Beneficial Effects of Trillium govanianum Rhizomes in Pain and Inflammation
Abstract
:1. Introduction
2. Results and Discussion
2.1. Acute Toxicity
2.1.1. In Vivo Toxicity Test
2.1.2. In Vitro Toxicity Test
2.2. Anti-Inflammatory Activity
2.3. Analgesic Activity
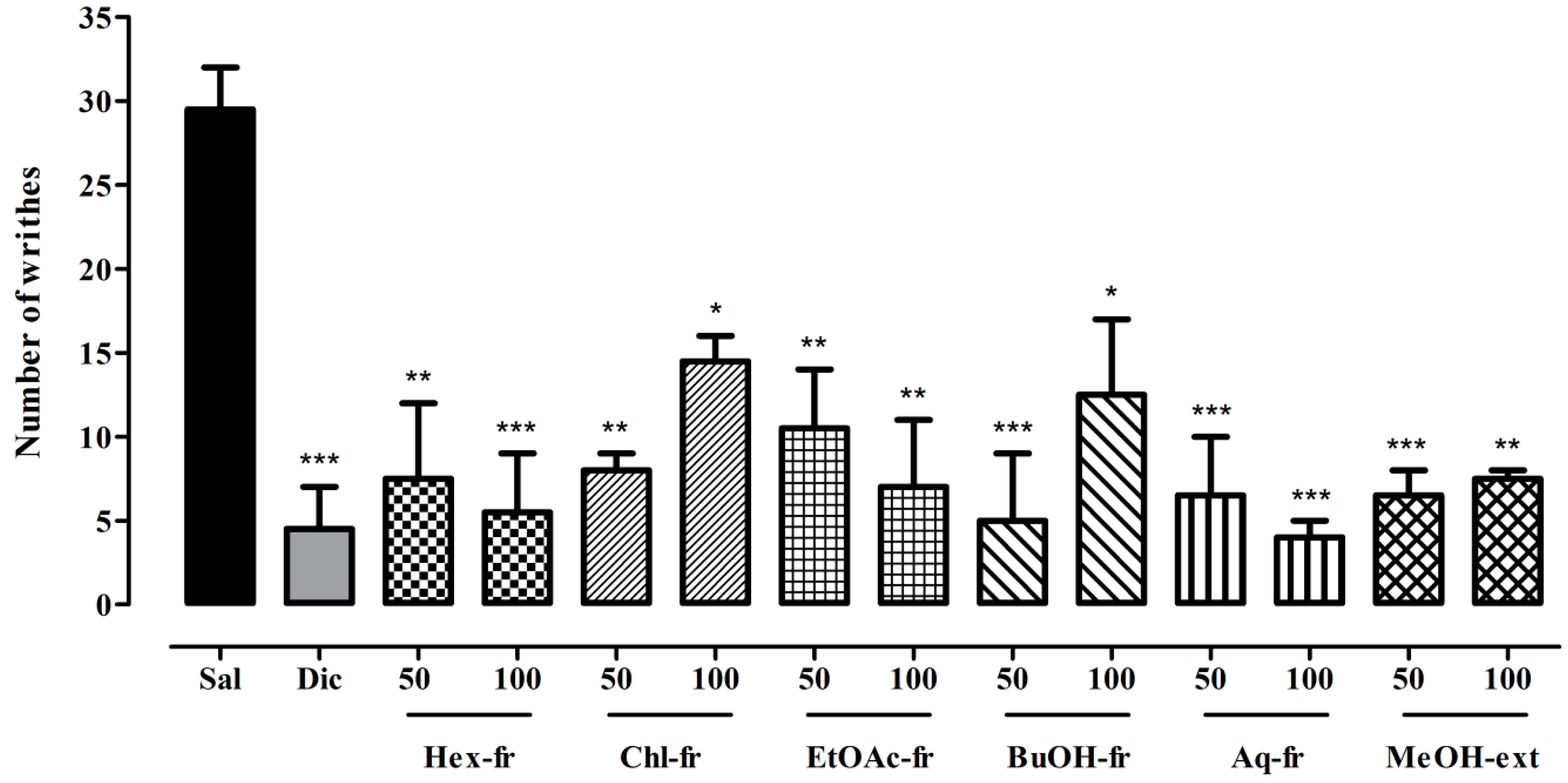
2.3.1. Abdominal Constriction Assay (Tonic Visceral Chemically-Induced Nociception)
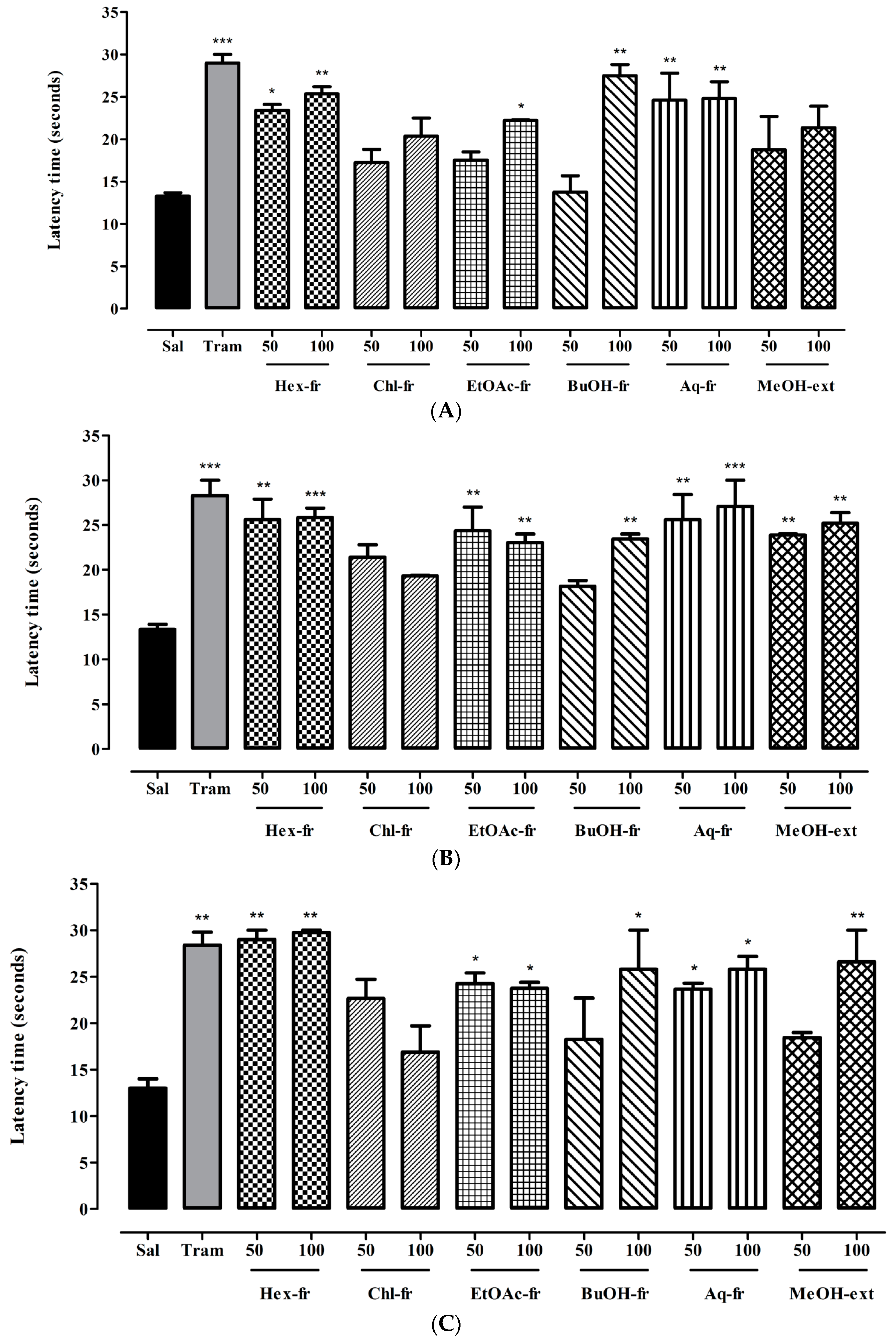
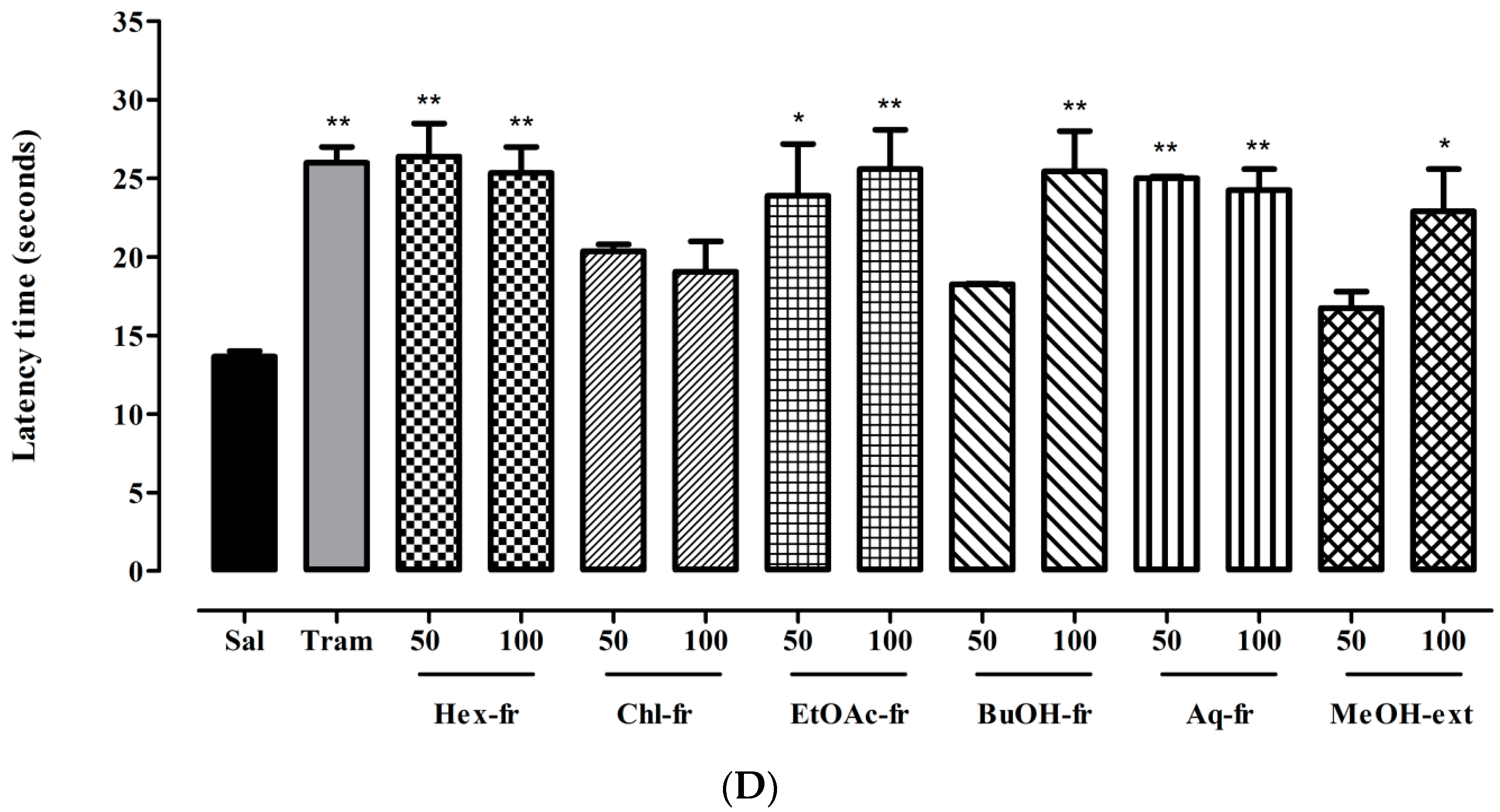
2.3.2. Hot Plate Test (Acute Phasic Thermal Nociception)
3. Experimental Section
3.1. Plant Material
3.2. Extraction and Fractionation
3.3. Animals
3.4. Acute Toxicity Test
3.4.1. In Vivo Toxicity
3.4.2. In Vitro Toxicity
3.5. Anti-Inflammatory Assay
3.5.1. Carrageenan Induced Paw Edema Assay
3.5.2. Chemiluminescence Assay
3.6. Analgesic Activity
3.6.1. Abdominal Constriction Assay
3.6.2. Hot Plate Test
3.7. Statistical Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| MeOH-ext | methanol extract |
| Chl-fr | chloroform fraction |
| EtOAc-fr | ethyl acetate fraction |
| BuOH-fr | n-butanol fraction |
| Hex-fr | n-hexane fraction |
| Aq-fr | aqueous fraction |
| COX-2 | cyclooxygenase-2 |
| HBSS++ | Hanks balanced salt solution |
| iNOS | inducible nitric oxide synthase |
| RLU | relative light units |
| ROS | reactive oxygen species |
| SOZ | serum opsonized zymosan |
| PG | prostaglandins |
| NO | nitric oxide |
| HOCl | hypochlorous acid |
| SOD | superoxide dismutase |
| IL | interleukin |
References
- Munuswamy, H.; Thirunavukkarasu, T.; Rajamani, S.; Elumalai, E.K.; Ernest, D. A review on antimicrobial efficacy of some traditional medicinal plants in Tamilnadu. J. Acute Dis. 2013, 2, 99–105. [Google Scholar] [CrossRef]
- Patwardhan, B.; Warude, D.; Pushpangadan, P.; Bhatt, N. Ayurveda and traditional Chinese medicine: A comparative overview. Evid. Based. Complement. Altern. Med. 2005, 2, 465–473. [Google Scholar] [CrossRef] [PubMed]
- Ficker, C.E.; Smith, M.L.; Susiarti, S.; Leaman, D.J.; Irawati, C.; Arnason, J.T. Inhibition of human pathogenic fungi by members of Zingiberaceae used by the Kenyah (Indonesian Borneo). J. Ethnopharmacol. 2003, 85, 289–293. [Google Scholar] [CrossRef]
