Anti-Diabetic Activity and Metabolic Changes Induced by Andrographis paniculata Plant Extract in Obese Diabetic Rats
Abstract
:1. Introduction
2. Results
2.1. Amino Acid Profile of A. paniculata Leaf and Extract
2.2. Quantitative Analysis of Selected Chemical Markers via qNMR
2.3. Short–Term Toxicity Test on A. paniculata Leaf Water Extract
2.4. Oral Glucose Tolerance Test (OGTT) on A. paniculata Leaf Water Extract
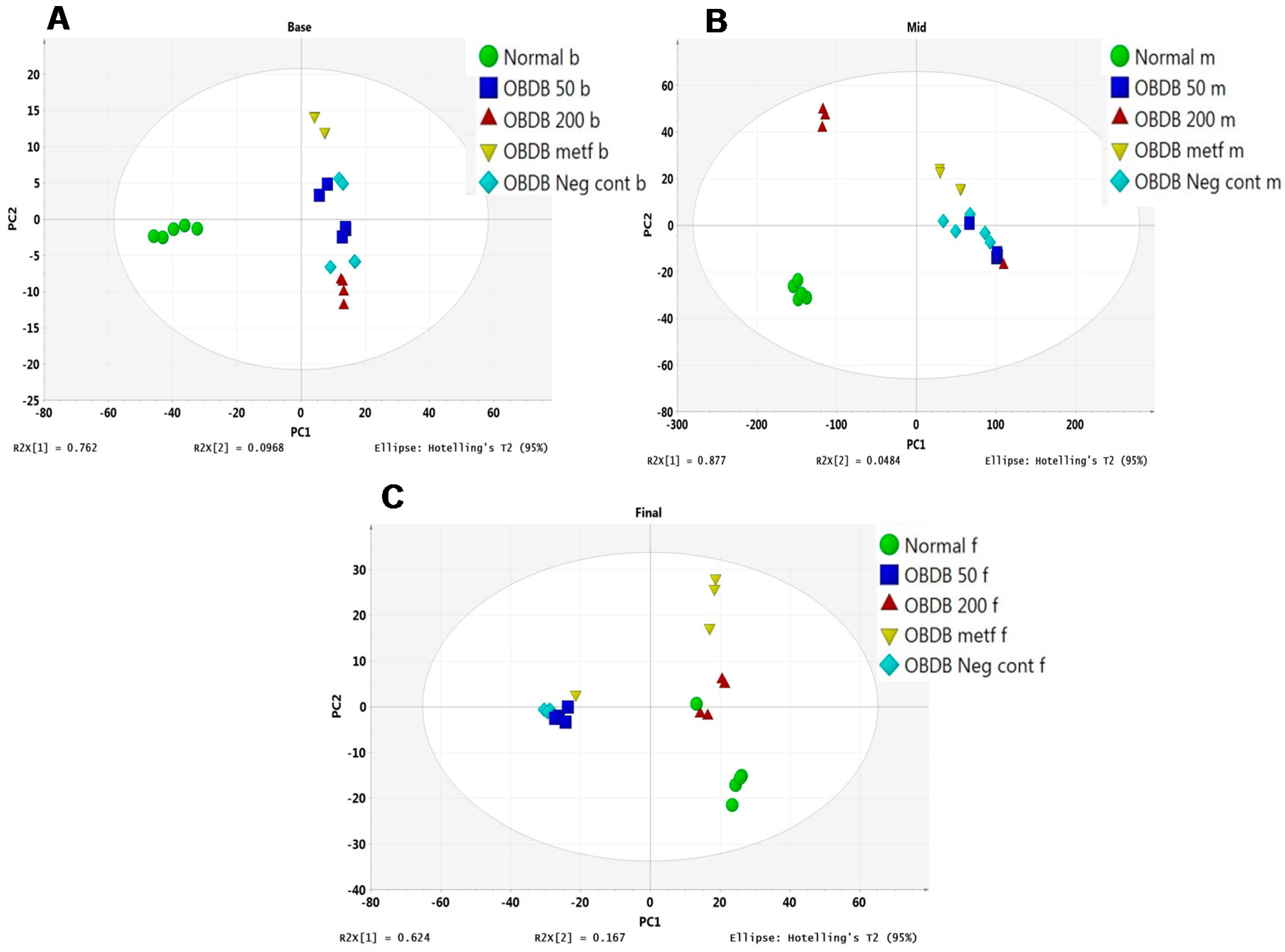
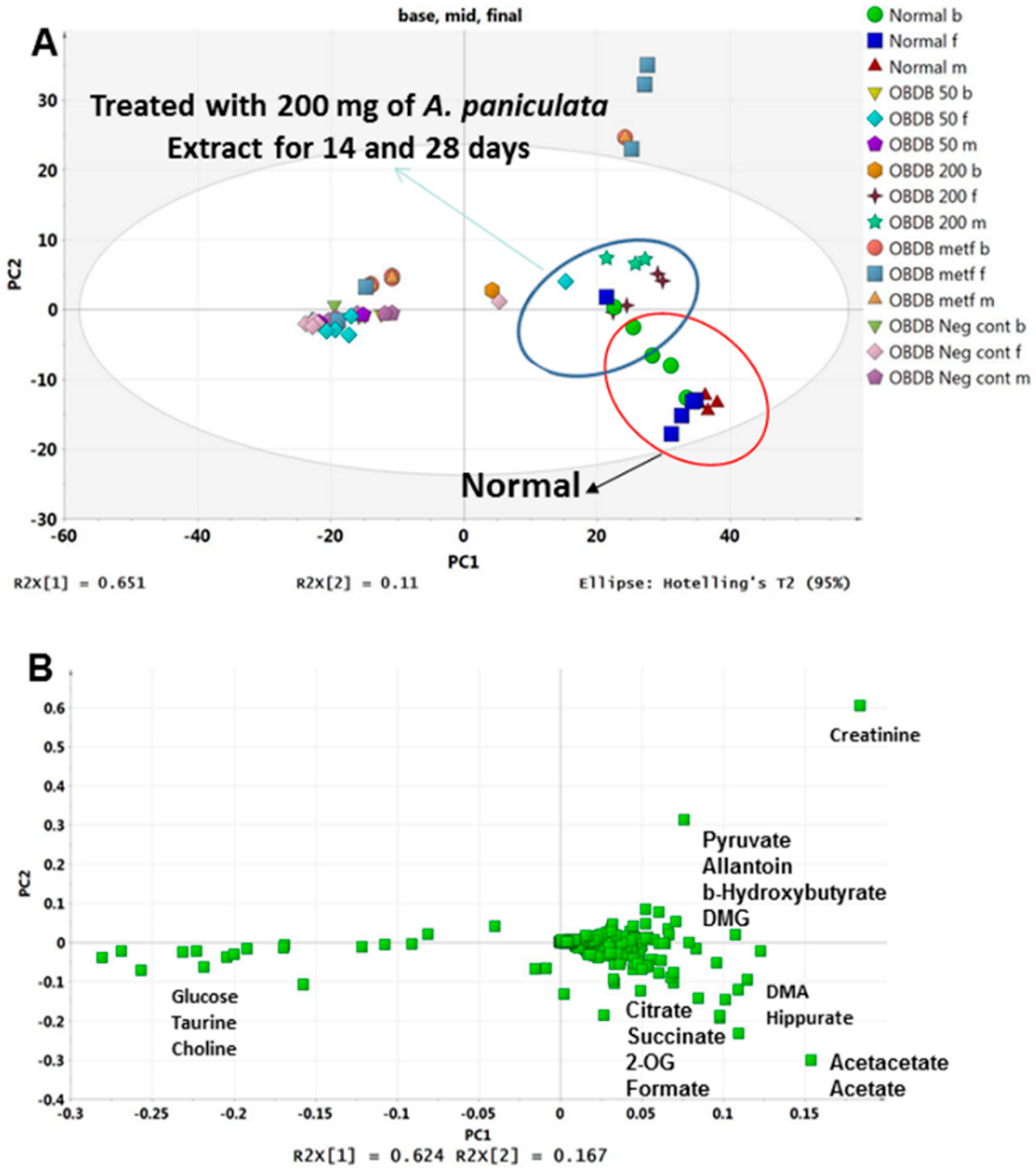
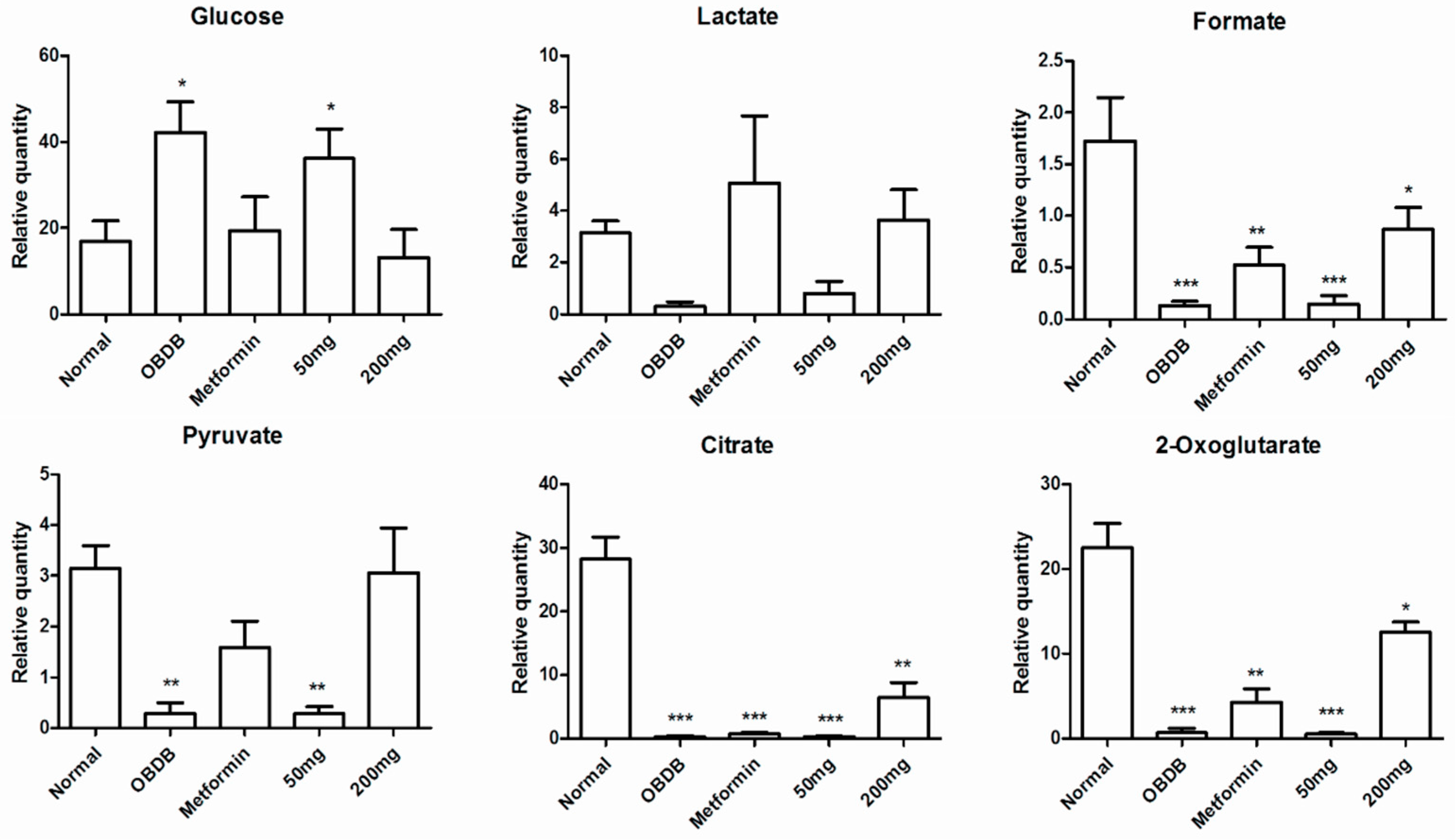
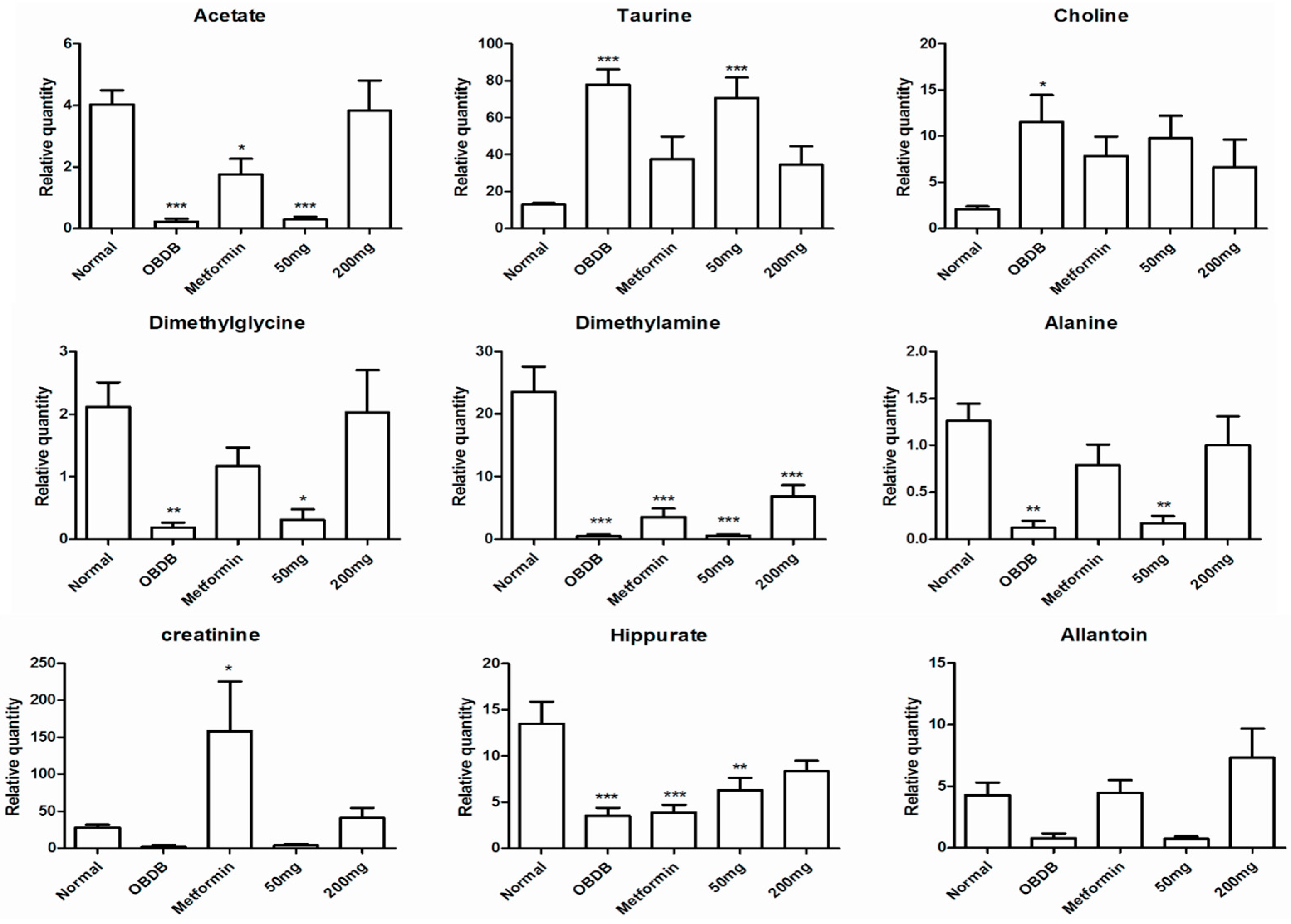
2.5. Metabolite Analysis of Urine Samples
3. Discussion
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Plant Material
4.2.1. Large-Scale Planting of A. paniculata (Harapan) for Metabolomics Analysis of Biofluids
4.2.2. Processing of Plant Material for Animal Biofluids Study
4.2.3. Preparation of A. paniculata Water Extract for Animal Study
4.3. Quantitative NMR Analysis of Selected Chemical Markers in A. paniculata (Harapan) Water Extract
4.4. Determination of Total Nitrogen Content (Kjeldahl Method) in A. paniculata (Harapan)
4.5. Amino Acid Analysis of A. paniculata (Harapan) Water Extract
4.6. Animals
4.7. Toxicity Test
4.8. Oral Glucose Tolerance Test
4.9. Animal Disease Models (Obese and Obese–Diabetic)
4.9.1. Administration of A. paniculata Water Extract to Experimental Animals
4.9.2. Extract Preparation and Dosage Calculation for Animal Study
4.10. 1H-NMR Metabolomics Analysis of Biofluid Samples
NMR Measurement and Data Processing
5. Conclusions
Supplementary Materials
Author Contributions
Conflicts of Interest
References
- Rang, H.P.; Dale, M.M.; Ritter, J.M.; Moore, P.K. Pharmacology, 5th ed.; Churchill Livingstone: London, UK, 2003. [Google Scholar]
- Wild, S.; Roglic, G.; Green, A.; Sicree, R.; King, H. Global prevalence of diabetes: Estimates for the year 2000 and projections for 2030. Diabetes Care 2004, 27, 1047–1053. [Google Scholar] [CrossRef] [PubMed]
- Shenfield, G. Metformin: Myths, misunderstandings and lessons from history. Aust. Prescr. 2013, 36, 38–39. [Google Scholar] [CrossRef]
- Okhuarobo, A.; Falodun, J.E.; Erharuyi, O.; Imieje, V.; Falodun, A.; Langer, P. Harnessing the medicinal properties of Andrographis paniculata for diseases and beyond: A review of its phytochemistry and pharmacology. Asian Pac. J. Trop. Dis. 2014, 4, 213–222. [Google Scholar] [CrossRef]
