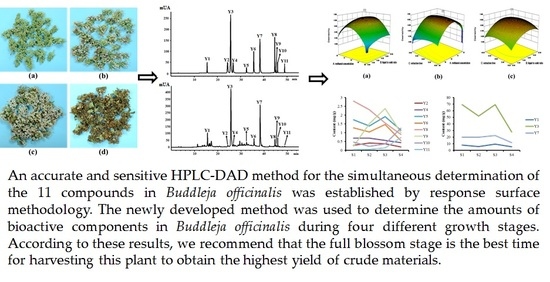
Optimization of the Extraction Conditions for Buddleja officinalis Maxim. Using Response Surface Methodology and Exploration of the Optimum Harvest Time
Abstract
:1. Introduction
2. Results and Discussion
2.1. Preliminary Experiments
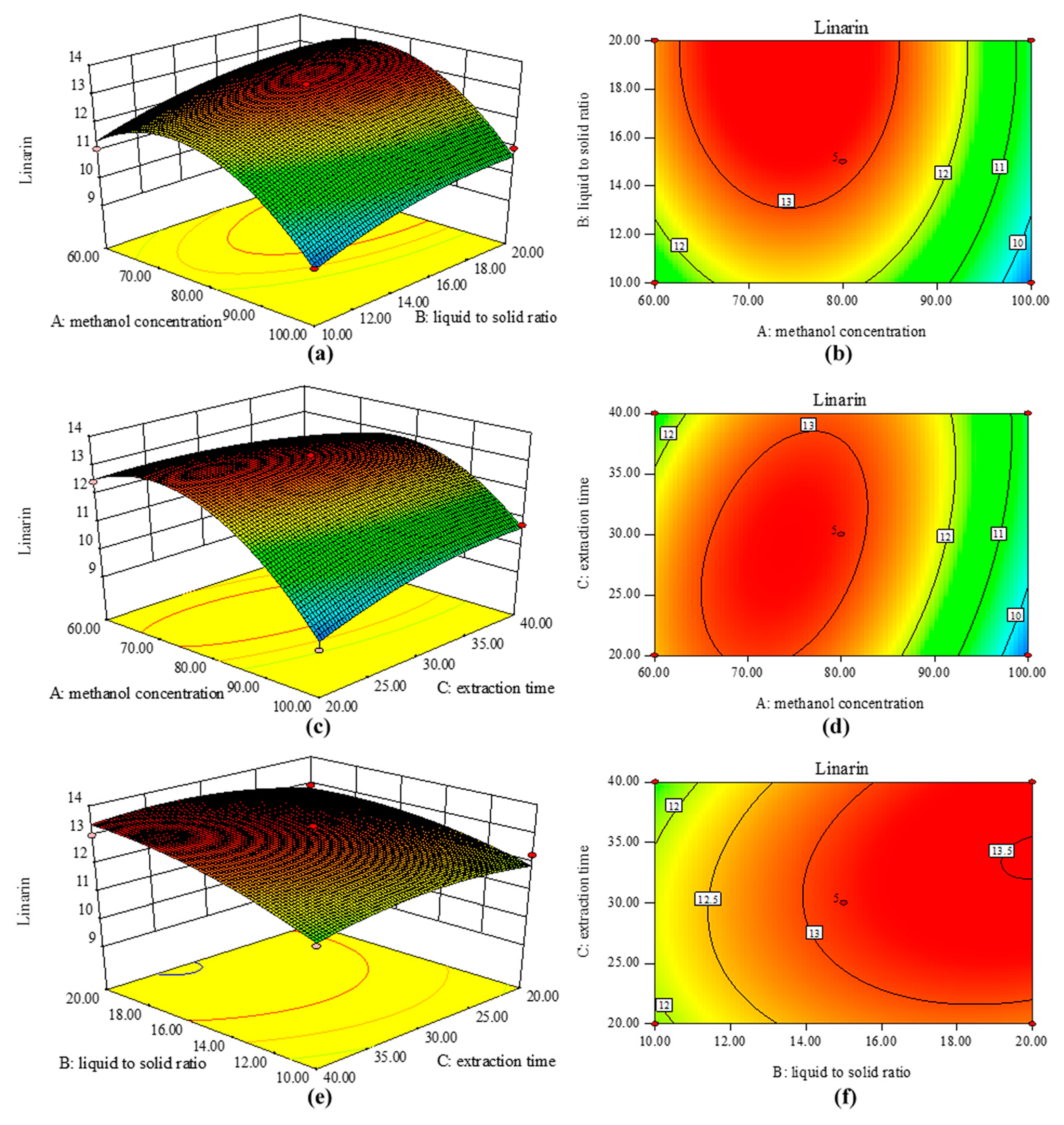
2.2. RSM Optimization
2.2.1. Fitting the Model
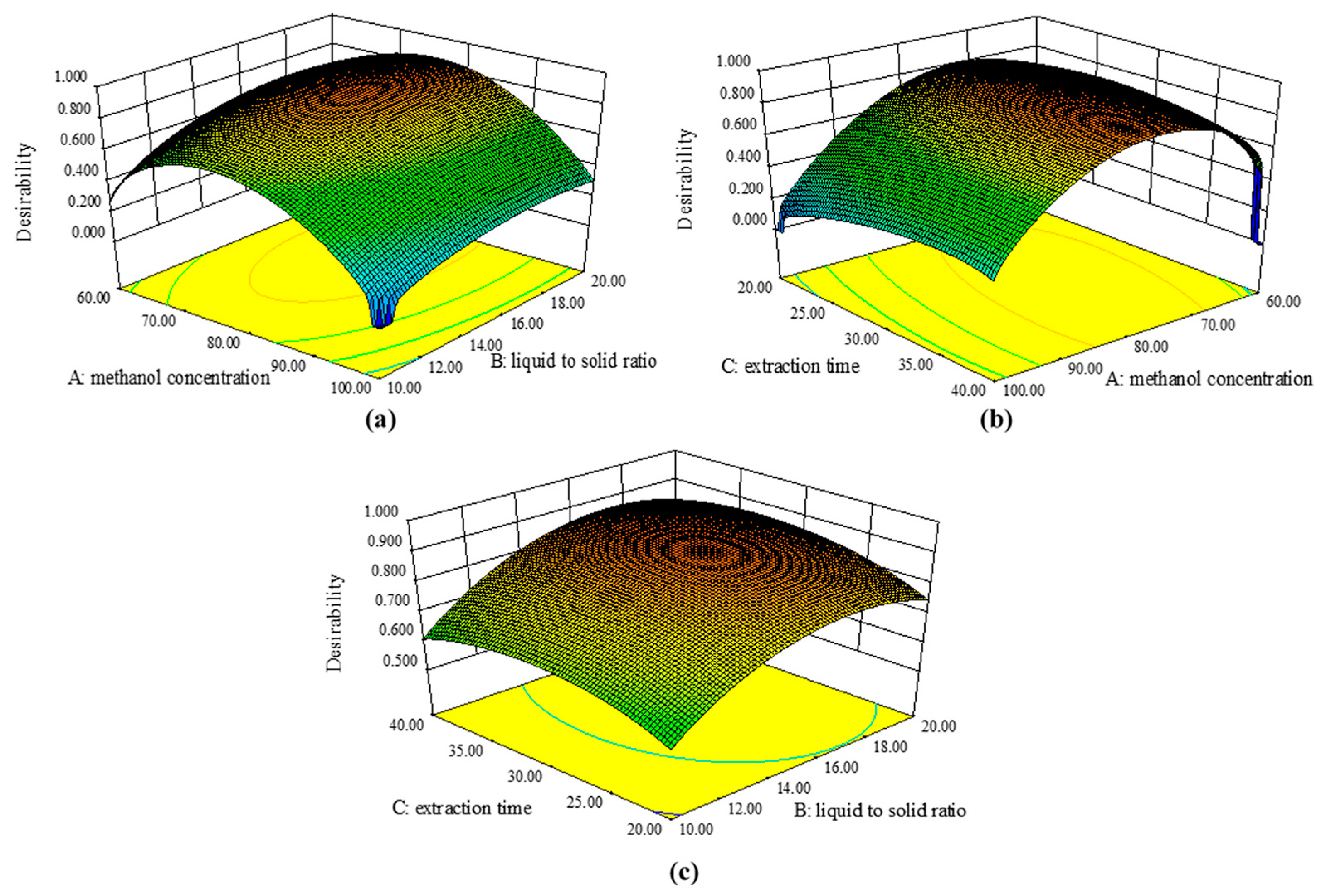
2.2.2. Optimization of Multiple Responses
2.2.3. Authentication of Predicted Model
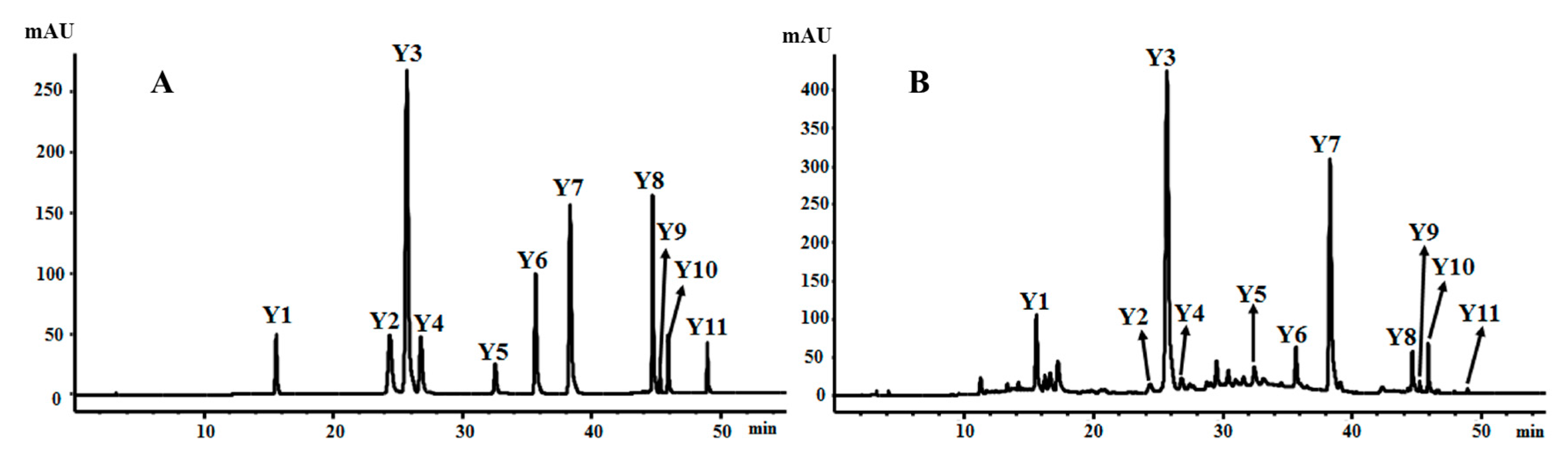
2.3. Validation of the HPLC-DAD Method
2.3.1. Linearity, LOD and LOQ
2.3.2. Precision
2.3.3. Repeatability
2.3.4. Stability
2.3.5. Accuracy
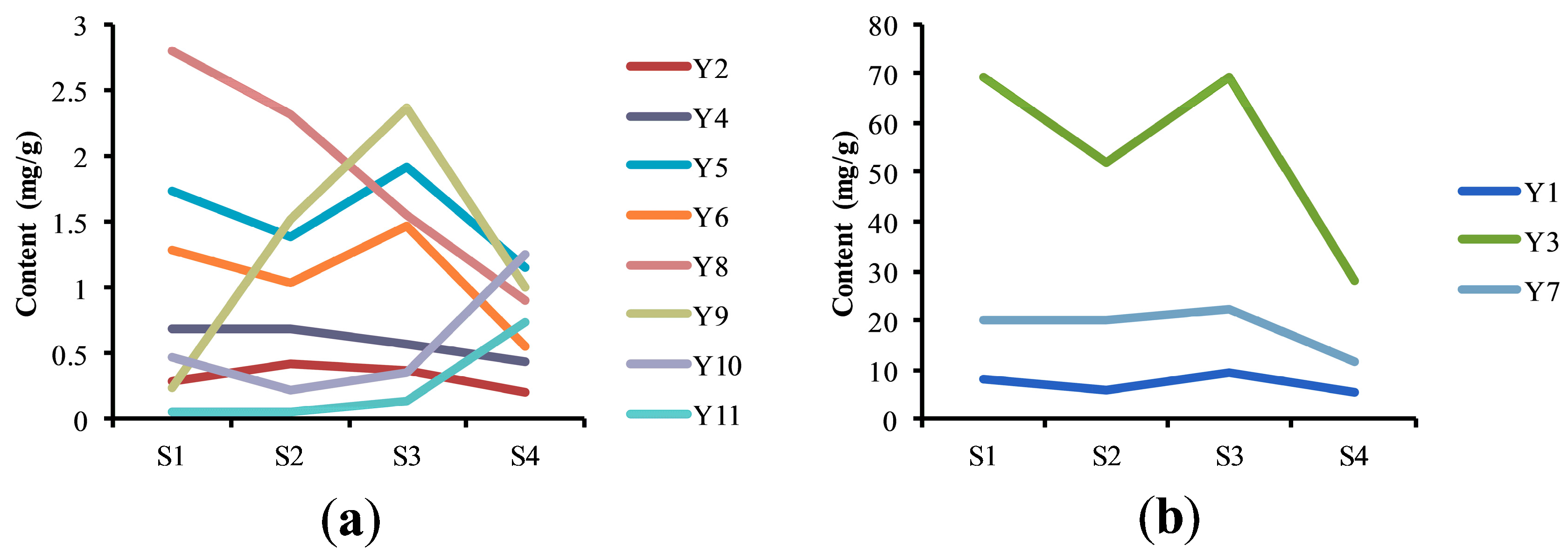
2.4. Analysis of Samples in Different Flowering Stages
3. Materials and Methods
3.1. Plant Samples
3.2. Chemicals and Reagents
3.3. Ultrasound-Assisted Extraction (UAE)
3.4. HPLC Analysis
3.5. Validation of the HPLC-DAD Method
3.5.1. Linearity, LOD and LOQ
3.5.2. Precision
3.5.3. Repeatability
3.5.4. Stability
3.5.5. Accuracy
3.6. Experimental Design
3.6.1. Preliminary Experiments
3.6.2. Response Surface Methodology
3.7. Validation of the Model
3.8. Statistical Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chinese Pharmacopoeia Committee. Pharmacopoeia of the People’s Republic of China; Chinese Medical Science and Technology Press: Beijing, China, 2015; Volume 1, p. 329. [Google Scholar]
- Piao, M.S.; Kim, M.R.; Lee, D.G.; Park, Y.; Hahm, K.S.; Moon, Y.H.; Woo, E.R. Antioxidative constituents from Buddleia officinalis. Arch. Pharm. Res. 2003, 26, 453. [Google Scholar] [PubMed]
- Lee, C.; Hwang, K.W.; Park, S.Y. A new stereoisomeric acetogenic glycoside from the flower buds of Buddleja officinalis. Bull. Korean Chem. Soc. 2014, 35, 2159. [Google Scholar] [CrossRef]
- Guo, H.Z.; Koike, K.; Li, W.; Satou, T.; Guo, D.; Nikaido, T. Saponins from the flower buds of Buddleja officinalis. J. Nat. Prod. 2004, 67, 10–13. [Google Scholar] [CrossRef] [PubMed]
