Nanotechnology Formulations for Antibacterial Free Fatty Acids and Monoglycerides
Abstract
:1. Introduction
2. Overview of Antimicrobial Lipids
2.1. Brief History
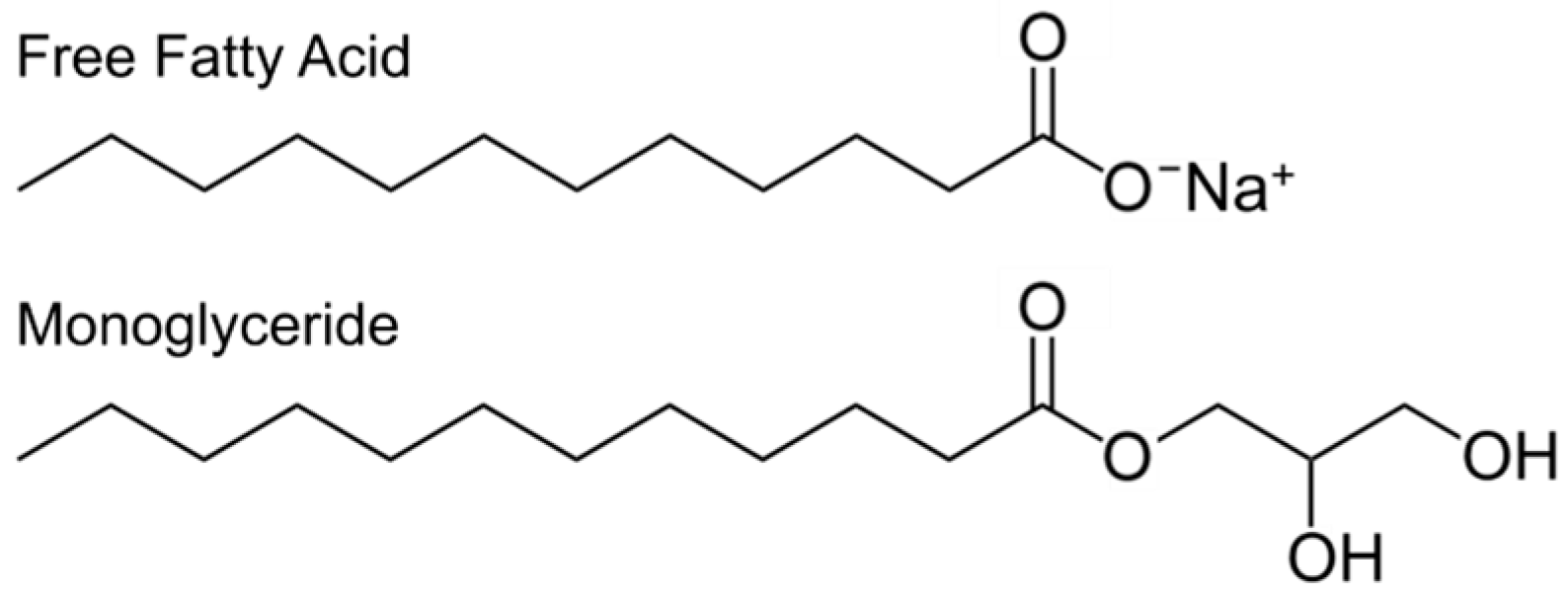
2.2. Classes of Antimicrobial Lipids
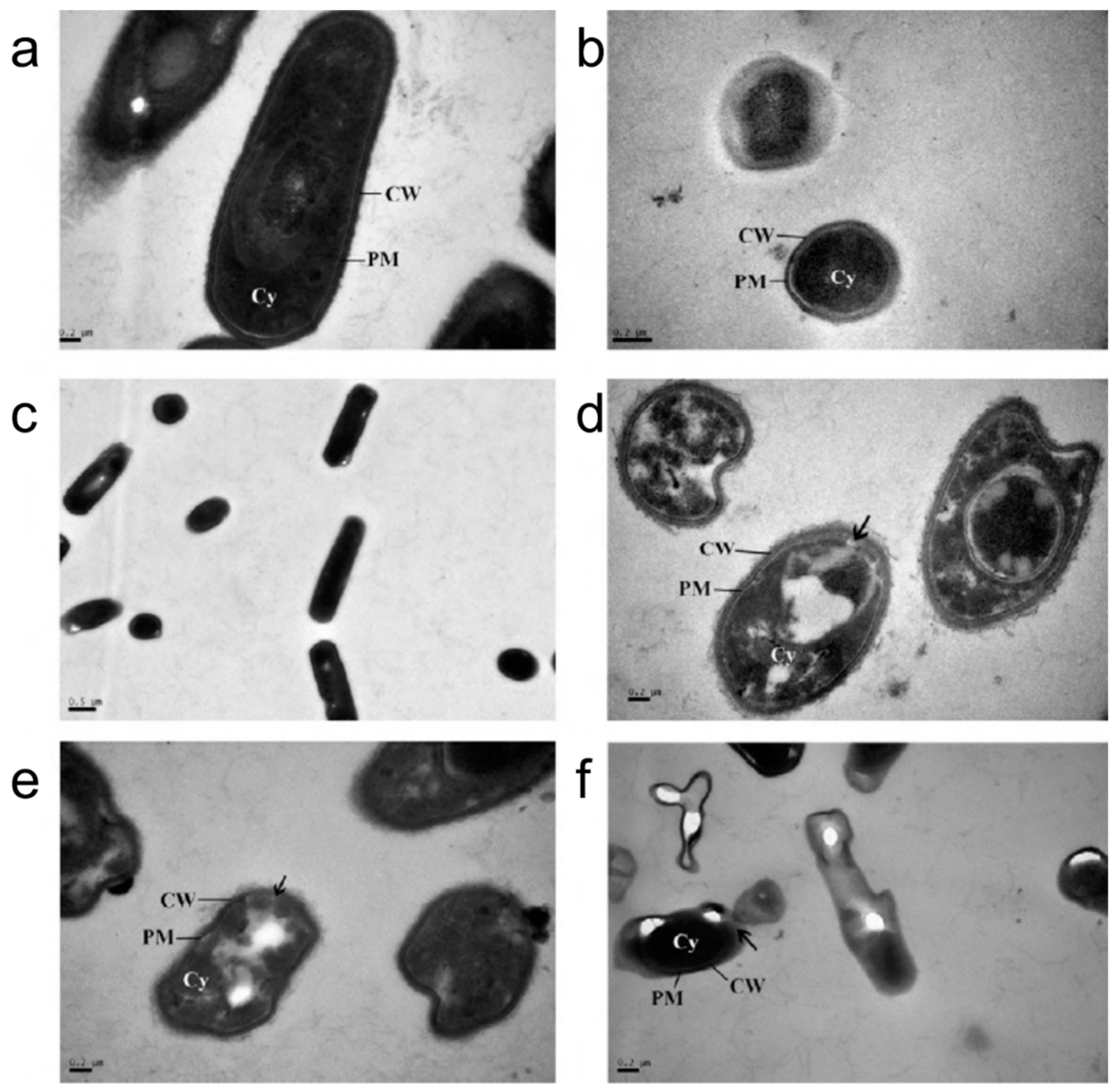
2.3. Activity Spectrum
2.4. Formulation Challenges
3. Nanotechnology Strategies
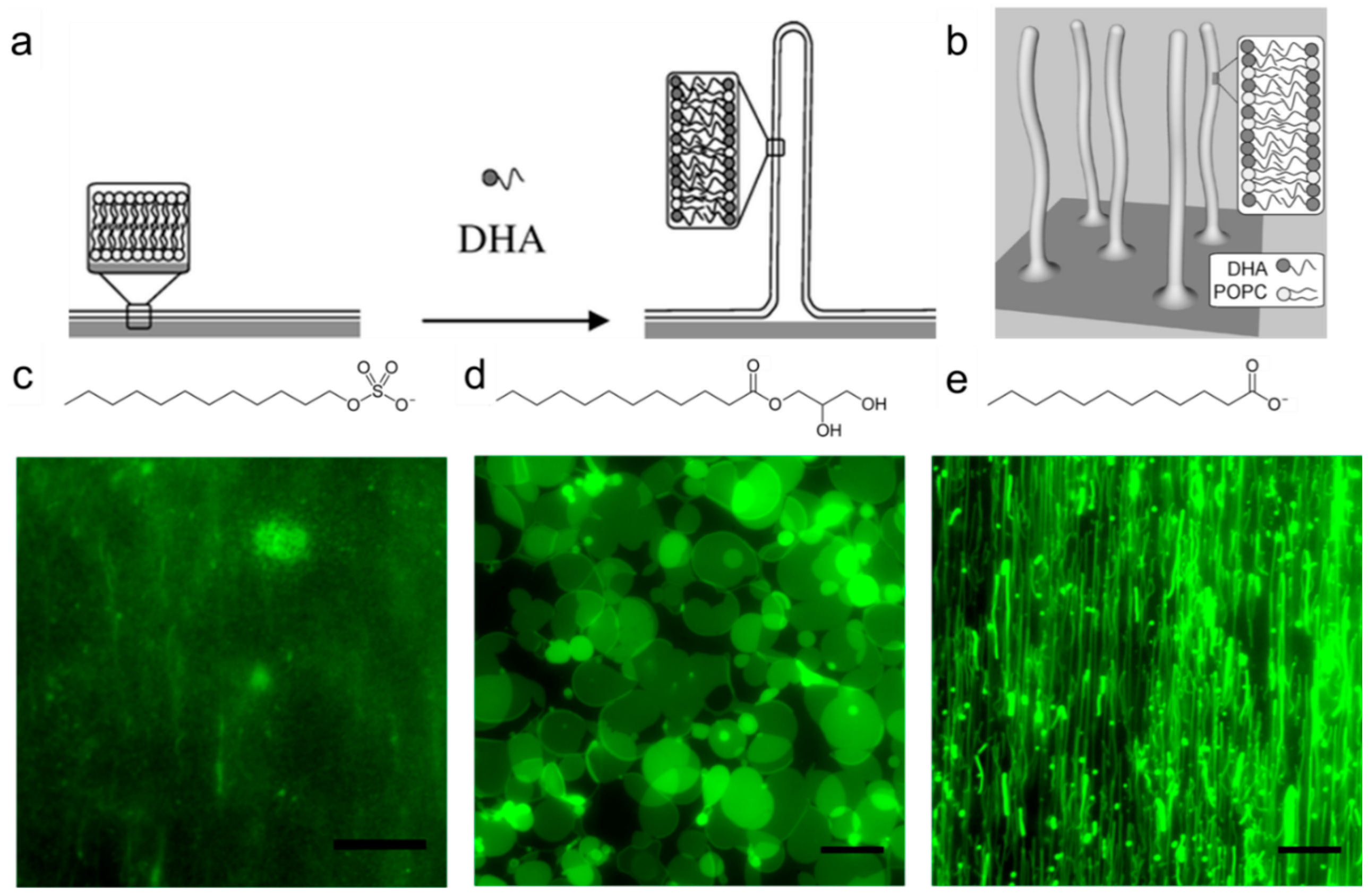
3.1. Emulsions
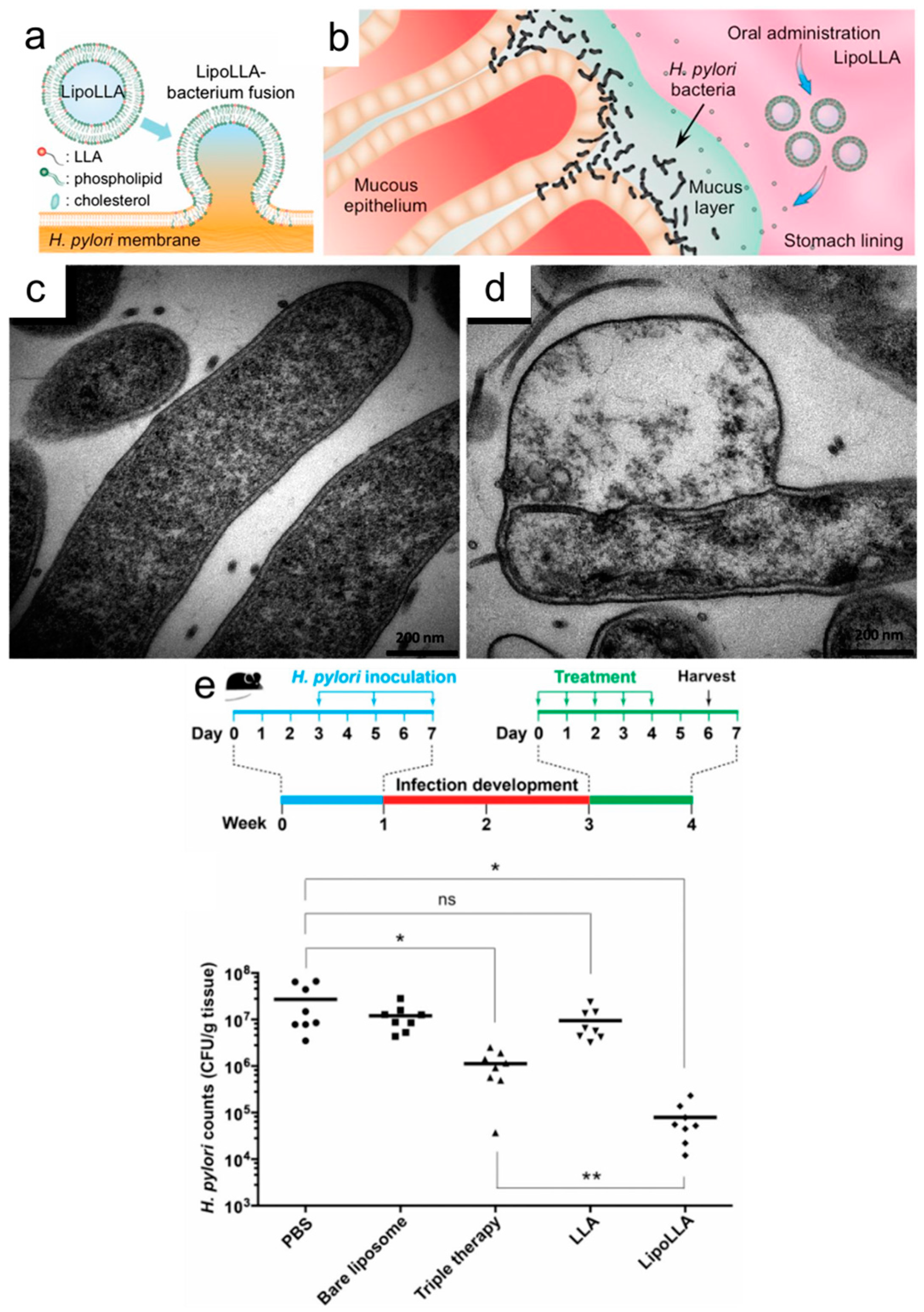
3.2. Liposomes
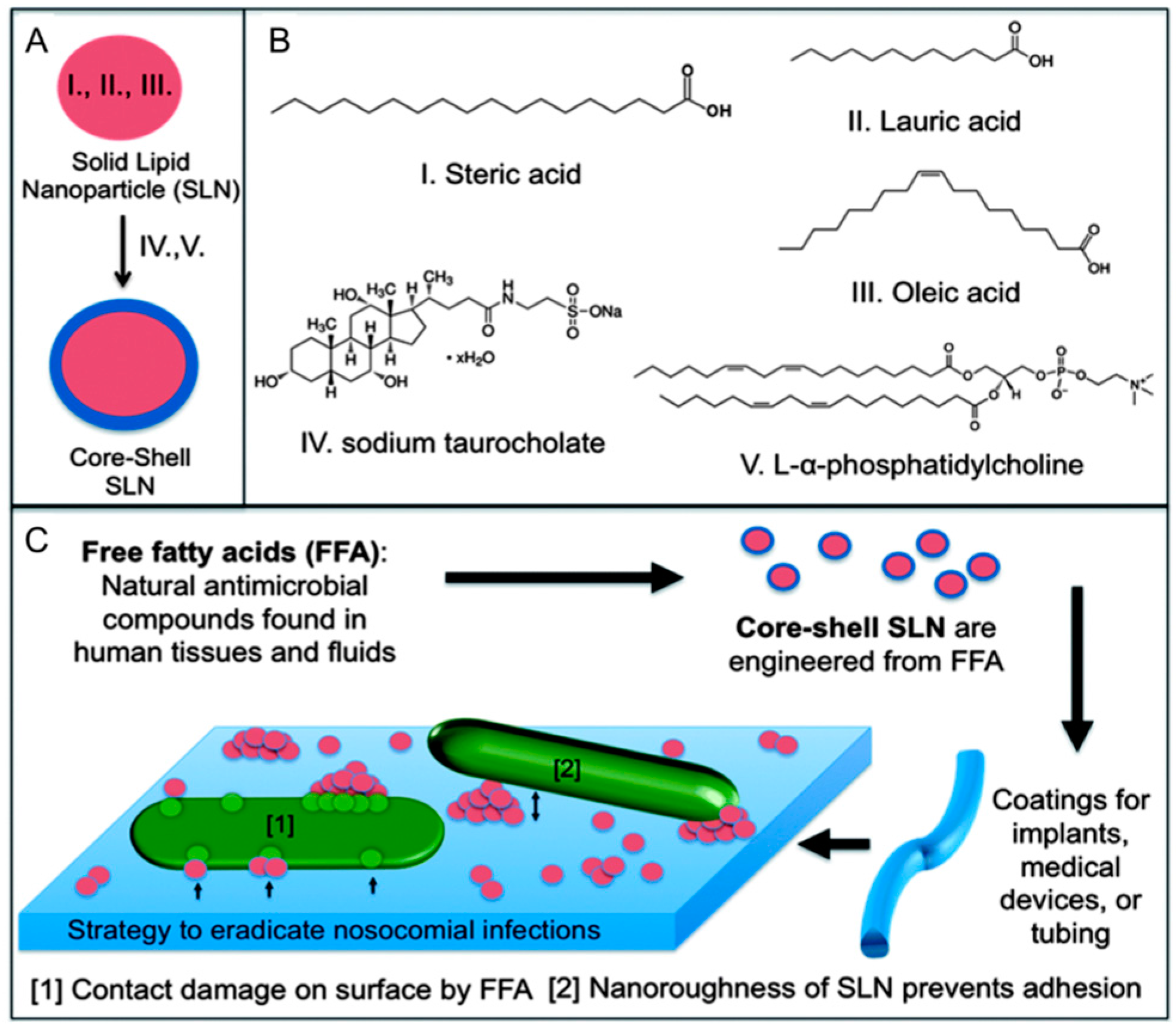
3.3. Solid Lipid Nanoparticles
3.4. Hydrogel
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Laxminarayan, R.; Duse, A.; Wattal, C.; Zaidi, A.K.; Wertheim, H.F.; Sumpradit, N.; Vlieghe, E.; Hara, G.L.; Gould, I.M.; Goossens, H. Antibiotic Resistance—The Need for Global Solutions. Lancet Infect. Dis. 2013, 13, 1057–1098. [Google Scholar] [CrossRef]
- Alanis, A.J. Resistance to Antibiotics: Are We in the Post-antibiotic Era? Arch. Med. Res. 2005, 36, 697–705. [Google Scholar] [CrossRef] [PubMed]
- Shlaes, D.M.; Sahm, D.; Opiela, C.; Spellberg, B. The FDA Reboot of Antibiotic Development. Antimicrob. Agents Chemother. 2013, 57, 4605–4607. [Google Scholar] [CrossRef] [PubMed]
- Suda, K.J.; Hicks, L.A.; Roberts, R.M.; Hunkler, R.J.; Danziger, L.H. A National Evaluation of Antibiotic Expenditures by Healthcare Setting in the United States, 2009. J. Antimicrob. Chemother. 2013, 68, 715–718. [Google Scholar] [CrossRef] [PubMed]
- Arias, C.A.; Murray, B.E. Antibiotic-resistant Bugs in the 21st Century—A Clinical Super-challenge. N. Engl. J. Med. 2009, 360, 439–443. [Google Scholar] [CrossRef] [PubMed]
- Cooper, M.A.; Shlaes, D. Fix the Antibiotics Pipeline. Nature 2011, 472, 32. [Google Scholar] [CrossRef] [PubMed]
- Priya, S.; Towse, A. New Drugs to Tackle Antimicrobial Resistance: Analysis of EU Policy Options; Office of Health Economics: London, UK, 2011. [Google Scholar]
- Laxminarayan, R.; Powers, J.H. Antibacterial R & D Incentives. Nat. Rev. Drug Discov. 2011, 10, 727–728. [Google Scholar] [PubMed]
- Fauci, A.S.; Marston, H.D. The Perpetual Challenge of Antimicrobial Resistance. JAMA 2014, 311, 1853–1854. [Google Scholar] [CrossRef] [PubMed]
- Nathan, C.; Cars, O. Antibiotic Resistance—Problems, Progress, and Prospects. N. Engl. J. Med. 2014, 371, 1761–1763. [Google Scholar] [CrossRef] [PubMed]
- Outterson, K.; Powers, J.H.; Daniel, G.W.; McClellan, M.B. Repairing the Broken Market for Antibiotic Innovation. Health Aff. 2015, 34, 277–285. [Google Scholar] [CrossRef] [PubMed]
- Mullard, A. 2014 FDA Drug Approvals. Nat. Rev. Drug Discov. 2015, 14, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Thamphiwatana, S.; Angsantikul, P.; Zhang, L. Nanoparticle Approaches against Bacterial Infections. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2014, 6, 532–547. [Google Scholar] [CrossRef] [PubMed]
- Ling, L.L.; Schneider, T.; Peoples, A.J.; Spoering, A.L.; Engels, I.; Conlon, B.P.; Mueller, A.; Schäberle, T.F.; Hughes, D.E.; Epstein, S. A New Antibiotic Kills Pathogens without Detectable Resistance. Nature 2015, 517, 455–459. [Google Scholar] [CrossRef] [PubMed]
