Boron Nitride Nanotubes: Recent Advances in Their Synthesis, Functionalization, and Applications
Abstract
:1. Introduction
2. Synthesis of BNNTs
2.1. Thermal Annealing and Chemical Vapor Deposition
2.2. Laser Vaporization with Pressurized Vapor/Condenser Method
2.3. Large Scale Synthesis by Inductive Thermal Plasma Method
2.4. Summary Remarks on Controlled Synthesis and Mass Production of BNNTs
- In contrast to the CVD method, all the plasma-based techniques (both laser plasma and inductive plasma) utilize very high synthesis temperatures (4000 K to 8000 K). These approaches led to the vaporization of B or BN solids into molten B droplets that induce the formation of BNNTs.
- High pressure environment (>1 atm) of nitrogen effectively facilitates and increases the formation of BNNTs from B droplets, due to higher collision rate of N2 or N radical with B droplets.
- A large temperature gradient (i.e., the high cooling rate) by drastic velocity profile or spatial profile (e.g., a solid condenser or quench rate) seems to enhance the formation small B droplets and hence further promotes long and small-diameter BNNTs formation.
- Apparently, the drawbacks of these plasma-based techniques are as follows:
- (a)
- Excessive high synthesis temperatures have led to serious contamination and clustering of BNNTs. This will make purification and dispersion of the fibril-like BNNTs challenging.
- (b)
- The plasma-based approaches required a customized chamber to generate high synthesis temperature, high pressure and rapid cooling conditions for mass production.
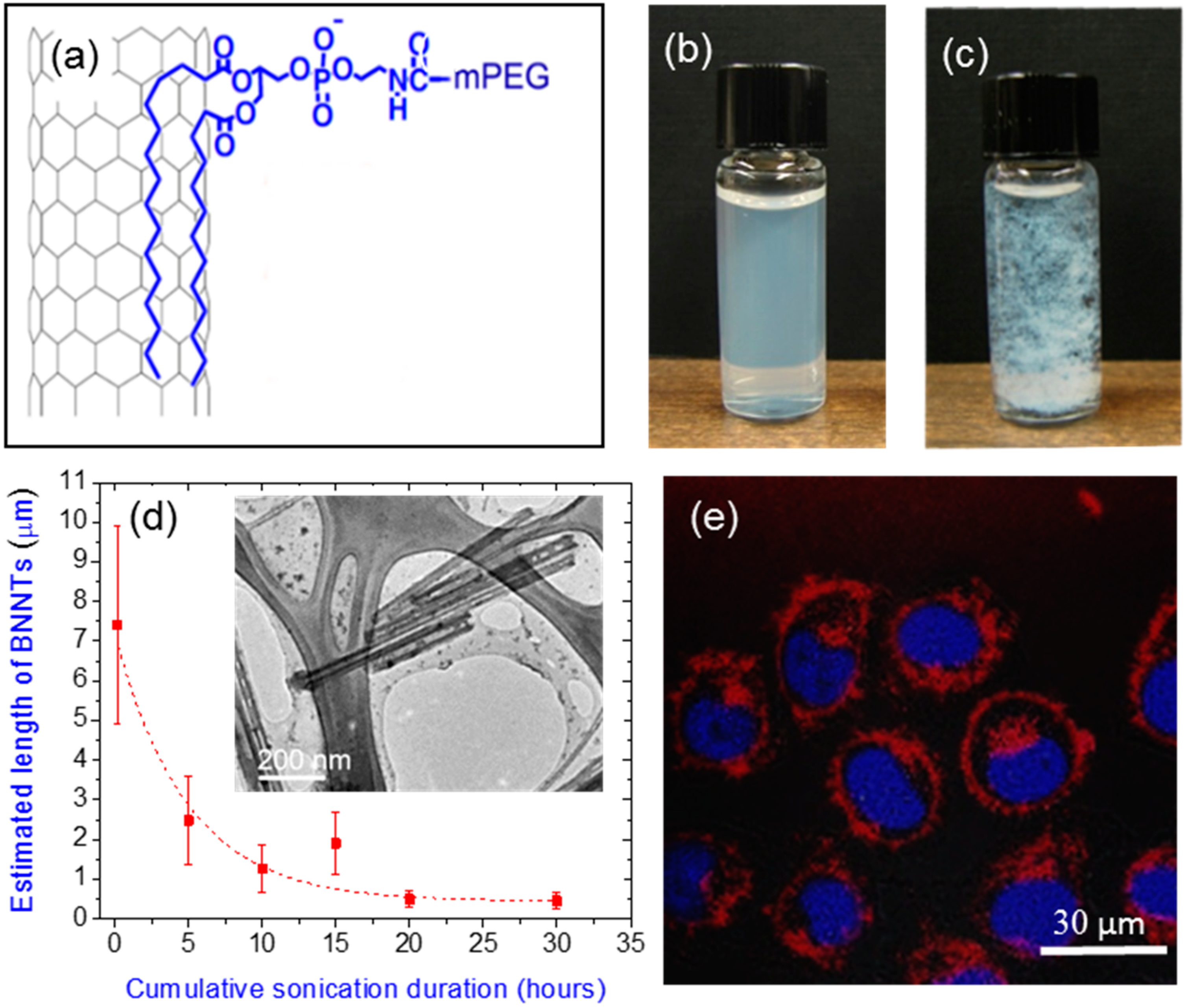
2.5. Purification, Dispersion and Functionalization of BNNTs
3. Non-Covalent Functionalization
4. Covalent Functionalization
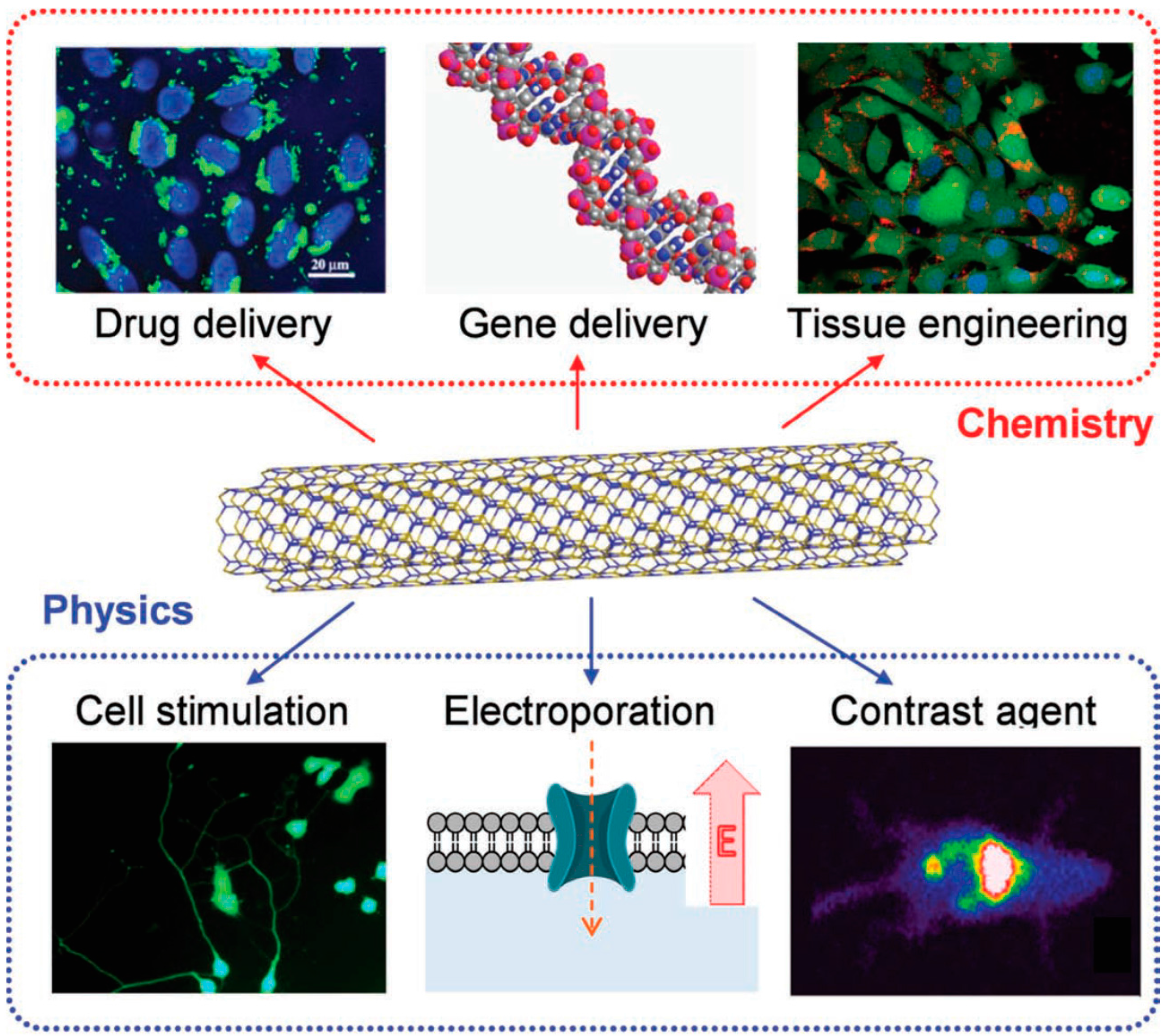
5. Potential Applications
5.1. Nano-Fillers for Composites
Remarks on Composite Application of BNNTs
5.2. Biomedical Applications
Remarks on Cytotoxicity of BNNTs in Biomedical Applications
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Rubio, A.; Corkill, J.L.; Cohen, M.L. Theory of graphitic boron nitride nanotubes. Phys. Rev. B 1994, 49. [Google Scholar] [CrossRef]
- Blase, X.; Rubio, A.; Louie, S.; Cohen, M. Stability and band gap constancy of boron nitride nanotubes. EPL-Europhys. Lett. 1994, 28. [Google Scholar] [CrossRef]
- Chopra, N.G.; Luyken, R.; Cherrey, K.; Crespi, V.H.; Cohen, M.L.; Louie, S.G.; Zettl, A. Boron nitride nanotubes. Science 1995, 269, 966–967. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.H.; Xie, M.; Kayastha, V.; Wang, J.; Yap, Y.K. Patterned growth of boron nitride nanotubes by catalytic chemical vapor deposition. Chem. Mater. 2010, 22, 1782–1787. [Google Scholar] [CrossRef]
- Chang, C.; Fennimore, A.; Afanasiev, A.; Okawa, D.; Ikuno, T.; Garcia, H.; Li, D.; Majumdar, A.; Zettl, A. Isotope effect on the thermal conductivity of boron nitride nanotubes. Phys. Rev. Lett. 2006, 97. [Google Scholar] [CrossRef] [PubMed]
