J.; Chan, R.; Wenk, M.R.; Hew, C.-L. Lipidomic study of intracellular singapore grouper iridovirus. Virology 2010, 399, 248–256. [Google Scholar] [CrossRef] [PubMed]
- Saito, H.; Ishikawa, S. Lipid classes and fatty acid profile of cultured and wild black rockfish, sebastes schlegeli. J. Oleo Sci. 2014, 63, 555–566. [Google Scholar] [CrossRef] [PubMed]
- Hawthorne, J.N.; Ansell, G.B. Phospholipids; Elsevier: Amsterdam, The Netherlands, 1982; Volume 4, p. 62. [Google Scholar]
- Ferioli, F.; Caboni, M.F. Composition of phospholipid fraction in raw chicken meat and pre-cooked chicken patties: Influence of feeding fat sources and processing technology. Eur. Food Res. Technol. 2010, 231, 117–126. [Google Scholar] [CrossRef]
- Lippi, G.; Mattiuzzi, C.; Cervellin, G. Meat consumption and cancer risk: A critical review of published meta-analyses. Crit. Rev. Oncol. Hematol. 2016, 97, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Bovalino, S.; Charleson, G.; Szoeke, C. The impact of red and processed meat consumption on cardiovascular disease risk in women. Nutrition 2016, 32, 349–354. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Palacios, T.; Ruiz, J.; Dewettinck, K.; Trung Le, T.; Antequera, T. Individual phospholipid classes from iberian pig meat as affected by diet. J. Agric. Food Chem. 2010, 58, 1755–1760. [Google Scholar] [CrossRef] [PubMed]
- Keller, J.D.; Kinsella, J.E. Phospholipid changes and lipid oxidation during cooking and frozen storage of raw ground beef. J. Food Sci. 1973, 38, 1200–1204. [Google Scholar] [CrossRef]
- Simopoulos, A.P. Omega-6/omega-3 essential fatty acid ratio and chronic diseases. Food Rev. Int. 2004, 20, 77–90. [Google Scholar] [CrossRef]
- Wang, Z.; Klipfell, E.; Bennett, B.J.; Koeth, R.; Levison, B.S.; DuGar, B.; Feldstein, A.E.; Britt, E.B.; Fu, X.; Chung, Y.-M.; et al. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature 2011, 472, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Gregory, J.C.; Org, E.; Buffa, J.A.; Gupta, N.; Wang, Z.; Li, L.; Fu, X.; Wu, Y.; Mehrabian, M. Gut microbial metabolite TMAO enhances platelet hyperreactivity and thrombosis risk. Cell 2016, 165, 111–124. [Google Scholar] [CrossRef] [PubMed]
- Wolk, A. Potential health hazards of eating red meat. J. Intern. Med. 2017, 281, 106–122. [Google Scholar] [CrossRef] [PubMed]
- Koeth, R.A.; Wang, Z.; Levison, B.S.; Buffa, J.A.; Org, E.; Sheehy, B.T.; Britt, E.B.; Fu, X.; Wu, Y.; Li, L.; et al. Intestinal microbiota metabolism of l-carnitine, a nutrient in red meat, promotes atherosclerosis. Nat. Med. 2013, 19, 576–585. [Google Scholar] [CrossRef] [PubMed]
- Couatre, D.; Bell, S. Is l-carnitine the link between red meat and heart disease? J. Nutr. Food Sci. 2013, 3. [Google Scholar] [CrossRef]
- Blank, M.L.; Cress, E.A.; Smith, Z.L.; Snyder, F. Meats and fish consumed in the american diet contain substantial amounts of ether-linked phospholipids. J. Nutr. 1992, 122, 1656–1661. [Google Scholar] [PubMed]
- Lecomte, M.; Bourlieu, C.; Michalski, M.-C. Nutritional Properties of Milk Lipids: Specific Function of the Milk Fat Globule in Dairy in Human Health and Disease across the Lifespan; Collier, R.J., Preedy, V.R., Eds.; Academic Press: Cambridge, MA, USA, 2017; pp. 435–452. [Google Scholar]
- Le, T.T.; Phan, T.T.Q.; Camp, J.V.; Dewettinck, K. Milk and dairy polar lipids: Occurence, purification, nutritional and technological properties. In Polar Lipids: Biology, Chemistry, and Technology; Ahmad, M.U., Xu, X., Eds.; AOCS Press: Urbana, IL, USA, 2015; pp. 91–143. [Google Scholar]