- Ficker, C.; Smith, M.; Akpagana, K.; Gbeassor, M.; Zhang, J.; Durst, T.; Assabgui, R.; Arnason, J. Bioassay guided isolation and identification of antifungal compounds from ginger. Phytother. Res. 2003, 17, 897–902. [Google Scholar] [CrossRef] [PubMed]
- Yokosuka, A.; Kawakami, S.; Haraguchi, M.; Mimaki, Y. Seven new triterpene glycosides from the pericarps of Stryphnodendron fissuratum. Phytochem. Lett. 2011, 4, 259–266. [Google Scholar] [CrossRef]
- Gupta, S. Prospects and perspectives of natural plants products in medicine. Indian. J. Pharmacol. 1994, 26, 1–12. [Google Scholar]
- Ahmad, F.; Rafeeq, A.K.; Shahid, R. Study of analgesic and anti-inflammatory activity from plant extracts of Lactuca scariola and Artemisia absinthium. J. Islam. Acad. Sci. 1992, 5, 111–114. [Google Scholar]
- Dina, T.A.; Rahman, M.A.; Ahmed, N.U.; Uddin, M.N. Analgesic and anti-inflammatory properties of Argyreia argentea methanol extract in animal model. J. Taibah Univ. Sci. 2010, 3, 1–7. [Google Scholar] [CrossRef]
- Muhammad, S.K. Diversity of Vascular Plants, Ethnobotany and Conservation Status of Ushairy Valley, District Dir, Upper NWFp Northern Pakistan. Ph.D. Thesis, Quaid-i-Azam University, Islamabad, Pakistan, 2011. Available online: http://eprints.hec.gov.pk/7497/ (accessed on 21 May 2016). [Google Scholar]
- Mahmood, A.; Mahmood, A.; Malik, R.N.; Shinwari, Z.K. Indigenous knowledge of medicinal plants from Gujranwala district, Pakistan. J. Ethnopharmacol. 2013, 148, 714–723. [Google Scholar] [CrossRef] [PubMed]
- Pant, S.; Samant, S. Ethnobotanical observations in the Mornaula reserve forest of Komoun, West Himalaya, India. Ethnobot. Leafl. 2010, 14, 193–217. [Google Scholar]
- Rani, S.; Rana, J.; Rana, P. Ethnomedicinal plants of Chamba district, Himachal Pradesh, India. J. Med. Plant Res. 2013, 7, 3147–3157. [Google Scholar]
- Gates, R.R. A systematic study of the North American genus Trillium, its variability, and its relation to Paris and Medeola. Ann. MI. Bot. Gard. 1917, 4, 43–92. [Google Scholar] [CrossRef]
- Yokosuka, A.; Mimaki, Y. Steroidal glycosides from the underground parts of Trillium erectum and their cytotoxic activity. Phytochemistry 2008, 69, 2724–2730. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Zou, K. Cytotoxicity of a plant steroidal saponin on human lung cancer cells. Asian Pac. J. Cancer Prev. 2011, 12, 513–517. [Google Scholar] [PubMed]
