Computer-Aided Drug Design Using Sesquiterpene Lactones as Sources of New Structures with Potential Activity against Infectious Neglected Diseases
Abstract
:1. Introduction
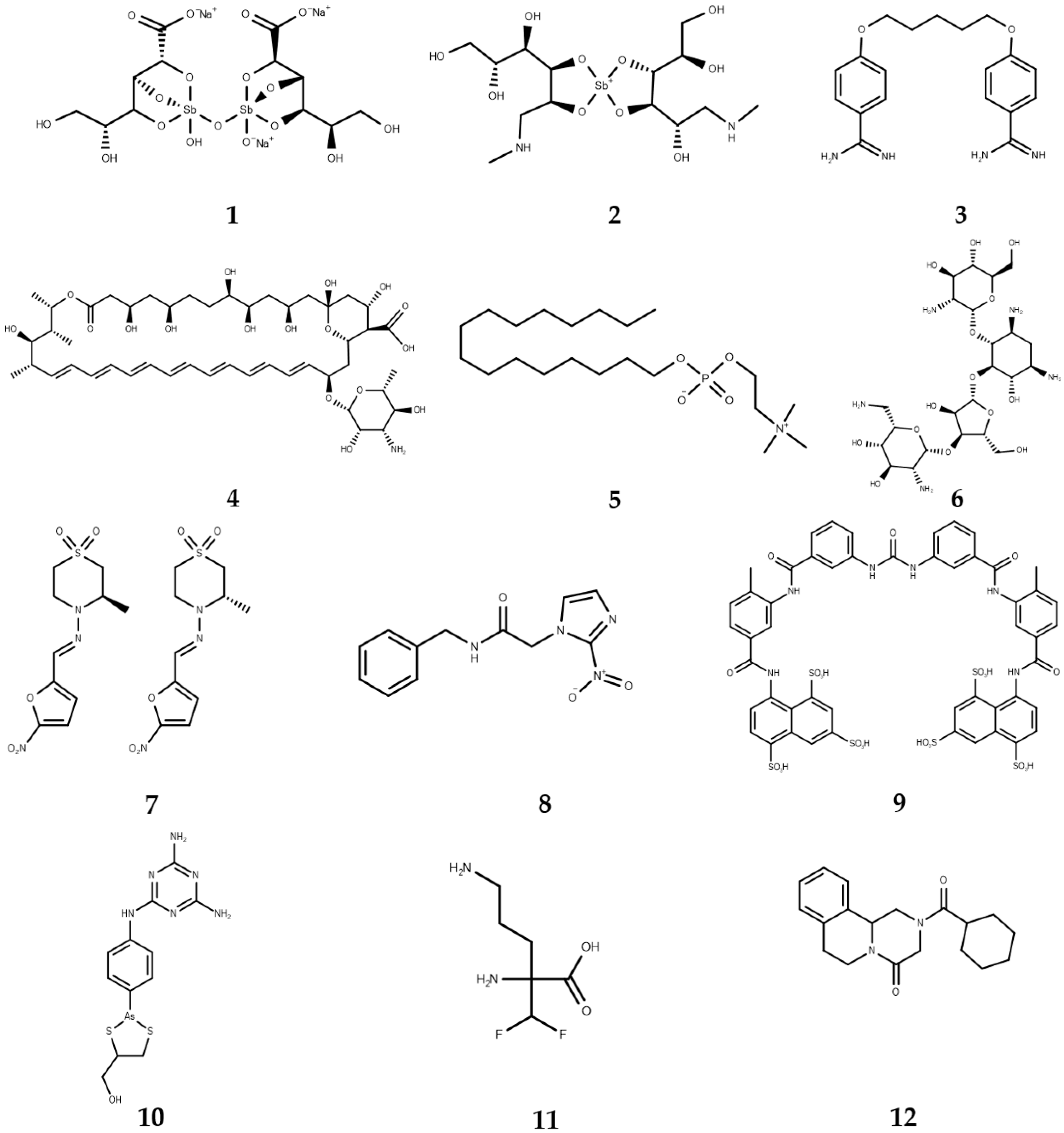
2. The Current Chemotherapy against Infectious NTDs
2.1. Leishmaniasis
2.2. Trypanosomiasis
2.3. Schistosomiasis
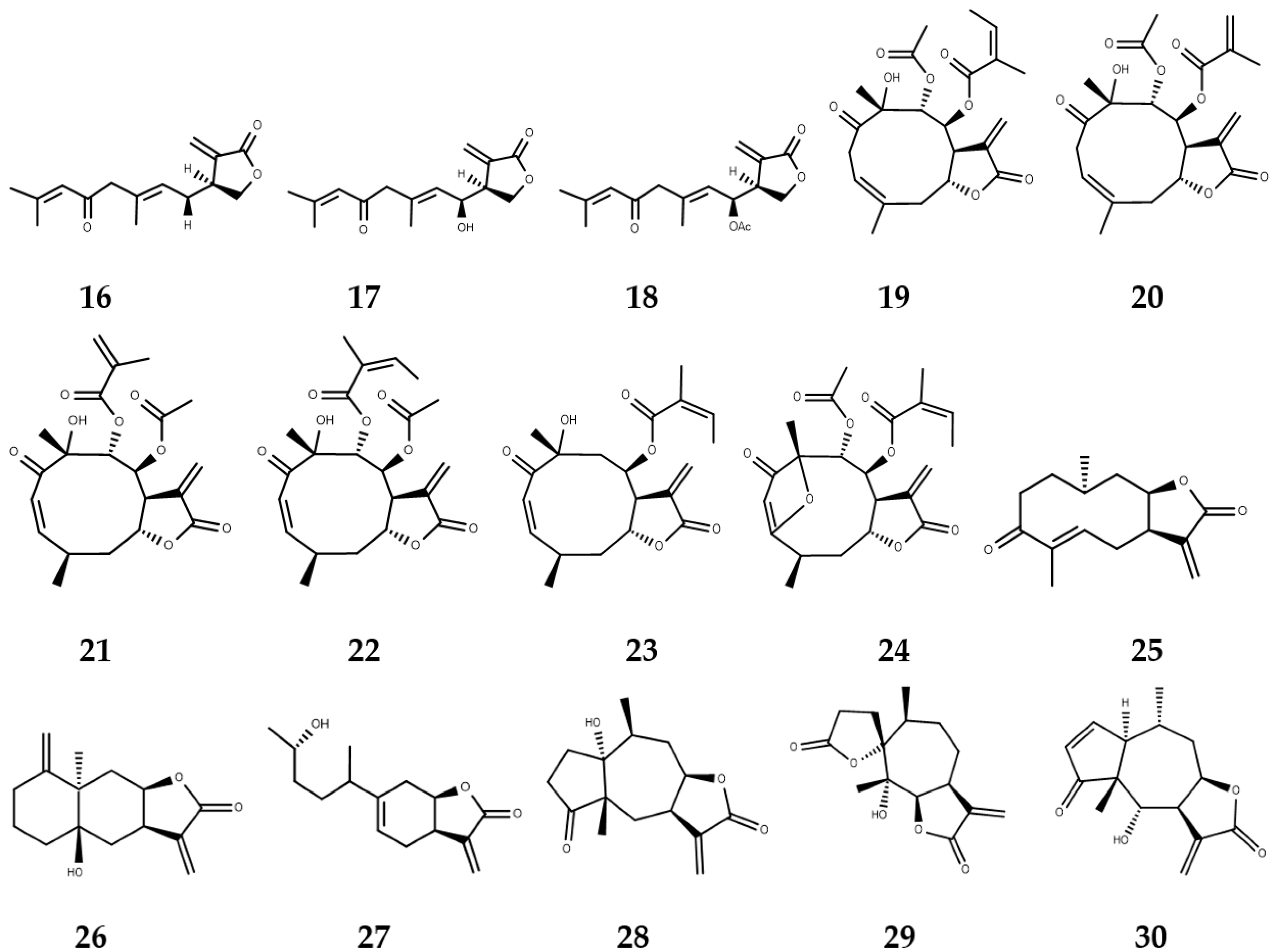
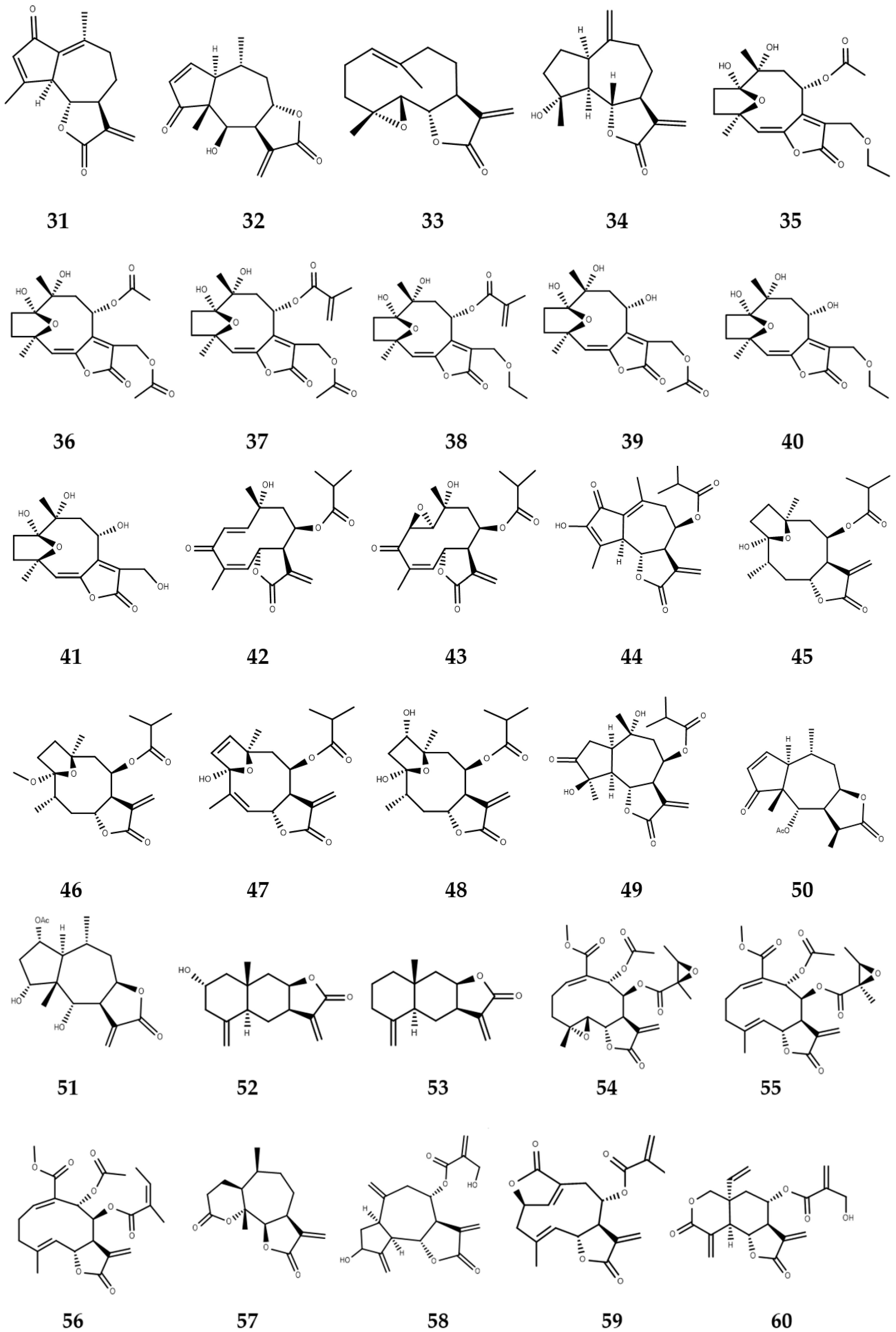
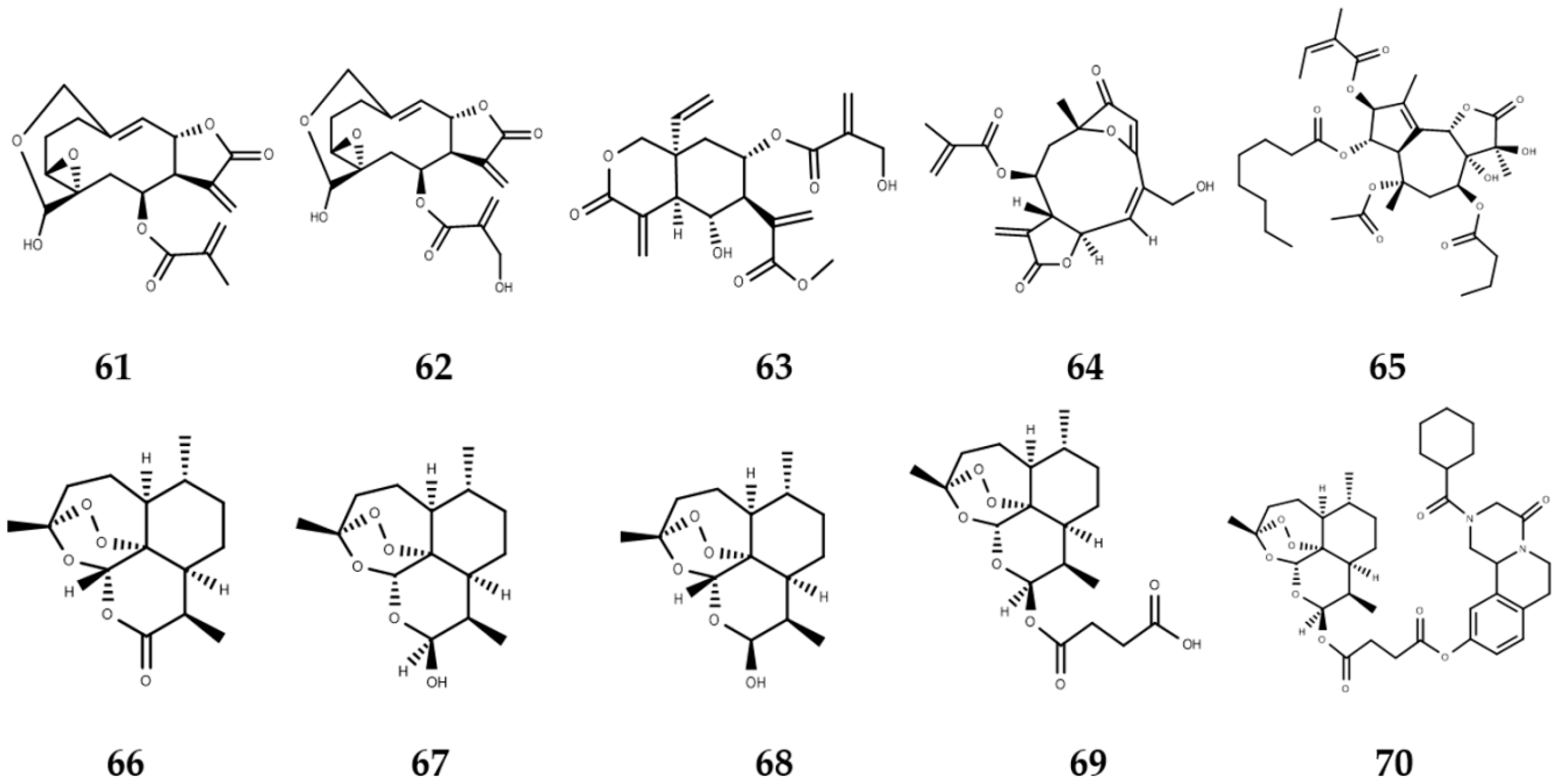
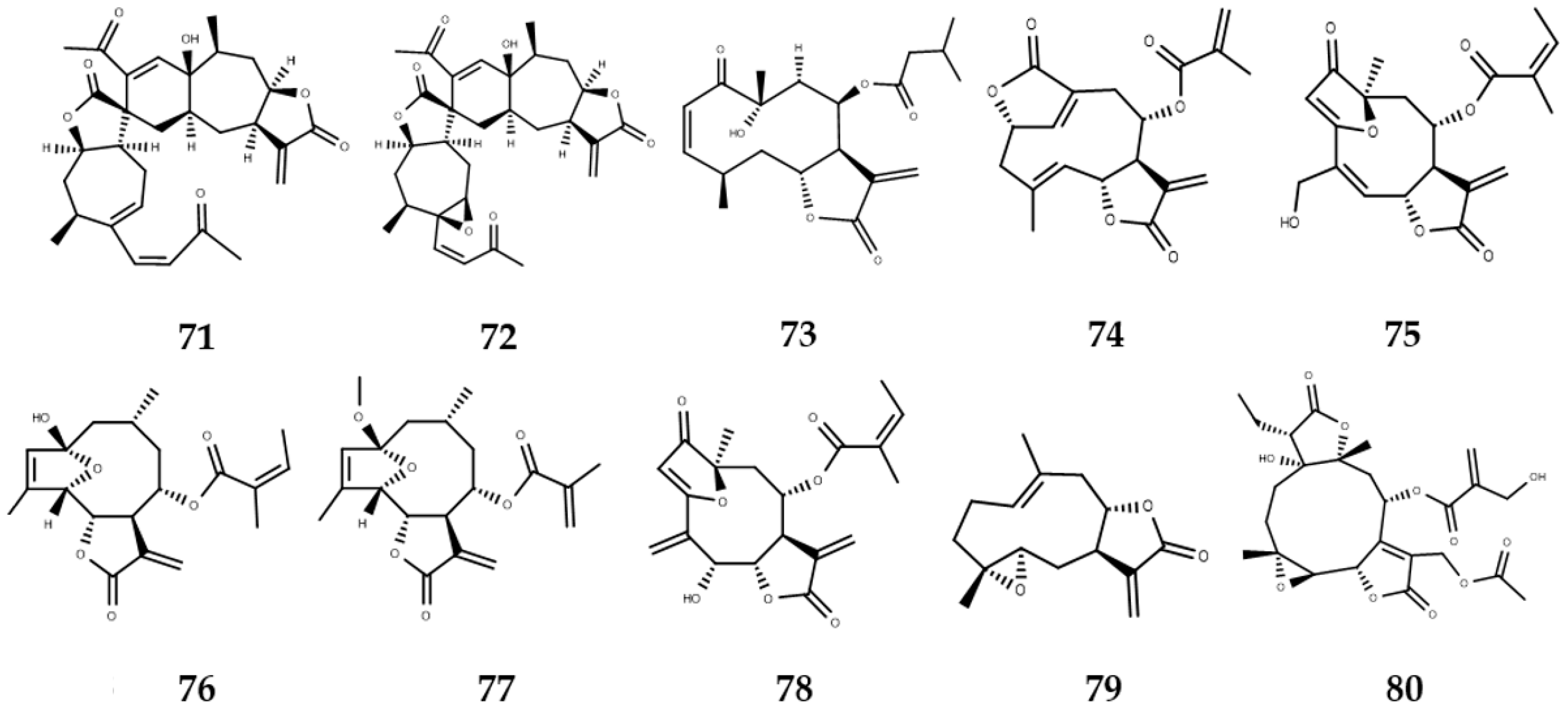
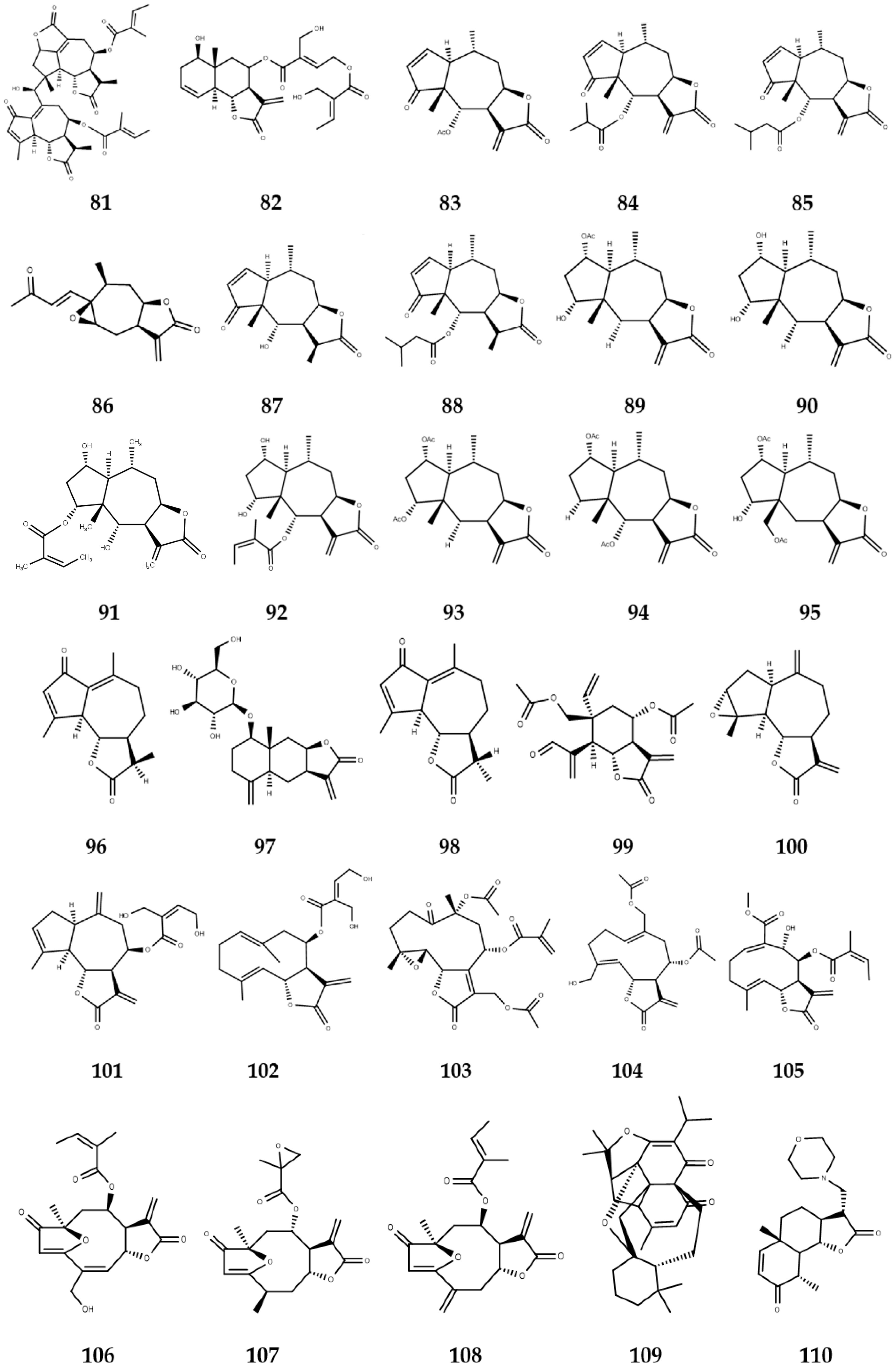
3. Sesquiterpene Lactones with Activity against NTDs
3.1. Sesquiterpene Lactones with Leishmanicidal Activity
3.2. Sesquiterpene Lactones with Trypanocidal Activity
3.3. Sesquiterpene Lactones with Anti-Schistosoma Activity