- Tai, B.H.; Cuong, N.M.; Nhiem, N.X.; Tung, N.H.; Quang, T.H.; Kim, Y.H. Buddlejasaponins from the flowers of Buddleja officinalis. Chem. Nat. Compd. 2011, 47, 467. [Google Scholar] [CrossRef]
- Xu, L.; Yao, X.H.; Zeng, J.G.; Peng, Q.H. Determination of 3 flavonoids in Buddleja officinalis Maxim. with HPLC. J. TCM Univ. Hunan 2008, 28, 21. [Google Scholar]
- Zhu, L.; Lei, P.; Liu, H.T.; Tan, R.Y.; Huang, Q. Simultaneous determination of acteoside and linarin in flos Buddlejae by RP-HPLC. Chin. J. Exp. Tradit. Med. Formulae 2014, 20, 76. [Google Scholar]
- Li, Y.; Li, S.; Lin, S.J.; Zhang, J.J.; Zhao, C.N.; Li, H.B. Microwave-assisted extraction of natural antioxidants from the exotic Gordonia axillaris fruit: Optimization and identification of phenolic compounds. Molecules 2017, 22, 1481. [Google Scholar] [CrossRef] [PubMed]
- Saurabh, C.K.; Gupta, S.; Variyar, P.S.; Sharma, A. Effect of addition of nanoclay, beeswax, tween-80 and glycerol on physicochemical properties of guar gum films. Ind. Crops Prod. 2016, 89, 109–118. [Google Scholar] [CrossRef]
- Bezerra, M.A.; Santelli, R.E.; Oliveira, E.P.; Villar, L.S.; Escaleira, L.A. Response surface methodology (RSM) as a tool for optimization in analytical chemistry. Talanta 2008, 76, 965–977. [Google Scholar] [CrossRef] [PubMed]
- Guo, L. Response surface optimization of ultrasonic-assisted extraction of yellow pigment from Buddleja officinalis Maxim. In Proceedings of the 2011 International Conference on New Technology of Agricultural, Zibo, China, 27–29 May 2011; pp. 1148–1151. [Google Scholar]
- Guo, L.; Zhu, W.C.; Liu, Y.T.; Wu, J.Y.; Zheng, A.Q.; Liu, Y.L. Response surface optimized extraction of flavonoids from Mimenghua and its antioxidant activities in vitro. Food Sci. Biotechnol. 2013, 22, 1285. [Google Scholar] [CrossRef]
