Influence of Harvest Season and Cultivar on the Variation of Phenolic Compounds Composition and Antioxidant Properties in Vaccinium ashei Leaves
Abstract
:1. Introduction
2. Results and Discussion
2.1. Influence of the Cultivar and Harvest Season on Phenolic Compounds Contents
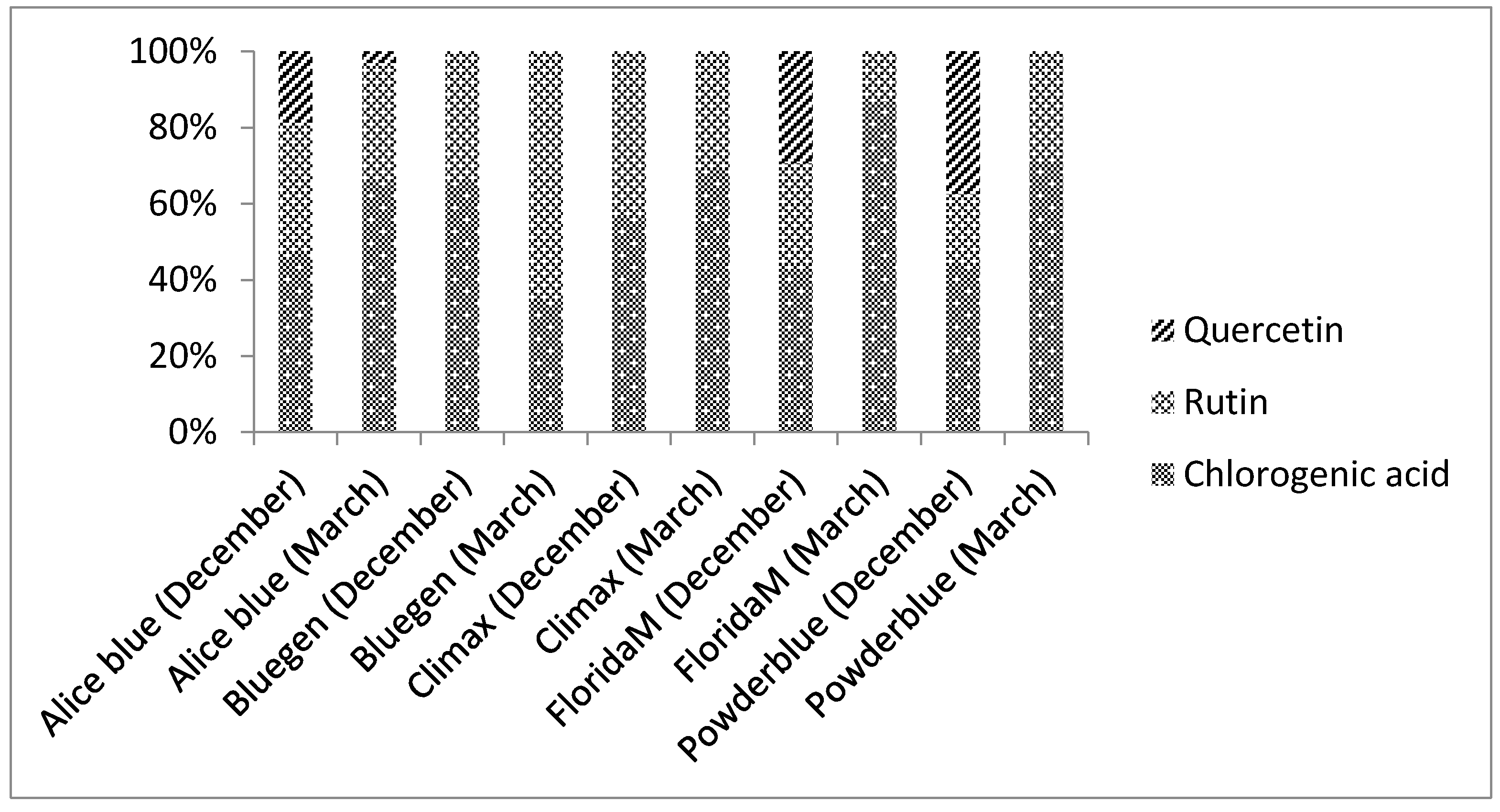
2.2. Phenolic Compounds Identification by HPLC
2.3. Influence of Cultivar and Harvest Season on Extracts Antioxidant Activity
3. Materials and Methods
3.1. Rabbiteye Blueberry Leaves
3.2. Reagents and Standards
3.3. Extracts Preparation
3.4. Total Flavonoids Content (TFC)
3.5. Total Polyphenolic Content (TPC)
3.6. Polyphenolics Qualitative Identification by HPLC-UV-DAD
3.7. Antioxidant Activity Analysis
3.7.1. DPPH Assay
3.7.2. ORAC Assay
3.8. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Nile, S.H.; Park, S.W. Edible berries: Bioactive components and their effect on human health. Nutrition 2014, 30, 134–144. [Google Scholar] [CrossRef] [PubMed]
- Zafra-stone, S.; Yasmin, T.; Bagchi, M.; Chatterjee, A.; Vinson, J.A.; Bagchi, D. Berry anthocyanins as novel antioxidants in human health and disease prevention. Mol. Nutr. Food Res. 2007, 51, 675–683. [Google Scholar] [CrossRef] [PubMed]
- Prencipe, F.P.; Bruni, R.; Guerrini, A.; Rossi, D.; Benvenuti, S.; Pellati, F. Metabolite profiling of polyphenols in Vaccinium berries and determination of their chemopreventive properties. J. Pharm. Biomed. Anal. 2014, 89, 257–267. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wu, J.; Wang, H.X.; Li, S.S.; Zheng, X.C.; Du, H.; Xu, Y.J.; Wang, L.S. Composition of phenolic compounds and antioxidant activity in the leaves of blueberry cultivars. J. Funct. Foods 2015, 16, 295–304. [Google Scholar] [CrossRef]
- Deng, Y.; Yang, G.; Yue, J.; Qian, B.; Liu, Z.; Wang, D.; Zhong, Y.; Zhao, Y. Influences of ripening stages and extracting solvents on the polyphenolic compounds, antimicrobial and antioxidant activities of blueberry leaf extracts. Food Control 2014, 38, 184–191. [Google Scholar] [CrossRef]
- Li, C.; Feng, J.; Huang, W.Y.; An, X.T. Composition of polyphenols and antioxidant activity of rabbiteye blueberry (Vaccinium ashei) in Nanjing. J. Agric. Food Chem. 2013, 61, 523–531. [Google Scholar] [CrossRef] [PubMed]
- Venskutonis, P.R.; Barnackas, S.; Kazernavičiūtė, R.; Maždžierienė, R.; Pukalskas, A.; Šipailienė, A.; Labokas, J.; Ložienė, C.; Abrutienė, G. Variations in antioxidant capacity and phenolics in leaf extracts isolated by different polarity solvents from seven blueberry (Vaccinium L.) genotypes at three phenological stages. Acta Physiol. Plant. 2016, 38, 33. [Google Scholar] [CrossRef]
- Zhu, L.; Liu, X.; Tan, J.; Wang, B. Influence of harvest season on antioxidant activity and constituents of Rabbiteye Blueberry (Vaccinium ashei) Leaves. J. Agric. Food Chem. 2013, 61, 11477–11483. [Google Scholar] [CrossRef] [PubMed]