- Chopra, N.G.; Zettl, A. Measurement of the elastic modulus of a multi-wall boron nitride nanotube. Solid State Commun. 1998, 105, 297–300. [Google Scholar] [CrossRef]
- Lee, C.H.; Drelich, J.; Yap, Y.K. Superhydrophobicity of boron nitride nanotubes grown on silicon substrates. Langmuir 2009, 25, 4853–4860. [Google Scholar] [CrossRef] [PubMed]
- Boinovich, L.B.; Emelyanenko, A.M.; Pashinin, A.S.; Lee, C.H.; Drelich, J.; Yap, Y.K. Origins of Thermodynamically Stable Superhydrophobicity of Boron Nitride Nanotubes Coatings. Langmuir 2012, 28, 1206–1216. [Google Scholar] [CrossRef] [PubMed]
- Tanur, A.E.; Wang, J.; Reddy, A.L.M.; Lamont, D.N.; Yap, Y.K.; Walker, G.C. Diameter-Dependent Bending Modulus of Individual Multiwall Boron Nitride Nanotubes. J. Phys. Chem. B 2013, 117, 4618–4625. [Google Scholar] [CrossRef] [PubMed]
- Yitao, D.; Wanlin, G.; Zhuhua, Z.; Bin, Z.; Chun, T. Electric-field-induced deformation in boron nitride nanotubes. J. Phys. D Appl. Phys. 2009, 42. [Google Scholar] [CrossRef]
- Kang, J.H.; Sauti, G.; Park, C.; Yamakov, V.I.; Wise, K.E.; Lowther, S.E.; Fay, C.C.; Thibeault, S.A.; Bryant, R.G. Multifunctional Electroactive Nanocomposites Based on Piezoelectric Boron Nitride Nanotubes. ACS Nano 2015, 9, 11942–11950. [Google Scholar] [CrossRef] [PubMed]
- Nakhmanson, S.; Calzolari, A.; Meunier, V.; Bernholc, J.; Nardelli, M.B. Spontaneous polarization and piezoelectricity in boron nitride nanotubes. Phys. Rev. B 2003, 67. [Google Scholar] [CrossRef]
- Dhungana, K.; Pati, R. Boron Nitride Nanotubes for Spintronics. Sensors 2014, 14, 17655–17685. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Lee, C.H.; Yap, Y.K. Recent advancements in boron nitride nanotubes. Nanoscale 2010, 2, 2028–2034. [Google Scholar] [CrossRef] [PubMed]
- Zhi, C.; Bando, Y.; Tang, C.; Golberg, D. Boron nitride nanotubes. Mater. Sci. Eng. R 2010, 70, 92–111. [Google Scholar] [CrossRef]
- Lee, C.H.; Qin, S.; Savaikar, M.A.; Wang, J.; Hao, B.; Zhang, D.; Banyai, D.; Jaszczak, J.A.; Clark, K.W.; Idrobo, J.-C.; et al. Room-Temperature Tunneling Behavior of Boron Nitride Nanotubes Functionalized with Gold Quantum Dots. Adv. Mater. 2013, 25, 4544–4548. [Google Scholar] [CrossRef] [PubMed]
- Hao, B.; Asthana, A.; Hazaveh, P.K.; Bergstrom, P.L.; Banyai, D.; Savaikar, M.A.; Jaszczak, J.A.; Yap, Y.K. New Flexible Channels for Room Temperature Tunneling Field Effect Transistors. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Parashar, V.; Durand, C.P.; Hao, B.; Amorim, R.G.; Pandey, R.; Tiwari, B.; Zhang, D.; Liu, Y.; Li, A.-P.; Yap, Y.K. Switching Behaviors of Graphene-Boron Nitride Nanotube Heterojunctions. Sci. Rep. 2015, 5. [Google Scholar] [CrossRef] [PubMed]
- Loiseau, A.; Willaime, F.; Demoncy, N.; Hug, G.; Pascard, H. Boron nitride nanotubes with reduced numbers of layers synthesized by arc discharge. Phys. Rev. Lett. 1996, 76. [Google Scholar] [CrossRef] [PubMed]