- Tan, X.J.; Li, Q.; Chen, X.H.; Wang, Z.W.; Shi, Z.Y.; Bi, K.S.; Jia, Y. Simultaneous determination of 13 bioactive compounds in Herba Artemisiae Scopariae (Yin Chen) from different harvest seasons by HPLC–DAD. J. Pharm. Biomed. Anal. 2008, 47, 847–853. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.J.; Mo, X.L.; Tang, X.Z.; Li, J.H.; Hu, M.B.; Yan, D.; Peng, W.; Wu, C.J. Extraction optimization, characterization, and bioactivities of polysaccharides from Pinelliae Rhizoma Praeparatum Cum Alumine employing ultrasound-assisted extraction. Molecules 2017, 22, 965. [Google Scholar] [CrossRef] [PubMed]
- Heleno, S.A.; Diz, P.; Prieto, M.A.; Barros, L.; Rodrigues, A.; Barreiro, M.F.; Ferreira, I.C.F.R. Optimization of ultrasound-assisted extraction to obtain mycosterolsfrom Agaricus bisporus L. by response surface methodology andcomparison with conventional Soxhlet extraction. Food Chem. 2016, 197, 1054–1063. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Yu, D.; Sun, H.F.; Zhang, Y.; Zhang, W.W.; Meng, F.J.; Du, X.W. Optimizing the extraction of anti-tumor alkaloids from the stem of Berberis amurensis by response surface methodology. Ind. Crops Prod. 2015, 69, 68–75. [Google Scholar] [CrossRef]
- Zhou, T.; Xu, D.P.; Lin, S.J.; Li, Y.; Zheng, J.; Zhou, Y.; Zhang, J.J.; Li, H.B. Ultrasound-assisted extraction and identification of natural antioxidants from the fruit of Melastoma sanguineum Sims. Molecules 2017, 22, 306. [Google Scholar] [CrossRef] [PubMed]
- Samavati, V. Polysaccharide extraction from Abelmoschus esculentus: Optimization by response surface methodology. Carbohydr. Polym. 2013, 95, 588–597. [Google Scholar] [CrossRef] [PubMed]
- Izadiyan, P.; Hemmateenejad, B. Multi-response optimization of factors affecting ultrasonic assisted extraction from Iranian basil using central composite design. Food Chem. 2016, 190, 864–870. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Cao, Y.L.; Jiang, J.G.; Lin, Q.S.; Chen, J.; Zhu, L. Response surface optimization of ultrasound-assisted flavonoids extraction from the flower of Citrus aurantium L. var. amara Engl. J. Sep. Sci. 2010, 33, 1349–1355. [Google Scholar] [PubMed]