- Hancock, R.E.; Sahl, H.-G. Antimicrobial and Host-defense Peptides as New Anti-infective Therapeutic Strategies. Nat. Biotechnol. 2006, 24, 1551–1557. [Google Scholar] [CrossRef] [PubMed]
- Afacan, N.J.; Yeung, A.T.; Pena, O.M.; Hancock, R.E. Therapeutic potential of host defense peptides in antibiotic-resistant infections. Curr. Pharm. Des. 2012, 18, 807–819. [Google Scholar] [CrossRef] [PubMed]
- Marr, A.K.; Gooderham, W.J.; Hancock, R.E. Antibacterial peptides for therapeutic use: Obstacles and realistic outlook. Curr. Opin. Pharmacol. 2006, 6, 468–472. [Google Scholar] [CrossRef] [PubMed]
- Uhlig, T.; Kyprianou, T.; Martinelli, F.G.; Oppici, C.A.; Heiligers, D.; Hills, D.; Calvo, X.R.; Verhaert, P. The emergence of peptides in the pharmaceutical business: From exploration to exploitation. EuPA Open Proteom. 2014, 4, 58–69. [Google Scholar] [CrossRef]
- Lehrer, R.I.; Ganz, T. Antimicrobial Peptides in Mammalian and Insect Host Defence. Curr. Opin. Immunol. 1999, 11, 23–27. [Google Scholar] [CrossRef]
- Brown, K.L.; Hancock, R.E. Cationic Host Defense (antimicrobial) Peptides. Curr. Opin. Immunol. 2006, 18, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Thormar, H. Antibacterial Effects of Lipids: Historical Review (1881 to 1960). In Lipids and Essential Oils as Antimicrobial Agents; John Wiley & Sons, Ltd: Hoboken, NJ, USA, 2011; pp. 25–45. [Google Scholar]
- Desbois, A.P.; Smith, V.J. Antibacterial Free Fatty Acids: Activities, Mechanisms of Action and Biotechnological Potential. Appl. Microbiol. Biotechnol. 2010, 85, 1629–1642. [Google Scholar] [CrossRef] [PubMed]
- Desbois, A.P. Potential applications of antimicrobial fatty acids in medicine, agriculture and other industries. Recent Pat. Anti Infect. Drug Discov. 2012, 7, 111–122. [Google Scholar] [CrossRef]
- Ramanathan, M.; Shrestha, L.K.; Mori, T.; Ji, Q.; Hill, J.P.; Ariga, K. Amphiphile nanoarchitectonics: From basic physical chemistry to advanced applications. Phys. Chem. Chem. Phys. 2013, 15, 10580–10611. [Google Scholar] [CrossRef] [PubMed]
- Nakanishi, W.; Minami, K.; Shrestha, L.K.; Ji, Q.; Hill, J.P.; Ariga, K. Bioactive nanocarbon assemblies: Nanoarchitectonics and applications. Nano Today 2014, 9, 378–394. [Google Scholar] [CrossRef]
- Ariga, K.; Ji, Q.; Nakanishi, W.; Hill, J.P.; Aono, M. Nanoarchitectonics: A new materials horizon for nanotechnology. Mater. Horiz. 2015, 2, 406–413. [Google Scholar] [CrossRef]
- Ariga, K.; Li, J.; Fei, J.; Ji, Q.; Hill, J.P. Nanoarchitectonics for Dynamic Functional Materials from Atomic-/Molecular-Level Manipulation to Macroscopic Action. Adv. Mater. 2016, 28, 1251–1286. [Google Scholar] [CrossRef] [PubMed]
- Nakatsuji, T.; Gallo, R.L. Antimicrobial Peptides: Old Molecules with New Ideas. J. Investig. Dermatol. 2011, 132, 887–895. [Google Scholar] [CrossRef] [PubMed]
- Drake, D.R.; Brogden, K.A.; Dawson, D.V.; Wertz, P.W. Thematic Review Series: Skin Lipids. Antimicrobial Lipids at the Skin Surface. J. Lipid Res. 2008, 49, 4–11. [Google Scholar] [CrossRef] [PubMed]
- Burtenshaw, J. Self-disinfection of the Skin: A Short Review and Some Original Observations. Br. Med. Bull. 1945, 3, 161–164. [Google Scholar] [PubMed]
