Large Scale Screening of Ethnomedicinal Plants for Identification of Potential Antibacterial Compounds
Abstract
:1. Introduction
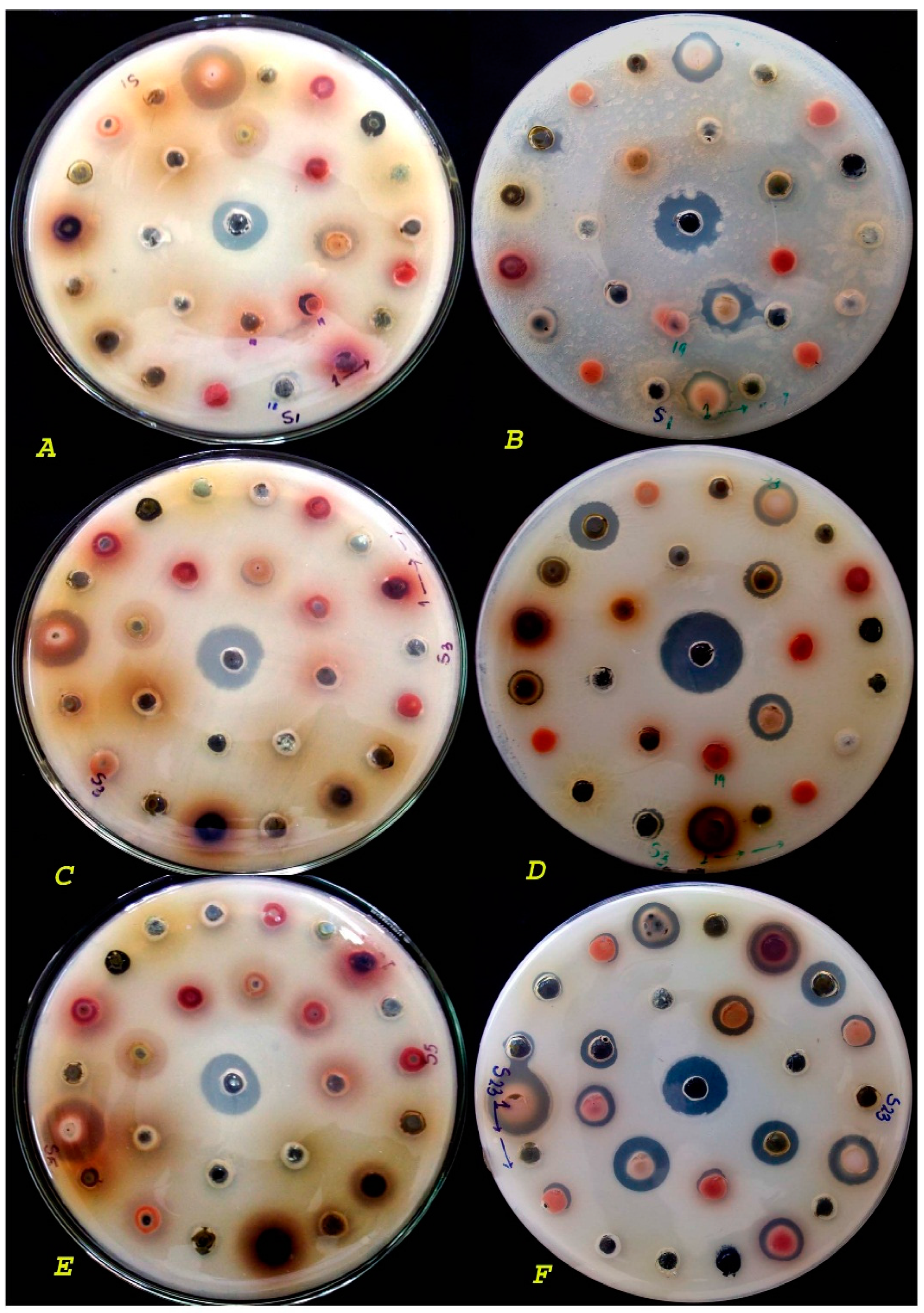
2. Results and Discussion
3. Experimental Section
3.1. Study Area
3.2. Processing
Preparation of Plant Extracts
3.3. Antibacterial Activity
3.3.1. Test Bacterial Strains
3.3.2. Maintenance of Bacteria
3.4. Antibiotics
3.5. Sensitivity Tests
3.6. Agar Cup Method
3.7. Minimum Inhibitory Concentration (MIC)
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| CFU | Colony forming unit |
| MDR | Multiple drug resistance |
| MHA | Muller-Hinton agar |
| MTCC | Microbial type culture collection |
| NA | Nutrient agar |
References
- Mohanta, T.K.; Occhipinti, A.; Atsbaha Zebelo, S.; Foti, M.; Fliegmann, J.; Bossi, S.; Maffei, M.E.; Bertea, C.M. Ginkgo biloba responds to herbivory by activating early signaling and direct defenses. PLoS ONE 2012, 7, e32822. [Google Scholar]
- Mohanta, T.K.; Tamboli, Y.; Zubaidha, P.K. Phytochemical and medicinal importance of Ginkgo biloba L. Nat. Prod. Res. 2014, 28, 746–752. [Google Scholar] [CrossRef] [PubMed]
- Verma, S.; Singh, S.P. Current and future status of herbal medicines. Vet. World 2008, 1, 347–350. [Google Scholar] [CrossRef]
- Dubey, N.K.; Kumar, R.; Tripathi, P. Global promotion of herbal medicine: India’s opportunity. Curr. Sci. 2004, 86, 37–41. [Google Scholar]
- Cown, M.M. Plant products as antimicrobial agents. Clin. Microbiol. Rev. 1999, 12, 564–582. [Google Scholar]
- Parekh, J.; Chanda, S.V. In vitro antimicrobial activity and phytochemical analysis of some Indian medicinal plants. Turk. J. Biotechnol. 2008, 31, 53–58. [Google Scholar]
- Fabry, W.; Okemo, P.O.; Ansorg, R. Antibacterial activity of East African medicinal plants. J. Ethnopharmacol. 1998, 60, 79–84. [Google Scholar] [CrossRef]
- Vlietinck, A.J.; Vanden Berghe, D.A. Can ethnopharmacology contribute to the development of antiviral drugs? J. Ethnopharmacol. 1991, 32, 141–153. [Google Scholar] [CrossRef]
- Fabricant, D.S.; Farnsworth, N.R. The Value of Plants Used in Traditional Medicine for Drug Discovery. Environ. Heal. 2001, 109, 69–75. [Google Scholar]
- Vuorela, P.; Leinonen, M.; Saikku, P.; Tammela, P.; Rauha, P.; Wennberg, T.; Vuorela, H. Natural Products in the Process of Finding New Drug Candidates. Curr. Med. Chem. 2004, 11, 1375–1389. [Google Scholar] [CrossRef]