- Teng, H.; Choi, Y.H. Optimization of ultrasonic-assisted extraction of bioactive alkaloid compounds from Rhizoma coptidis (Coptis chinensis Franch.) using response surface methodology. Food Chem. 2014, 142, 299–305. [Google Scholar] [CrossRef] [PubMed]
- Fu, S.; Zhang, J.; Li, T.; Wang, S.; Ding, W.J.; Zhao, M.M.; Du, Y.F.; Wang, Q.; Jia, J. Multi-responses extraction optimization based on response surface methodology combined with polarity switching HPLC–MS/MS for the simultaneous quantitation of 11 compounds in Cortex Fraxini: Application to four species of Cortex Fraxini and its 3 confusable species. J. Pharm. Biomed. 2014, 91, 210–221. [Google Scholar]
- Kong, D.X.; Li, Y.Q.; Bai, M.; He, H.J.; Liang, G.X.; Wu, H. Correlation between the dynamic accumulation of the main effective components and their associated regulatory enzyme activities at different growth stages in Lonicera japonica Thunb. Ind. Crops Prod. 2017, 96, 16–22. [Google Scholar] [CrossRef]
- Sheng, G.Q.; Zhang, J.R.; Pu, X.P.; Ma, J.; Li, C.L. Protective effect of verbascoside on 1-methyl-4-phenylpyridinium ion-induced neurotoxicity in PC12 cells. Eur. J. Pharmacol. 2002, 451, 119. [Google Scholar] [CrossRef]
- Wang, J.; Ma, C.H.; Wang, S.M. Effects of acteoside on lipopolysaccharide-induced inflammation in acute lung injury via regulation of NF-κB pathway in vivo and in vitro. Toxicol. Appl. Pharmacol. 2015, 285, 128–135. [Google Scholar]
- Yoou, M.S.; Kim, H.M.; Jeong, H.J. Acteoside attenuates TSLP-induced mast cell proliferation via down-regulating MDM2. Int. Immunopharmacol. 2015, 26, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.Y.; Quan, Q.M.; Qin, F.; Li, Y.X. Flavonoids content of Buddleja officinalis Maxim in different habitats. J. Nourthest For. Univ. 2014, 42, 50. [Google Scholar]