- Thormar, H.; Hilmarsson, H. The Role of Microbicidal Lipids in Host Defense against Pathogens and Their Potential as Therapeutic Agents. Chem. Phys. Lipids 2007, 150, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Fischer, C.L.; Blanchette, D.R.; Brogden, K.A.; Dawson, D.V.; Drake, D.R.; Hill, J.R.; Wertz, P.W. The Roles of Cutaneous Lipids in Host Defense. Biochim. Biophys. Acta 2014, 1841, 319–322. [Google Scholar] [CrossRef] [PubMed]
- Aminov, R.I. A Brief History of the Antibiotic Era: Lessons Learned and Challenges for the Future. Front. Microbiol. 2010, 1, 134. [Google Scholar] [CrossRef] [PubMed]
- Heerklotz, H. Interactions of Surfactants with Lipid Membranes. Q. Rev. Biophys. 2008, 41, 205–264. [Google Scholar] [CrossRef] [PubMed]
- Hammer, K.A.; Carson, C.F. Antibacterial and Antifungal Activities of Essential Oils. In Lipids and Essential Oils as Antimicrobial Agents; John Wiley & Sons, Ltd: Hoboken, NJ, USA, 2011; pp. 255–306. [Google Scholar]
- Kabara, J.; Vrable, R.; Jie, M.L.K. Antimicrobial Lipids: Natural and Synthetic Fatty Acids and Monoglycerides. Lipids 1977, 12, 753–759. [Google Scholar] [CrossRef] [PubMed]
- Kabara, J.J. Structure-function relationships of surfactants as antimicrobial agents. J. Soc. Cosmet. Chem. 1978, 29, 733–741. [Google Scholar]
- Kabara, J.J. Antimicrobial Agents Derived from Fatty Acids. J. Am. Oil Chem. Soc. 1984, 61, 397–403. [Google Scholar] [CrossRef]
- Kabara, J.J.; Swieczkowski, D.M.; Conley, A.J.; Truant, J.P. Fatty Acids and Derivatives as Antimicrobial Agents. Antimicrob. Agents Chemother. 1972, 2, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Conley, A.J.; Kabara, J.J. Antimicrobial Action of Esters of Polyhydric Alcohols. Antimicrob. Agents Chemother. 1973, 4, 501–506. [Google Scholar] [CrossRef] [PubMed]
- Kabara, J.J. GRAS Antimicrobial Agents for Cosmetic Products. J. Soc. Cosmet. Chem. 1980, 31, 1–10. [Google Scholar]
- Nakatsuji, T.; Kao, M.C.; Fang, J.-Y.; Zouboulis, C.C.; Zhang, L.; Gallo, R.L.; Huang, C.-M. Antimicrobial Property of Lauric Acid against Propionibacterium acnes: Its Therapeutic Potential for Inflammatory Acne Vulgaris. J. Investig. Dermatol. 2009, 129, 2480–2488. [Google Scholar] [CrossRef] [PubMed]
- Galbraith, H.; Miller, T.; Paton, A.; Thompson, J. Antibacterial activity of long chain fatty acids and the reversal with calcium, magnesium, ergocalciferol and cholesterol. J. Appl. Bacteriol. 1971, 34, 803–813. [Google Scholar] [CrossRef] [PubMed]
- Nazari, M.; Kurdi, M.; Heerklotz, H. Classifying Surfactants with Respect to Their Effect on Lipid Membrane Order. Biophys. J. 2012, 102, 498–506. [Google Scholar] [CrossRef] [PubMed]
- Bergsson, G.; Arnfinnsson, J.; Steingrímsson, Ó.; Thormar, H. Killing of Gram-positive cocci by Fatty Acids and Monoglycerides. APMIS 2001, 109, 670–678. [Google Scholar] [CrossRef] [PubMed]
- Thid, D.; Benkoski, J.J.; Svedhem, S.; Kasemo, B.; Gold, J. DHA-induced changes of supported lipid membrane morphology. Langmuir 2007, 23, 5878–5881. [Google Scholar] [CrossRef] [PubMed]
- Yoon, B.K.; Jackman, J.A.; Kim, M.C.; Cho, N.-J. Spectrum of membrane morphological responses to antibacterial fatty acids and related surfactants. Langmuir 2015, 31, 10223–10232. [Google Scholar] [CrossRef] [PubMed]