- Ono, M.; Sugita, F.; Shigematsu, S.; Takamura, C.; Yoshimitsu, H.; Miyashita, H.; Ikeda, T.; Nohara, T. Three new steroid glycosides from the underground parts of Trillium kamtschaticum. Chem. Pharm. Bull. 2007, 55, 1093–1096. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Liu, C.; Xiao, D.; Han, J.; Yue, Z.; Sun, Y.; Fan, L.; Zhang, F.; Meng, J.; Zhang, R. Trillium tschonoskii steroidal saponins suppress the growth of colorectal cancer cells in vitro and in vivo. J. Ethnopharmacol. 2015, 168, 136–145. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Zou, K.; Xiong, B. The rhizome of Trillium tschonoskii Maxim. Extract induces apoptosis in human lung cancer cells. Z. Naturforsch. A Phys. Sci. 2011, 66, 477–484. [Google Scholar] [CrossRef]
- Fu, L.K.; Jin, J.M. China Plant Red Data Book-Rare and Endangered Plants; Science Press: Beijing, China, 1992. [Google Scholar]
- Wang, J.; Zou, K.; Zhang, Y.; Liu, C.; Wu, J.; Zhou, Y.; Dan, F.; Zhang, Y. An 18-norspirostanol saponin with inhibitory action against COX-2 production from the underground part of Trillium tschonoskii. Chem. Pharm. Bull. 2007, 55, 679–681. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.l.; Zou, K.; Wang, J.Z.; Zhu, L.B.; Zhou, Y.; Yang, J. Study on the anti-inflammatory, analgesic and thrombosis effects of extract of Trillium tschonoskii Maxim. Lishizhen Med. Mater. Med. Res. 2008, 19, 1178–1180. [Google Scholar]
- Deni, B. The Royal Horticultural Society Encyclopedia of Herbs and Their Uses. Dorling Kindersley B 1996, 424, 116–122. [Google Scholar]
- Rahman, S.U.; Ismail, M.; Shah, M.R.; Adhikari, A.; Anis, I.; Ahmad, M.S.; Khurram, M. Govanoside A, a new steroidal saponin from rhizomes of Trillium govanianum. Steroids 2015, 104, 270–275. [Google Scholar] [CrossRef] [PubMed]
- Rahman, S.U.; Ismail, M.; Khurram, M.; Haq, I.U. Pharmacognostic and ethnomedicinal studies on Trillium govanianum. Pak. J. Bot. 2015, 47, 187–192. [Google Scholar]
- Rahman, S.U.; Ismail, M.; Shah, M.R.; Iriti, M.; Shahid, M. GC/MS analysis, free radical scavenging, anticancer and β-glucuronidase inhibitory activities of Trillium govanianum rhizomes. Bangladesh J. Pharmacol. 2015, 10, 577–583. [Google Scholar] [CrossRef]