- Jarukamjorn, K.; Nemoto, N. Pharmacological Aspects of Andrographis paniculata on health and its major diterpenoid constituent Andrographolide. J. Health Sci. 2008, 54, 340–381. [Google Scholar] [CrossRef]
- Niranjan, A.; Tewari, S.K.; Lehri, A. Biological activities of Kalmegh (Andrographis paniculata Nees) and its active principles—A review. Indian J. Nat. Prod. Resour. 2010, 1, 125–135. [Google Scholar]
- Akbar, S. Andrographis paniculata: A Review of Pharmacological Activities and Clinical Effects. Altern. Med. Rev. 2001, 16, 66–77. [Google Scholar]
- Subramanian, R.; Asmawi, M.Z.; Sadikun, A. A bitter plant with a sweet future? A comprehensive review of an oriental medicinal plant: Andrographis paniculata. Phytochem. Rev. 2012, 11, 39–75. [Google Scholar] [CrossRef]
- Hossain, M.S.; Urbi, Z.; Sule, A.; Rahman, K.M.H. Andrographis paniculata (Burm. f.) Wall. ex Nees: A Review of Ethnobotany, Phytochemistry, and Pharmacology. Sci. World J. 2014. [Google Scholar] [CrossRef] [PubMed]
- Jada, S.R.; Matthews, C.; Saad, M.S.; Hamzah, A.S.; Lajis, N.H.; Stevens, M.F.; Stanslas, J. Benzylidene derivatives of andrographolide inhibit growth of breast and colon cancer cells in vitro by inducing G1 arrest and apoptosis. Br. J. Pharmacol. 2008, 155, 641–654. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.C.; Chan, T.K.; Ng, D.S.W.; Sagineedu, S.R.; Stanslas, J.; Wong, F.W.S. Andrographolide and its analogues: Versatile bioactive molecules for combating inflammation and cancer. Clin. Exp. Pharmacol. Physiol. 2012, 39, 300–310. [Google Scholar] [CrossRef] [PubMed]
- Dai, G.F.; Xu, H.W.; Wang, J.F.; Liu, F.W.; Liu, H.M. Studies on the novel á-glucosidase inhibitory activity and structure-activity relationships for andrographolide analogue. Bioorg. Med. Chem. Lett. 2006, 16, 2710–2713. [Google Scholar] [CrossRef] [PubMed]
- Fiehn, O. Metabolomics-the link between genotypes and phenotypes. Plant Mol. Biol. 2002, 48, 155–171. [Google Scholar] [CrossRef] [PubMed]
- Kinross, J.M.; Holmes, E.; Darzi, A.W.; Nicholson, J.K. Metabolic phenotyping for monitoring surgical patients. Lancet 2011, 377, 1817–1819. [Google Scholar] [CrossRef]