- Vorum, H.; Brodersen, R.; Kragh-Hansen, U.; Pedersen, A.O. Solubility of Long-chain Fatty Acids in Phosphate Buffer at pH 7.4. Biochim. Biophys. Acta 1992, 1126, 135–142. [Google Scholar] [CrossRef]
- Yang, D.; Pornpattananangkul, D.; Nakatsuji, T.; Chan, M.; Carson, D.; Huang, C.-M.; Zhang, L. The Antimicrobial Activity of Liposomal Lauric Acids against Propionibacterium acnes. Biomaterials 2009, 30, 6035–6040. [Google Scholar] [CrossRef] [PubMed]
- Al-Adham, I.; Khalil, E.; Al-Hmoud, N.; Kierans, M.; Collier, P. Microemulsions are membrane-active, antimicrobial, self-preserving systems. J. Appl. Microbiol. 2000, 89, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Thormar, H.; Hilmarsson, H.; Bergsson, G. Stable concentrated emulsions of the 1-monoglyceride of capric acid (monocaprin) with microbicidal activities against the food-borne bacteria Campylobacter jejuni, Salmonella spp., and Escherichia coli. Appl. Environ. Microbiol. 2006, 72, 522–526. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.; Feng, F.; Huang, B. Physicochemical Characterization and Evaluation of a Microemulsion System for Antimicrobial Activity of Glycerol Monolaurate. Int. J. Pharm. 2006, 321, 171–175. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Shen, Y.; Bao, Y.; He, Y.; Feng, F.; Zheng, X. Characterization and synergistic antimicrobial activities of food-grade dilution-stable microemulsions against Bacillus subtilis. Food Res. Int. 2008, 41, 495–499. [Google Scholar] [CrossRef]
- Zhang, H.; Shen, Y.; Weng, P.; Zhao, G.; Feng, F.; Zheng, X. Antimicrobial activity of a food-grade fully dilutable microemulsion against Escherichia coli and Staphylococcus aureus. Int. J. Food Microbiol. 2009, 135, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.; Zhang, M.; Huang, B.I.N.; Liu, J.U.N.; Hu, H.; Feng, F. Enhancement of Antimicrobial Activities by the Food-Grade Monolaurin Microemulsion System. J. Food Process Eng. 2009, 32, 104–111. [Google Scholar] [CrossRef]
- Feng, F.; Zhang, H.; Sha, S.; Lu, Z.; Shen, Y.; Zheng, X. Characterization and Antimicrobial Evaluation of Dilution-Stable Microemulsions Against Stenotrophomonas maltrophilia. J. Dispers. Sci. Technol. 2009, 30, 503–509. [Google Scholar] [CrossRef]
- Zhang, H.; Cui, Y.; Zhu, S.; Feng, F.; Zheng, X. Characterization and Antimicrobial Activity of a Pharmaceutical Microemulsion. Int. J. Pharm. 2010, 395, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Petra, Š.; Věra, K.; Iva, H.; Petr, H.; Zdeňka, K.; Leona, B. Formulation, Antibacterial Activity, and Cytotoxicity of 1-monoacylglycerol Microemulsions. Eur. J. Lipid Sci. Technol. 2014, 116, 448–457. [Google Scholar] [CrossRef]
- Barenholz, Y.C. Doxil®—the First FDA-approved Nano-drug: Lessons Learned. J. Controll. Release 2012, 160, 117–134. [Google Scholar] [CrossRef] [PubMed]
- Beaulac, C.; Clement-Major, S.; Hawari, J.; Lagacé, J. Eradication of Mucoid Pseudomonas aeruginosa with Fluid Liposome-encapsulated Tobramycin in an Animal Model of Chronic Pulmonary Infection. Antimicrob. Agents Chemother. 1996, 40, 665–669. [Google Scholar] [PubMed]