- Pawliczak, R. The role of radical oxygen species in airway inflammation. Pol. Merkur. Lekarski. 2003, 14, 493–496. [Google Scholar] [PubMed]
- Cuzzocrea, S.; Mazzon, E.; Dugo, L.; Serraino, I.; Ciccolo, A.; Centorrino, T.; De Sarro, A.; CAPUTI, A.P. Protective effects of n-acetylcysteine on lung injury and red blood cell modification induced by carrageenan in the rat. FASEB J. 2001, 15, 1187–1200. [Google Scholar] [CrossRef] [PubMed]
- Salvemini, D.; Wang, Z.-Q.; Bourdon, D.M.; Stern, M.K.; Currie, M.G.; Manning, P.T. Evidence of peroxynitrite involvement in the carrageenan-induced rat paw edema. Eur. J. Pharmacol. 1996, 303, 217–220. [Google Scholar] [CrossRef]
- Fridovich, I. Superoxide anion radical (O2), superoxide dismutases, and related matters. J. Biol. Chem. 1997, 272, 18515–18517. [Google Scholar] [CrossRef] [PubMed]
- Uttara, B.; Singh, A.V.; Zamboni, P.; Mahajan, R. Oxidative stress and neurodegenerative diseases: A review of upstream and downstream antioxidant therapeutic options. Curr. Neuropharmacol. 2009, 7, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Sultana, N.; Arayne, M.S.; Naz, A.; Mesaik, M.A. Identification of anti-inflammatory and other biological activities of 3-carboxamide, 3-carbohydrazide and ester derivatives of gatifloxacin. Chem. Cent. J. 2013, 7, 6. [Google Scholar] [CrossRef] [PubMed]
- Gene, R.M.; Segura, L.; Adzet, T.; Marin, E.; Iglesias, J. Heterotheca inuloides: Anti-inflammatory and analgesic effect. J. Ethnopharmacol. 1998, 16, 157–162. [Google Scholar] [CrossRef]
- Salvemini, D.; Wang, Z.Q.; Wyatt, P.S.; Bourdon, D.M.; Marino, M.H.; Manning, P.T.; Currie, M.G. Nitric oxide: A key mediator in the early and late phase of carrageenan-induced rat paw inflammation. Br. J. Pharmacol. 1996, 118, 829–838. [Google Scholar] [CrossRef] [PubMed]
- Ianaro, A.; O’donnell, C.; Di Rosa, M.; Liew, F. A nitric oxide synthase inhibitor reduces inflammation, down-regulates inflammatory cytokines and enhances interleukin-10 production in carrageenin-induced oedema in mice. Immunology 1994, 82, 370–375. [Google Scholar] [PubMed]