- Eloff, J.N.; Katerere, D.R.; McGaw, L.J. The biological activity and chemistry of the southern African Combretaceae. J. Ethnopharmacol. 2008, 119, 686–699. [Google Scholar] [CrossRef] [PubMed]
- Pauw, E.; Eloff, J. Which tree orders in southern Africa have the highest antimicrobial activity and selectivity against bacterial and fungal pathogens of animals? BMC Complement. Altern. Med. 2014, 14. [Google Scholar] [CrossRef] [PubMed]
- Lewis, K.; Ausubel, F.M. Prospects for plant-derived antibacterials. Nat. Biotech. 2006, 24, 1504–1507. [Google Scholar] [CrossRef] [PubMed]
- Panda, S.K. Ethno-medicinal uses and screening of plants for antibacterial activity from Similipal Biosphere Reserve, Odisha, India. J. Ethnopharmacol. 2014, 151, 158–175. [Google Scholar] [CrossRef] [PubMed]
- Padhi, L.; Panda, S. Antibacterial activity of Eleutherine bulbosa (Miller) Urban (Iridaceae) against multidrug resistant bacteria. J. Acute Med. 2015, 5, 53–61. [Google Scholar] [CrossRef] [Green Version]
- Silva, G.; Lee, I.; Kinghor, A. Special problems with the extraction of plants. In Methods in Biotechnology; Cannel, R., Ed.; Humana Press Inc: Totowa, NJ, USA, 1998; pp. 343–363. [Google Scholar]
- Panda, S.K.; Niranjan, P.; Gunanidhi, S.; Bastia, A.K.; Dutta, S.K. Anti-diarrheal activities of medicinal plants of Similipal Biosphere Reserve, Odisha, India. Int. J. Med. Aromat. Plants 2012, 2, 123–134. [Google Scholar]
- Lin, J.; Opoku, A.R.; Geheeb-Keller, M.; Hutchings, A.D.; Terblanche, S.E.; Jäger, A.K.; Van Staden, J. Preliminary screening of some traditional zulu medicinal plants for anti-inflammatory and anti-microbial activities. J. Ethnopharmacol. 1999, 68, 267–274. [Google Scholar] [CrossRef]
- Romero, C.D.; Chopin, S.F.; Buck, G.; Martinez, E.; Garcia, M.; Bixby, L. Antibacterial properties of common herbal remedies of the southwest. J. Ethnopharmacol. 2005, 99, 253–257. [Google Scholar] [CrossRef] [PubMed]
- Nikaido, H. Outer membrane In Escherichia coli and Salmonella. In Cellular and Molecular Biology; Neidhardt, F.C., Ed.; ASM Press: Washington, DC, USA, 1996; pp. 29–47. [Google Scholar]
- Yerra, R.; Gupta, M.; Mazumder, U. In Vitro Lipid Peroxidation and Antimicrobial Activity of Mucuna pruriens Seeds. Iran. J. Pharmacol. Ther. 2005, 4, 32–35. [Google Scholar]