- Ehlenfeldt, M.K.; Prior, R.L. Oxygen radical absorbance capacity (ORAC) and phenolic and anthocyanin concentrations in fruit and leaf tissues of highbush blueberry. J. Agric. Food Chem. 2001, 49, 2222–2227. [Google Scholar] [CrossRef] [PubMed]
- Piljac-Zegarac, J.; Belscak, A; Piljac, A. Antioxidant capacity and polyphenolic content of blueberry (Vaccinium corymbosum L.) leaf infusions. J. Med. Food 2009, 12, 608–614. [Google Scholar] [CrossRef] [PubMed]
- Vyas, P.; Kalidindi, S.; Chibrikova, L.; Igamberdiev, A.U.; Weber, J.T. Chemical analysis and effect of blueberry and lingonberry fruits and leaves against glutamate-mediated excitotoxicity. J. Agric. Food Chem. 2013, 61, 7769–7776. [Google Scholar] [CrossRef] [PubMed]
- Ferlemi, A.V.; Lamari, F.N. Berry Leaves: An alternative source of bioactive natural products of nutritional and medicinal value. Antioxidants 2016, 5, 17. [Google Scholar] [CrossRef] [PubMed]
- Agati, G.; Azzarello, E.; Pollastri, S.; Tattini, M. Plant science flavonoids as antioxidants in plants: Location and functional significance. Plant Sci. 2012, 196, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Hokkanen, J.; Mattila, S.; Jaakola, L.; Pirttilä, A.M.; Tolonen, A. Identification of phenolic compounds from lingonberry (Vaccinium vitis-idaea L.), Bilberry (Vaccinium myrtillus L.) andHybrid Bilberry (Vaccinium x intermedium Ruthe L.) Leaves. J. Agric. Food Chem. 2009, 57, 9437–9447. [Google Scholar] [CrossRef] [PubMed]
- Routray, W.; Orsat, V. Variation of phenolic profile and antioxidant activity of North American highbush blueberry leaves with variation of time of harvest and cultivar. Ind. Crop. Prod. 2014, 62, 147–155. [Google Scholar] [CrossRef]
- Tiwari, U.; Cummins, E. Factors influencing levels of phytochemicals in selected fruit and vegetables during pre- and post-harvest food processing operations. Food Res. Int. 2013, 50, 497–506. [Google Scholar] [CrossRef]
- Costa, G.; Grangeia, H.; Figueirinha, A.; Figueiredo, I.V.; Batista, M.T. Influence of harvest date and material quality on polyphenolic content and antioxidant activity of Cymbopogon citratus infusion. Ind. Crop. Prod. 2016, 83, 738–745. [Google Scholar] [CrossRef]
- Calixto, J.B. Efficacy, safety, quality control, marketing and regulatory guidelines for herbal medicines (phytotherapeutic agents). Braz. J. Med. Biol. Res. 2000, 33, 179–189. [Google Scholar] [CrossRef] [PubMed]
- Mcintyre, K.L.; Harris, C.S.; Saleem, A.; Beaulieu, L.P.; Ta, C.A.; Haddad, P.S.; Arnason, J.T. Seasonal Phytochemical Variation of Anti-Glycation Principles in Lowbush Blueberry (Vaccinium angustifolium). Planta Med. 2009, 75, 286–292. [Google Scholar] [CrossRef] [PubMed]
- Pervin, M.; Hasnat, M.A.; Lim, B.O. Antibacterial and antioxidant activities of Vaccinium corymbosum L. leaf extract. Asian. Pac. J. Trop. Dis. 2013, 3, 444–453. [Google Scholar] [CrossRef]