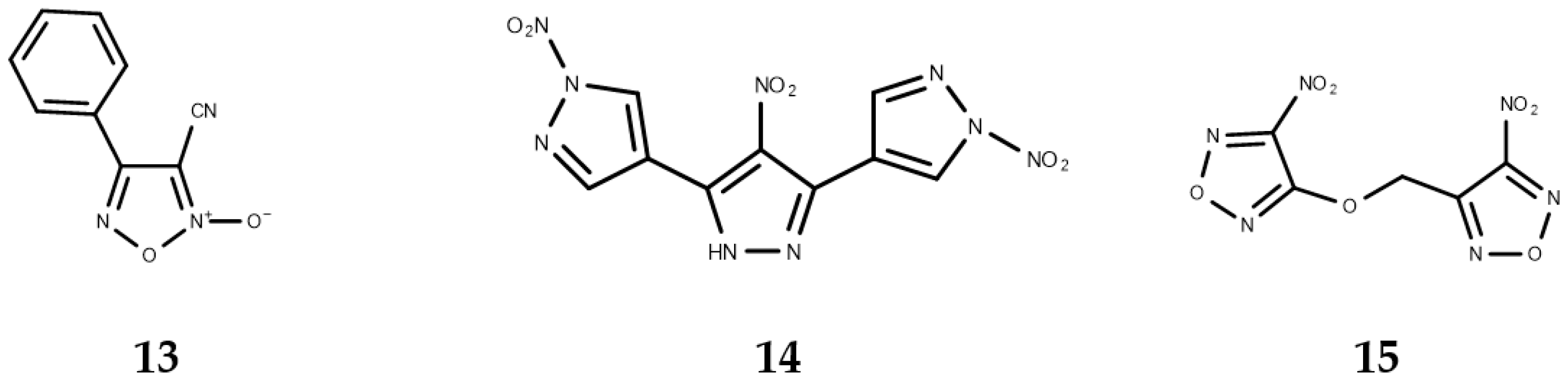
4. CADD Studies against NTDs
4.1. Anti-Leishmania In Silico Studies
4.2. Anti-T. brucei and Anti-T. cruzi In Silico Studies
5. Conclusions and Perspectives
Acknowledgments
Author Contributions
Conflicts of Interest
References
- World Health Organization (WHO). Investing to Overcome the Global Impact of Neglected Tropical Diseases: Third Who Report on Neglected Diseases; WHO Document Production Services: Geneva, Switzerland, 2015. [Google Scholar]
- GBD 2013 Mortality and Causes of Death Collaborators. Global, regional, and national age–sex specific all-cause and cause-specific mortality for 240 causes of death, 1990–2013: A systematic analysis for the global burden of disease study 2013. Lancet 2015, 385, 117–171. [Google Scholar]