- Seibert, K.; Zhang, Y.; Leahy, K.; Hauser, S.; Masferrer, J.; Perkins, W.; Lee, L.; Isakson, P. Pharmacological and biochemical demonstration of the role of cyclooxygenase 2 in inflammation and pain. Proc. Natl. Acad. Sci. USA 1994, 91, 12013–12017. [Google Scholar] [CrossRef] [PubMed]
- Fantone, J.C.; Ward, P. Role of oxygen-derived free radicals and metabolites in leukocyte-dependent inflammatory reactions. Am. J. Pathol. 1982, 107, 395–418. [Google Scholar] [PubMed]
- Huang, W.H.; An-Rong, L.; Ching-Huey, Y. Antioxidative and anti-inflammatory activities of polyhydroxyflavonoids of Scutellaria baicalensis GEORGI. Biosci. Biotechnol. Biochem. 2006, 70, 2371–2380. [Google Scholar] [CrossRef] [PubMed]
- Punitha, D.; Udhayasankar, M.R.; Danya, U.; Arumugasamy, K.; Shalimol, A. Anti-inflammatory Activity of Characterized Compound Diosgenin Isolated from Tinospora malabarica Miers in Ann. (Menispermaceae) in Animal Model. Int. J. Herb. Med. 2013, 1, 76–78. [Google Scholar]
- Ferreira, S.; Lorenzetti, B.B.; Castro, M.S.A.; Correa, F. Antialgic effect of aspirin-like drugs and the inhibition of prostaglandin synthesis. In The Recognition of Anti-rheumatic Drugs; MTP Press Ltd.: Lancaster, PA, USA, 1978; pp. 25–37. [Google Scholar]
- Hosseinzadeh, H.; Younesi, H.M. Antinociceptive and anti-inflammatory effects of Crocus sativus L. stigma and petal extracts in mice. BMC Pharmacol. 2002, 2, 7. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.-R.; Zhou, H.; Jiang, Z.-H.; Wong, Y.F.; Liu, L. In vivo anti-inflammatory and analgesic activities of a purified saponin fraction derived from the root of Ilex pubescens. Biol. Pharm. Bull. 2008, 31, 643–650. [Google Scholar] [CrossRef] [PubMed]
- Navarro, P.; Giner, R.M.; Recio, M.C.; Máñez, S.; Cerdá-Nicolás, M.; Rı́os, J.-L. In vivo anti-inflammatory activity of saponins from Bupleurum rotundifolium. Life Sci. 2001, 68, 1199–1206. [Google Scholar] [CrossRef]
- Shao, B.; Hongzhu, G.; Yajun, C.; Min, Y.; Han, J.; Guo, D. Steroidal saponins from Smilax china and their anti-inflammatory activities. Phytochemistry 2007, 68, 623–630. [Google Scholar] [CrossRef] [PubMed]
- Agnihotri, S.; Wakode, S.; Agnihotri, A. An overview on anti-inflammatory properties and chemo-profiles of plants used in traditional medicine. Indian J. Nat. Prod. Resour. 2010, 1, 150–167. [Google Scholar]