- Kuete, V.; Nguemeving, J.R.; Beng, V.P.; Azebaze, A.G.B.; Etoa, F.-X.; Meyer, M.; Bodo, B.; Nkengfack, A.E. Antimicrobial activity of the methanolic extracts and compounds from Vismia laurentii De Wild (Guttiferae). J. Ethnopharmacol. 2007, 109, 372–379. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, I.; Mehmood, Z.; Mohammad, F. Screening of some Indian medicinal plants for their antimicrobial properties. J. Ethnopharmacol. 1998, 62, 183–193. [Google Scholar] [CrossRef]
- Valsaraj, R.; Pushpangadan, P.; Smitt, U.W.; Adsersen, A.; Nyman, U. Antimicrobial screening of selected medicinal plants from India. J. Ethnopharmacol. 1997, 58, 75–83. [Google Scholar] [CrossRef]
- Samy, R.P.; Ignacimuthu, S. Antibacterial activity of some folklore medicinal plants used by tribals in Western Ghats of India. J. Ethnopharmacol. 2000, 69, 63–71. [Google Scholar] [CrossRef]
- Srinivasan, D.; Nathan, S.; Suresh, T.; Lakshmana Perumalsamy, P. Antimicrobial activity of certain Indian medicinal plants used in folkloric medicine. J. Ethnopharmacol. 2001, 74, 217–220. [Google Scholar] [CrossRef]
- Ahmad, I.; Beg, A.Z. Antimicrobial and phytochemical studies on 45 Indian medicinal plants against multi-drug resistant human pathogens. J. Ethnopharmacol. 2001, 74, 113–123. [Google Scholar] [CrossRef]
- Jeevan Ram, A.; Bhakshu, L.M.; Venkata Raju, R.R. In vitro antimicrobial activity of certain medicinal plants from Eastern Ghats, India, used for skin diseases. J. Ethnopharmacol. 2004, 90, 353–357. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.P.; Chauhan, N.S.; Padh, H.; Rajani, M. Search for antibacterial and antifungal agents from selected Indian medicinal plants. J. Ethnopharmacol. 2006, 107, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Parekh, J.; Chanda, S.V. Antibacterial activity of aqueous and alcoholic extracts of 34 Indian medicinal plants against some Staphylococcus species. Turk. J. Biol. 2008, 32, 63–71. [Google Scholar]
- Ahmad, I.; Aqil, F. In vitro efficacy of bioactive extracts of 15 medicinal plants against ESβL-producing multidrug-resistant enteric bacteria. Microbiol. Res. 2007, 162, 264–275. [Google Scholar] [CrossRef] [PubMed]
- Posadzki, P.; Watson, L.K.; Ernst, E. Adverse effects of herbal medicines: An overview of systematic reviews. Clin. Med. J. R. Coll. Phys. Lond. 2013, 13, 7–12. [Google Scholar] [CrossRef]
- Methods to study antimicrobial and antioxidant properties of medicinal plants. In Advances in Natural Products; Panda, S. (Ed.) Studium Press LLC: Houston, TX, USA, 2015; pp. 179–230.