- Feasey, N.; Wansbrough-Jones, M.; Mabey, D.C.W.; Solomon, A.W. Neglected tropical diseases. Br. Med. Bull. 2010, 93, 179–200. [Google Scholar] [CrossRef] [PubMed]
- WHO. Neglected Tropical Diseases. Available online: http://www.who.int/neglected_diseases/diseases/en/ (accessed on 10 September 2016).
- Khare, S.; Nagle, A.S.; Biggart, A.; Lai, Y.H.; Liang, F.; Davis, L.C.; Barnes, S.W.; Mathison, C.J.; Myburgh, E.; Gao, M.Y.; et al. Proteasome inhibition for treatment of leishmaniasis, chagas disease and sleeping sickness. Nature 2016, 537, 229–233. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Research priorities for chagas disease, human african trypanosomiasis and leishmaniasis. World Health Organ. Tech. Rep. Ser. 2012, 975, 1–100. [Google Scholar]
- De Luca, V.; Salim, V.; Atsumi, S.M.; Yu, F. Mining the biodiversity of plants: A revolution in the making. Science 2012, 336, 1658–1661. [Google Scholar] [CrossRef] [PubMed]
- Bohm, B.A.; Stuessy, T.F. Flavonoids of the Sunflower Family (Asteraceae); Springer: Vienna, Austria, 2001. [Google Scholar]
- Tu, Y. The discovery of artemisinin (qinghaosu) and gifts from Chinese medicine. Nat. Med. 2011, 17, 1217–1220. [Google Scholar] [CrossRef] [PubMed]
- Nobel Prize. The Nobel Prize in Physiology or Medicine 2015. Press Release: Stockholm, Sweden, 2015; Available online: https://www.nobelprize.org/nobel_prizes/medicine/laureates/2015/press.html (accessed on 11 September 2016).