- Boué, J.; Blanpied, C.; Brousset, P.; Vergnolle, N.; Dietrich, G. Endogenous opioid-mediated analgesia is dependent on adaptive T cell response in mice. J. Immunol. 2011, 186, 5078–5084. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, H.; Shimoda, A.; Ishii, K.; Kadokawa, T. Central and peripheral analgesic action of non-acidic non-steroidal anti-inflammatory drugs in mice and rats. Arch. Int. Pharmacodyn. Ther. 1986, 282, 16–25. [Google Scholar] [PubMed]
- Collier, H.; Dinneen, L.; Johnson, C.A.; Schneider, C. The abdominal constriction response and its suppression by analgesic drugs in the mouse. Br. J. Pharmacol. Chemother. 1968, 32, 295–310. [Google Scholar] [CrossRef] [PubMed]
- Tjølsen, A.; Rosland, J.H.; Berge, O.-G.; Hole, K. The increasing-temperature hot-plate test: An improved test of nociception in mice and rats. J. Pharmacol. Methods 1991, 25, 241–250. [Google Scholar] [CrossRef]
- Thangaraj, P. Pharmacological Assays of Plant-Based Natural Products; Springe Nature: Basel, Switzerland, 2016. [Google Scholar] [CrossRef]
- Khan, H.; Saeed, M.; Khan, M.A.; Dar, A.; Khan, I. The antinociceptive activity of Polygonatum verticillatum rhizomes in pain models. J. Ethnopharmacol. 2010, 127, 521–527. [Google Scholar] [CrossRef] [PubMed]
- Baydoun, E.; Bibi, M.; Iqbal, M.A.; Atia-tul, W.; Farran, D.; Smith, C.; Sattar, S.A.; Rahman, A.U.; Choudhary, M.I. Microbial transformation of anticancer steroid exemestane and cytotoxicity of its metabolites against cancer cell lines. Chem. Cent. J. 2013, 7. [Google Scholar] [CrossRef] [PubMed]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Morris, C.J. Carrageenan-induced paw edema in the rat and mouse. Inflamm. Protoc. 2003, 225, 115–121. [Google Scholar]
- Helfand, S.L.; Werkmeister, J.; Roder, J.C. Chemiluminescence response of human natural killer cells. I. The relationship between target cell binding, chemiluminescence, and cytolysis. J. Exp. Med. 1982, 156, 492–505. [Google Scholar] [CrossRef] [PubMed]
- O’Callaghan, J.P.; Holtzman, S.G. Quantification of the analgesic activity of narcotic antagonists by a modified hot-plate procedure. J. Pharmacol. Exp. Ther. 1975, 192, 497–505. [Google Scholar] [PubMed]
- Sample Availability: Not available.