Sample Availability: Samples of the compounds Y2, Y3, Y4, Y6, Y7, Y10, Y11 are available from the authors. |


| Run | Independent Variables a | Dependent Variables b | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| X1 | X2 | X3 | Y1 | Y2 | Y3 | Y4 | Y5 | Y6 | Y7 | Y8 | Y9 | Y10 | Y11 | |
| 1 | 80 (0) | 15 (0) | 30 (0) | 5.02 | 0.99 | 27.25 | 0.60 | 0.98 | 2.30 | 13.30 | 1.45 | 0.98 | 0.60 | 0.13 |
| 2 | 80 (0) | 10 (−1) | 40 (1) | 4.55 | 0.98 | 25.63 | 0.54 | 0.95 | 2.11 | 11.55 | 1.34 | 0.94 | 0.55 | 0.12 |
| 3 | 80 (0) | 15 (0) | 30 (0) | 5.22 | 1.04 | 27.44 | 0.60 | 1.11 | 2.31 | 13.23 | 1.40 | 1.01 | 0.59 | 0.13 |
| 4 | 60 (−1) | 15 (0) | 20 (−1) | 5.18 | 0.99 | 24.53 | 0.57 | 1.21 | 2.29 | 12.43 | 1.30 | 0.97 | 0.52 | 0.12 |
| 5 | 80 (0) | 15 (0) | 30 (0) | 4.77 | 0.97 | 26.71 | 0.53 | 1.07 | 2.22 | 12.80 | 1.43 | 1.00 | 0.56 | 0.13 |
| 6 | 80 (0) | 15 (0) | 30 (0) | 5.25 | 1.07 | 27.69 | 0.65 | 1.15 | 2.33 | 13.28 | 1.51 | 0.96 | 0.61 | 0.13 |
| 7 | 80 (0) | 20 (1) | 40 (1) | 4.95 | 1.04 | 26.53 | 0.61 | 1.11 | 2.26 | 12.99 | 1.34 | 1.01 | 0.59 | 0.13 |
| 8 | 60 (−1) | 20 (1) | 30 (0) | 4.45 | 1.09 | 23.31 | 0.74 | 1.35 | 2.26 | 12.67 | 1.25 | 1.01 | 0.53 | 0.13 |
| 9 | 60 (−1) | 10 (−1) | 30 (0) | 4.20 | 1.07 | 21.00 | 0.74 | 1.27 | 2.06 | 11.08 | 1.22 | 0.91 | 0.43 | 0.11 |
| 10 | 100 (1) | 10 (−1) | 30 (0) | 3.29 | 0.57 | 24.39 | 0.16 | 0.47 | 1.53 | 9.36 | 1.29 | 0.72 | 0.49 | 0.11 |
| 11 | 60 (−1) | 15 (0) | 40 (1) | 4.62 | 1.09 | 22.72 | 0.75 | 1.39 | 2.23 | 11.92 | 1.21 | 0.99 | 0.49 | 0.13 |
| 12 | 80 (0) | 15 (0) | 30 (0) | 5.06 | 1.03 | 27.07 | 0.57 | 1.15 | 2.29 | 13.19 | 1.49 | 1.01 | 0.59 | 0.14 |
| 13 | 100 (1) | 20 (1) | 30 (0) | 4.03 | 0.64 | 25.89 | 0.18 | 0.61 | 1.74 | 10.94 | 1.36 | 0.74 | 0.50 | 0.12 |
| 14 | 100 (1) | 15 (0) | 20 (−1) | 3.43 | 0.51 | 23.88 | 0.16 | 0.42 | 1.41 | 8.92 | 1.22 | 0.64 | 0.45 | 0.12 |
| 15 | 100 (1) | 15 (0) | 40 (1) | 4.25 | 0.63 | 26.47 | 0.18 | 0.67 | 1.78 | 10.74 | 1.44 | 0.81 | 0.53 | 0.11 |