- Arenal, R.; Stephan, O.; Cochon, J.-L.; Loiseau, A. Root-growth mechanism for single-walled boron nitride nanotubes in laser vaporization technique. J. Am. Chem. Soc. 2007, 129, 16183–16189. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.P.; Sun, X.S.; Lee, C.S.; Bello, I.; Lee, S.T.; Gu, H.D.; Leung, K.M.; Zhou, G.W.; Dong, Z.F.; Zhang, Z. Synthesis of boron nitride nanotubes by means of excimer laser ablation at high temperature. Appl. Phys. Lett. 1998, 72, 1966–1968. [Google Scholar] [CrossRef]
- Lee, R.; Gavillet, J.; de La Chapelle, M.L.; Loiseau, A.; Cochon, J.-L.; Pigache, D.; Thibault, J.; Willaime, F. Catalyst-free synthesis of boron nitride single-wall nanotubes with a preferred zig-zag configuration. Phys. Rev. B 2001, 64. [Google Scholar] [CrossRef]
- Laude, T.; Matsui, Y.; Marraud, A.; Jouffrey, B. Long ropes of boron nitride nanotubes grown by a continuous laser heating. Appl. Phys. Lett. 2000, 76, 3239–3241. [Google Scholar] [CrossRef]
- Han, W.; Bando, Y.; Kurashima, K.; Sato, T. Synthesis of boron nitride nanotubes from carbon nanotubes by a substitution reaction. Appl. Phys. Lett. 1998, 73, 3085–3087. [Google Scholar] [CrossRef]
- Han, W.-Q.; Mickelson, W.; Cumings, J.; Zettl, A. Transformation of BxCyNz nanotubes to pure BN nanotubes. Appl. Phys. Lett. 2002, 81, 1110–1112. [Google Scholar] [CrossRef]
- Lourie, O.R.; Jones, C.R.; Bartlett, B.M.; Gibbons, P.C.; Ruoff, R.S.; Buhro, W.E. CVD Growth of Boron Nitride Nanotubes. Chem. Mater. 2000, 12, 1808–1810. [Google Scholar] [CrossRef]
- Kim, M.J.; Chatterjee, S.; Kim, S.M.; Stach, E.A.; Bradley, M.G.; Pender, M.J.; Sneddon, L.G.; Maruyama, B. Double-Walled Boron Nitride Nanotubes Grown by Floating Catalyst Chemical Vapor Deposition. Nano Lett. 2008, 8, 3298–3302. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.; Bando, Y.; Sato, T.; Kurashima, K. A novel precursor for synthesis of pure boron nitride nanotubes. Chem. Commun. 2002. [Google Scholar] [CrossRef]
- Zhi, C.; Bando, Y.; Tan, C.; Golberg, D. Effective precursor for high yield synthesis of pure BN nanotubes. Solid State Commun. 2005, 135, 67–70. [Google Scholar] [CrossRef]
- Lee, C.H.; Wang, J.; Kayatsha, V.K.; Huang, J.Y.; Yap, Y.K. Effective growth of boron nitride nanotubes by thermal chemical vapor deposition. Nanotechnology 2008, 19. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Chadderton, L.T.; Gerald, J.F.; Williams, J.S. A solid-state process for formation of boron nitride nanotubes. Appl. Phys. Lett. 1999, 74, 2960–2962. [Google Scholar] [CrossRef]
- Li, L.H.; Chen, Y.; Glushenkov, A.M. Synthesis of boron nitride nanotubes by boron ink annealing. Nanotechnology 2010, 21. [Google Scholar] [CrossRef] [PubMed]
- Golberg, D.; Bando, Y.; Tang, C.; Zhi, C. Boron nitride nanotubes. Adv. Mater. 2007, 19, 2413–2432. [Google Scholar] [CrossRef]
- Kalay, S.; Yilmaz, Z.; Sen, O.; Emanet, M.; Kazanc, E.; Çulha, M. Synthesis of boron nitride nanotubes and their applications. Beilstein J. Nanotechnol. 2015, 6, 84–102. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Lin, J.; Tang, C.; Bando, Y.; Zhi, C.; Zhai, T.; Dierre, B.; Sekiguchi, T.; Golberg, D. Bulk synthesis, growth mechanism and properties of highly pure ultrafine boron nitride nanotubes with diameters of sub-10 nm. Nanotechnology 2011, 22. [Google Scholar] [CrossRef] [PubMed]