- Beaulac, C.; Sachetelli, S.; Lagace, J. In-vitro bactericidal Efficacy of Sub-MIC Concentrations of Liposome-encapsulated Antibiotic against Gram-negative and Gram-positive Bacteria. J. Antimicrob. Chemother. 1998, 41, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Desjardins, A.; Chen, T.; Khalil, H.; Sayasith, K.; Lagace, J. Differential Behaviour of Fluid Liposomes toward Mammalian Epithelial Cells and Bacteria: Restriction of Fusion to Bacteria. J. Drug Target. 2002, 10, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Nikaido, H. Prevention of Drug Access to Bacterial Targets: Permeability Barriers and Active Efflux. Science 1994, 264, 382–388. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.W.; Thamphiwatana, S.; Zhang, L.; Obonyo, M. Mechanism of Antibacterial Activity of Liposomal Linolenic Acid against Helicobacter pylori. PLoS ONE 2015, 10, e0116519. [Google Scholar] [CrossRef] [PubMed]
- Pornpattananangkul, D.; Fu, V.; Thamphiwatana, S.; Zhang, L.; Chen, M.; Vecchio, J.; Gao, W.; Huang, C.M.; Zhang, L. In vivo Treatment of Propionibacterium acnes Infection with Liposomal Lauric Acids. Adv. Healthc. Mater. 2013, 2, 1322–1328. [Google Scholar] [CrossRef] [PubMed]
- Thamphiwatana, S.; Gao, W.; Obonyo, M.; Zhang, L. In vivo Treatment of Helicobacter pylori Infection with Liposomal Linolenic Acid Reduces Colonization and Ameliorates Inflammation. Proc. Natl. Acad. Sci. USA 2014, 111, 17600–17605. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.-M.; Chen, C.-H.; Pornpattananangkul, D.; Zhang, L.; Chan, M.; Hsieh, M.-F.; Zhang, L. Eradication of Drug Resistant Staphylococcus aureus by Liposomal Oleic Acids. Biomaterials 2011, 32, 214–221. [Google Scholar] [CrossRef] [PubMed]
- Obonyo, M.; Zhang, L.; Thamphiwatana, S.; Pornpattananangkul, D.; Fu, V.; Zhang, L. Antibacterial Activities of Liposomal Linolenic Acids against Antibiotic-resistant Helicobacter pylori. Mol. Pharm. 2012, 9, 2677–2685. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.H.; Nakatsuji, T.; Zouboulis, C.C.; Zhang, L.; Huang, C.M. An Innate Bactericidal Oleic Acid Effective against Skin Infection of Methicillin-resistant Staphylococcus aureus: A Therapy Concordant with Evolutionary Medicine. J. Microbiol. Biotechnol. 2011, 21, 391–399. [Google Scholar] [PubMed]
- Mbandi, E.; Brywig, M.; Shelef, L.A. Antilisterial Effects of Free Fatty Acids and Monolaurin in Beef Emulsions and Hot Dogs. Food Microbiol. 2004, 21, 815–818. [Google Scholar] [CrossRef]
- Zhang, H.; Lu, Z.; Zhang, L.; Bao, Y.; Zhan, X.; Feng, F.; Zheng, X. Antifungal Activity of a Food-grade Dilution-stable Microemulsion against Aspergillus niger. Lett. Appl. Microbiol. 2008, 47, 445–450. [Google Scholar] [CrossRef] [PubMed]
- Taylor, E.N.; Kummer, K.M.; Dyondi, D.; Webster, T.J.; Banerjee, R. Multi-scale Strategy to Eradicate Pseudomonas aeruginosa on Surfaces Using Solid Lipid Nanoparticles Loaded with Free Fatty Acids. Nanoscale 2014, 6, 825–832. [Google Scholar] [CrossRef] [PubMed]
- Silva, E.L.; Carneiro, G.; de Araújo, L.A.; de Jesus, M.; Trindade, V.; Yoshida, M.I.; Oréfice, R.L.; de Macêdo Farias, L.; de Carvalho, M.A.R.; Santos, S.G.D. Solid Lipid Nanoparticles Loaded with Retinoic Acid and Lauric Acid as an Alternative for Topical Treatment of Acne Vulgaris. J. Nanosci. Nanotechnol. 2015, 15, 792–799. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, E.M. Hydrogel: Preparation, characterization, and applications. J. Adv. Res. 2013, 6, 105–121. [Google Scholar] [CrossRef] [PubMed]
