Abstract
The purpose of this study was to explore a series of Passerini reactions on a biocatalytically derived enantiopure azetidine-2-carboxyaldehyde in order to obtain, in a diastereoselective manner, polyfunctionalised derivatives having the potential to be cyclized to chiral bridged bicyclic nitrogen heterocycles. While diastereoselectivity was poor under classical Passerini conditions, a significant increase of diastereoselectivity (up to 76:24) was gained by the use of zinc bromide as promoter. The methodology has a broad scope and yields are always good.
1. Introduction
Isocyanide-based multicomponent reactions [1,2] represent a very powerful tool in diversity-oriented synthesis, allowing the introduction of several diversity inputs in a single step. However, the use of these reactions for the stereoselective obtainment of non-aromatic chiral heterocycles endowed with several stereogenic centres remains challenging. In principle, this task may be reached by using chiral substrates and by combining the multicomponent step with a subsequent cyclization process. However, the diastereochemical control of the new stereogenic centre by the pre-existing ones is in most cases very poor, unless an intramolecular variant is exploited. So far only chiral amines (for the Ugi reaction) [3,4,5,6,7,8] and very specific carboxylic acids (for the Passerini reaction) [9] have brought about significant levels of diastereoselection. On the contrary, little or no success has been obtained with chiral carbonyl compounds. Moreover, racemization/epimerization is usually observed in the Ugi reaction with aldehydes possessing an α-stereogenic carbon [10,11], probably because of imine-enamine equilibria. From this point of view, the Passerini reaction seems more useful, since it is highly stereoconservative, even with stereochemical labile α-chiral aldehydes [12].
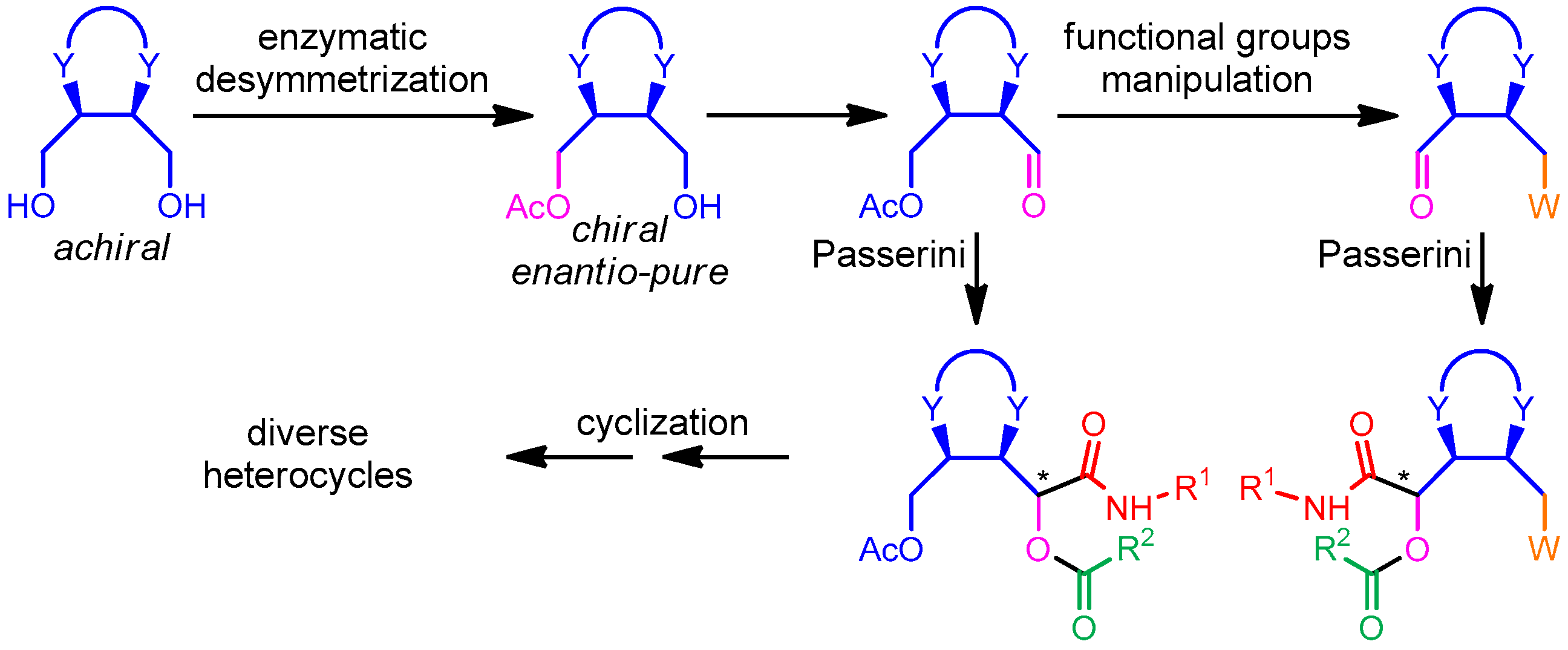
We have devised a general strategy, depicted in Scheme 1, to access chiral enantio-pure heterocycles in a diversity-oriented manner [13]. We mean to exploit, in Passerini reactions, a series of α-chiral aldehydes derived from enzymatic desymmetrization of meso cyclic diols. This strategy offers various benefits: (1) the chiral aldehydes are accessible in a very convenient way and high e.e. using low-cost and “green” catalysts; (2) both enantiomers are accessible, using the complementary acylation and hydrolysis processes; (3) apart from the different rings, diversity can also be achieved by introducing, through functional group manipulations, a variety of alternative appendages, indicated as W in Scheme 1 and (4) the additional arm (a masked CH2OH group or a CH2W group) can be exploited for post-MCR cyclizations leading to a variety of non-aromatic heterocyclic scaffolds (exploration of scaffold diversity). These cyclizations may involve not only the CH2OAc or CH2W, but also the secondary amide NH and other possible additional moieties installed into the carboxylic or isonitrile components. We have recently published a first “proof of concepts” of this strategy, using erythritol derived building blocks [14].