- Amir, P.; Chunyi, Z.; Yoshio, B.; Tomonobu, N.; Dmitri, G. A comprehensive analysis of the CVD growth of boron nitride nanotubes. Nanotechnology 2012, 23. [Google Scholar] [CrossRef]
- Li, L.; Liu, X.; Li, L.; Chen, Y. High yield BNNTs synthesis by promotion effect of milling-assisted precursor. Microelectron. Eng. 2013, 110, 256–259. [Google Scholar] [CrossRef]
- Li, L.H.; Chen, Y.; Glushenkov, A.M. Boron nitride nanotube films grown from boron ink painting. J. Mater. Chem. 2010, 20, 9679–9683. [Google Scholar] [CrossRef]
- Li, L.; Li, L.H.; Chen, Y.; Dai, X.J.; Xing, T.; Petravic, M.; Liu, X. Mechanically activated catalyst mixing for high-yield boron nitride nanotube growth. Nanoscale Res. Lett. 2012. [Google Scholar] [CrossRef] [PubMed]
- Golberg, D.; Bando, Y.; Eremets, M.; Takemura, K.; Kurashima, K.; Yusa, H. Nanotubes in boron nitride laser heated at high pressure. Appl. Phys. Lett. 1996, 69, 2045–2047. [Google Scholar] [CrossRef]
- Zhou, G.W.; Zhang, Z.; Bai, Z.G. Catalyst effects on formation of boron nitride nano-tubules synthesized by laser ablation. Solid State Commun. 1999, 109, 555–559. [Google Scholar] [CrossRef]
- Wang, J.; Kayastha, V.K.; Yap, Y.K.; Fan, Z.; Lu, J.G.; Pan, Z.; Ivanov, I.N.; Puretzky, A.A.; Geohegan, D.B. Low temperature growth of boron nitride nanotubes on substrates. Nano Lett. 2005, 5, 2528–2532. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.W.; Jordan, K.C.; Park, C.; Kim, J.-W.; Lillehei, P.T.; Crooks, R.; Harrison, J.S. Very long single-and few-walled boron nitride nanotubes via the pressurized vapor/condenser method. Nanotechnology 2009, 20. [Google Scholar] [CrossRef] [PubMed]
- Tiano, A.L.; Park, C.; Lee, J.W.; Luong, H.H.; Gibbons, L.J.; Chu, S.-H.; Applin, S.; Gnoffo, P.; Lowther, S.; Kim, H.J.; et al. Boron nitride nanotube: Synthesis and applications. In Proc. SPIE 9060, Nanosensors, Biosensors, and Info-Tech Sensors and Systems 2014; Varadan, V.K., Ed.; SPIE: San Diego, CA, USA, 2014; Volume 9060. [Google Scholar] [CrossRef]
- Gnoffo, P.A.; Fay, C.C. Laser Vaporization and Plume Chemistry in a Boron Nitride Nanotube Production Rig. J. Thermophys. Heat Tranf. 2013, 27, 369–381. [Google Scholar] [CrossRef]
- Kim, K.S.; Kingston, C.T.; Hrdina, A.; Jakubinek, M.B.; Guan, J.; Plunkett, M.; Simard, B. Hydrogen-Catalyzed, Pilot-Scale Production of Small-Diameter Boron Nitride Nanotubes and Their Macroscopic Assemblies. ACS Nano 2014, 8, 6211–6220. [Google Scholar] [CrossRef] [PubMed]
- Fathalizadeh, A.; Pham, T.; Mickelson, W.; Zettl, A. Scaled Synthesis of Boron Nitride Nanotubes, Nanoribbons, and Nanococoons Using Direct Feedstock Injection into an Extended-Pressure, Inductively-Coupled Thermal Plasma. Nano Lett. 2014, 14, 4881–4886. [Google Scholar] [CrossRef] [PubMed]
- BNNT, LLC. Available online: http://www.bnnt.com/ (accessed on 5 August 2016).