- Kristmundsdóttir, T.; Árnadóttir, S.G.; Bergsson, G.; Thormar, H. Development and evaluation of microbicidal hydrogels containing monoglyceride as the active ingredient. J. Pharm. Sci. 1999, 88, 1011–1015. [Google Scholar] [CrossRef] [PubMed]
- Thormar, H.; Bergsson, G.; Gunnarsson, E.; Georgsson, G.; Witvrouw, M.; Steingrimsson, O.; de Clercq, E.; Kristmundsdóttir, T. Hydrogels containing monocaprin have potent microbicidal activities against sexually transmitted viruses and bacteria in vitro. Sex. Transm. Infect. 1999, 75, 181–185. [Google Scholar] [CrossRef] [PubMed]
- Thorgeirsdottir, T.; Thormar, H.; Kristmundsdottir, T. The influence of formulation variables on stability and microbicidal activity of monoglyceride monocaprin. J. Drug Deliv. Sci. Technol. 2005, 15, 233–236. [Google Scholar] [CrossRef]
- Kristmundsdóttir, T.; Sigurdsson, P.; Thormar, H. Effect of buffers on the properties of microbicidal hydrogels containing monoglyceride as the active ingredient. Drug Dev. Ind. Pharm. 2003, 29, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Thorgeirsdóttir, T.Ó.; Kjøniksen, A.-L.; Knudsen, K.D.; Kristmundsdóttir, T.; Nyström, B. Viscoelastic and structural properties of pharmaceutical hydrogels containing monocaprin. Eur. J. Pharm. Biopharm. 2005, 59, 333–342. [Google Scholar] [CrossRef] [PubMed]
- Neyts, J.; Kristmundsdottir, T.; De Clercq, E.; Thormar, H. Hydrogels containing monocaprin prevent intravaginal and intracutaneous infections with HSV-2 in mice: Impact on the search for vaginal microbicides. J. Med. Virol. 2000, 61, 107–110. [Google Scholar] [CrossRef]
- Skulason, S.; Holbrook, W.P.; Thormar, H.; Gunnarsson, G.B.; Kristmundsdottir, T. A study of the clinical activity of a gel combining monocaprin and doxycycline: A novel treatment for herpes labialis. J. Oral Pathol. Med. 2012, 41, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Duggan, A.; Atherton, J.; Cockayne, A.; Balsitis, M.; Evison, S.; Hale, T.; Hawkey, C.; Spiller, R. Clarification of the link between polyunsaturated fatty acids and Helicobacter pylori-associated duodenal ulcer disease: A dietary intervention study. Br. J. Nutr. 1997, 78, 515–522. [Google Scholar] [CrossRef] [PubMed]
- Frieri, G.; Pimpo, M.T.; Palombieri, A.; Melideo, D.; Marcheggiano, A.; Caprilli, R.; D’Alessandro, A.; Seri, S. Polyunsaturated fatty acid dietary supplementation: An adjuvant approach to treatment of Helicobacter pylori infection. Nutr. Res. 2000, 20, 907–916. [Google Scholar] [CrossRef]
- Dayrit, C.S. Coconut oil in health and disease: Its and monolaurin’s potential as cure for HIV/AIDS. Indian Coconut J. 2000, 31, 19–24. [Google Scholar]
| Antimicrobial Lipid | Organism | Effects | Reference |
|---|---|---|---|
| Ethyl oleate | E. coli | • Bactericidal activity against Ps. aeruginosa and S. aureus, and induced membrane alterations against Ps. Aeruginosa. | [50] |
| Ps. aeruginosa | |||
| S. aureus | |||
| Monocaprin | C. jejuni | • ~1 mM monocaprin emulsions caused a greater than 6- to 7-log10 reduction in viable bacterial count of C. jejuni within 1 min. • Antimicrobial activity of monocaprin emulsions against C. coli, C. lari, Salmonella spp. and E. coli. was demonstrated. | [51] |