| Dose (mg/kg) | Total Number of Mice = 6 | Percent Lethality | LD50 (mg/kg) | |
|---|---|---|---|---|
| No. of Dead Mice | No. of Survived Mice | |||
| 150 | 0 | 6 | 0 | 2030.4 (1488.8–3069.0) * |
| 500 | 0 | 6 | 0 | |
| 1000 | 1 | 5 | 16 | |
| 1500 | 2 | 4 | 33 | |
| 3000 | 5 | 1 | 83 | |
| 6000 | 6 | 0 | 100 | |
| Sample | IC50 (µg/mL) |
|---|---|
| MeOH-ext | 7.89 ± 0.43 |
| Hex-fr | 5.78 ± 0.34 |
| Chl-fr | 3.69 ± 0.77 |
| EtOAc-fr | 3.95 ± 0.25 |
| BuOH-fr | 2.59 ± 0.14 |
| Aq-fr | 3.07 ± 0.29 |
| Cycloheximide | 0.73 ± 0.12 |
| Sample | Dose (mg/kg) | 1st h | 2nd h | 3rd h | 4th h | 5th h |
|---|---|---|---|---|---|---|
| Diclofenac sodium | 10 | 27.3 ± 2.7 *** | 47.6 ± 0.8 *** | 67.0 ± 2.5 *** | 70.6 ± 0.6 *** | 74.3 ± 0.6 *** |
| MeOH-ext | 50 | 8.0 ± 1.7 * | 18.0 ± 1.7 ** | 22.6 ± 2.3 *** | 32.0 ± 5.2 *** | 34.0 ± 5.2 *** |
| 100 | 12.0 ± 1.1 ** | 44.3 ± 4.6 *** | 65.0 ± 4.6 *** | 63.6 ± 1.7 *** | 66.3 + 4.6 *** | |
| 200 | 19.0 ± 2.3 * | 44.6 ± 3.8 *** | 62.6 ± 3.7 *** | 62.6 ± 1.4 *** | 64.6 ± 4.0 *** | |
| Chl-fr | 25 | 4.0 ± 1.0 * | 8.5 ± 0.5 * | 21.3 ± 3.5 ** | 35.0 ± 1.0 *** | 42.5 ± 2.5 *** |
| 50 | 4.0 ± 2.0 ** | 18.8 ± 1.0 ** | 20.3 ± 2.0 ** | 43.0 ± 1.0 *** | 58.0 ± 1.0 *** | |
| 100 | 12.1 ± 3.0 * | 21.5 ± 0.5 ** | 43.5 ± 3.5 *** | 45.0 ± 1.0 *** | 63.5 ± 0.5 *** | |
| EtOAc-fr | 25 | 3.5 ± 0.5 * | 9.5 ± 0.5 * | 18.0 ± 3.0 ** | 18.5 ± 1.5 ** | 39.5 ± 0.5 *** |
| 50 | 10.5 ± 0.5 * | 16.0 ± 1.0 ** | 29.5 ± 0.5 *** | 30.5 ± 2.5 *** | 44.5 ± 3.5 *** | |
| 100 | 9.5 ± 0.5 * | 22.5 ± 1.5 ** | 30.5 ± 0.5 *** | 33.0 ± 2.0 *** | 47.5 ± 0.5 *** | |
| BuOH-fr | 25 | 12.9 ± 3.7 * | 14.3 ± 5.5 * | 35.2 ± 1.0 *** | 58.0 ± 1.5 *** | 61.0 ± 3.4 *** |