- Raymor Industries Inc. Available online: http://www.raymor.com/our-products/ (accessed on 5 August 2016).
- Chen, H.; Chen, Y.; Yu, J.; Williams, J.S. Purification of boron nitride nanotubes. Chem. Phys. Lett. 2006, 425, 315–319. [Google Scholar] [CrossRef]
- Zhi, C.; Bando, Y.; Tang, C.; Honda, S.; Sato, K.; Kuwahara, H.; Golberg, D. Purification of Boron Nitride Nanotubes through Polymer Wrapping. J. Phys. Chem. B 2006, 110, 1525–1528. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.H.; Zhang, D.; Yap, Y.K. Functionalization, dispersion, and cutting of boron nitride nanotubes in water. J. Phys. Chem. C 2011, 116, 1798–1804. [Google Scholar] [CrossRef]
- Liao, Y.; Chen, Z.; Connell, J.W.; Fay, C.C.; Park, C.; Kim, J.-W.; Lin, Y. Chemical Sharpening, Shortening, and Unzipping of Boron Nitride Nanotubes. Adv. Funct. Mater. 2014, 24, 4497–4506. [Google Scholar] [CrossRef]
- Tiano, A.L.; Gibbons, L.; Tsui, M.; Applin, S.I.; Silva, R.; Park, C.; Fay, C.C. Thermodynamic approach to boron nitride nanotube solubility and dispersion. Nanoscale 2016, 8, 4348–4359. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Chen, Y.; Li, C.P.; Zhang, H.; Williams, J.S.; Liu, Y.; Liu, Z.; Ringer, S.P. Eu-doped Boron Nitride Nanotubes as a Nanometer-Sized Visible-Light Source. Adv. Mater. 2007, 19, 1845–1848. [Google Scholar] [CrossRef]
- Tang, C.; Bando, Y.; Huang, Y.; Yue, S.; Gu, C.; Xu, F.; Golberg, D. Fluorination and Electrical Conductivity of BN Nanotubes. J. Am. Chem. Soc. 2005, 127, 6552–6553. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.L.; Bai, X.D.; Liu, K.H.; Xu, Z.; Golberg, D.; Bando, Y.; Wang, E.G. Direct Synthesis of B−C−N Single-Walled Nanotubes by Bias-Assisted Hot Filament Chemical Vapor Deposition. J. Am. Chem. Soc. 2006, 128, 6530–6531. [Google Scholar] [CrossRef] [PubMed]
- Iyyamperumal, E.; Wang, S.; Dai, L. Vertically Aligned BCN Nanotubes with High Capacitance. ACS Nano 2012, 6, 5259–5265. [Google Scholar] [CrossRef] [PubMed]
- Pham, T.; Fathalizadeh, A.; Shevitski, B.; Turner, S.; Aloni, S.; Zettl, A. A Universal Wet-Chemistry Route to Metal Filling of Boron Nitride Nanotubes. Nano Lett. 2016, 16, 320–325. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Dorozhkin, P.S.; Bando, Y.; Golberg, D. Controllable Modification of SiC Nanowires Encapsulated in BN Nanotubes. Adv. Mater. 2005, 17, 545–549. [Google Scholar] [CrossRef]
- Wu, J.; Yin, L. Platinum Nanoparticle Modified Polyaniline-Functionalized Boron Nitride Nanotubes for Amperometric Glucose Enzyme Biosensor. ACS Appl. Mater. Interfaces 2011, 3, 4354–4362. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Lin, J.; Bando, Y.; Tang, C.; Zhi, C.; Shi, Y.; Takayama-Muromachi, E.; Golberg, D. BN nanotubes coated with uniformly distributed Fe3O4 nanoparticles: Novel magneto-operable nanocomposites. J. Mater. Chem. 2010, 20, 1007–1011. [Google Scholar] [CrossRef]
- Zhi, C.; Bando, Y.; Tang, C.; Golberg, D. SnO2 Nanoparticle-Functionalized Boron Nitride Nanotubes. J. Phys. Chem. B 2006, 110, 8548–8550. [Google Scholar] [CrossRef] [PubMed]
- Velayudham, S.; Lee, C.H.; Xie, M.; Blair, D.; Bauman, N.; Yap, Y.K.; Green, S.A.; Liu, H. Noncovalent Functionalization of Boron Nitride Nanotubes with Poly(p-phenylene-ethynylene)s and Polythiophene. ACS Appl. Mater. Interfaces 2010, 2, 104–110. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Sawada, T.; Zhi, C.; Bando, Y.; Golberg, D.; Serizawa, T. Dispersion of Boron Nitride Nanotubes in Aqueous Solution by Simple Aromatic Molecules. J. Nanosci. Nanotechnol. 2014, 14, 3028–3033. [Google Scholar] [CrossRef] [PubMed]
- Zhi, C.; Bando, Y.; Tang, C.; Xie, R.; Sekiguchi, T.; Golberg, D. Perfectly Dissolved Boron Nitride Nanotubes Due to Polymer Wrapping. J. Am. Chem. Soc. 2005, 127, 15996–15997. [Google Scholar] [CrossRef] [PubMed]
- Zhi, C.; Bando, Y.; Wang, W.; Tang, C.; Kuwahara, H.; Golberg, D. Molecule Ordering Triggered by Boron Nitride Nanotubes and “Green” Chemical Functionalization of Boron Nitride Nanotubes. J. Phys. Chem. C 2007, 111, 18545–18549. [Google Scholar] [CrossRef]
- Zhi, C.Y.; Bando, Y.; Tang, C.C.; Huang, Q.; Golberg, D. Boron nitride nanotubes: Functionalization and composites. J. Mater. Chem. 2008, 18, 3900–3908. [Google Scholar] [CrossRef]