| 16 | 80 (0) | 10 (−1) | 20 (−1) | 4.93 | 0.89 | 26.23 | 0.41 | 0.94 | 2.14 | 12.26 | 1.28 | 0.95 | 0.56 | 0.12 |
| 17 | 80 (0) | 20 (1) | 20 (−1) | 4.80 | 0.94 | 26.57 | 0.51 | 1.05 | 2.22 | 12.92 | 1.29 | 1.00 | 0.58 | 0.13 |
| Compound | Equations a |
|---|---|
| 1 | Y1 = 5.06 − 0.43X1 + 0.16X2 + 3.750 × 10−3X3 + 0.12X1X2 + 0.35X1X3 + 0.13X2X3 − 0.75X12 − 0.32X22 + 0.061X32 |
| 2 | Y2 = 1.02 − 0.24X1 + 0.025X2 + 0.051X3 + 0.013X1X2 + 5.000 × 10−3X1X3 + 2.500 × 10−3X2X3 − 0.17X12 − 0.010X22 − 0.047X32 |
| 3 | Y3 = 27.21 + 1.1X1 + 0.63X2 + 0.016X3 − 0.21X1X2 + 1.10X1X3 + 0.14X2X3 − 2.71X12 − 0.87X22 − 0.12X32 |
| 4 | Y4 = 0.59 − 0.26X1 + 0.024X2 + 0.054X3 + 5.000 × 10−3X1X2 − 0.040X1X3 − 7.500 × 10−3X2X3 − 0.12X12 − 0.016X22 − 0.056X32 |
| 5 | Y5 = 1.09 − 0.38X1 + 0.061X2 + 0.063X3 + 0.015X1X2 + 0.018X1X3 + 0.013X2X3 − 0.13X12 − 0.039X22 − 0.041X32 |
| 6 | Y6 = 2.29 − 0.30X1 + 0.080X2 + 0.040X3 + 2.500 × 10−3X1X2 + 0.11X1X3 + 0.017X2X3 − 0.32X12 − 0.069X22 − 0.039X32 |
| 7 | Y7 = 13.16 − 1.02X1 + 0.66X2 + 0.084X3 − 2.500 × 10−3X1X2 + 0.58X1X3 + 0.19X2X3 − 1.79X12 − 0.36X22 − 0.37X32 |
| 8 | Y8 = 1.46 + 0.041X1 + 0.014X2 + 0.030X3 + 0.010X1X2 + 0.078X1X3 − 2.500 × 10−3X2X3 − 0.098X12 − 0.078X22 − 0.066X32 |
| 9 | Y9 = 0.99 − 0.12X1 + 0.030X2 + 0.024X3 − 0.020X1X2 + 0.038X1X3 + 5.000 × 10−3X2X3 − 0.13X12 − 0.012X22 − 4.750 × 10−3X32 |
| 10 | Y10 = 0.59 + 0.000X1 + 0.021X2 + 6.250 × 10−3X3 − 0.023X1X2 + 0.028X1X3 + 5.000 × 10 −3X2X3 − 0.088X12 − 0.015X22 − 5.000 × 10−3X32 |
| 11 | Y11 = 0.13 − 3.750 × 10−3X1 + 6.250 × 10−3X2 + 0.000X3 − 2.500 × 10−3X1X2 − 5.000 × 10−3X1X3 + 0.000X2X3 − 9.750 × 10−3X12 − 4.750 × 10−3X22 − 2.250 × 10−3X32 |
| Source | Y1 | Y2 | Y3 | Y4 | Y5 | Y6 | Y7 | Y8 | Y9 | Y10 | Y11 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Regression coefficients | X1 | 32.53 ** | 475.37 ** | 27.07 ** | 282.41 ** | 221.76 ** | 154.40 ** | 59.16 ** | 10.48 * | 122.79 ** | 0.000 | 7.50 * |