- Zhang, A.; Sun, H.; Wang, X. Saliva metabolomics opens door to biomarker discovery, disease diagnosis, and treatment. Appl. Biochem. Biotechnol. 2012, 168, 1718–1727. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.; Sun, H.; Yan, G.; Wang, P.; Wang, X. Metabolomics for Biomarker Discovery: Moving to the Clinic. BioMed Res. Int. 2015. [Google Scholar] [CrossRef] [PubMed]
- Emwas, A.H.; Luchinat, C.; Turano, P.; Tenori, L.; Roy, R.; Salek, R.M.; Ryan, D.; Merzaban, J.S.; Kaddurah-Daouk, R.; Zeri, A.C.; et al. Tandardizing the experimental conditions for using urine in NMR-based metabolomic studies with a particular focus on diagnostic studies: A review. Metabolomics 2015, 11, 872–894. [Google Scholar] [CrossRef] [PubMed]
- Reo, N.V. NMR-based metabolomics. Drug Chem. Toxicol. 2002, 25, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Jankevics, A.; Liepinsh, E.; Liepinsh, E.; Vilskersts, R.; Grinberga, S.; Pugovics, O.; Dambrova, M. Metabolomic studies of experimental diabetic urine samples by 1H NMR spectroscopy and LC/MS method. Chemometr. Intell. Lab. 2009, 97, 11–17. [Google Scholar] [CrossRef]
- Salek, R.M.; Maguire, M.L.; Bentley, E.; Rubtsov, D.V.; Hough, T.; Cheeseman, M.; Nunez, D.; Sweatman, B.C.; Haselden, J.N.; Cox, R.D.; et al. A metabolomic comparison of urinary changes in type 2 diabetes in mouse, rat, and human. Physiol. Genom. 2007, 29, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Gowda, G.A.N.; Asiago, V.; Shanaiah, N.; Barbas, C.; Raftery, D. Correlative and quantitative 1H NMR-based metabolomics reveals specific metabolic pathway disturbances in diabetic rats. Anal. Biochem. 2008, 383, 76–84. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Gao, H.; Zhao, Y.; Lin, D. Metabonomic analysis of the therapeutic effect of Zhibai Dihuang Pill in treatment of streptozotocin-induced diabetic nephropathy. J. Ethnopharmacol. 2012, 142, 647–656. [Google Scholar] [CrossRef] [PubMed]
- Villareal, D.T.; Apovian, C.M.; Kushner, R.F.; Klein, S. Obesity in older adults: Technical review and position statement of the American Society for Nutrition and NAASO, The Obesity Society. Am. J. Clin. Nutr. 2005, 82, 923–934. [Google Scholar] [CrossRef] [PubMed]