| C. coli | |||
| C. lari | |||
| Salmonella spp. | |||
| E. coli | |||
| Glycerol monolaurate | B. subtilis | • Increased antibacterial activity of GML emulsions versus free GML against B. subtilis and E. coli. | [52] |
| E. coli | |||
| Glycerol monolaurate | B. subtilis | • Demonstrated antibacterial effect of emulsions against B. subtilis, with improved activity in the presence of sodium lactate salt. | [53] |
| Glycerol monolaurate | E. coli | • Complete loss of viability of E. coli or S. aureus cells within 1 min caused by highly concentrated emulsions, and slow kinetics observed with 10-times diluted emulsions. • Induced release of nucleic acids due to bacterial membrane damage. | [54] |
| S. aureus | |||
| Glycerol monolaurate | B. subtilis | • GML emulsions are more potent against B. subtilis whereas free GML is stronger against E. coli. | [55] |
| E. coli | |||
| Glycerol monolaurate | S. maltophilia | • Greater antibacterial activity of GML emulsions against S. maltophilia versus the ceftazidime antibiotic, and enhanced activity with sodium benzoate as a hydrotrope. | [56] |
| E. coli | |||
| Glycerol monolaurate | B. subtilis | • Complete loss of viability of E. coli, S. aureus and B. subtilis cells within 1 min caused by GML emulsions. • Emulsions damaged bacterial cell walls. | [57] |
| E. coli | |||
| S. aureus | |||
| 1-monoacylglycerol (1-MAG) of capric (C10:0), undecanoic (C11:0), lauric (C12:0),, myristic (C14:0) acids | B. cereus | • Enhanced antimicrobial activities of emulsions against Gram-negative strains versus 1-MAGs and opposite trend was observed with Gram-positive stains. • Best antibacterial activity against both bacterial types was observed with 1-MAG C12:0 emulsions. • 10 mg/L concentration was determined to be the limit for moderate toxicity (40%–60% cell survival). | [58] |
| B. subtilis | |||
| E. faecalis | |||
| S. aureus | |||
| M. luteus | |||
| C. freundii | |||
| E. coli | |||
| P. aeruginosa | |||
| S. entérica | |||
| S. marcescens |
| Antimicrobial Lipid | Organism | Study Design | Effects | Reference |
|---|---|---|---|---|
| Linolenic acid, Stearic acid, Oleic acid | H. pylori | In Vitro | • Demonstrated efficacy of LipoLLA with MBC value of 200 µg/mL. | [65] |
| • Significant effect of LipoLLA on increasing outer membrane permeability of H. pylori. | ||||
| Linolenic acid | H. pylori | In Vitro | • Effective in killing both spiral and coccoid forms of the bacteria based on membrane disruption. | [66] |
| • LipoLLA has higher barrier to development of drug-resistant strains than free LLA. | ||||
| Lauric acid | P. acnes | In Vitro | • MBC value of LipoLA at 51 µg/mL against P. acnes. | [50] |
| • Established importance of critical molar fraction of free fatty acids in LipoFAs. | ||||
| Linolenic acid | H. pylori | In Vivo | • MBC values for LipoLLA and LLA of 65 µg/mL and 80 µg/mL, respectively. | [67] |
| • Significant efficacy of LipoLLA in vivo with excellent biocompatibility. | ||||
| Lauric acid | P. acnes | In Vivo | • Effective therapeutic efficacy of 2 mg/mL LipoLA in topical formulation. | [68] |