| X2 | 4.34 | 5.32 | 8.39 * | 2.27 | 5.72 * | 11.17 * | 24.80 ** | 1.16 | 7.52 * | 10.22 * | 20.83 ** | |
| X3 | 2.460 × 10−3 | 22.37 ** | 6.449 × 10−3 | 11.62 * | 5.96 * | 2.79 | 0.40 | 5.54 | 4.71 | 0.88 | 0.000 | |
| X1X2 | 1.31 | 0.67 | 0.43 | 0.050 | 0.17 | 5.452 × 10−3 | 1.786 × 10−3 | 0.31 | 1.67 | 5.73 * | 1.67 | |
| X1X3 | 10.41 * | 0.11 | 12.74 ** | 3.22 | 0.23 | 10.08 * | 9.69 * | 18.49 ** | 5.87 * | 8.56 * | 6.67 * | |
| X2X3 | 1.54 | 0.027 | 0.21 | 0.11 | 0.12 | 0.27 | 1.09 | 0.019 | 0.10 | 0.28 | 0.000 | |
| X12 | 52.41 ** | 125.77 ** | 81.53 ** | 29.85 ** | 13.26 ** | 96.24 ** | 96.09 ** | 31.12 ** | 79.82 ** | 91.17 ** | 26.68 ** | |
| X22 | 9.25 * | 0.45 | 8.43 * | 0.56 | 1.19 | 4.34 | 3.90 | 19.72 ** | 0.66 | 2.68 | 6.33 * | |
| X32 | 0.34 | 10.11 * | 0.16 | 6.70 * | 1.35 | 1.38 | 4.12 | 13.90 ** | 0.099 | 0.30 | 1.42 | |
| Model | 12.70 ** | 71.78 ** | 15.88 ** | 37.69 ** | 27.88 ** | 31.68 ** | 22.74 ** | 12.01 ** | 24.99 ** | 13.64 ** | 8.20 ** | |
| Lack of fit | 1.56 | 0.036 | 5.09 | 1.05 | 1.10 | 4.78 | 6.38 | 0.20 | 3.42 | 1.02 | 0.42 | |
| - | X1 (%) | X2 (mL/g) | X3 (min) | Contents (mg/g) a | Desirability | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Y1 | Y2 | Y3 | Y4 | Y5 | Y6 | Y7 | Y8 | Y9 | Y10 | Y11 | |||||
| Predicted | 76 | 34.1:1 | 32.79 | 5.12 | 1.08 | 26.90 | 0.66 | 1.20 | 2.37 | 13.49 | 1.43 | 1.03 | 0.59 | 0.14 | 0.90 |
| Experimental | 76 | 34:1 | 33.00 | 5.18 | 1.07 | 28.20 | 0.65 | 1.15 | 2.43 | 13.57 | 1.44 | 1.01 | 0.60 | 0.13 | |
| Relative Error (%) | 1.17 | 0.64 | 4.84 | 2.22 | 4.55 | 2.48 | 0.60 | 0.94 | 2.17 | 1.39 | 3.97 | ||||
| Analytes | Regression Equation | R2 | Linear Range (μg/mL) | LOD (ng/mL) | LOQ (ng/mL) |
|---|---|---|---|---|---|
| Echinacoside | y = 12,180.9575x − 3.3677 | 1.0000 | 2.41–154 | 35.5 | 128.9 |
| Luteolin-7-O-rutinoside | y = 13,590.9832x − 2.3474 | 0.9999 | 0.95–61 | 27.0 | 88.7 |