- Chadwick, M.; Trewin, H.; Gawthrop, F.; Wagstaff, C. Sesquiterpenoids lactones: Benefits to plants and people. Int. J. Mol. Sci. 2013, 14, 12780–12805. [Google Scholar] [CrossRef] [PubMed]
- Scotti, M.T.; Emerenciano, V.; Ferreira, M.J.; Scotti, L.; Stefani, R.; da Silva, M.S.; Mendonca Junior, F.J. Self-organizing maps of molecular descriptors for sesquiterpene lactones and their application to the chemotaxonomy of the asteraceae family. Molecules 2012, 17, 4684–4702. [Google Scholar] [CrossRef] [PubMed]
- Hristozov, D.; Da Costa, F.B.; Gasteiger, J. Sesquiterpene lactones-based classification of the family asteraceae using neural networks and k-nearest neighbors. J. Chem. Inf. Model. 2007, 47, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Da Costa, F.B.; Terfloth, L.; Gasteiger, J. Sesquiterpene lactone-based classification of three asteraceae tribes: A study based on self-organizing neural networks applied to chemosystematics. Phytochemistry 2005, 66, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Scotti, M.T.; Fernandes, M.B.; Ferreira, M.J.; Emerenciano, V.P. Quantitative structure-activity relationship of sesquiterpene lactones with cytotoxic activity. Bioorg. Med. Chem. 2007, 15, 2927–2934. [Google Scholar] [CrossRef] [PubMed]
- Heilmann, J.; Wasescha, M.R.; Schmidt, T.J. The influence of glutathione and cysteine levels on the cytotoxicity of helenanolide type sesquiterpene lactones against KB cells. Bioorg. Med. Chem. 2001, 9, 2189–2194. [Google Scholar] [CrossRef]
- Woerdenbag, H.J.; Merfort, I.; Passreiter, C.M.; Schmidt, T.J.; Willuhn, G.; van Uden, W.; Pras, N.; Kampinga, H.H.; Konings, A.W. Cytotoxicity of flavonoids and sesquiterpene lactones from arnica species against the GLC4 and the COLO 320 cell lines. Planta Med. 1994, 60, 434–437. [Google Scholar] [CrossRef] [PubMed]
- Chappuis, F.; Sundar, S.; Hailu, A.; Ghalib, H.; Rijal, S.; Peeling, R.; Alvar, J.; Boelaert, M. Visceral leishmaniasis: What are the needs for diagnosis, treatment and control? Nat. Rev. Microbiol. 2007, 11, 873–882. [Google Scholar] [CrossRef] [PubMed]
- Pearson, R.D.; Sousa, A.Q. Clinical spectrum of leishmaniasis. Clin. Infect. Dis. 1996, 22, 1–13. [Google Scholar] [CrossRef] [PubMed]
- McGwire, B.S.; Satoskar, A.R. Leishmaniasis: Clinical syndromes and treatment. QJM 2014, 107, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Sundar, S.; Rai, M. Laboratory diagnosis of visceral leishmaniasis. Clin. Diagn. Lab. Immunol. 2002, 9, 951–958. [Google Scholar] [CrossRef] [PubMed]
- Belo, V.S.; Struchiner, C.J.; Barbosa, D.S.; Nascimento, B.W.; Horta, M.A.; da Silva, E.S.; Werneck, G.L. Risk factors for adverse prognosis and death in American visceral leishmaniasis. PLoS Negl. Trop. Dis. 2014, 8, e2982. [Google Scholar] [CrossRef] [PubMed]
- Vasconcellos Ede, C.; Pimentel, M.I.; Schubach Ade, O.; de Oliveira Rde, V.; Azeredo-Coutinho, R.B.; Silva Fda, C.; Salgueiro Mde, M.; Moreira, J.S.; Madeira Mde, F.; Baptista, C.; et al. Intralesional meglumine antimoniate for treatment of cutaneous leishmaniasis patients with contraindication to systemic therapy from Rio De Janeiro (2000 to 2006). Am. J. Trop. Med. Hyg. 2012, 87, 257–260. [Google Scholar] [CrossRef] [PubMed]
- Yesilova, Y.; Surucu, H.A.; Ardic, N.; Aksoy, M.; Yesilova, A.; Oghumu, S.; Satoskar, A.R. Meglumine antimoniate is more effective than sodium stibogluconate in the treatment of cutaneous leishmaniasis. J. Dermatol. Treat. 2016, 27, 83–87. [Google Scholar] [CrossRef] [PubMed]
- Asilian, A.; Sadeghinia, A.; Faghihi, G.; Momeni, A.; Amini Harandi, A. The efficacy of treatment with intralesional meglumine antimoniate alone, compared with that of cryotherapy combined with the meglumine antimoniate or intralesional sodium stibogluconate, in the treatment of cutaneous leishmaniasis. Ann. Trop. Med. Parasitol. 2003, 97, 493–498. [Google Scholar] [CrossRef] [PubMed]
- Sundar, S.; Singh, A. Recent developments and future prospects in the treatment of visceral leishmaniasis. Ther. Adv. Infect. Dis. 2016, 3, 98–109. [Google Scholar] [CrossRef] [PubMed]
- Lira, R.; Sundar, S.; Makharia, A.; Kenney, R.; Gam, A.; Saraiva, E.; Sacks, D. Evidence that the high incidence of treatment failures in Indian Kala-Azar is due to the emergence of antimony-resistant strains of Leishmania donovani. J. Infect. Dis. 1999, 180, 564–567. [Google Scholar] [CrossRef] [PubMed]
- Sundar, S. Drug resistance in Indian visceral leishmaniasis. Trop. Med. Int. Health 2001, 6, 849–854. [Google Scholar] [CrossRef] [PubMed]
- Burrows, J.N.; Elliott, R.L.; Kaneko, T.; Mowbray, C.E.; Waterson, D. The role of modern drug discovery in the fight against neglected and tropical diseases. Med. Chem. Comm. 2014, 5, 688–700. [Google Scholar] [CrossRef]
- Croft, S.L.; Coombs, G.H. Leishmaniasis—Current chemotherapy and recent advances in the search for novel drugs. Trends Parasitol. 2003, 19, 502–508. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.; Kumar, M.; Singh, R.K. Leishmaniasis: Current status of available drugs and new potential drug targets. Asian Pac. J. Trop. Med. 2012, 5, 485–497. [Google Scholar] [CrossRef]
- Clementi, A.; Battaglia, G.; Floris, M.; Castellino, P.; Ronco, C.; Cruz, D.N. Renal involvement in leishmaniasis: A review of the literature. NDT Plus 2011, 4, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Tarleton, R.L. Chagas disease: A solvable problem, ignored. Trends Mol. Med. 2016, 22, 835–838. [Google Scholar] [CrossRef] [PubMed]