- Dewettinck, K.; Rombaut, R.; Thienpont, N.; Le, T.T.; Messens, K.; Van Camp, J. Nutritional and technological aspects of milk fat globule membrane material. Int. Dairy J. 2008, 18, 436–457. [Google Scholar] [CrossRef]
- Lopez, C. Milk fat globules enveloped by their biological membrane: Unique colloidal assemblies with a specific composition and structure. Curr. Opin. Colloid Interface Sci. 2011, 16, 391–404. [Google Scholar] [CrossRef]
- Lopez, C.; Briard-Bion, V.; Ménard, O. Polar lipids, sphingomyelin and long-chain unsaturated fatty acids from the milk fat globule membrane are increased in milks produced by cows fed fresh pasture based diet during spring. Food Res. Int. 2014, 58, 59–68. [Google Scholar] [CrossRef]
- Rodríguez-Alcalá, L.M.; Fontecha, J. Major lipid classes separation of buttermilk, and cows, goats and ewes milk by high performance liquid chromatography with an evaporative light scattering detector focused on the phospholipid fraction. J. Chromatogr. A 2010, 1217, 3063–3066. [Google Scholar] [CrossRef] [PubMed]
- Graves, E.L.F.; Beaulieu, A.D.; Drackley, J.K. Factors affecting the concentration of sphingomyelin in bovine milk. J. Dairy Sci. 2007, 90, 706–715. [Google Scholar] [CrossRef]
- Wiking, L.; Nielsen, J.H.; Båvius, A.K.; Edvardsson, A.; Svennersten-Sjaunja, K. Impact of milking frequencies on the level of free fatty acids in milk, fat globule size, and fatty acid composition. J. Dairy Sci. 2006, 89, 1004–1009. [Google Scholar] [CrossRef]
- Singh, H. The milk fat globule membrane—A biophysical system for food applications. Curr. Opin. Colloid Interface Sci. 2006, 11, 154–163. [Google Scholar] [CrossRef]
- Bitman, J.; Wood, D.L. Changes in milk fat phospholipids during lactation. J. Dairy Sci. 1990, 73, 1208–1216. [Google Scholar] [CrossRef]
- Rodríguez-Alcalá, L.; Castro-Gómez, P.; Felipe, X.; Noriega, L.; Fontecha, J. Effect of processing of cow milk by high pressures under conditions up to 900 mpa on the composition of neutral, polar lipids and fatty acids. Food Sci. Technol. 2015, 62, 265–270. [Google Scholar] [CrossRef]
- Lopez, C.; Briard-Bion, V.; Menard, O.; Rousseau, F.; Pradel, P.; Besle, J.-M. Phospholipid, sphingolipid, and fatty acid compositions of the milk fat globule membrane are modified by diet. J. Agric. Food Chem. 2008, 56, 5226–5236. [Google Scholar] [CrossRef] [PubMed]
- Guerra, E.; Verardo, V.; Caboni, M.F. Determination of bioactive compounds in cream obtained as a by-product during cheese-making: Influence of cows’ diet on lipid quality. Int. Dairy J. 2015, 42, 16–25. [Google Scholar] [CrossRef]
- Pimentel, L.; Gomes, A.; Pintado, M.; Rodríguez-Alcalá, L.M. Isolation and analysis of phospholipids in dairy foods. J. Anal. Methods Chem. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Logan, A.; Cocks, B.G.; Rochfort, S. Seasonal variation of polar lipid content in bovine milk. Food Chem. 2017, 237, 865–869. [Google Scholar] [CrossRef] [PubMed]
- Gallier, S.; Gragson, D.; Cabral, C.; Jiménez-Flores, R.; Everett, D.W. Composition and fatty acid distribution of bovine milk phospholipids from processed milk products. J. Agric. Food Chem. 2010, 58, 10503–10511. [Google Scholar] [CrossRef] [PubMed]
- Fong, B.Y.; Norris, C.S.; MacGibbon, A.K.H. Protein and lipid composition of bovine milk-fat-globule membrane. Int. Dairy J. 2007, 17, 275–288. [Google Scholar] [CrossRef]