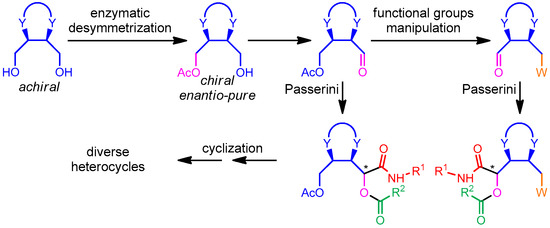
Scheme 1.
General strategy of combining biocatalysis with the Passerini reaction. “*” represents the new stereogenic centre.
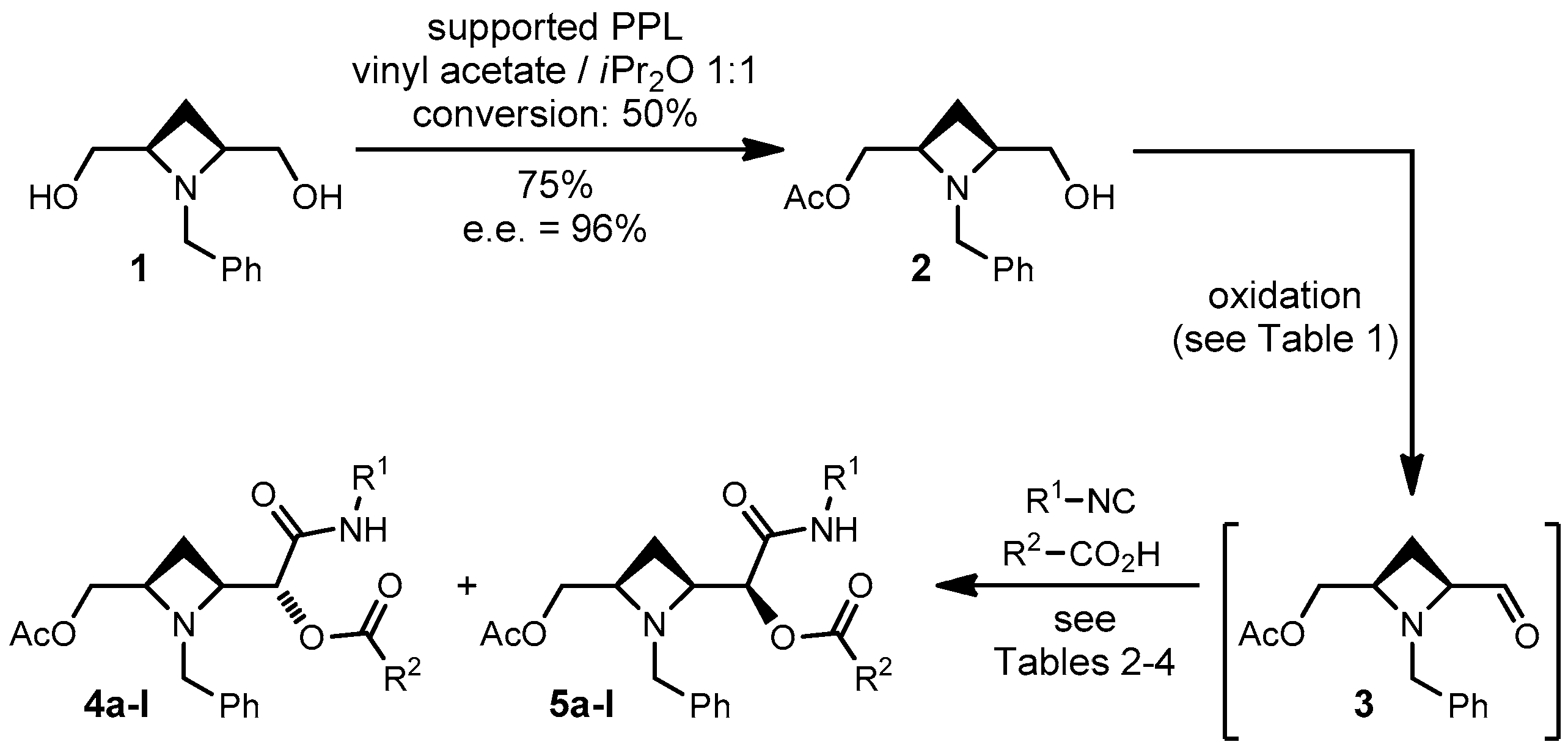
In this paper we report our preliminary results involving a different meso diol, that is 2,4-azetidine-dimethanol 1 (Scheme 2).