- Bermudez, J.; Davies, C.; Simonazzi, A.; Real, J.P.; Palma, S. Current drug therapy and pharmaceutical challenges for chagas disease. Acta Trop. 2016, 156, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Castro, J.A.; de Mecca, M.M.; Bartel, L.C. Toxic side effects of drugs used to treat chagas’ disease (American trypanosomiasis). Hum. Exp. Toxicol. 2006, 25, 471–479. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, A.R.L.; Hecht, M.M.; Guimaro, M.C.; Sousa, A.O.; Nitz, N. Pathogenesis of chagas’ disease: Parasite persistence and autoimmunity. Clin. Microbiol. Rev. 2011, 24, 592–630. [Google Scholar] [CrossRef] [PubMed]
- Apt, W. Current and developing therapeutic agents in the treatment of chagas disease. Drug Des. Dev. Ther. 2010, 4, 243–253. [Google Scholar] [CrossRef]
- Viotti, R.; Vigliano, C.; Lococo, B.; Alvarez, M.G.; Petti, M.; Bertocchi, G.; Armenti, A. Side effects of benznidazole as treatment in chronic chagas disease: Fears and realities. Expert Rev. Anti Infect. Ther. 2009, 7, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Brun, R.; Blum, J.; Chappuis, F.; Burri, C. Human African trypanosomiasis. Lancet 2010, 375, 148–159. [Google Scholar] [CrossRef]
- Jones, A.J.; Avery, V.M. Future treatment options for human African trypanosomiasis. Expert Rev. Anti Infect. Ther. 2015, 13, 1429–1432. [Google Scholar] [CrossRef] [PubMed]
- Nok, A.J. Arsenicals (melarsoprol), pentamidine and suramin in the treatment of human African trypanosomiasis. Parasitol. Res. 2003, 90, 71–79. [Google Scholar] [PubMed]
- Babokhov, P.; Sanyaolu, A.O.; Oyibo, W.A.; Fagbenro-Beyioku, A.F.; Iriemenam, N.C. A current analysis of chemotherapy strategies for the treatment of human African trypanosomiasis. Pathog. Glob. Health 2013, 107, 242–252. [Google Scholar] [CrossRef] [PubMed]
- Raber, G.; Raber, T.; Raml, R.; Murko, M.; Magnes, C.; Francesconi, K.A. Determination of the trypanocidal drug melarsoprol and its conversion products in biological fluids with HPLC–ICPMS/ESMS. Talanta 2013, 116, 876–881. [Google Scholar] [CrossRef] [PubMed]
- Chappuis, F. Melarsoprol-free drug combinations for second-stage gambian sleeping sickness: The way to go. Clin. Infect. Dis. 2007, 45, 1443–1445. [Google Scholar] [CrossRef] [PubMed]
- Chappuis, F.; Udayraj, N.; Stietenroth, K.; Meussen, A.; Bovier, P.A. Eflornithine is safer than melarsoprol for the treatment of second-stage trypanosoma brucei gambiense human african trypanosomiasis. Clin Infect Dis 2005, 41, 748–751. [Google Scholar] [CrossRef] [PubMed]
- Colley, D.G.; Bustinduy, A.L.; Secor, W.E.; King, C.H. Human schistosomiasis. Lancet 2014, 383, 2253–2264. [Google Scholar] [CrossRef]
- Zoni, A.C.; Catala, L.; Ault, S.K. Schistosomiasis prevalence and intensity of infection in latin america and the caribbean countries, 1942–2014: A systematic review in the context of a regional elimination goal. PLoS Negl. Trop. Dis. 2016, 10. [Google Scholar] [CrossRef] [PubMed]
- Fenwick, A.; Jourdan, P. Schistosomiasis elimination by 2020 or 2030? Int. J. Parasitol. 2016, 46, 385–388. [Google Scholar] [CrossRef] [PubMed]
- Cioli, D.; Pica-Mattoccia, L.; Basso, A.; Guidi, A. Schistosomiasis control: Praziquantel forever? Mol. Biochem. Parasit. 2014, 195, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Wang, L.; Liang, Y.S. Susceptibility or resistance of praziquantel in human schistosomiasis: A review. Parasitol. Res. 2012, 111, 1871–1877. [Google Scholar] [CrossRef] [PubMed]
- Alonso, D.; Muñoz, J.; Gascón, J.; Valls, M.E.; Corachan, M. Failure of standard treatment with praziquantel in two returned travelers with schistosoma haematobium infection. Am. J. Trop. Med. Hyg. 2006, 74, 342–344. [Google Scholar] [PubMed]
- Fallon, P.G.; Doenhoff, M.J. Drug-resistant schistosomiasis: Resistance to praziquantel and oxamniquine induced in schistosoma mansoni in mice is drug specific. Am. J. Trop. Med. Hyg. 1994, 51, 83–88. [Google Scholar] [PubMed]
- Sayed, A.A.; Simeonov, A.; Thomas, C.J.; Inglese, J.; Austin, C.P.; Williams, D.L. Identification of oxadiazoles as new drug leads for the control of schistosomiasis. Nat. Med. 2008, 14, 407–412. [Google Scholar] [CrossRef] [PubMed]
- Rai, G.; Sayed, A.A.; Lea, W.A.; Luecke, H.F.; Chakrapani, H.; Prast-Nielsen, S.; Jadhav, A.; Leister, W.; Shen, M.; Inglese, J.; et al. Structure mechanism insights and the role of nitric oxide donation guide the development of oxadiazole-2-oxides as therapeutic agents against schistosomiasis. J. Med. Chem. 2009, 52, 6474–6483. [Google Scholar] [CrossRef] [PubMed]
- Melo-Filho, C.C.; Dantas, R.F.; Braga, R.C.; Neves, B.J.; Senger, M.R.; Valente, W.C.; Rezende-Neto, J.M.; Chaves, W.T.; Muratov, E.N.; Paveley, R.A.; et al. Qsar-driven discovery of novel chemical scaffolds active against Schistosoma Mansoni. J. Chem. Inf. Model. 2016, 56, 1357–1372. [Google Scholar] [CrossRef] [PubMed]
- Karioti, A.; Skaltsa, H.; Kaiser, M.; Tasdemir, D. Trypanocidal, leishmanicidal and cytotoxic effects of anthecotulide-type linear sesquiterpene lactones from Anthemis Auriculata. Phytomedicine 2009, 16, 783–787. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.K.; Fronczek, F.R.; Burandt, C.L.; Zjawiony, J.K. Antileishmanial Germacranolides from Calea Zacatechichi. Planta Med. 2011, 77, 749–753. [Google Scholar] [CrossRef] [PubMed]
- Avolio, F.; Rimando, A.M.; Cimmino, A.; Andolfi, A.; Jain, S.; Tekwani, B.L.; Evidente, A. Inuloxins A-D and derivatives as antileishmanial agents: Structure-activity relationship study. J. Antibiot. 2014, 67, 597–601. [Google Scholar] [CrossRef] [PubMed]