- Hariri, N.; Thibault, L. High-fat diet-induced obesity in animal models. Nutr. Res. Rev. 2010, 23, 270–299. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, K.; Viswanad, B.; Asrat, L.; Kaul, C.L.; Ramarao, P. Combination of high-fat diet-fed and low-dose streptozotocin-treated rat: A model for type 2 diabetes and pharmacological screening. Pharmacol. Res. 2005, 52, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Duggan, G.E.; Hittel, D.S.; Hughey, C.C.; Weljie, A.; Vogel, H.J.; Shearer, J. Differentiating short- and long-term effects of diet in the obese mouse using 1H-nuclear magnetic resonance metabolomics. Diabetes Obes. Metab. 2011, 13, 859–862. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Yang, S.O.; Kim, H.S.; Kim, Y.; Park, T.; Choi, H.K. 1H-nuclear magnetic resonance spectroscopy-based metabolic assessment in a rat model of obesity induced by a high-fat diet. Anal. Bioanal. Chem. 2009, 395, 1117–1124. [Google Scholar] [CrossRef] [PubMed]
- Serkova, N.J.; Jackman, M.; Brown, J.L.; Liu, T.; Hirose, R.; Roberts, J.P.; Maher, J.J.; Niemann, C.U. Metabolic profiling of livers and blood from obese Zucker rats. J. Hepatol. 2006, 44, 956–962. [Google Scholar] [CrossRef] [PubMed]
- Waldram, A.; Holmes, E.; Wang, Y.; Rantalainen, M.; Wilson, I.D.; Tuohy, K.M. Top-down systems biology modeling of host metabotype-microbiome associations in obese rodents. J. Proteome Res. 2009, 8, 2361–2375. [Google Scholar] [CrossRef] [PubMed]
- Jansson, P.A.; Larsson, A.; Smith, U.; Lonnroth, P. Lactate release from the subcutaneous tissue in lean and obese men. J. Clin. Investig. 1994, 93, 240–246. [Google Scholar] [CrossRef] [PubMed]
- Newgard, C.B.; An, J.; Bain, J.R.; Muehlbauer, M.J.; Stevens, R.D.; Lien, L.F. A branched-chain amino acid-related metabolic signature that differentiates obese and lean humans and contributes to insulin resistance. Cell Metab. 2009, 9, 311–326. [Google Scholar] [CrossRef] [PubMed]
- Amaral, S.; Moreno, A.J.; Santos, M.S.; Seica, R.; Ramalho-Santos, J. Effects of hyperglycemia on sperm and testicular cells of Goto-Kakizaki and streptozotocin-treated rat models for diabetes. Theriogenology 2006, 66, 2056–2067. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.C.; Zhang, X.D.; Liao, S.X.; Gao, H.C.; Wang, H.Y.; Lin, D.H. A metabonomic comparison of urinary changes in Zucker and GK rats. J. Biomed. Biotechnol. 2010. [Google Scholar] [CrossRef] [PubMed]