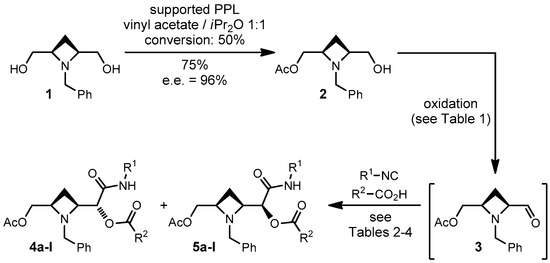
Scheme 2.
Diastereoselective Passerini reactions on aldehyde 3.
2. Results
2.1. Optimization of Oxidation-Passerini on a Model Compound
Meso diol 1 was prepared in four steps from glutaric acid as previously described by us [15], and efficiently desymmetrized with pig pancreatic lipase supported on Celite [15]. The resulting monoacetate 2 was converted into aldehyde 3, which was not isolated but immediately subjected to a Passerini reaction in CH2Cl2. For the first optimization studies we used t-butyl isocyanide and acetic acid as the other two components.
Oxidation of 2 was found out to be troublesome, probably because of the presence of a tertiary amine. In previous works [10,14] we have found that the method employing catalytic TEMPO (2,2,6,6-tetramethylpiperidin-1-yl)oxyl radical) and stoichiometric BAIB (bis-acetoxyiodosobenzene) was particularly useful for generating aldehydes to be used in “one-pot” oxidative Passerini reactions [16]. The only drawback is that the acid component is necessarily acetic acid, generated during the oxidation. However, in this case, this system proved to be unfit (Table 1). The yields of Passerini reaction were unsatisfactory and substantial amounts of unoxidized alcohol 2 were recovered. By increasing the length of the oxidation step (entries 1–3) we could not avoid the residual presence of 2 while the overall yield dropped. Thus we explored other oxidation methods. Also IBX (2-iodoxybenzoic acid), another reagent often used for one-pot oxidative Passerini reactions [16,17,18,19,20], in our hands performed only poorly (entries 4–5) again failing to quantitatively convert 2.

Table 1.
Optimization of oxidation conditions 1.
Eventually we found out that the Swern oxidation was in this case the best one (entries 6–8). After extractive work-up, the resulting crude aldehyde 3 is pure enough to be used as such without any purification, and no epimerization was detected, even using Et3N as the base. Furthermore, this oxidation method allows to use any carboxylic acid as input in the Passerini reaction and, contrary to one-pot methodologies, consents to change solvent and other reaction conditions of the Passerini reaction.
Unfortunately, although the overall yield was good, diastereoselectivity was only poor. In all instances the 4a:5a ratio was only 57:43 and this ratio was not improved by lowering the temperature.
Thus we undertook a deep study of alternative reaction conditions in order to improve the d.r. of the reaction. In particular we thought that a Lewis acid could be able to impose a more rigid transition state through its chelation by the basic nitrogen and the carbonyl oxygen. Table 2 shows a screening of Lewis acids. In some cases (entries 2, 4, 5, 11) the diastereomeric ratio was increased, but at the expense of reaction cleanness. Interestingly, with SnCl4 (entry 5) a reversal of diastereoselectivity was obtained. On the other hand, zinc (II) derivatives afforded full conversions and clean crude products, although with moderate diastereoselectivity (entries 12–15). However, comparison of entries 12 and 13 made us think that prompt solubility of the Lewis acid may be an important issue. The only difference between these two experiments is that in entry 13 we used a preformed solution of Lewis acid.

Table 2.
Effect of various Lewis acids on diastereoselectivity 1.
Thus we selected zinc bromide (more soluble than ZnCl2) and performed a solvent screening (Table 3). As expected, the d.r. was significantly higher in solvent where fast dissolution of ZnBr2 takes place, like CH3CN or THF. Finally, a decrease of the operating temperature brought about a slight increase in yield and d.r. We selected conditions of entry 8 as the best ones.

Table 3.
Optimization of ZnBr2 mediated Passerini 1.
2.2. Scope of the Reaction
The scope of the reaction was then examined (Table 4), using different isocyanides and carboxylic acids. For comparison, about half of the products were also prepared using the standard Passerini conditions (reaction in CH2Cl2 at room temperature without any Lewis acid). As can be seen, yields are always excellent under both conditions, although slightly better without the Lewis acid.

Table 4.
Scope of Passerini reaction of aldehyde 3.
On the other hand, the diastereomeric ratios are persistently improved by the addition of ZnBr2, reaching values up to 76:24. These ratios may be considered good, if one takes into account the typical poor diastereoselectivity of Passerini reactions and the low steric requirements of linear isocyanides. We performed also two experiments with chiral, enantiomerically pure acids (the two enantiomers of N-Fmoc valine) in order to see if diastereoselectivity could be controlled by the chiral acid as well. The results (entries 10, 11) indicate a very little effect of the absolute configuration of the acid on the d.r. and confirms once again the incapacity of chiral carboxylic acids, with very few exceptions [9], to direct the stereochemical course of isocyanide-based multicomponent reactions. In nearly all cases the two diastereomers 4 and 5 could be separated by chromatography (ΔRf > 0.05), with the exception of 4–5h. Thus, despite the not exceptional diastereoselectivity, this allows the obtainment of these multifunctionalised adducts possessing 3 stereogenic centres in high e.e. and diastereomeric purity.