- Antunes, L.E.C.; Gonçalves, E.D.; Ristow, N.C.; Carpenedo, S.; Trevisan, R. Phenology, production and quality of blueberry cultivars. Pesq. Agropec. Bras. 2008, 43, 1011–1015. [Google Scholar] [CrossRef]
- Percival, D.; Mackenzie, J.L. Use of plant growth regulators to increase polyphenolic compounds in the wild blueberry. Can. J. Plant Sci. 2007, 87, 333–336. [Google Scholar] [CrossRef]
- Skupień, K.; Oszmiański, J.; Kostrzewa-Nowak, D.; Tarasiuk, J. In vitro antileukaemic activity of extracts from berry plant leaves against sensitive and multidrug resistant HL60 cells. Cancer Lett. 2006, 236, 282–291. [Google Scholar] [CrossRef] [PubMed]
- Ferlemi, A.V.; Mermigki, P.G.; Makri, O.E.; Anagnostopoulos, D.; Koulakiotis, N.S.; Margarity, M.; Tsarbopoulos, A.; Georgakopoulos, C.D.; Lamari, F.N. Cerebral area differential redox response of neonatal rats to selenite-induced oxidative stress and to concurrent administration of Highbush Blueberry leaf polyphenols. Neurochem. Res. 2015, 11, 2280–2292. [Google Scholar] [CrossRef] [PubMed]
- Sato, Y.; Itagaki, S.; Kurokawa, T.; Ogura, J.; Kobayashi, M.; Hirano, T.; Sugawara, M.; Iseki, K. In vitro and in vivo antioxidant properties of chlorogenic acid and caffeic acid. Int. J. Pharm. 2011, 403, 136–138. [Google Scholar] [CrossRef] [PubMed]
- Zielinska, D.; Szawara-Nowak, D.; ZielinskI, H. Determination of the antioxidant activity of rutin and its contribution to the antioxidant capacity of diversified buckwheat origin material by updated analytical strategies. Pol. J. Food Nutr. Sci. 2010, 60, 315–321. [Google Scholar]
- Shi, G.; Rao, L.; Yu, H.; Xiang, H.; Pen, G.; Long, S.; Yang, C. Yeast-cell-based microencapsulation of chlorogenic acid as a water-soluble antioxidant. J. Food Eng. 2007, 80, 1060–1067. [Google Scholar] [CrossRef]
- Ganeshpurkar, A.; Saluja, A.K. The Pharmacological Potential of Rutin. Saudi Pharm J. 2017, 25, 149–164. [Google Scholar] [CrossRef] [PubMed]
- Grace, S.C.; Logan, B.A.; Adams, W.W. Seasonal differences in foliar content of chlorogenic acid, a phenylpropanoid antioxidant, in Mahonia repens. Plant Cell Environ. 1998, 21, 513–521. [Google Scholar] [CrossRef]
- Ramakrishna, A.; Ravishankar, G.A. Influence of abiotic stress signals on secondary metabolites in plants. Plant Signal. Behav. 2011, 6, 1720–1731. [Google Scholar] [PubMed]
- Wang, H.; Guo, X.; Hu, X.; Li, T.; Fu, X.; Liu, R.H. Comparison of phytochemical profiles, antioxidant and cellular antioxidant activities in different varieties of Blueberry (Vaccinium spp.). Food Chem. 2017, 15, 773–781. [Google Scholar] [CrossRef] [PubMed]