- Laegreid, A.; Kolsto otnass, A.-B.; Fuglesang, J. Human and bovine milk: Comparison of ganglioside composition and enterotoxin- inhibitory activity. Pediatr. Res. 1986, 20, 416–421. [Google Scholar] [CrossRef] [PubMed]
- Rivas-Serna, I.M.; Polakowski, R.; Shoemaker, G.K.; Mazurak, V.C.; Clandinin, M.T. Profiling gangliosides from milk products and other biological membranes using lc/ms. J. Food Compos. Anal. 2015, 44, 45–55. [Google Scholar] [CrossRef]
- Cinque, B.; Di Marzio, L.; Centi, C.; Di Rocco, C.; Riccardi, C.; Grazia Cifone, M. Sphingolipids and the immune system. Pharmacol. Res. 2003, 47, 421–437. [Google Scholar] [CrossRef]
- Deguchi, H.; Yegneswaran, S.; Griffin, J.H. Sphingolipids as bioactive regulators of thrombin generation. J. Biol. Chem. 2004, 279, 12036–12042. [Google Scholar] [CrossRef] [PubMed]
- Pettus, B.; Chalfant, C.; Hannun, Y. Sphingolipids in inflammation: Roles and implications. Curr. Mol. Med. 2004, 4, 405–418. [Google Scholar] [CrossRef] [PubMed]
- Vesper, H.; Schmelz, E.-M.; Nikolova-Karakashian, M.N.; Dillehay, D.L.; Lynch, D.V.; Merrill, A.H. Sphingolipids in food and the emerging importance of sphingolipids to nutrition. J. Nutr. 1999, 129, 1239–1250. [Google Scholar] [PubMed]
- Lemonnier, L.A.; Dillehay, D.L.; Vespremi, M.J.; Abrams, J.; Brody, E.; Schmelz, E.M. Sphingomyelin in the suppression of colon tumors: Prevention versus intervention. Arch. Biochem. Biophys. 2003, 419, 129–138. [Google Scholar] [CrossRef] [PubMed]
- Schmelz, E.M.; Dillehay, D.L.; Webb, S.K.; Reiter, A.; Adams, J.; Merrill, A.H., Jr. Sphingomyelin consumption suppresses aberrant colonic crypt foci and increases the proportion of adenomas versus adenocarcinomas in cf1 mice treated with 1,2-dimethylhydrazine: Implications for dietary sphingolipids and colon carcinogenesis. Cancer Res. 1996, 56, 4936–4941. [Google Scholar] [PubMed]
- Mazzei, J.C.; Zhou, H.; Brayfield, B.P.; Hontecillas, R.; Bassaganya-Riera, J.; Schmelz, E.M. Suppression of intestinal inflammation and inflammation-driven colon cancer in mice by dietary sphingomyelin: Importance of peroxisome proliferator-activated receptor γ expression. J. Nutr. Biochem. 2011, 22, 1160–1171. [Google Scholar] [CrossRef] [PubMed]
- Schmelz, E.M. Sphingolipids in the chemoprevention of colon cancer. Front. Biosci. 2004, 9, 2632–2639. [Google Scholar] [CrossRef] [PubMed]
- Babahosseini, H.; Roberts, P.C.; Schmelz, E.M.; Agah, M. Bioactive sphingolipid metabolites modulate ovarian cancer cell structural mechanics. Integr. Biol. 2013, 5, 1385–1392. [Google Scholar] [CrossRef] [PubMed]
- Simon, K.W.; Tait, L.; Miller, F.; Cao, C.; Davy, K.P.; LeRoith, T.; Schmelz, E.M. Suppression of breast xenograft growth and progression in nude mice: Implications for the use of orally administered sphingolipids as chemopreventive agents against breast cancer. Food Funct. 2010, 1, 90–98. [Google Scholar] [CrossRef] [PubMed]
- Lecomte, M.; Couëdelo, L.; Meugnier, E.; Plaisancié, P.; Létisse, M.; Benoit, B.; Gabert, L.; Penhoat, A.; Durand, A.; Pineau, G.; et al. Dietary emulsifiers from milk and soybean differently impact adiposity and inflammation in association with modulation of colonic goblet cells in high-fat fed mice. Mol. Nutr. Food Res. 2016, 60, 609–620. [Google Scholar] [CrossRef] [PubMed]
- Noh, S.K.; Koo, S.I. Milk sphingomyelin is more effective than egg sphingomyelin in inhibiting intestinal absorption of cholesterol and fat in rats. J. Nutr. 2004, 134, 2611–2616. [Google Scholar] [PubMed]