2.3. Establishment of Relative Configuration
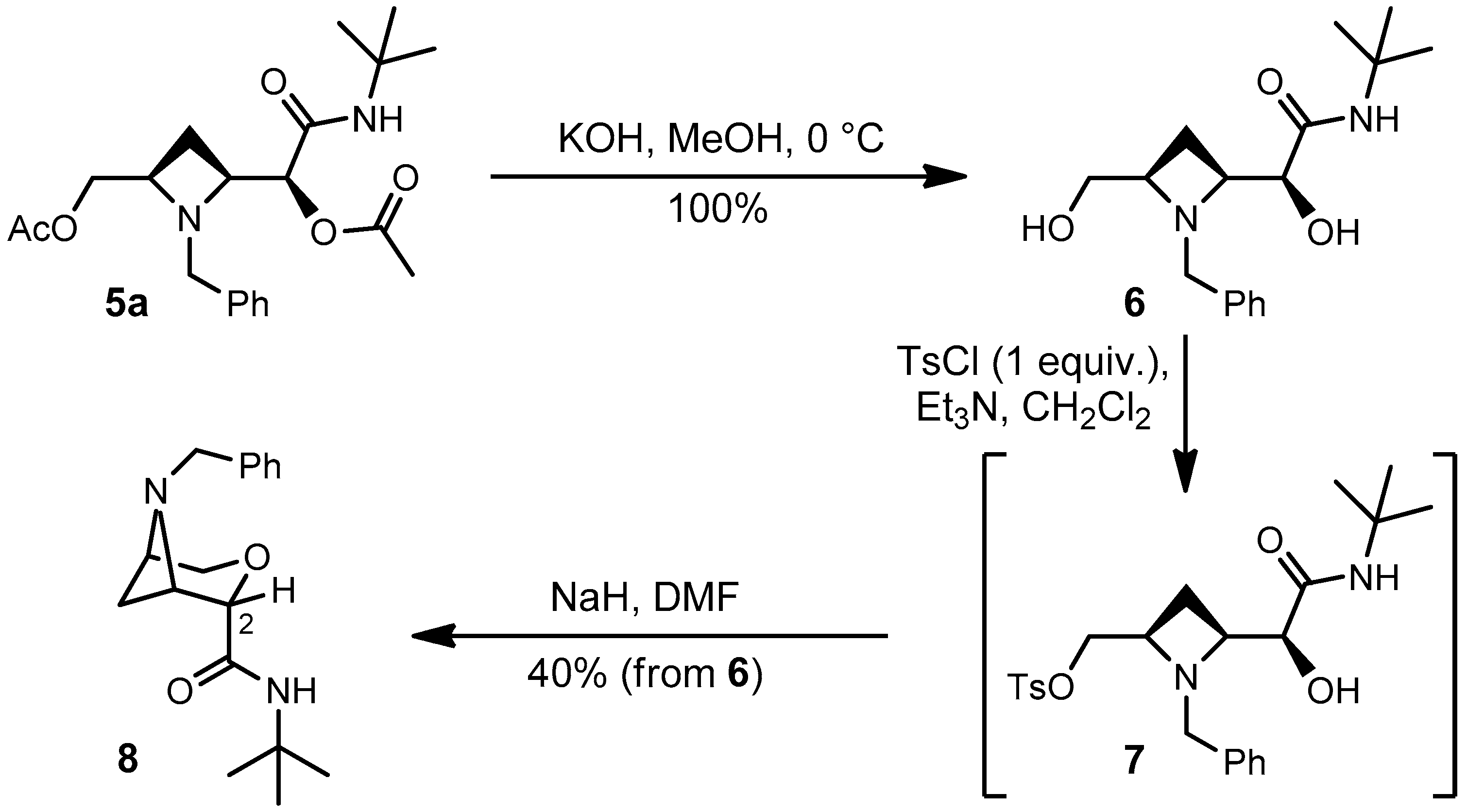
It is our aim to exploit adducts 4a–l and 5a–l for a variety of cyclization reactions affording interesting bridged bicyclic heterocycles, but this is outside the scope of the present paper. However, we report here a very preliminary (unoptimized) cyclization, which has allowed to demonstrate the relative configuration of major and minor adducts 4a and 5a. Compound 5a has been first hydrolysed in quantitative yield to diol 6 (Scheme 3). The latter was reacted with p-toluenesulphonyl chloride leading to selective tosylation. 1H-NMR of the crude showed that tosylation had involved only the primary alcohol. Treatment of the crude tosylate 7 with NaH gave, in moderate yield, compound 8 as a first example of strained bicyclic system. The relative configuration of 8 was demonstrated by nOe experiments. In particular, a 5.9%nOe between the benzylic CH2 and H-2 is particularly diagnostic, being not possible for the other diastereomer. From the relative configuration of 8 we have deduced the configuration of 5a, in the reasonable assumption that the configuration was retained.