- Sulsen, V.P.; Frank, F.M.; Cazorla, S.I.; Anesini, C.A.; Malchiodi, E.L.; Freixa, B.; Vila, R.; Muschietti, L.V.; Martino, V.S. Trypanocidal and leishmanicidal activities of sesquiterpene lactones from Ambrosia tenuifolia sprengel (asteraceae). Antimicrob. Agents Chemother. 2008, 52, 2415–2419. [Google Scholar] [CrossRef] [PubMed]
- Barrera, P.A.; Jimenez-Ortiz, V.; Tonn, C.; Giordano, O.; Galanti, N.; Sosa, M.A. Natural sesquiterpene lactones are active against leishmania mexicana. J. Parasitol. 2008, 94, 1143–1149. [Google Scholar] [CrossRef] [PubMed]
- Barrera, P.; Sulsen, V.P.; Lozano, E.; Rivera, M.; Beer, M.F.; Tonn, C.; Martino, V.S.; Sosa, M.A. Natural sesquiterpene lactones induce oxidative stress in leishmania mexicana. Evid. Based Complement. Altern. Med. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Tiuman, T.S.; Ueda-Nakamura, T.; Garcia Cortez, D.A.; Dias Filho, B.P.; Morgado-Diaz, J.A.; de Souza, W.; Nakamura, C.V. Antileishmanial activity of parthenolide, a sesquiterpene lactone isolated from Tanacetum parthenium. Antimicrob. Agents Chemother. 2005, 49, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Tiuman, T.S.; Ueda-Nakamura, T.; Alonso, A.; Nakamura, C.V. Cell death in amastigote forms of leishmania amazonensis induced by parthenolide. BMC Microbiol. 2014, 14. [Google Scholar] [CrossRef] [PubMed]
- Rabito, M.F.; Britta, E.A.; Pelegrini, B.L.; Scariot, D.B.; Almeida, M.B.; Nixdorf, S.L.; Nakamura, C.V.; Ferreira, I.C. In vitro and in vivo antileishmania activity of sesquiterpene lactone-rich dichloromethane fraction obtained from Tanacetum parthenium (L.) schultz-bip. Exp. Parasitol. 2014, 143, 18–23. [Google Scholar] [CrossRef] [PubMed]
- Girardi, C.; Fabre, N.; Paloque, L.; Ramadani, A.P.; Benoit-Vical, F.; Gonzalez-Aspajo, G.; Haddad, M.; Rengifo, E.; Jullian, V. Evaluation of antiplasmodial and antileishmanial activities of herbal medicine pseudelephantopus spiralis (less.) cronquist and isolated hirsutinolide-type sesquiterpenoids. J. Ethnopharmacol. 2015, 170, 167–174. [Google Scholar] [PubMed]
- de Toledo, J.S.; Ambrosio, S.R.; Borges, C.H.; Manfrim, V.; Cerri, D.G.; Cruz, A.K.; Da Costa, F.B. In vitro leishmanicidal activities of sesquiterpene lactones from tithonia diversifolia against leishmania braziliensis promastigotes and amastigotes. Molecules 2014, 19, 6070–6079. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, T.J.; Brun, R.; Willuhn, G.; Khalid, S.A. Anti-trypanosomal activity of helenalin and some structurally related sesquiterpene lactones. Planta Med. 2002, 68, 750–751. [Google Scholar] [CrossRef] [PubMed]
- Jimenez, V.; Kemmerling, U.; Paredes, R.; Maya, J.D.; Sosa, M.A.; Galanti, N. Natural sesquiterpene lactones induce programmed cell death in Trypanosoma cruzi: A new therapeutic target? Phytomedicine 2014, 21, 1411–1418. [Google Scholar] [CrossRef] [PubMed]
- Frank, F.M.; Ulloa, J.; Cazorla, S.I.; Maravilla, G.; Malchiodi, E.L.; Grau, A.; Martino, V.; Catalan, C.; Muschietti, L.V. Trypanocidal activity of smallanthus sonchifolius: Identification of active sesquiterpene lactones by bioassay-guided fractionation. Evid. Based Complement. Altern. Med 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Cogo, J.; Caleare, A.d.O.; Ueda-Nakamura, T.; Filho, B.P.D.; Ferreira, I.C.P.; Nakamura, C.V. Trypanocidal activity of guaianolide obtained from Tanacetum parthenium (L.) Schultz-Bip. And its combinational effect with benznidazole. Phytomedicine 2012, 20, 59–66. [Google Scholar] [PubMed]
- Sulsen, V.P.; Puente, V.; Papademetrio, D.; Batlle, A.; Martino, V.S.; Frank, F.M.; Lombardo, M.E. Mode of action of the sesquiterpene lactones psilostachyin and psilostachyin C on Trypanosoma cruzi. PLoS ONE 2016, 11, e0150526. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, S.; Kaiser, M.; Brun, R.; Hamburger, M.; Adams, M. Cynaropicrin: The first plant natural product with in vivo activity against trypanosoma brucei. Planta Med. 2012, 78, 553–556. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, C.F.; Batista Dda, G.; de Araujo, J.S.; Batista, M.M.; Lionel, J.; de Souza, E.M.; Hammer, E.R.; da Silva, P.B.; de Mieri, M.; Adams, M.; et al. Activities of psilostachyin a and cynaropicrin against Trypanosoma cruzi in vitro and in vivo. Antimicrob. Agents Chemother. 2013, 57, 5307–5314. [Google Scholar] [CrossRef] [PubMed]
- Zahari, Z.; Jani, N.A.; Amanah, A.; Latif, M.N.; Majid, M.I.; Adenan, M.I. Bioassay-guided isolation of a sesquiterpene lactone of deoxyelephantopin from elephantopus scaber linn. Active on trypanosome brucei rhodesience. Phytomedicine 2014, 21, 282–285. [Google Scholar] [PubMed]
- Jisaka, M.; Kawanaka, M.; Sugiyama, H.; Takegawa, K.; Huffman, M.A.; Ohigashi, H.; Koshimizu, K. Antischistosomal activities of sesquiterpene lactones and steroid glucosides from vernonia amygdalina, possibly used by wild chimpanzees against parasite-related diseases. Biosci. Biotechnol. Biochem. 1992, 56, 845–846. [Google Scholar] [CrossRef] [PubMed]
- Barth, L.R.; Fernandes, A.P.M.; RibeiroPaes, J.T.; Rodrigues, V. Effects of goyazensolide during in vitro cultivation of schistosoma mansoni. Mem. Inst. Oswaldo Cruz 1997, 92, 427–429. [Google Scholar] [CrossRef] [PubMed]