- Huang, Q.; Sandanayaka, A.S.D.; Bando, Y.; Zhi, C.Y.; Ma, R.Z.; Shen, G.Z.; Golberg, D.; Zhao, J.C.; Araki, Y.; Ito, O.; et al. Donor–Acceptor Nanoensembles Based on Boron Nitride Nanotubes. Adv. Mater. 2007, 19, 934–938. [Google Scholar] [CrossRef]
- Wang, W.; Bando, Y.; Zhi, C.; Fu, W.; Wang, E.; Golberg, D. Aqueous noncovalent functionalization and controlled near-surface carbon doping of multiwalled boron nitride nanotubes. J. Am. Chem. Soc. 2008, 130, 8144–8145. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.; Zhi, C.; Bando, Y.; Golberg, D.; Serizawa, T. Noncovalent Functionalization of Boron Nitride Nanotubes in Aqueous Media Opens Application Roads in Nanobiomedicine. Nanobiomedicine 2014, 20, 1575–1581. [Google Scholar] [CrossRef]
- Weng, Q.; Wang, X.; Wang, X.; Bando, Y.; Golberg, D. Functionalized hexagonal boron nitride nanomaterials: Emerging properties and applications. Chem. Soc. Rev. 2016, 45, 3989–4012. [Google Scholar] [CrossRef] [PubMed]
- Oostingh, G.J.; Casals, E.; Italiani, P.; Colognato, R.; Stritzinger, R.; Ponti, J.; Pfaller, T.; Kohl, Y.; Ooms, D.; Favilli, F. Problems and challenges in the development and validation of human cell-based assays to determine nanoparticle-induced immunomodulatory effects. Part. Fibre Toxicol. 2011, 8. [Google Scholar] [CrossRef] [PubMed]
- ShiraláFernando, K. Solubilization of boron nitride nanotubes. Chem. Commun. 2005, 3670–3672. [Google Scholar]
- Zhi, C.; Bando, Y.; Tang, C.; Golberg, D. Immobilization of proteins on boron nitride nanotubes. J. Am. Chem. Soc. 2005, 127, 17144–17145. [Google Scholar] [CrossRef] [PubMed]
- Ciofani, G.; Danti, S.; D’Alessandro, D.; Moscato, S.; Menciassi, A. Assessing cytotoxicity of boron nitride nanotubes: Interference with the MTT assay. Biochem. Biophys. Res. Commun. 2010, 394, 405–411. [Google Scholar] [CrossRef] [PubMed]
- Del Turco, S.; Ciofani, G.; Cappello, V.; Gemmi, M.; Cervelli, T.; Saponaro, C.; Nitti, S.; Mazzolai, B.; Basta, G.; Mattoli, V. Cytocompatibility evaluation of glycol-chitosan coated boron nitride nanotubes in human endothelial cells. Colloids Surf. B 2013, 111, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Ciofani, G.; Ricotti, L.; Danti, S.; Moscato, S.; Nesti, C.; D’Alessandro, D.; Dinucci, D.; Chiellini, F.; Pietrabissa, A.; Petrini, M.; et al. Investigation of interactions between poly-l-lysine-coated boron nitride nanotubes and C2C12 cells: up-take, cytocompatibility, and differentiation. Int. J. Nanomed. 2010, 5, 285–298. [Google Scholar] [CrossRef]
- Serena, D.; Gianni, C.; Stefania, M.; Delfo, D.A.; Elena, C.; Claudia, N.; Rosaria, B.; Giovanni, B.; Andrea, P.; Michele, L.; et al. Boron nitride nanotubes and primary human osteoblasts: In vitro compatibility and biological interactions under low frequency ultrasound stimulation. Nanotechnology 2013, 24. [Google Scholar] [CrossRef]
- Ciofani, G.; Raffa, V.; Menciassi, A.; Cuschieri, A. Cytocompatibility, interactions, and uptake of polyethyleneimine-coated boron nitride nanotubes by living cells: Confirmation of their potential for biomedical applications. Biotechnol. Bioeng. 2008, 101, 850–858. [Google Scholar] [CrossRef] [PubMed]
- Ciofani, G.; Danti, S.; Genchi, G.G.; Mazzolai, B.; Mattoli, V. Boron Nitride Nanotubes: Biocompatibility and Potential Spill-Over in Nanomedicine. Small 2013, 9, 1672–1685. [Google Scholar] [CrossRef] [PubMed]
- Horváth, L.; Magrez, A.; Golberg, D.; Zhi, C.; Bando, Y.; Smajda, R.; Horváth, E.; Forró, L.; Schwaller, B. In Vitro Investigation of the Cellular Toxicity of Boron Nitride Nanotubes. ACS Nano 2011, 5, 3800–3810. [Google Scholar] [CrossRef] [PubMed]
- Soares, D.C.F.; Ferreira, T.H.; Ferreira, C.d.A.; Cardoso, V.N.; de Sousa, E.M.B. Boron nitride nanotubes radiolabeled with 99mTc: Preparation, physicochemical characterization, biodistribution study, and scintigraphic imaging in Swiss mice. Int. J. Pharm. 2012, 423, 489–495. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Wu, P.; Rousseas, M.; Okawa, D.; Gartner, Z.; Zettl, A.; Bertozzi, C.R. Boron nitride nanotubes are noncytotoxic and can be functionalized for interaction with proteins and cells. J. Am. Chem. Soc. 2009, 131, 890–891. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.; Zhi, C.; Bando, Y.; Golberg, D.; Serizawa, T. Isolation of individual boron nitride nanotubes via peptide wrapping. J. Am. Chem. Soc. 2010, 132, 4976–4977. [Google Scholar] [CrossRef] [PubMed]