| Acteoside | y = 15,782.4634x − 20.6464 | 1.0000 | 13.30–834 | 56.2 | 174.3 |
| Luteolin-7-O-glucoside | y = 26,416.3027x − 4.8503 | 0.9999 | 0.40–25.5 | 38.8 | 107.3 |
| Apigenin-7-O-glucuronide | y = 20,583.5424x − 2.8324 | 0.9998 | 0.91–58 | 40.6 | 115.4 |
| Neobudofficide | y = 15,239.2623x − 1.2074 | 1.0000 | 2.52–161.5 | 24.7 | 86.5 |
| Linarin | y = 17,508.4610x − 2.2655 | 1.0000 | 6.09–390 | 63.2 | 218.1 |
| N1,N5,N10-(E)-tri-p-coumaroylspermidine | y = 18,198.3921x − 18.5268 | 0.9990 | 3.91–125 | 32.6 | 61.3 |
| Crocin III | y = 73,715.0130x − 19.4425 | 0.9999 | 3.97–127 | 45.8 | 136.2 |
| Apigenin | y = 40,454x − 0.6379 | 0.9999 | 0.98–31.25 | 35.7 | 131.8 |
| acacetin | y = 24,216.6307x − 3.9914 | 0.9996 | 0.45–14.5 | 28.4 | 99.7 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xie, G.; Li, R.; Han, Y.; Zhu, Y.; Wu, G.; Qin, M. Optimization of the Extraction Conditions for Buddleja officinalis Maxim. Using Response Surface Methodology and Exploration of the Optimum Harvest Time. Molecules 2017, 22, 1877. https://doi.org/10.3390/molecules22111877
Xie G, Li R, Han Y, Zhu Y, Wu G, Qin M. Optimization of the Extraction Conditions for Buddleja officinalis Maxim. Using Response Surface Methodology and Exploration of the Optimum Harvest Time. Molecules. 2017; 22(11):1877. https://doi.org/10.3390/molecules22111877
Chicago/Turabian StyleXie, Guoyong, Ran Li, Yu Han, Yan Zhu, Gang Wu, and Minjian Qin. 2017. "Optimization of the Extraction Conditions for Buddleja officinalis Maxim. Using Response Surface Methodology and Exploration of the Optimum Harvest Time" Molecules 22, no. 11: 1877. https://doi.org/10.3390/molecules22111877
APA StyleXie, G., Li, R., Han, Y., Zhu, Y., Wu, G., & Qin, M. (2017). Optimization of the Extraction Conditions for Buddleja officinalis Maxim. Using Response Surface Methodology and Exploration of the Optimum Harvest Time. Molecules, 22(11), 1877. https://doi.org/10.3390/molecules22111877