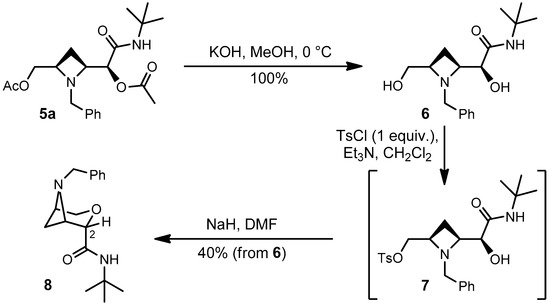
Scheme 3.
Cyclization of Passerini adduct 5a to bridged bicyclic system 8.
Finally, similarities at tlc and 1H-NMR indicate that the major product is always 4a–l. Actually, apart from 4–5h (no separation), a comparison between the thin layer chromatographies revealed the Rf of 5 to be always higher than the one of 4. Furthermore, in the proton spectra, δ of CHOAc is always downfield for 5 with respect to 4 (0.45 > Δδ > 0.18), whereas δ of both diastereotopic CH2OAc is always downfield for 4 compared to 5.
3. Discussion
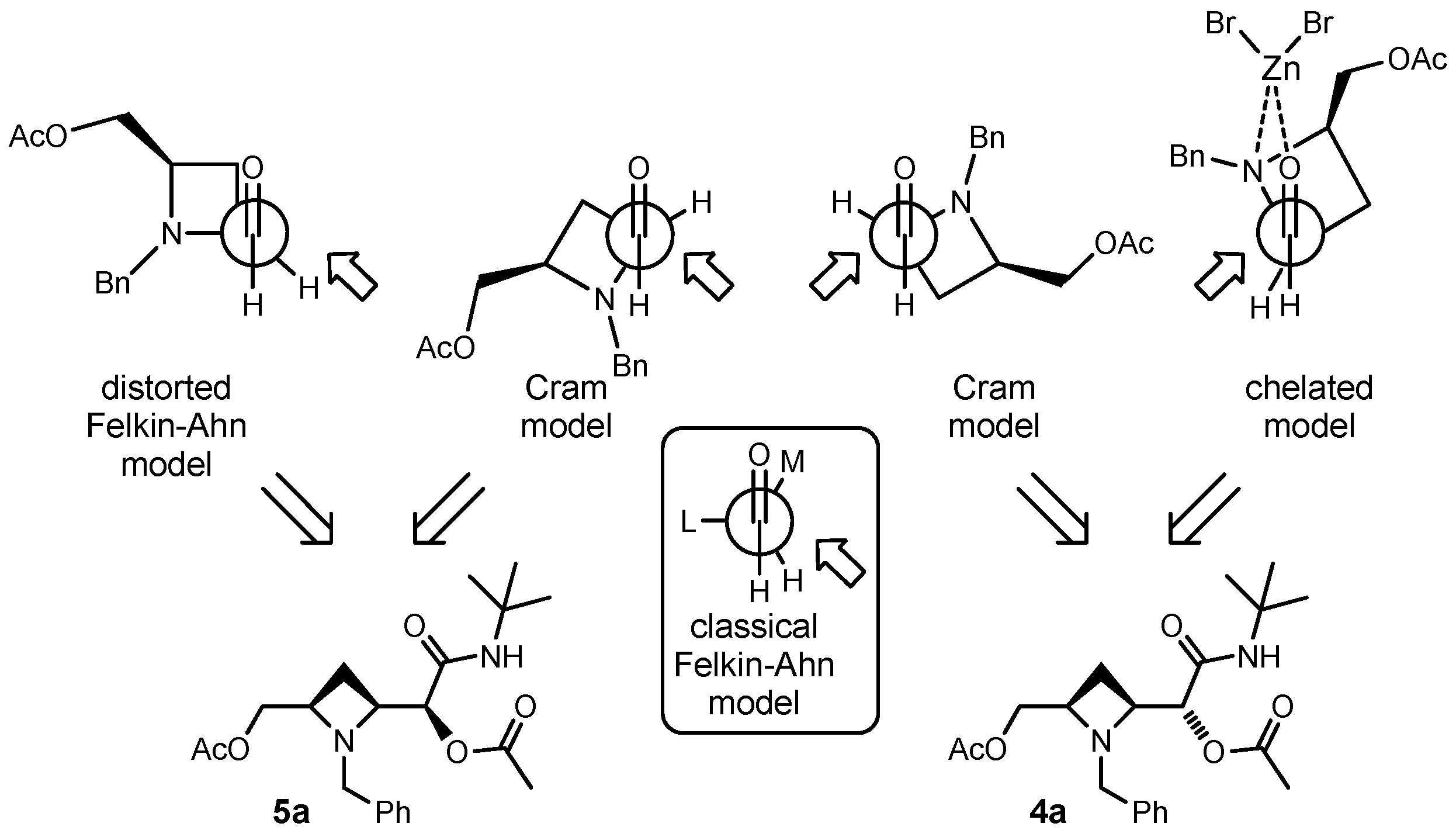
Having established the relative configuration, we may try to rationalise the results. In the absence of Lewis acid, taking into account the typical asymmetric induction achieved with α-aminoaldehydes, one would have expected a diastereoselection dictated by the Felkin-Ahn model, where the nitrogen plays the role of “large” group (Scheme 4). Applying this model, 5, and not 4, would have been the major adduct. However, while in acyclic situations the angles among the three bonds in the background are 109.5° (that become 120° in Newman projection), the presence of the azetidine ring alters the situation, since the bond angles inside the ring are expected to be around 90°. Thus, if the C–N bond is orthogonal to the C=O bond, as predicted by Felkin-Ahn model for stereoelectronic reasons, the C-C bond of azetidine should be eclipsed with the C=O, leading to unfavourable steric interactions. In this case, the two Cram models depicted in Scheme 4 seem more reasonable. The one on the right should prevail, since the steric bias around the carbonyl oxygen is less for a trivalent nitrogen than for a tetravalent carbon. Note that in the commonly accepted mechanism of Passerini reaction, protonation of the aldehyde oxygen by the carboxylic acid takes place concurrently with isocyanide addition. Anyway, the competition between the two Cram conformations and the Felkin-Ahn one can explain, together with the low bulkiness of incoming isocyanide, the low diastereoselection achieved.