- Zhi, C.; Bando, Y.; Tang, C.; Honda, S.; Sato, K.; Kuwahara, H.; Golberg, D. Covalent functionalization: towards soluble multiwalled boron nitride nanotubes. Angew. Chem. Int. Ed. 2005, 44, 7932–7935. [Google Scholar] [CrossRef] [PubMed]
- Huang, Q.; Bando, Y.; Zhi, C.; Golberg, D.; Kurashima, K.; Xu, F.; Gao, L. Chemical peeling and branching of boron nitride nanotubes in dimethyl sulfoxide. Angew. Chem. Int. Ed. 2006, 45, 2044–2047. [Google Scholar] [CrossRef] [PubMed]
- Pal, S.; Vivekchand, S.; Govindaraj, A.; Rao, C. Functionalization and solubilization of BN nanotubes by interaction with Lewis bases. J. Mater. Chem. 2007, 17, 450–452. [Google Scholar] [CrossRef]
- Ciofani, G.; Genchi, G.G.; Liakos, I.; Athanassiou, A.; Dinucci, D.; Chiellini, F.; Mattoli, V. A simple approach to covalent functionalization of boron nitride nanotubes. J. Colloid Interface Sci. 2012, 374, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Shin, H.; Guan, J.; Zgierski, M.Z.; Kim, K.S.; Kingston, C.T.; Simard, B. Covalent Functionalization of Boron Nitride Nanotubes via Reduction Chemistry. ACS Nano 2015, 9, 12573–12582. [Google Scholar] [CrossRef] [PubMed]
- Zhi, C.; Bando, Y.; Tang, C.; Honda, S.; Kuwahara, H.; Golberg, D. Boron nitride nanotubes/polystyrene composites. J. Mater. Res. 2006, 21, 2794–2800. [Google Scholar] [CrossRef]
- Huang, Q.; Bando, Y.; Xu, X.; Nishimura, T.; Zhi, C.; Tang, C.; Xu, F.; Gao, L.; Golberg, D. Enhancing superplasticity of engineering ceramics by introducing BN nanotubes. Nanotechnology 2007, 18. [Google Scholar] [CrossRef]
- Xue, Y.; Jiang, B.; Bourgeois, L.; Dai, P.; Mitome, M.; Zhang, C.; Yamaguchi, M.; Matveev, A.; Tang, C.; Bando, Y.; et al. Aluminum matrix composites reinforced with multi-walled boron nitride nanotubes fabricated by a high-pressure torsion technique. Mater. Des. 2015, 88, 451–460. [Google Scholar] [CrossRef]
- Yamaguchi, M.; Meng, F.; Firestein, K.; Tsuchiya, K.; Golberg, D. Powder metallurgy routes toward aluminum boron nitride nanotube composites, their morphologies, structures and mechanical properties. Mater. Sci. Eng. A 2014, 604, 9–17. [Google Scholar] [CrossRef]
- Zhi, C.; Bando, Y.; Terao, T.; Tang, C.; Kuwahara, H.; Golberg, D. Towards Thermoconductive, Electrically Insulating Polymeric Composites with Boron Nitride Nanotubes as Fillers. Adv. Funct. Mater. 2009, 19, 1857–1862. [Google Scholar] [CrossRef]
- Sauti, G.; Park, C.; Kang, J.H.; Kim, J.; Harrison, J.S.; Smith, M.W.; Jordan, K.; Lowther, S.E.; Lillehei, P.T.; Thibeault, S.A. Boron nitride and boron nitride nanotube materials for radiation shielding. US Patent US20130119316 A1, 16 May 2013. [Google Scholar]
- Sears, V.F. Neutron Scattering Lengths and Cross Section. Neutron News 1992, 3, 29–37. [Google Scholar] [CrossRef]
- Gao, J.; Xu, B. Applications of nanomaterials inside cells. Nano Today 2009, 4, 37–51. [Google Scholar] [CrossRef]
- Genchi, G.G.; Ciofani, G. Bioapplications of boron nitride nanotubes. Nanomedicine 2015, 10, 3315–3319. [Google Scholar] [CrossRef] [PubMed]
- Lahiri, D.; Rouzaud, F.; Richard, T.; Keshri, A.K.; Bakshi, S.R.; Kos, L.; Agarwal, A. Boron nitride nanotube reinforced polylactide–polycaprolactone copolymer composite: Mechanical properties and cytocompatibility with osteoblasts and macrophages in vitro. Acta Biomater. 2010, 6, 3524–3533. [Google Scholar] [CrossRef] [PubMed]
- Lahiri, D.; Singh, V.; Benaduce, A.P.; Seal, S.; Kos, L.; Agarwal, A. Boron nitride nanotube reinforced hydroxyapatite composite: Mechanical and tribological performance and in-vitro biocompatibility to osteoblasts. J. Mech. Behav. Biomed. 2011, 4, 44–56. [Google Scholar] [CrossRef] [PubMed]
- Shuai, C.; Gao, C.; Feng, P.; Xiao, T.; Yu, K.; Deng, Y.; Peng, S. Boron Nitride Nanotubes Reinforce Tricalcium Phosphate Scaffolds and Promote the Osteogenic Differentiation of Mesenchymal Stem Cells. J. Biomed. Nanotechnol. 2016, 12, 934–947. [Google Scholar] [CrossRef] [PubMed]
- Gianni, C.; Barbara, M.; Virgilio, M. Recent Advancements in Boron Nitride Nanotube Biomedical Research. In Nanotubes and Nanosheets; Chen, Y., Ed.; CRC Press: Boca Raton, FL, USA, 2015; pp. 575–586. [Google Scholar]