| 50 | 18.8 ± 3.4 *** | 46.6 ± 4.9 *** | 68.6 ± 2.3 *** | 66.0 ± 1.7 *** | 69.0 ± 4.3 *** | |
| 100 | 25.6 ± 1.7 *** | 47.6 ± 2.1 *** | 68.6 ± 1.4 *** | 70.3 ± 2.6 *** | 72.6 ± 3.9 *** |
| Samples | IC50 ± SD (µg/mL) |
|---|---|
| MeOH-ext | 30.8 ± 7.0 |
| Hex-fr | 107.1 ± 38.4 |
| Chl-fr | 81.6 ± 24.6 |
| EtOAc-fr | 114.8 ± 2.3 |
| BuOH-fr | 16.5 ± 7.5 |
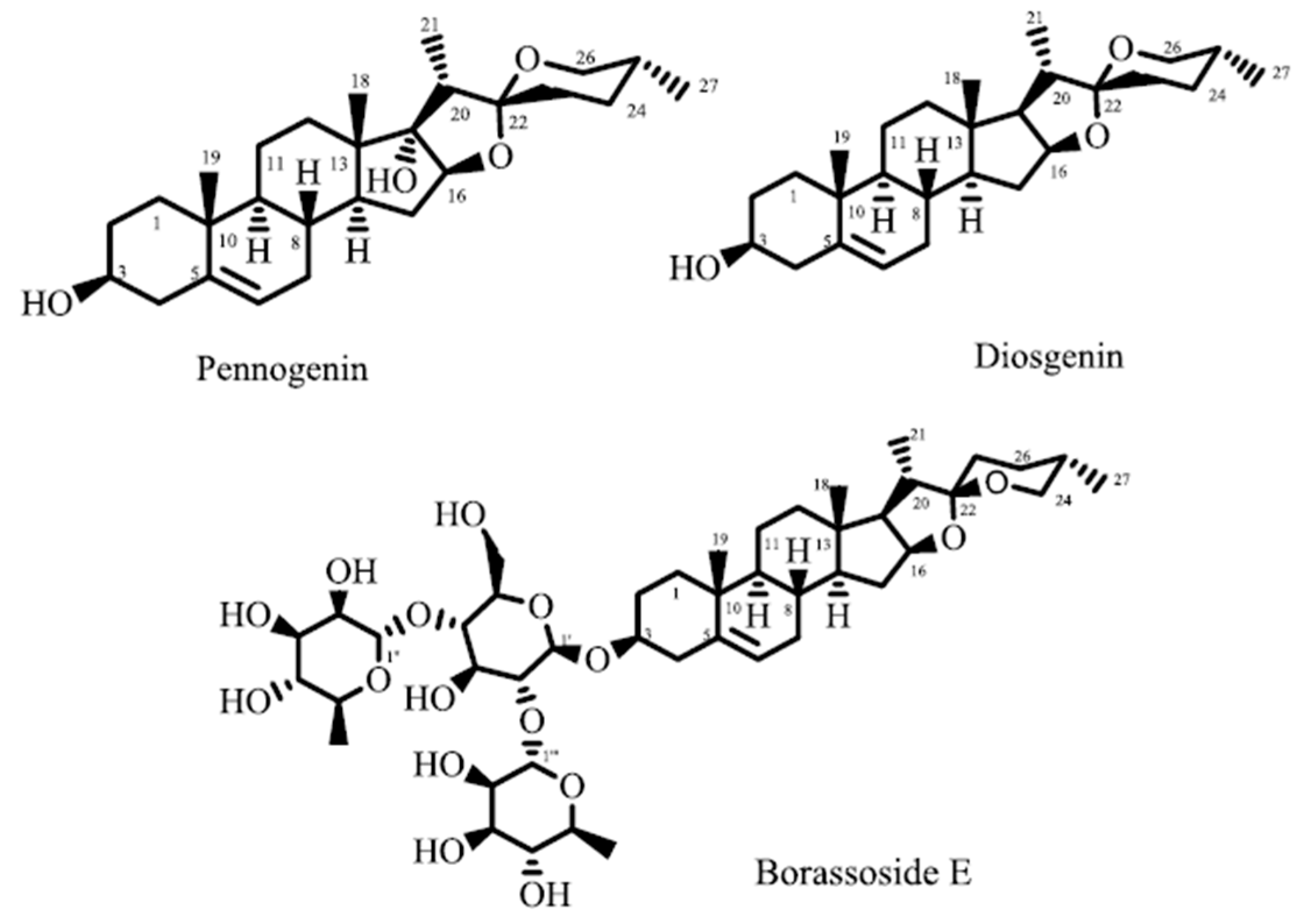
| Pennogenin | 5.0 ± 0.8 |
| Borassoside E | 31.5 ± 6.6 |
| Diosgenin | 53.2 ± 2.7 |
| Ibuprofen (Positive control) | 11.2 ± 1.9 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ur Rahman, S.; Adhikari, A.; Ismail, M.; Raza Shah, M.; Khurram, M.; Shahid, M.; Ali, F.; Haseeb, A.; Akbar, F.; Iriti, M. Beneficial Effects of Trillium govanianum Rhizomes in Pain and Inflammation. Molecules 2016, 21, 1095. https://doi.org/10.3390/molecules21081095
Ur Rahman S, Adhikari A, Ismail M, Raza Shah M, Khurram M, Shahid M, Ali F, Haseeb A, Akbar F, Iriti M. Beneficial Effects of Trillium govanianum Rhizomes in Pain and Inflammation. Molecules. 2016; 21(8):1095. https://doi.org/10.3390/molecules21081095
Chicago/Turabian StyleUr Rahman, Shafiq, Achyut Adhikari, Muhammad Ismail, Muhammad Raza Shah, Muhammad Khurram, Muhammad Shahid, Farman Ali, Abdul Haseeb, Fazal Akbar, and Marcello Iriti. 2016. "Beneficial Effects of Trillium govanianum Rhizomes in Pain and Inflammation" Molecules 21, no. 8: 1095. https://doi.org/10.3390/molecules21081095
APA StyleUr Rahman, S., Adhikari, A., Ismail, M., Raza Shah, M., Khurram, M., Shahid, M., Ali, F., Haseeb, A., Akbar, F., & Iriti, M. (2016). Beneficial Effects of Trillium govanianum Rhizomes in Pain and Inflammation. Molecules, 21(8), 1095. https://doi.org/10.3390/molecules21081095