- Deng, W.J.; Nie, S.; Dai, J.; Wu, J.R.; Zeng, R. Proteome, phosphoproteome, and hydroxyproteome of liver mitochondria in diabetic rats at early pathogenic stages. Mol. Cell. Proteome 2010, 9, 100–116. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Xie, Z.; Lin, J.; Song, H.; Wang, Q.; Wang, K.; Su, M.; Qiu, Y.; Zhao, T.; Song, K.; et al. Transcriptomic and metabonomic profiling of obesity-prone and obesity-resistant rats under high fat diet. J. Proteome Res. 2008, 7, 4775–4783. [Google Scholar] [CrossRef] [PubMed]
- Simpson, D.P. Citrate excretion: A window on renal metabolism. Am. J. Physiol. 1983, 244, F223–F234. [Google Scholar] [PubMed]
- Souto, G.; Donapetry, C.; Calvino, J.; Adeva, M.M. Metabolic acidosis-induced insulin resistance and cardiovascular risk. Metab. Syndr. Relat. Disord. 2011, 9, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Cupisti, A.; Meola, M.; D’Alessandro, C.; Bernabini, G.; Pasquali, E.; Carpi, A.; Barsotti, G. Insulin resistance and low urinary citrate excretion in calcium stone formers. Biomed. Pharmacother. 2007, 61, 86–90. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Liu, X.; Xie, L.; Gao, H.; Lin, D. 1H NMR-based metabonomic analysis of metabolic changes in streptozotocin-induced diabetic rats. Anal. Sci. 2010, 26, 1277–1282. [Google Scholar] [CrossRef] [PubMed]
- Waters, N.J.; Waterfield, C.J.; Farrant, R.D.; Holmes, E.; Nicholson, J.K. Metabonomic deconvolution of embedded toxicity: Application to thioacetamide hepato- and nephrotoxicity. Chem. Res. Toxicol. 2005, 18, 639–654. [Google Scholar] [CrossRef] [PubMed]
- Belfiore, F.; Iannello, S.; Campione, R.; Volpicelli, G. Metabolic effects of high glucose concentrations: Inhibition of hepatic pyruvate kinase. Diabetes Res. 1989, 10, 183–186. [Google Scholar] [PubMed]
- Vats, V.; Yadav, S.P.; Grover, J.K. Effect of T. foenumgraecum on glycogen content of tissues and the key enzymes of carbohydrate metabolism. J. Ethnopharmacol. 2003, 85, 237–242. [Google Scholar] [CrossRef]
- Walker, J.B. Metabolic control of creatine biosynthesis. II. Restoration of transamidinase activity following creatine repression. J. Biol. Chem. 1961, 236, 493–498. [Google Scholar] [PubMed]