- Conway, V.; Couture, P.; Gauthier, S.; Pouliot, Y.; Lamarche, B. Effect of buttermilk consumption on blood pressure in moderately hypercholesterolemic men and women. Nutrition 2014, 30, 116–119. [Google Scholar] [CrossRef] [PubMed]
- Conway, V.; Gauthier, S.F.; Pouliot, Y. Effect of cream pasteurization, microfiltration and enzymatic proteolysis on in vitro cholesterol-lowering activity of buttermilk solids. Dairy Sci. Technol. 2010, 90, 449–460. [Google Scholar] [CrossRef]
- Lordan, R.; Zabetakis, I. Ovine and caprine lipids promoting cardiovascular health in milk and its derivatives. Adv. Dairy Res. 2017, 5. [Google Scholar] [CrossRef]
- Thorning, T.K.; Raben, A.; Tholstrup, T.; Soedamah-Muthu, S.S.; Givens, I.; Astrup, A. Milk and dairy products: Good or bad for human health? An assessment of the totality of scientific evidence. Food Nutr. Res. 2016, 60, 32527. [Google Scholar] [CrossRef] [PubMed]
- Markey, O.; Vasilopoulou, D.; Givens, D.I.; Lovegrove, J.A. Dairy and cardiovascular health: Friend or foe? Nutr. Bull. 2014, 39, 161–171. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Astrup, A.; Lovegrove, J.A.; Gijsbers, L.; Givens, D.I.; Soedamah-Muthu, S.S. Milk and dairy consumption and risk of cardiovascular diseases and all-cause mortality: Dose–response meta-analysis of prospective cohort studies. Eur. J. Epidemiol. 2017, 32, 269–287. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; Cho, W. The consumption of dairy products is associated with reduced risks of obesity and metabolic syndrome in korean women but not in men. Nutrients 2017, 9, 630. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Xun, P.; Wan, Y.; He, K.; Cai, W. Long-term association between dairy consumption and risk of childhood obesity: A systematic review and meta-analysis of prospective cohort studies. Eur. J. Clin. Nutr. 2016, 70, 414–423. [Google Scholar] [CrossRef] [PubMed]
- Gijsbers, L.; Ding, E.L.; Malik, V.S.; de Goede, J.; Geleijnse, J.M.; Soedamah-Muthu, S.S. Consumption of dairy foods and diabetes incidence: A dose-response meta-analysis of observational studies. Am. J. Clin. Nutr. 2016, 103, 1111–1124. [Google Scholar] [CrossRef] [PubMed]
- Alexander, D.D.; Bylsma, L.C.; Vargas, A.J.; Cohen, S.S.; Doucette, A.; Mohamed, M.; Irvin, S.R.; Miller, P.E.; Watson, H.; Fryzek, J.P. Dairy consumption and cvd: A systematic review and meta-analysis. Br. J. Nutr. 2016, 115, 737–750. [Google Scholar] [CrossRef] [PubMed]
- De Goede, J.; Soedamah-Muthu, S.S.; Pan, A.; Gijsbers, L.; Geleijnse, J.M. Dairy consumption and risk of stroke: A systematic review and updated dose–response meta-analysis of prospective cohort studies. J. Am. Heart Assoc. 2016, 5, e002787. [Google Scholar] [CrossRef] [PubMed]
- Van Hoogevest, P.; Wendel, A. The use of natural and synthetic phospholipids as pharmaceutical excipients. Eur. J. Lipid Sci. Technol. 2014, 116, 1088–1107. [Google Scholar] [CrossRef] [PubMed]
- Andersen, C.J.; Blesso, C.N.; Lee, J.; Barona, J.; Shah, D.; Thomas, M.J.; Fernandez, M.L. Egg consumption modulates hdl lipid composition and increases the cholesterol-accepting capacity of serum in metabolic syndrome. Lipids 2013, 48, 557–567. [Google Scholar] [CrossRef] [PubMed]
- Noh, S.K.; Koo, S.I. Egg sphingomyelin lowers the lymphatic absorption of cholesterol and α-tocopherol in rats. J. Nutr. 2003, 133, 3571–3576. [Google Scholar] [PubMed]
- Duivenvoorden, I.; Voshol, P.J.; Rensen, P.C.; van Duyvenvoorde, W.; Romijn, J.A.; Emeis, J.J.; Havekes, L.M.; Nieuwenhuizen, W.F. Dietary sphingolipids lower plasma cholesterol and triacylglycerol and prevent liver steatosis in apoe* 3leiden mice. Am. J. Clin. Nutr. 2006, 84, 312–321. [Google Scholar] [PubMed]