- Cardeñosa, V.; Girones-vilaplana, A.; Muriel, J.L.; Moreno, D.A.; Morenoo-Rojas, J.M. Influence of genotype, cultivation system and irrigation regime on antioxidant capacity capacity and selected phenolics of blueberry (Vaccinium corymbosum L.). Food Chem. 2016, 202, 276–283. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, D.; Agustinah, W.; Woods, F.; Coneva, E.; Vinson, E.; Shetty, K. In vitro screening and evaluation of phenolic antioxidant-linked anti-hyperglycemic functions of rabbiteye blueberry (Vaccinium ashei) cultivars. J. Berry Res. 2017, 7, 163–177. [Google Scholar] [CrossRef]
- Yang, J.; Guo, J.; Yuan, J. In vitro antioxidant properties of rutin. Food Sci Technol. 2008, 41, 1060–1066. [Google Scholar] [CrossRef]
- Araruna, M.K.A.; Santos, K.K.A.; Costa, J.G.M.; Coutinho, H.D.M.; Boligon, A.A.; Stefanello, S.T.; Athayde, M.L.; Saraiva, R.A.; Rocha, J.B.T.; Kerntopf, M.R.; et al. Phenolic composition and in vitro activity of the Brazilian fruit tree Caryocar coriaceum Wittm. Eur. J. Integr. Med. 2013, 5, 178–183. [Google Scholar] [CrossRef]
- Woisky, R.G.; Salatino, A. Analysis of própolis: some parameters and procedures for chemical quality control. J. Apic. Res. 1998, 37, 99–105. [Google Scholar] [CrossRef]
- Chandra, S.; Mejia, E.G. Polyphenolic Compounds, antioxidant capacity, and quinone reductase activity of an aqueous extract of Ardisia compressa in Comparison to Mate (Ilex paraguariensis) and Green (Camellia sinensis) Teas. J. Agric. Food Chem. 2004, 52, 3583–3589. [Google Scholar] [CrossRef] [PubMed]
- Evaristo, I.M.; Leitão, M.C. Identificação e quantificação por DAD-HPLC, da fracção fenólica contida em folhas de Quercus suber L. Silva Lusitana 2001, 9, 135–141. [Google Scholar]
- Mensor, L.L.; Menezes, F.S.; Leitão, G.G.; Reis, A.S.; dos Santos, T.C.; Coube, C.S.; Leitão, S.G. Screening of Brazilian plant extracts for antioxidant activity by the use of DPPH free radical method. Phytother. Res. 2001, 15, 127–130. [Google Scholar] [CrossRef] [PubMed]
- Ou, B.; Hampsch-Woodill, M.; Prior, R.L. Development and validation of an improved oxygen radical absorbance capacity assay using fluorescein as the fluorescent probe. J. Agric. Food Chem. 2001, 49, 4619–4626. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Not available. |

| Cultivars | TPC (mg/g) 1 | TFC (µg/g) 2 | ||
|---|---|---|---|---|
| December | March | December | March | |
| Bluegem | 75.4 ± 0.6 a | 170 ± 2 a | 18.9 ± 0.2 a | 45.2 ± 0.5 a |
| Powderblue | 79 ± 1 b | 154 ± 1 b | 24.3 ± 0.3 b | 48.7 ± 0.7 b |
| Clímax | 133.6 ± 0.4 c | 185 ± 1 c | 32.3 ± 0.2 c | 49.8 ± 0.8 b |
| FloridaM | 93.1 ± 0.5 d | 166 ± 1 d | 21.3 ± 0.2d | 38.3 ± 0.8 c |
| Aliceblue | 110 ± 2 e | 222 ± 1 e | 19.5 ± 0.3 a | 39.1 ± 0.6 d |
| Means ± SD | 98 ± 21 | 179 ± 26 | 23 ± 5 | 44 ± 5 |
| Cultivars/Compounds | Clímax | Aliceblue | Bluegem | Powderblue | FloridaM | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| December | March | December | March | December | March | December | March | December | March | |