- Konishi, F. The Relationship Of Urinary 17-Hydroxycorticosteroids to Creatinine in Obesity. Metabolism 1964, 13, 847–851. [Google Scholar] [CrossRef]
- Schirra, H.J.; Anderson, C.G.; Wilson, W.J.; Kerr, L.; Craik, D.J.; Waters, M.J.; Lichanska, A.M. Altered metabolism of growth hormone receptor mutant mice: A combined NMR metabonomics and microarray study. PLoS ONE 2008, 3, e2764. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.E.; Onesti, G.; Ramirez, O.; Brest, A.N.; Swartz, C. Creatinine clearance in renal disease. A reappraisal. Br. Med. J. 1969, 4, 11–14. [Google Scholar] [CrossRef] [PubMed]
- Sugino, N. Evaluation of renal function in chronic renal disease with special reference to creatinine. Nihon Jinzo Gakkai Shi 1990, 32, 745–750. [Google Scholar] [PubMed]
- Kumar, A.; Kapoor, S.; Gupta, R.C. Comparison of Urinary Protein: Creatinine Index and Dipsticks for Detection of Microproteinuria in Diabetes Mellitus Patients. J. Clin. Diagn. Res. 2013, 7, 622–626. [Google Scholar] [CrossRef] [PubMed]
- Hansen, S.H. The role of taurine in diabetes and the development of diabetic complications. Diabetes Metab. Res. Rev. 2001, 17, 330–346. [Google Scholar] [CrossRef] [PubMed]
- Nakaya, Y.; Minami, A.; Harada, N.; Sakamoto, S.; Niwa, Y.; Ohnaka, M. Taurine improves insulin sensitivity in the Otsuka Long-Evans Tokushima Fatty rat, a model of spontaneous type 2 diabetes. Am. J. Clin. Nutr. 2000, 71, 54–58. [Google Scholar] [PubMed]
- Klein, M.; Dorn, C.; Saugspier, M.; Hellerbrand, C.; Oefner, P.; Gronwald, W. Discrimination of steatosis and NASH in mice using nuclear magnetic resonance spectroscopy. Metabolomics 2011, 7, 237–246. [Google Scholar] [CrossRef]
- Rubio-Aliaga, I.; Roos, B.; Sailer, M.; McLoughlin, G.A.; Boekschoten, M.V.; van Erk, M.; Bachmair, E.M.; Van Schothorst, E.M.; Keijer, J.; Coort, S.L.; et al. Alterations in hepatic one-carbon metabolism and related pathways following a high-fat dietary intervention. Physiol. Genom. 2011, 43, 408–416. [Google Scholar] [CrossRef] [PubMed]
- Martensson, J.; Hermansson, G. Sulfur amino acid metabolism in juvenile-onset nonketotic and ketotic diabetic patients. Metabolism 1984, 33, 425–428. [Google Scholar] [CrossRef]