- Mohanta, T.; Patra, J.; Rath, S. Evaluation of antimicrobial activity and phytochemical screening of oils and nuts of Semicarpus anacardium. Sci. Res. Essay 2007, 2, 486–490. [Google Scholar]
- Bauer, A.W.; Kirby, W.M.; Sherris, J.C.; Turck, M. Antibiotic susceptibility testing by a standardized single disk method. Am. J. Clin. Pathol. 1966, 45, 493–496. [Google Scholar] [PubMed]
- Sample Availability: Samples of the plant extracts are available from the authors.

| Scrutiny | No. of Extracts Reported as Antibacterial (%) | ||
|---|---|---|---|
| Element | Methanol Extract | Aqueous Extract | |
| Total number of plant species tested—22 | Gram positive | 146 (56.58%) | 89 (34.49%) |
| Total number of Genus tested—177 | Gram negative | 137 (53.10%) | 102 (39.53%) |
| Total number of family tested—83 | B. cereus | 108 (41.86%) | 50 (19.37%) |
| Total number of parts tested = 258 | S. aureus | 124 (48.06%) | 76 (29.45%) |
| Leaves-125; Bark-19; Whole part-08; Stem-04 | E. coli | 68 (26.35%) | 45 (17.44%) |
| Root-04; Rhizome-03; Fruit-03 and Bulb-01 | S. typhimurium | 65 (25.19%) | 41 (15.89%) |
| Total number of methanol extracts active—165 | S. dysentriae | 50 (19.37%) | 22 (8.52%) |
| Total number of aqueous extracts active—127 | S. flexneri | 66 (25.58%) | 28 (10.85%) |
| Number of species do not show activity—90 species | S. sonnei | 47 (18.21%) | 24 (9.30%) |
| Number of extracts do not show activity | V. cholerae | 72 (27.90%) | 38 (14.72%) |
| (93 methanol + 131 aqueous = 224) | Zone ≥ 20 mm | 10 (3.87%) | 0 |
| Total number of family show activity—68 | Zone 15–20 mm | 34 (13.17%) | 9 (3.48%) |
| Total number of family do not show activity—15 | Zone < 15 | 160 (62.01%) | 121 (46.89%) |
| Plant Description | Zone of Inhibition in mm | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| PU | E | Bc | Sa | Ec | St | Sd | Sf | Ss | Vc | |
| Acanthaceae | ||||||||||
| Andrographis paniculata (Burm. f.) Nees | Lf | A | 14 | 12 | 11 | 10 | 12 | - | 14 | - |
| M | 12 | 12 | 14 | 13 | - | 12 | 16 | - | ||
| St | A | 12 | 12 | 12 | 12 | - | - | - | - | |
| M | 12 | 14 | 16 | - | 10 | 14 | 15 | 10 | ||
| Barleria cristata L. | Lf | A | 12 | 12 | - | - | - | - | - | - |
| M | 14 | 18 | - | - | - | - | - | - | ||
| Adhatoda vasica Nees | Lf | A | 11 | 10 | - | 12 | 12 | - | 12 | 11 |
| M | 14 | 12 | 10 | 10 | 10 | 10 | 12 | 10 | ||
| Acoraceae | ||||||||||
| Acorus calamus L. | Rh | A | - | - | - | - | - | - | - | 09 |
| M | 12 | 18 | - | - | - | 10 | - | 12 | ||
| Alangiaceae | ||||||||||
| Alangium salvifolium (C.B.Clarke) W.W.Sm. & Cave | Lf | A | 12 | 10 | 10 | - | - | - | - | - |
| M | 14 | 16 | 12 | 12 | 12 | 12 | - | - | ||
| Alpinia galangal (Linn.) Wild. | Lf | A | - | - | - | - | - | - | - | - |
| M | 14 | 12 | 10 | 10 | - | 16 | - | 14 | ||
| Amaranthaceae | ||||||||||