- Chakravarthy, M.V.; Lodhi, I.J.; Yin, L.; Malapaka, R.R.V.; Xu, H.E.; Turk, J.; Semenkovich, C.F. Identification of a physiologically relevant endogenous ligand for pparα in liver. Cell 2009, 138, 476–488. [Google Scholar] [CrossRef] [PubMed]
- Ratliff, J.C.; Mutungi, G.; Puglisi, M.J.; Volek, J.S.; Fernandez, M.L. Eggs modulate the inflammatory response to carbohydrate restricted diets in overweight men. Nutr. Metab. 2008, 5, 6. [Google Scholar] [CrossRef] [PubMed]
- Zdrojewicz, Z.; Herman, M.; Starostecka, E. Hen’s egg as a source of valuable biologically active substances. Postepy Higieny I Medycyny Doswiadczalnej 2016, 70, 751–759. [Google Scholar] [CrossRef] [PubMed]
- Sakakima, Y.; Hayakawa, A.; Nagasaka, T.; Nakao, A. Prevention of hepatocarcinogenesis with phosphatidylcholine and menaquinone-4: In vitro and in vivo experiments. J. Hepatol. 2007, 47, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Hu, C. The role of platelet-activating factor (paf) in fertilization and implantation. Reprod. Contracept. 1991, 11, 7–10. [Google Scholar]
- Tou, J.C.; Jaczynski, J.; Chen, Y.-C. Krill for human consumption: Nutritional value and potential health benefits. Nutr. Rev. 2007, 65, 63–77. [Google Scholar] [CrossRef] [PubMed]
- Hjaltason, B.; Haraldsson, G.G. Fish Oils and Lipids from Marine Sources; Gunstone, F.D., Ed.; Woodhead Publishing Limited: Cambridge, UK, 2006. [Google Scholar]
- Phleger, C.F.; Nelson, M.M.; Mooney, B.D.; Nichols, P.D. Interannual and between species comparison of the lipids, fatty acids and sterols of antarctic krill from the us amlr elephant island survey area. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2002, 131, 733–747. [Google Scholar] [CrossRef]
- Winther, B.; Hoem, N.; Berge, K.; Reubsaet, L. Elucidation of phosphatidylcholine composition in krill oil extracted from euphausia superba. Lipids 2011, 46, 25–36. [Google Scholar] [CrossRef] [PubMed]
- Henna Lu, F.S.; Nielsen, N.S.; Timm-Heinrich, M.; Jacobsen, C. Oxidative stability of marine phospholipids in the liposomal form and their applications. Lipids 2011, 46, 3–23. [Google Scholar] [CrossRef] [PubMed]
- Frankel, E.N. Lipid Oxidation, 1st ed.; Oily Press: High Wycombe, UK, 1998. [Google Scholar]
- Kamal-Eldin, A.; Yanishlieva, N.V. N-3 fatty acids for human nutrition: Stability considerations. Eur. J. Lipid Sci. Technol. 2002, 104, 825–836. [Google Scholar] [CrossRef]
- Tanaka, T.; Tokumura, A.; Tsukatani, H. Platelet-activating factor (paf)-like phospholipids formed during peroxidation of phosphatidylcholines from different foodstuffs. Biosci. Biotechnol. Biochem. 1995, 59, 1389–1393. [Google Scholar] [CrossRef] [PubMed]
- Maligan, J.M.; Estiasih, T.; Kusnadi, J. Structured phospholipids from commercial soybean lecithin containing omega-3 fatty acids reduces atherosclerosis risk in male sprague dawley rats which fed with an atherogenic diet. World Acad. Sci. Eng. Technol. 2012, 69, 552–558. [Google Scholar]
- Song, J.-H.; Inoue, Y.; Miyazawa, T. Oxidative stability of docosahexaenoic acid-containing oils in the form of phospholipids, triacylglycerols, and ethyl esters. Biosci. Biotechnol. Biochem. 1997, 61, 2085–2088. [Google Scholar] [CrossRef] [PubMed]
- Lu, F.S.H.; Nielsen, N.S.; Baron, C.P.; Jacobsen, C. Marine phospholipids: The current understanding of their oxidation mechanisms and potential uses for food fortification. Crit. Rev. Food Sci. Nutr. 2017, 57, 2057–2070. [Google Scholar] [CrossRef] [PubMed]