- Szabo, A.; Kenesei, E.; Korner, A.; Miltenyi, M.; Szucs, L.; Nagy, I. Changes in plasma and urinary amino acid levels during diabetic ketoacidosis in children. Diabetes Res. Clin. Pract. 1991, 12, 91–97. [Google Scholar] [CrossRef]
- Merheb, M.; Daher, R.T.; Nasrallah, M.; Sabra, R.; Ziyadeh, F.N.; Barada, K. Taurine intestinal absorption and renal excretion test in diabetic patients: A pilot study. Diabetes Care 2007, 30, 2652–2654. [Google Scholar] [CrossRef] [PubMed]
- McManus, M.L.; Churchwell, K.B.; Strange, K. Regulation of cell volume in health and disease. N. Engl. J. Med. 1995, 333, 1260–1266. [Google Scholar] [PubMed]
- Wu, X.W.; Lee, C.C.; Muzny, D.M.; Caskey, C.T. Urate oxidase: Primary structure and evolutionary implications. Proc. Natl. Acad. Sci. USA 1989, 86, 9412–9416. [Google Scholar] [CrossRef] [PubMed]
- Tsushima, Y.; Nishizawa, H.; Tochino, Y.; Nakatsuji, H.; Sekimoto, R.; Nagao, H. Uric acid secretion from adipose tissue and its increase in obesity. J. Biol. Chem. 2013, 288, 27138–27149. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Wang, J.; Kong, L. Quantitative analysis of four major diterpenoids in Andrographis paniculata by 1H NMR and its application for quality control of commercial preparations. J. Pharm. Biomed. 2012, 70, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Hainida, E.; Amin, I.; Normah, H.; Mohd-Esa, N. Nutritional and amino acid contents of differently treated Roselle (Hibiscus sabdariffa L.) seeds. Food Chem. 2008, 111, 906–911. [Google Scholar] [CrossRef]
- Jalil, A.M.; Ismail, A.; Pei, C.P.; Hamid, M.; Kamaruddin, S.H.S. Effects of cocoa extract on glucometabolism, oxidative stress, and antioxidant enzymes in obese-diabetic (Ob-db) rats. J. Agric. Food Chem. 2008, 56, 7877–7884. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; He, T.; Lu, Q.; Shang, J.; Sun, H.; Zhang, L. Asiatic acid preserves beta cell mass and mitigates hyperglycemia in streptozocin-induced diabetic rats. Diabetes Metab. Res. Rev. 2010, 26, 448–454. [Google Scholar] [CrossRef] [PubMed]
- Maulidiani.; Abas, F.; Khatib, A.; Perumal, V.; Suppaiah, V.; Ismail, A.; Hamid, M.; Shaari, K.; Lajis, N.H. Metabolic alterations in obese diabetes rats upon treatment with Centella asiatica extract. J. Ethnopharmacol. 2016, 180, 60–69. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Not Available.