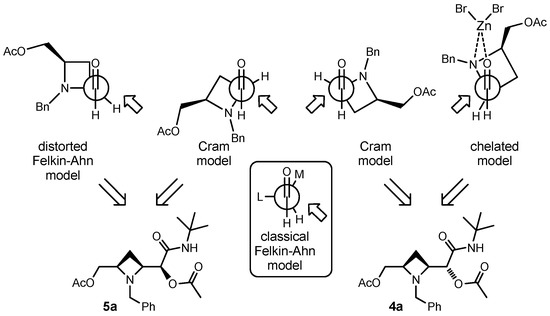
Scheme 4.
Models for rationalisation of results.
In the presence of zinc bromide, coordination of the metal by the nitrogen and by the carbonyl oxygen should result in a chelated model, which correctly predicts 4 as the major product, and this may explain the increase in diastereoselectivity.
Although the final diastereomeric ratios achieved are still only moderate, we have demonstrated, once again, the possibility, at least with chiral aldehydes bearing heteroatoms, to increase the diastereoselectivity using Lewis acids as catalysts or mediators. The high yields, the possibility, in nearly all cases, to separate the two diastereomers, and the high potential of adducts 4–5 for undergoing a variety of cyclization reactions leading to new, bridged, chiral heterocycles (such as 8), make the here presented methodology quite valuable for the synthesis of libraries to be applied in medicinal chemistry. Researches directed towards the obtainment of such heterocycles in a diversity-oriented way are in progress and will be reported in due course. It should be noted that the tertiary amine may be either cleaved to a secondary amine by debenzylation or alkylated to a quaternary cation. Therefore, we think that the bridged rigid systems that can be obtained through cyclization protocols may be useful also as chiral organocatalysts in the form of secondary amines (imminium-enamine catalysis), tertiary amines (base catalysis) or quaternary salts (phase-transfer catalysis).
4. Materials and Methods
4.1. General Remarks
Column chromatographies were done with the “flash” methodology using 220–400 mesh silica. Petroleum ether (40–60 °C) is abbreviated as PE. All reactions using dry solvents were carried out under a nitrogen (or argon if specified) atmosphere. Diol 1 and alcohol 2 were prepared as previously described [15]. Characterization of all new compounds is reported in the Supplementary Material.
4.2. Synthesis of ((2R,4S)-1-Benzyl-4-formylazetidin-2-yl)methyl Acetate (3)
A solution of DMSO (212 μL, 2.99 mmol) in DCM (1.42 mL) was added over 15 min to a stirred solution of oxalyl chloride (133 μL, 1.57 mmol) in DCM (2.90 mL) at −78 °C. Upon completion of the addition, the mixture was stirred at −78 °C for 15 min, followed by addition of a solution of alcohol (2R,4S) 2 (314 mg, 1.26 mmol) in DCM (0.8 mL) over 10 min at −78 °C and the resulting mixture was stirred for 15 min. Then Et3N (350 μL, 2.52 mmol) was added dropwise over 10 min. The resulting mixture was allowed to warm to 0 °C and stirred at 0 °C for 30 min. The reaction was quenched by addition of water (10 mL). The layers were separated and the aqueous phase was extracted with DCM (2 × 10 mL). The combined organic layers were washed with aqueous NaHCO3 (5%) (20 mL) extracted 3 times (10 mL DCM each) and finally with brine, dried over sodium sulphate and concentrated to afford 322 mg of a yellow orange oil. The crude was directly employed in the next Passerini reactions.
4.3. General Procedure for Passerini Reaction under Classical Conditions
A solution of crude aldehyde 3 in dry CH2Cl2 (0.5 M) was treated with the carboxylic acid (1.2 eq) at r.t. After 2 min the isocyanide (1.2 eq) was added. The resulting solution was stirred at r.t. for 1 h, and then quenched with aqueous NaHCO3 (5%). Extraction with CH2Cl2 (3 × 10 mL), drying with Na2SO4, and concentration gave a crude product that was purified by flash column chromatography on silica gel with PE/Et2O or n-hexane/Et2O mixtures.