- Şen, Ö.; Emanet, M.; Çulha, M. Chapter 3-Biocompatibility evaluation of boron nitride nanotubes. In Boron Nitride Nanotubes in Nanomedicine; Ciofani, G., Mattoli, V., Eds.; William Andrew Publishing: Boston, MA, USA, 2016; pp. 41–58. [Google Scholar]
- Ciofani, G.; Danti, S.; Nitti, S.; Mazzolai, B.; Mattoli, V.; Giorgi, M. Biocompatibility of boron nitride nanotubes: An up-date of in vivo toxicological investigation. Int. J. Pharm. 2013, 444, 85–88. [Google Scholar] [CrossRef] [PubMed]
- Salvetti, A.; Rossi, L.; Iacopetti, P.; Li, X.; Nitti, S.; Pellegrino, T.; Mattoli, V.; Golberg, D.; Ciofani, G. In vivo biocompatibility of boron nitride nanotubes: Effects on stem cell biology and tissue regeneration in planarians. Nanomedicine 2015, 10, 1911–1922. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Li, L.H.; Ramakrishnan, S.; Dai, X.J.; Nicholas, K.; Chen, Y.; Chen, Z.; Liu, X. Controlling Wettability of Boron Nitride Nanotube Films and Improved Cell Proliferation. J. Phys. Chem. C 2012, 116, 18334–18339. [Google Scholar] [CrossRef]
- Barth, R.F.; Soloway, A.H.; Fairchild, R.G. Boron Neutron Capture Therapy of Cancer. Cancer Res. 1990, 50, 1061–1070. [Google Scholar] [CrossRef] [PubMed]
- Coderre, J.A.; Morris, G.M. The Radiation Biology of Boron Neutron Capture Therapy. Radiat. Res. 1999, 151, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Barth, R.F.; Coderre, J.A.; Vicente, M.G.H.; Blue, T.E. Boron Neutron Capture Therapy of Cancer: Current Status and Future Prospects. Clin. Cancer Res. 2005, 11, 3987–4002. [Google Scholar] [CrossRef] [PubMed]
- Barth, R.F. Boron neutron capture therapy at the crossroads: Challenges and opportunities. Appl. Radiat. Isotopes 2009, 67, S3–S6. [Google Scholar] [CrossRef] [PubMed]
- Buzatu, D.A.; Wilkes, J.G.; Miller, D.; Darsey, J.A.; Heinze, T.; Birls, A.; Beger, R. Nanotubes for Cancer Therapy and Diagnostics. US Patent US20110027174 A1, 3 February 2011. [Google Scholar]
- Ciofani, G.; Raffa, V.; Menciassi, A.; Cuschieri, A. Folate Functionalized Boron Nitride Nanotubes and their Selective Uptake by Glioblastoma Multiforme Cells: Implications for their Use as Boron Carriers in Clinical Boron Neutron Capture Therapy. Nanoscale Res. Lett. 2009, 4, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Menichetti, L.; De Marchi, D.; Calucci, L.; Ciofani, G.; Menciassi, A.; Forte, C. Boron nitride nanotubes for boron neutron capture therapy as contrast agents in magnetic resonance imaging at 3 T. Appl. Radiat. Isotopes 2011, 69, 1725–1727. [Google Scholar] [CrossRef] [PubMed]
- Rocca, A.; Marino, A.; Del Turco, S.; Cappello, V.; Parlanti, P.; Pellegrino, M.; Golberg, D.; Mattoli, V.; Ciofani, G. Pectin-coated boron nitride nanotubes: In vitro cyto-/immune-compatibility on RAW 264.7 macrophages. Biochim. Biophys. Acta 2016, 1860, 775–784. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Yague, M.A.; Larrañaga, A.; Gladkovskaya, O.; Stanley, A.; Tadayyon, G.; Guo, Y.; Sarasua, J.-R.; Tofail, S.A.M.; Zeugolis, D.I.; Pandit, A.; et al. Effects of Polydopamine Functionalization on Boron Nitride Nanotube Dispersion and Cytocompatibility. Bioconjugate Chem. 2015, 26, 2025–2037. [Google Scholar] [CrossRef] [PubMed]



© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, C.H.; Bhandari, S.; Tiwari, B.; Yapici, N.; Zhang, D.; Yap, Y.K. Boron Nitride Nanotubes: Recent Advances in Their Synthesis, Functionalization, and Applications. Molecules 2016, 21, 922. https://doi.org/10.3390/molecules21070922
Lee CH, Bhandari S, Tiwari B, Yapici N, Zhang D, Yap YK. Boron Nitride Nanotubes: Recent Advances in Their Synthesis, Functionalization, and Applications. Molecules. 2016; 21(7):922. https://doi.org/10.3390/molecules21070922
Chicago/Turabian StyleLee, Chee Huei, Shiva Bhandari, Bishnu Tiwari, Nazmiye Yapici, Dongyan Zhang, and Yoke Khin Yap. 2016. "Boron Nitride Nanotubes: Recent Advances in Their Synthesis, Functionalization, and Applications" Molecules 21, no. 7: 922. https://doi.org/10.3390/molecules21070922
APA StyleLee, C. H., Bhandari, S., Tiwari, B., Yapici, N., Zhang, D., & Yap, Y. K. (2016). Boron Nitride Nanotubes: Recent Advances in Their Synthesis, Functionalization, and Applications. Molecules, 21(7), 922. https://doi.org/10.3390/molecules21070922