| Pathway | Metabolite | Chemical Shift (ppm) |
|---|---|---|
| Glucose metabolism | Glucose | 4.64 (d, J = 8.0 Hz), 5.24 (d, J = 3.5 Hz) |
| Pyruvate | 2.32 (s) | |
| Lactate | 1.32 (d, J = 7.5 Hz) | |
| Formate | 8.40 (s) | |
| TCA Cycle | Citrate | 2.56 (d, J = 16.0 Hz), 2.68 (d, J = 16.0 Hz) |
| Fumarate | 6.52 (s) | |
| Succinate | 2.40 (s) | |
| 2-Oxoglutarate | 3.00 (t, J = 7.0) | |
| Fatty acid metabolism | β-Hydroxy butyrate | 0.96 (t, J = 7.5 Hz) |
| Acetoacetate | 2.28 (s) | |
| Acetone | 2.24 (s) | |
| Acetate | 1.96 (s) | |
| Choline metabolsim | Choline | 3.20 (s) |
| Dimethylglycine | 2.92 (s) | |
| Amino acid metabolism | Malonate | 3.12 (s) |
| Taurine | 3.44 (t, J = 6.5 Hz); 3.27 (t, J = 6.5 Hz) | |
| Alanine | 1.48 (d, J = 7.5 Hz) | |
| Hippurate | 7.52 (t, J = 7.5 Hz); 3.98 (d, J = 7.5 Hz) | |
| Creatine metabolism | Creatine | 3.02 (s); 3.92 (s) |
| Creatinine | 4.04 (s), 3.04 (s) | |
| Urea | 5.8 (br s) | |
| TMA metabolsim | Dimethylamine | 2.72 (s) |
| Trimethylamine | 2.88 (s) | |
| Purine metabolsim | Allantoin | 5.4 (s), 6.02 (s) |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akhtar, M.T.; Bin Mohd Sarib, M.S.; Ismail, I.S.; Abas, F.; Ismail, A.; Lajis, N.H.; Shaari, K. Anti-Diabetic Activity and Metabolic Changes Induced by Andrographis paniculata Plant Extract in Obese Diabetic Rats. Molecules 2016, 21, 1026. https://doi.org/10.3390/molecules21081026
Akhtar MT, Bin Mohd Sarib MS, Ismail IS, Abas F, Ismail A, Lajis NH, Shaari K. Anti-Diabetic Activity and Metabolic Changes Induced by Andrographis paniculata Plant Extract in Obese Diabetic Rats. Molecules. 2016; 21(8):1026. https://doi.org/10.3390/molecules21081026
Chicago/Turabian StyleAkhtar, Muhammad Tayyab, Mohamad Syakir Bin Mohd Sarib, Intan Safinar Ismail, Faridah Abas, Amin Ismail, Nordin Hj Lajis, and Khozirah Shaari. 2016. "Anti-Diabetic Activity and Metabolic Changes Induced by Andrographis paniculata Plant Extract in Obese Diabetic Rats" Molecules 21, no. 8: 1026. https://doi.org/10.3390/molecules21081026
APA StyleAkhtar, M. T., Bin Mohd Sarib, M. S., Ismail, I. S., Abas, F., Ismail, A., Lajis, N. H., & Shaari, K. (2016). Anti-Diabetic Activity and Metabolic Changes Induced by Andrographis paniculata Plant Extract in Obese Diabetic Rats. Molecules, 21(8), 1026. https://doi.org/10.3390/molecules21081026