4.4. General Procedure for Passerini Reaction with ZnBr2
A solution of ZnBr2 (1.0 eq) in dry THF (0.2 M) was placed at −20 °C under nitrogen atmosphere and the isocyanide (1.1 eq) was added. After stirring for 10 min, a solution of the crude aldehyde (1.0 eq) and carboxylic acid (1.1 eq) in THF (0.2 M) was added dropwise. The resulting solution (0.4 M) was stirred at −20 °C for 18 h. The reaction was quenched with aqueous NaHCO3 (5%) (10 mL) and the organic phase was extracted with ethyl acetate (2 × 10 mL), dried over sodium sulphate, and concentrated. The residue was purified by flash column chromatography on silica gel with PE/Et2O or n-hexane/Et2O mixtures.
4.5 Synthesis of (2S)-2-((2S,4R)-1-Benzyl-4-(hydroxymethyl)azetidin-2-yl)-N-(tert-butyl)-2-hydroxyacetamide (6)
A solution of 5a (170 mg, 0.44 mmol) in MeOH (870 μL) was treated, at 0 °C with a solution of KOH (73 mg, 1.31 mmol) in MeOH (1.3 mL). After stirring for 1 h, the reaction was quenched with aqueous NH4Cl (5%) (4 mL) and concentrated under vacuum, in order to remove methanol. The mixture was taken up with saturated aqueous NH4Cl and extracted with AcOEt (10 mL × 3). The organic phase was washed with brine, dried over Na2SO4, and concentrated under vacuum to afford 137 mg of yellow-white, analytically pure, crystals (quant. yield).
4.6 Synthesis of (1S,2S,5R)-6-Benzyl-N-(tert-butyl)-3-oxa-6-azabicyclo[3.1.1]heptane-2-carboxamide (8)
A solution of diol 6 (50 mg, 0.16 mmol) in CH2Cl2 (5 mL) was cooled to 0 °C and treated with Et3N (82 μL, 0.60 mmol), N,N-dimethylaminopyridine (DMAP) (2 mg, 16 µmol) and 4-toluenesulphonyl chloride (31 mg, 0.16 mmol). The mixture was allowed to reach room temperature for 24 h, then it was treated with saturated aqueous NaHCO3 (5%, 20 mL) and extracted with CH2Cl2 (50 + 25 mL). The combined organic phases were washed with brine (20 mL), dried (Na2SO4), and concentrated to give the crude tosylate 7 as a yellow oil. Rf = 0.48 (PE/Et2O 1:3). The crude product could be used in the next step without further purification. To a solution of crude 7 in dry DMF (8 mL) at r.t., sodium hydride (8 mg, 60% of a dispersion in paraffine, 0.20 mmol) was added. The mixture was stirred for 1 h at r.t., then treated with saturated aqueous NH4Cl (40 mL) and extracted with Et2O (50 mL × 2). The combined organic phases were washed with brine (10 mL), dried (Na2SO4), and concentrated. The crude product was eluted from a column of silica gel with PE/Et2O 1:3 to give 8 (19 mg, 40%) as an amorphous solid.
Supplementary Materials
The following are available online at: http://www.mdpi.com/1420-3049/21/9/1153/s1. Full characterization of new compounds and copies of 1H- and 13C-NMR spectra.
Acknowledgments
We thank University of Genova for funding, Erasmus+ for sponsorship to A. Bozzano, Compagnia San Paolo for purchasing of the NMR instrument, and Annegret Laub for performing part of HRMS spectra.
Author Contributions
R.R., L.B., A.B. and L.W. conceived and designed the experiments; A.B., M.S. and L.M. performed the experiments; L.M. and L.W. supervised the work of A.B.; R.R. supervised the work of M.S.; R.R., L.B., L.M. and A.B. analyzed the data; R.R., L.B. and L.M. wrote the paper.
Conflicts of Interest
The authors declare no conflict of interest. The founding sponsors had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, and in the decision to publish the results.