- Yanishlieva, N.V.; Marinova, E.M. Stabilisation of edible oils with natural antioxidants. Eur. J. Lipid Sci. Technol. 2001, 103, 752–767. [Google Scholar] [CrossRef]
- Lyberg, A.-M.; Fasoli, E.; Adlercreutz, P. Monitoring the oxidation of docosahexaenoic acid in lipids. Lipids 2005, 40, 969. [Google Scholar] [CrossRef] [PubMed]
- Janssen, C.I.F.; Zerbi, V.; Mutsaers, M.P.C.; de Jong, B.S.W.; Wiesmann, M.; Arnoldussen, I.A.C.; Geenen, B.; Heerschap, A.; Muskiet, F.A.J.; Jouni, Z.E.; et al. Impact of dietary n-3 polyunsaturated fatty acids on cognition, motor skills and hippocampal neurogenesis in developing c57bl/6j mice. J. Nutr. Biochem. 2015, 26, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Schuchardt, J.P.; Hahn, A. Bioavailability of long-chain omega-3 fatty acids. Prostaglandins Other Lipid Mediat. 2013, 89, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Runau, F.; Arshad, A.; Isherwood, J.; Norris, L.; Howells, L.; Metcalfe, M.; Dennison, A. Potential for proteomic approaches in determining efficacy biomarkers following administration of fish oils rich in omega-3 fatty acids. Nutr. Clin. Pract. 2015, 30, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Hjorth, E.; Vedin, I.; Eriksdotter, M.; Freund-Levi, Y.; Wahlund, L.-O.; Cederholm, T.; Palmblad, J.; Schultzberg, M. Effects of n-3 fa supplementation on the release of proresolving lipid mediators by blood mononuclear cells: The omegad study. J. Lipid Res. 2015, 56, 674–681. [Google Scholar] [CrossRef] [PubMed]
- Hung, M.-C.; Shibasaki, K.; Yoshida, R.; Sato, M.; Imaizumi, K. Learning behaviour and cerebral protein kinase c, antioxidant status, lipid composition in senescence-accelerated mouse: Influence of a phosphatidylcholine–vitamin b12 diet. Br. J. Nutr. 2007, 86, 163–171. [Google Scholar] [CrossRef]
- Torres, G.J.; Durán, A.S. Phospholipids: Properties and health effects. Nutr. Hosp. 2014, 31, 76–83. [Google Scholar]
- Jung, Y.Y.; Nam, Y.; Park, Y.S.; Lee, H.S.; Hong, S.A.; Kim, B.K.; Park, E.S.; Chung, Y.H.; Jeong, J.H. Protective effect of phosphatidylcholine on lipopolysaccharide-induced acute inflammation in multiple organ injury. Korean J. Physiol. Pharmacol. 2013, 17, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Vicenova, M.; Nechvatalova, K.; Chlebova, K.; Kucerova, Z.; Leva, L.; Stepanova, H.; Faldyna, M. Evaluation of in vitro and in vivo anti-inflammatory activity of biologically active phospholipids with anti-neoplastic potential in porcine model. BMC Complement. Altern. Med. 2014, 14, 339. [Google Scholar] [CrossRef] [PubMed]
- Moncada, S.; Higgs, E.A. Arachidonate metabolism in blood cells and the vessel wall. Baillieres Clin. Haematol. 1986, 15, 273–292. [Google Scholar]
- Mori, T.A.; Beilin, L.J.; Burke, V.; Morris, J.; Ritchie, J. Interactions between dietary fat, fish, and fish oils and their effects on platelet function in men at risk of cardiovascular disease. Arterioscler. Thromb. Vasc. Biol. 1997, 17, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Mori, T.A.; Vandongen, R.; Mahanian, F.; Douglas, A. Plasma lipid levels and platelet and neutrophil function in patients with vascular disease following fish oil and olive oil supplementation. Metabolism 1992, 41, 1059–1067. [Google Scholar] [CrossRef]
- Gioxari, A.; Kaliora, A.C.; Marantidou, F.; Panagiotakos, D.P. Intake of ω-3 polyunsaturated fatty acids in patients with rheumatoid arthritis: A systematic review and meta-analysis. Nutrition 2017. [Google Scholar] [CrossRef] [PubMed]