- Martins, S.M.; Torres, C.R.; Ferreira, S.T. Inhibition of the ecto-ATPdiphosphohydrolase of schistosoma mansoni by thapsigargin. Biosci. Rep. 2000, 20, 369–381. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, C.N.F.; de Oliveira, R.N.; Frezza, T.F.; Rehder, V.L.G.; Allegretti, S.M. Tegument of schistosoma mansoni as a therapeutic target. In Parasitic Diseases—Schistosomiasis; InTech: Rijeka, Croatia, 2013. [Google Scholar]
- Neves, B.; Andrade, C.; Cravo, P. Natural products as leads in schistosome drug discovery. Molecules 2015, 20. [Google Scholar] [CrossRef] [PubMed]
- Madbouly, N.A.; Shalash, I.R.; El Deeb, S.O.; El Amir, A.M. Effect of artemether on cytokine profile and egg induced pathology in murine schistosomiasis Mansoni. J. Adv. Res. 2015, 6, 851–857. [Google Scholar] [CrossRef] [PubMed]
- Dong, L.L.; Duan, W.W.; Chen, J.L.; Sun, H.; Qiao, C.H.; Xia, C.M. An artemisinin derivative of praziquantel as an orally active antischistosomal agent. PLoS ONE 2014, 9, e112163. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, T.J.; Da Costa, F.B.; Lopes, N.P.; Kaiser, M.; Brun, R. In silico prediction and experimental evaluation of furanoheliangolide sesquiterpene lactones as potent agents against trypanosoma brucei rhodesiense. Antimicrob. Agents Chemother. 2014, 58, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Trossini, G.H.; Maltarollo, V.G.; Schmidt, T.J. Hologram qsar studies of antiprotozoal activities of sesquiterpene lactones. Molecules 2014, 19, 10546–10562. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, T.J.; Nour, A.M.; Khalid, S.A.; Kaiser, M.; Brun, R. Quantitative structure—Antiprotozoal activity relationships of sesquiterpene lactones. Molecules 2009, 14, 2062–2076. [Google Scholar] [CrossRef] [PubMed]
- Bernal, F.A.; Coy-Barrera, E. In-silico analyses of sesquiterpene-related compounds on selected leishmania enzyme-based targets. Molecules 2014, 19, 5550–5569. [Google Scholar] [CrossRef] [PubMed]
- Sosa, A.M.; Amaya, S.; Salamanca Capusiri, E.; Gilabert, M.; Bardon, A.; Gimenez, A.; Vera, N.R.; Borkosky, S.A. Active sesquiterpene lactones against leishmania amazonensis and leishmania Braziliensis. Nat. Prod. Res. 2016, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Ogungbe, I.V.; Setzer, W.N. In-silico leishmania target selectivity of antiparasitic terpenoids. Molecules 2013, 18, 7761–7847. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, T.J.; Heilmann, J. Quantitative structure-cytotoxicity relationships of sesquiterpene lactones derived from partial charge (q)-based fractional accessible surface area descriptors (q_frasas). Quant. Struct. Act. Relationsh. 2002, 21, 276–287. [Google Scholar] [CrossRef]
- Schmidt, T. Quantitative structure-cytotoxicity relationships within a series of helenanolide type sesquiterpene lactones. Pharm. Pharmacol. Lett. 1999, 9, 9–13. [Google Scholar]
- Sacconnay, L.; Angleviel, M.; Randazzo, G.M.; Marçal Ferreira Queiroz, M.; Ferreira Queiroz, E.; Wolfender, J.-L.; Carrupt, P.-A.; Nurisso, A. Computational studies on sirtuins from Trypanosoma cruzi: Structures, conformations and interactions with phytochemicals. PLoS Negl. Trop. Dis. 2014, 8, e2689. [Google Scholar] [CrossRef] [PubMed]
- Todeschini, R.; Gramatica, P.; Provenzani, R.; Marengo, E. Weighted holistic invariant molecular descriptors. Part 2. Theory development and applications on modeling physicochemical properties of polyaromatic hydrocarbons. Chemom. Intell. Lab. 1995, 27, 221–229. [Google Scholar]
- Fabian, L.; Sulsen, V.; Frank, F.; Cazorla, S.; Malchiodi, E.; Martino, V.; Lizarraga, E.; Catalan, C.; Moglioni, A.; Muschietti, L.; et al. In silico study of structural and geometrical requirements of natural sesquiterpene lactones with trypanocidal activity. Mini Rev. Med. Chem. 2013, 13, 1407–1414. [Google Scholar] [CrossRef] [PubMed]
- Cruciani, G.; Crivori, P.; Carrupt, P.A.; Testa, B. Molecular fields in quantitative structure–permeation relationships: The volsurf approach. J. Mol. Struct. 2000, 503, 17–30. [Google Scholar] [CrossRef]
- Hu, Z.; Wahl, J.; Hamburger, M.; Vedani, A. Molecular mechanisms of endocrine and metabolic disruption: An in silico study on antitrypanosomal natural products and some derivatives. Toxicol. Lett. 2016, 252, 29–41. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Herrera Acevedo, C.; Scotti, L.; Feitosa Alves, M.; Formiga Melo Diniz, M.D.F.; Scotti, M.T. Computer-Aided Drug Design Using Sesquiterpene Lactones as Sources of New Structures with Potential Activity against Infectious Neglected Diseases. Molecules 2017, 22, 79. https://doi.org/10.3390/molecules22010079
Herrera Acevedo C, Scotti L, Feitosa Alves M, Formiga Melo Diniz MDF, Scotti MT. Computer-Aided Drug Design Using Sesquiterpene Lactones as Sources of New Structures with Potential Activity against Infectious Neglected Diseases. Molecules. 2017; 22(1):79. https://doi.org/10.3390/molecules22010079
Chicago/Turabian StyleHerrera Acevedo, Chonny, Luciana Scotti, Mateus Feitosa Alves, Margareth De Fátima Formiga Melo Diniz, and Marcus Tullius Scotti. 2017. "Computer-Aided Drug Design Using Sesquiterpene Lactones as Sources of New Structures with Potential Activity against Infectious Neglected Diseases" Molecules 22, no. 1: 79. https://doi.org/10.3390/molecules22010079
APA StyleHerrera Acevedo, C., Scotti, L., Feitosa Alves, M., Formiga Melo Diniz, M. D. F., & Scotti, M. T. (2017). Computer-Aided Drug Design Using Sesquiterpene Lactones as Sources of New Structures with Potential Activity against Infectious Neglected Diseases. Molecules, 22(1), 79. https://doi.org/10.3390/molecules22010079