| Chlorogenic acid | 15.87 ± 0.03 a | 9.8 ± 0.01 b | 15.58 ± 0.05 a | 18.21 ± 0.05 c | 7.41 ± 0.02 d | 2.03 ± 0.03 e | 8.76 ± 0.01 f | 21.28 ± 0.05 g | 17.3 ± 0.3 h | 14.11 ± 0.07 i |
| Rutin | 12.13 ± 0.02 a | 4.38 ± 0.01 b | 11.3 ± 0.1 c | 8.64 ± 0.09 d | 3.73 ± 0.08 e | 15.8 ± 0.1 f | 3.6 ± 0.1 g | 8.9 ± 0.2 d | 11.42 ± 0.02 c | 2.59 ± 0.04 g |
| Quercetin | N.D. | N.D. | 6.20 ± 0.09 a | 0.83 ± 0.02 b | N.D. | N.D. | 7.4 + 0.2c | N.D. | 11.9 ± 0.2 d | N.D. |
| Cultivar | IC50 for DPPH (µg/mL) | ORAC Values (mmol Trolox/100 g) | ||
|---|---|---|---|---|
| December | March | December | March | |
| Bluegem | 105 ± 2 a | 5.80 ± 0.04 a | 211 ± 6 a | 431 ± 8 a |
| Powderblue | 60.1 ± 0.2 b | 12.1 ± 0.6 b | 374 ± 2 b | 202 ± 9 b |
| Clímax | 25 ± 2 c | 12.39 ± 0.02 b | 341 ± 5 c | 283 ± 6 c |
| Florida M | 25.6 ± 0.1 c | 16.0 ± 0.2 c | 178 ± 5 d | 181 ± 9 d |
| Aliceblue | 24.5 ± 0.2 c | 12.8 ± 0.2 b | 245 ± 5 e | 338 ± 4 e |
| Means ± SD | 48 ± 35 | 11 ± 4 | 270 ± 84 | 287 ± 102.01 |
| DPPH | ORAC | |||
|---|---|---|---|---|
| December | March | December | March | |
| TPC | 0.76 | 0.43 a | 0.25 | 0.40 |
| TFC | 0.30 | 0.08 | 0.69 | 0.05 |
| Chlorogenic acid | 0.99 | 0.65 a | 0.24 a | 0.69 a |
| Rutin | 0.98 | 0.83 | 0.21 a | 0.80 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cezarotto, V.S.; Giacomelli, S.R.; Vendruscolo, M.H.; Vestena, A.S.; Cezarotto, C.S.; Da Cruz, R.C.; Maurer, L.H.; Ferreira, L.M.; Emanuelli, T.; Cruz, L. Influence of Harvest Season and Cultivar on the Variation of Phenolic Compounds Composition and Antioxidant Properties in Vaccinium ashei Leaves. Molecules 2017, 22, 1603. https://doi.org/10.3390/molecules22101603
Cezarotto VS, Giacomelli SR, Vendruscolo MH, Vestena AS, Cezarotto CS, Da Cruz RC, Maurer LH, Ferreira LM, Emanuelli T, Cruz L. Influence of Harvest Season and Cultivar on the Variation of Phenolic Compounds Composition and Antioxidant Properties in Vaccinium ashei Leaves. Molecules. 2017; 22(10):1603. https://doi.org/10.3390/molecules22101603
Chicago/Turabian StyleCezarotto, Verciane Schneider, Sandro Rogério Giacomelli, Maria Helena Vendruscolo, Angélica Signor Vestena, Caroll Schneider Cezarotto, Ritiel Corrêa Da Cruz, Luana Haselein Maurer, Luana Mota Ferreira, Tatiana Emanuelli, and Letícia Cruz. 2017. "Influence of Harvest Season and Cultivar on the Variation of Phenolic Compounds Composition and Antioxidant Properties in Vaccinium ashei Leaves" Molecules 22, no. 10: 1603. https://doi.org/10.3390/molecules22101603
APA StyleCezarotto, V. S., Giacomelli, S. R., Vendruscolo, M. H., Vestena, A. S., Cezarotto, C. S., Da Cruz, R. C., Maurer, L. H., Ferreira, L. M., Emanuelli, T., & Cruz, L. (2017). Influence of Harvest Season and Cultivar on the Variation of Phenolic Compounds Composition and Antioxidant Properties in Vaccinium ashei Leaves. Molecules, 22(10), 1603. https://doi.org/10.3390/molecules22101603