- Lewkowicz, N.; Lewkowicz, P.; Kurnatowska, A.; Tchórzewski, H. Biological action and clinical application of shark liver oil. Polski Merkuriusz Lekarski 2006, 20, 598–601. [Google Scholar] [PubMed]
- Schaefer, M.B.; Ott, J.; Mohr, A.; Bi, M.H.; Grosz, A.; Weissmann, N.; Ishii, S.; Grimminger, F.; Seeger, W.; Mayer, K. Immunomodulation by n-3- versus n-6-rich lipid emulsions in murine acute lung injury—Role of platelet-activating factor receptor. Crit. Care Med. 2007, 35, 544–554. [Google Scholar] [CrossRef] [PubMed]
- Misso, N.L.A.; Thompson, P.J. Fish oil supplementation inhibits platelet aggregation and ATP release induced by platelet-activating factor and other agonists. Platelets 1995, 6, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Woodman, R.J.; Mori, T.A.; Burke, V.; Puddey, I.B.; Barden, A.; Watts, G.F.; Beilin, L.J. Effects of purified eicosapentaenoic acid and docosahexaenoic acid on platelet, fibrinolytic and vascular function in hypertensive type 2 diabetic patients. Atherosclerosis 2003, 166, 85–93. [Google Scholar] [CrossRef]
- Sperling, R.I.; Robin, J.L.; Kylander, K.A.; Lee, T.H.; Lewis, R.A.; Austen, K.F. The effects of n-3 polyunsaturated fatty acids on the generation of platelet-activating factor-acether by human monocytes. J. Immunol. 1987, 139, 4186–4191. [Google Scholar] [PubMed]
- Nasopoulou, C.; Karantonis, H.C.; Andriotis, M.; Demopoulos, C.A.; Zabetakis, I. Antibacterial and anti-paf activity of lipid extracts from sea bass (Dicentrarchus labrax) and gilthead sea bream (Sparus aurata). Food Chem. 2008, 111, 433–438. [Google Scholar] [CrossRef] [PubMed]
- Nasopoulou, C.; Stamatakis, G.; Demopoulos, C.A.; Zabetakis, I. Effects of olive pomace and olive pomace oil on growth performance, fatty acid composition and cardio protective properties of gilthead sea bream (Sparus aurata) and sea bass (Dicentrarchus labrax). Food Chem. 2011, 129, 1108–1113. [Google Scholar] [CrossRef] [PubMed]
- Galanos, D.S.; Kapoulas, V.M. Isolation of polar lipids from triglyceride mixtures. J. Lipid Res. 1962, 3, 134–136. [Google Scholar]
- Nasopoulou, C.; Gogaki, V.; Stamatakis, G.; Papaharisis, L.; Demopoulos, C.; Zabetakis, I. Evaluation of the in vitro anti-atherogenic properties of lipid fractions of olive pomace, olive pomace enriched fish feed and gilthead sea bream (Sparus aurata) fed with olive pomace enriched fish feed. Mar. Drugs 2013, 11, 3676–3688. [Google Scholar] [CrossRef] [PubMed]
| Studied Food and Components | Type of Study | Results |
|---|---|---|
| PLs of red and white wine, musts, grape-skins, and yeast | In vitro studies in washed rabbit platelets (WRPs) and in U937 macrophages In vivo postprandial dietary interventions studies in humans | Inhibition of platelet aggregation and modulation of PAF-metabolism towards reduced PAF-levels [73,74,75,76,77,78] |
| PLs of fish(Sea bass, sea bream, salmon, etc.) | In vitro studies in WRPs, human platelet rich plasma (hPRP) and in human mesangial cells (HMCs). In vivo studies in hyperlipidaemic rabbits | Inhibition of platelet aggregation, modulation of PAF-metabolism towards reduced PAF-levels and reduction of the thickness of atherosclerotic lesions in hypercholesterolaemic rabbits [79,80,81,82,83,84,85,86,87] Unpublished data for Salmon-PLs |