| • No irritation of normal mouse skin by LipoLLA. | ||||
| Oleic acid | S. aureus (MRSA) | In Vivo | • 12-fold increase in in vitro efficacy of OA in liposomal formulation versus free OA. | [69] |
| • High in vivo efficacy of LipoOA in treatment of MRSA skin infections. |
| Antimicrobial Agents | Organism | Study Design | Effects | Reference |
|---|---|---|---|---|
| Caprylic acid, Capric acid, Undecylenic acid, Lauric acid, Myristic acid, Palmitoleic acid, Oleic acid, 1-monoglyceride of each fatty acid | Herpes simplex virus type 1 (HSV-1) | In Vitro | • Monocaprin, the 1-monoglyceride of capric acid, had the most potent antiviral activity compounds against HSV-1. | [75] |
| • Significant antiviral activity of hydrogel formulation against HSV-1 with > 106-fold inactivation in 1 min. | ||||
| • Reduced cytotoxicity in human cells with monocaprin-containing hydrogel compared to commercial spermicidal product. | ||||
| Monocaprin | Herpes simplex virus type 2 (HSV-2), HIV-1, C. trachomatis, N. gonorrhoeae | In Vitro | • Potent inactivation efficacy against HSV-2, HIV-1, N. gonorrhoeae, and C. Trachomatis. | [76] |
| • No toxicity of the monocaprin-containing hydrogel against rabbit vaginal mucosa. | ||||
| Monocaprin | Herpes simplex virus (HSV-1) | In Vitro | • Enhanced stability of monocaprin in pharmaceutical formulations in the presence of carbomer 974P. | [77] |
| • Reduced effect on HSV-1 inactivation with increasing amount of polysorbate 20 surfactant. | ||||
| Monocaprin | Herpes simplex virus (HSV-1) | In Vitro | • Insignificant effect of buffer on antiviral activity of hydrogel formulation against HSV-1. | [78] |
| • Decreased hydrogel viscosity in the presence of buffers. | ||||
| Monocaprin | N.A. | In Vitro | • Significant effect of solution pH on structural and rheological properties of hydrogels. | [79] |
| Monocaprin | Herpes simplex virus (HSV-2) | In Vivo | • Potent antiviral activity of hydrogel formulation against intravaginal and intracutaneous HSV-2 infection in mice without skin irritation. | [80] |
| Monocaprin Doxycyclin | Herpes labialis | In Vivo | • Effective clinical treatment of hydrogel formulation containing monocaprin and doxycyclin for treatment of Herpes labialis. | [81] |
| • Significant decrease in healing time and pain versus free monocaprin and placebo hydrogel. |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jackman, J.A.; Yoon, B.K.; Li, D.; Cho, N.-J. Nanotechnology Formulations for Antibacterial Free Fatty Acids and Monoglycerides. Molecules 2016, 21, 305. https://doi.org/10.3390/molecules21030305
Jackman JA, Yoon BK, Li D, Cho N-J. Nanotechnology Formulations for Antibacterial Free Fatty Acids and Monoglycerides. Molecules. 2016; 21(3):305. https://doi.org/10.3390/molecules21030305
Chicago/Turabian StyleJackman, Joshua A., Bo Kyeong Yoon, Danlin Li, and Nam-Joon Cho. 2016. "Nanotechnology Formulations for Antibacterial Free Fatty Acids and Monoglycerides" Molecules 21, no. 3: 305. https://doi.org/10.3390/molecules21030305
APA StyleJackman, J. A., Yoon, B. K., Li, D., & Cho, N.-J. (2016). Nanotechnology Formulations for Antibacterial Free Fatty Acids and Monoglycerides. Molecules, 21(3), 305. https://doi.org/10.3390/molecules21030305