References
- Wessjohann, L.A.; Kaluderovic, G.; Neves Filho, R.A.W.; Morejon, M.C.; Lemanski, G.; Ziegler, T. The Ugi reaction. In Science of Synthesis: Multicomponent Reactions, Vol. 1; Müller, T.J.J., Ed.; Thieme: Stuttgart, Germany, 2013; pp. 415–497. [Google Scholar]
- Riva, R.; Banfi, L.; Basso, A. The Passerini Reaction. In Science of Synthesis: Multicomponent Reactions, Vol. 1; Müller, T.J.J., Ed.; Thieme: Stuttgart, Germany, 2013; pp. 327–414. [Google Scholar]
- Banfi, L.; Basso, A.; Chiappe, C.; De Moliner, F.; Riva, R.; Sonaglia, L. Development of a stereoselective Ugi reaction starting from an oxanorbornene beta-amino acid derivative. Org. Biomol. Chem. 2012, 10, 3819–3829. [Google Scholar] [CrossRef] [PubMed]
- Basso, A.; Banfi, L.; Riva, R.; Guanti, G. A novel highly selective chiral auxiliary for the asymmetric synthesis of l- and d-α-amino acid derivatives via a multicomponent Ugi reaction. J. Org. Chem. 2005, 70, 575–579. [Google Scholar] [CrossRef] [PubMed]
- Hebach, C.; Kazmaier, U. Via Ugi reactions to conformationally fixed cyclic peptides. Chem. Commun. 2003. [Google Scholar] [CrossRef]
- Nenajdenko, V.G.; Gulevich, A.V.; Chernichenko, K.Y.; Sokolova, N.V.; Balenkova, E.S. R-α-Phenylglycinol and R-α-phenylglycinamide as novel chiral templates in diastereoselective Ugi reaction. Mendeleev Commun. 2011, 21, 245–246. [Google Scholar] [CrossRef]
- Ross, G.F.; Herdtweck, E.; Ugi, I. Stereoselective U-4CRs with 1-amino-5-desoxy-5-thio-2,3,4-O-isobutanoyl-β-d-xylopyranose—An effective and selectively removable chiral auxiliary. Tetrahedron 2002, 58, 6127–6133. [Google Scholar] [CrossRef]
- Caputo, S.; Basso, A.; Moni, L.; Riva, R.; Rocca, V.; Banfi, L. Diastereoselective Ugi reaction of chiral 1,3-aminoalcohols derived from an organocatalytic Mannich reaction. Beilstein J. Org. Chem. 2016, 12, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Frey, R.; Galbraith, S.G.; Guelfi, S.; Lamberth, C.; Zeller, M. First examples of a highly stereoselective Passerini reaction: A new access to enantiopure mandelamides. Synlett 2003. [Google Scholar] [CrossRef]
- Moni, L.; Banfi, L.; Basso, A.; Carcone, L.; Rasparini, M.; Riva, R. Ugi and Passerini Reactions of Biocatalytically Derived Chiral Aldehydes: Application to the Synthesis of Bicyclic Pyrrolidines and of Antiviral Agent Telaprevir. J. Org. Chem. 2015, 80, 3411–3428. [Google Scholar] [CrossRef] [PubMed]
- Kelly, G.L.; Lawrie, K.W.M.; Morgan, P.; Willis, C.L. Ugi four component condensations using aldehydes with an asymmetric centre at C-2. Tetrahedron Lett. 2000, 41, 8001–8005. [Google Scholar] [CrossRef]
- Morana, F.; Basso, A.; Riva, R.; Rocca, V.; Banfi, L. The homo-PADAM Protocol: Stereoselective and Operationally Simple Synthesis of α-Oxo- or α-Hydroxy-gamma-acylaminoamides and Chromanes. Chem. Eur. J. 2013, 19, 4563–4569. [Google Scholar] [CrossRef] [PubMed]
- Banfi, L.; Basso, A.; Moni, L.; Riva, R. The Alternative Route to Enantiopure Multicomponent Reaction Products: Biocatalytic or Organocatalytic Enantioselective Production of Inputs for Multicomponent Reactions. Eur. J. Org. Chem. 2014, 2014, 2005–2015. [Google Scholar] [CrossRef]
- Moni, L.; Banfi, L.; Basso, A.; Martino, E.; Riva, R. Diastereoselective Passerini Reaction of Biobased Chiral Aldehydes: Divergent Synthesis of Various Polyfunctionalized Heterocycles. Org. Lett. 2016, 18, 1638–1641. [Google Scholar] [CrossRef] [PubMed]
- Guanti, G.; Riva, R. Synthesis of chiral non-racemic azetidines by lipase-catalysed acetylations and their transformation into amino alcohols: Precursors of chiral catalysts. Tetrahedron Asymm. 2001, 12, 605–618. [Google Scholar] [CrossRef]
- Moni, L.B.L.; Riva, R.; Basso, A. External Oxidant-based Multi Component Reactions. Synthesis 2016, in press. [Google Scholar]
- De Moliner, F.; Crosignani, S.; Banfi, L.; Riva, R.; Basso, A. Synthesis of 5-Carboxamide-oxazolines with a Passerini-Zhu/Staudinger-Aza-Wittig Two-Step Protocol. J. Comb. Chem. 2010, 12, 613–616. [Google Scholar] [CrossRef] [PubMed]
- De Moliner, F.; Crosignani, S.; Galatini, A.; Riva, R.; Basso, A. Novel Application of alpha-Azido Aldehydes in Multicomponent Reactions: Synthesis of Triazolo-Fused Dihydrooxazinones via a Passerini Reaction-Dipolar Cycloaddition Strategy. Acs Comb. Sci. 2011, 13, 453–457. [Google Scholar] [CrossRef] [PubMed]
- Henze, M.; Kreye, O.; Brauch, S.; Nitsche, C.; Naumann, K.; Wessjohann, L.A.; Westermann, B. Photoaffinity-Labeled Peptoids and Depsipeptides by Multicomponent Reactions. Synthesis 2010. [Google Scholar] [CrossRef]
- Ngouansavanh, T.; Zhu, J.P. Alcohols in isonitrile-based multicomponent reaction: Passerini reaction of alcohols in the presence of O-iodoxybenzoic acid. Angew. Chem. Int. Ed. Engl. 2006, 45, 3495–3497. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds 4a–l and 5a–l are available from the authors.
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).