| PLs of olive oil and olive pomace | In vitro studies in WRPs and in HMCs. In vivo study in hyperlipidaemic rabbits | Inhibition of platelet aggregation and modulation of PAF-metabolism towards reduced PAF-levels and reduction of the thickness of atherosclerotic lesions in hypercholesterolaemic rabbits and regression of the already formed atherosclerotic lesions [87,88,89,90,91] |
| PLs of seed oils (soybean, corn, sunflower, and sesame oil) | In vitro studies in WRPs | Inhibition of platelet aggregation [88] |
| PLs of Hen egg | In vitro studies in WRPs | Inhibition of platelet aggregation [92] |
| PLs of dairy products (milk, yoghurt, cheese, etc.) | In vitro studies in WRPs and in hPRP | Inhibition of platelet aggregation [93,94,95] unpublished data for bovine, ovine and caprine milk, yogurt and cheese |
| PLs * | Total PLs 1 | PC 2 | PE 2 | PI 2 | PS 2 | SM 2 |
|---|---|---|---|---|---|---|
| Egg | ||||||
| Egg yolk [8,113,114,123,124] | 28–33 | 65–75 | 10–20 | 0.5–2.0 | - | 2–5 |
| Meat | ||||||
| Chicken Liver [113,124] | 43–47 | 42–48 | 30–34 | - | 5–7 | 10–12 |
| Chicken Breast [113,124] | 67–70 | 48–52 | 23–25 | - | 12–14 | 7–9 |
| Beef [113,124,125,126] | 14–18 | 58–65 | 20–30 | 5–7 | 2–4 | 5–7 |
| Pork [113,124,127,128,129] | 55–63 | 20–34 | - | 1–8 | 1.2–6 | |
| Sheep-Lamb [129] | 42 | 38–55 | 25–31 | - | - | 4–7 |
| Rabbit [127,129] | 23 | 51–65 | 20–24 | 4 | 4–8 | - |
| Pigeon [127,129] | 28–66 | 33–49 | 26–46 | 2–8 | 3–5 | 3–5 |
| Duck (muscle) [130] | 30–45 | 25–30 | 5–10 | trace | trace | 1–2 |
| Turkey [129] | 33–80 | 38–60 | 30–42 | - | - | 2–7 |
| Dairy Products | ||||||
| Cow’s Milk [3,8,113,124] | 0.3–1.1 | 20–40 | 20–42 | 0.6–12 | 2–11 | 18–35 |
| Ewes’ milk [131] | 0.2–1.0 | 26–28 | 26–40 | 4–7 | 4–11 | 22–30 |
| Goat milk [131] | 0.2–1.0 | 27–32 | 20–42 | 4–10 | 3–14 | 16–30 |
| Marine Products | ||||||
| General Marine Composition [8,113,124,132,133,134] | 2–95 | 45–90 | 5–35 | 1–6 | 1–11 | 1–15 |
| Squid [113,124] | 64–67 | 70–75 | 8–12 | 6–8 | 7–11 | |
| Cod [113,124,129] | 24–30 | 50–77 | 12–25 | 3–4 | 4–6 | 5–11 |
| Salmon roe [135] | 30 | 80 | 13 | 4 | trace | 3 |
| Salmon [129,136] | 45–50 | 50–62 | 10–40 | 5–7 | 1–7 | 0.2–1 |
| Gilthead Sea Bream (muscle) [137,138] | 1–5 | 45–60 | 20–30 | 5–8 | 3–4 | 2–5 |
| Sea Bass (muscle) [139] | 62 | 20 | 7 | 4 | 3.4 | |
| Sea Bass (egg) [140] | 10–22 | 11–15 | 12–14 | 47–66 | - | 5–18 |
| Trout (muscle) [127,129] | 12–19 | 66 | 21–25 | 2 | 4 | 2 |
| Surgeonfish (muscle) [141] | 9 | 56 | 29 | 7 | 4 | - |
| Grouper [142] | 29–48 | 4–13 | 10–18 | 2–4 | 11–14 | |
| Black Rockfish [143] | 3–20 | 30–60 | 20–40 | trace | trace | trace |
| Molluscs [144] | 35–50 | 21–37 | 4–6 | 5–12 | 5–17 | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lordan, R.; Tsoupras, A.; Zabetakis, I. Phospholipids of Animal and Marine Origin: Structure, Function, and Anti-Inflammatory Properties. Molecules 2017, 22, 1964. https://doi.org/10.3390/molecules22111964
Lordan R, Tsoupras A, Zabetakis I. Phospholipids of Animal and Marine Origin: Structure, Function, and Anti-Inflammatory Properties. Molecules. 2017; 22(11):1964. https://doi.org/10.3390/molecules22111964
Chicago/Turabian StyleLordan, Ronan, Alexandros Tsoupras, and Ioannis Zabetakis. 2017. "Phospholipids of Animal and Marine Origin: Structure, Function, and Anti-Inflammatory Properties" Molecules 22, no. 11: 1964. https://doi.org/10.3390/molecules22111964
APA StyleLordan, R., Tsoupras, A., & Zabetakis, I. (2017). Phospholipids of Animal and Marine Origin: Structure, Function, and Anti-Inflammatory Properties. Molecules, 22(11), 1964. https://doi.org/10.3390/molecules22111964