| Achyranthes aspera L. | Wp | A | - | - | - | 11 | - | - | - | 09 |
| M | 14 | 12 | 12 | 12 | - | - | - | 08 | ||
| Achyranthes bidentata L. Blume | Wp | A | - | - | - | - | - | - | - | - |
| M | 12 | 12 | - | - | - | - | - | - | ||
| Cyathula prostrata L. Blume | Lf | A | - | - | - | - | - | - | - | - |
| M | - | - | - | - | - | - | - | 10 | ||
| Anacardiaceae | ||||||||||
| Buchanania lanzan Spreng | Bk | A | 15 | 12 | - | - | - | - | - | - |
| M | 16 | 14 | - | 13 | - | 12 | 14 | 10 | ||
| Lannea coromandelica (Houtt.) Merr. | Bk | A | 12 | 12 | - | 09 | 10 | - | - | - |
| M | - | 12 | - | 14 | 10 | 10 | - | 10 | ||
| Mangifera indica L. | Lf | A | - | - | - | - | - | - | - | - |
| M | 10 | 14 | - | - | - | - | - | - | ||
| Semecarpus anacardium L.f. | Fr | A | 11 | 14 | - | 12 | - | - | - | - |
| M | 12 | 15 | - | 13 | - | 14 | - | 11 | ||
| Spondias pinnata (L.f.) Kurz | Lf | A | - | - | 10 | - | - | - | - | - |
| M | 10 | 14 | 11 | 12 | - | 14 | - | 13 | ||
| Annonaceae | ||||||||||
| Annona reticulata L. | Lf | A | - | - | - | - | - | 12 | - | 12 |
| M | 12 | 12 | - | - | 12 | 13 | - | 12 | ||
| Annona squamosa L. | Lf | A | - | 12 | - | - | - | - | 12 | - |
| M | 13 | 16 | - | - | 12 | - | 14 | 12 | ||
| Apiaceae | ||||||||||
| Centella asiatica (L.) Urb. | Wp | A | 12 | 12 | 10 | - | - | - | - | 10 |
| M | 13 | 14 | 10 | - | 12 | - | 12 | 14 | ||
| Eryngium foetidum L. | Lf | A | 09 | 12 | 12 | - | 13 | - | 11 | - |
| M | 10 | 14 | 13 | - | 13 | - | 12 | - | ||
| St | A | 11 | 13 | 12 | - | - | - | 09 | - | |
| M | 12 | 18 | 14 | - | 12 | 12 | 11 | 10 | ||
| Apocyanaceae | ||||||||||
| Alstonia scholaris (L.) R.Br. | Lf | A | - | - | - | - | - | - | - | - |
| M | 14 | 11 | - | - | - | 10 | - | 12 | ||
| Alstonia venenata R.Br. | Lf | A | - | - | - | - | - | - | - | - |
| M | 12 | - | - | - | - | 14 | - | 10 | ||
| Holarrhena antidysenterica Wall ex. A.DC. | Lf | A | 18 | 12 | 12 | 14 | - | - | 11 | - |
| M | 15 | 12 | 12 | 14 | - | 12 | 12 | 12 | ||
| Ichnocarpus frutescens (L.) W.T.Aiton | Lf | A | - | - | - | - | - | - | - | - |
| M | 12 | 11 | - | - | - | - | 12 | - | ||
| Rauvolfia serpentina (L.) Benth. ex Kurz | Rt | A | - | - | - | - | - | - | - | - |
| M | - | - | 10 | - | - | - | 12 | - | ||
| Araceae | ||||||||||
| Colocasia esculenta (L.) Schott | Rh | A | - | - | 09 | - | 12 | - | - | 09 |
| M | - | - | 12 | 12 | 14 | - | - | 12 | ||
| Aristolochiaceae | ||||||||||
| Aristolochia indica L. | Lf | A | - | 12 | - | - | - | - | - | - |
| M | 12 | 10 | - | - | 10 | 10 | - | - | ||
| Asclepiadaceae | ||||||||||
| Calotropis procera (Aiton) Dryand. | Lt | A | - | 12 | - | - | - | - | - | 12 |
| M | - | 14 | - | - | - | - | - | 12 | ||
| Pergularia demia (Forssk.) Chiov. | Lf | A | - | - | - | - | - | - | 12 | - |
| M | 11 | 12 | - | - | - | - | 13 | 11 | ||
| Hemidesmus indicus (L.) R. Br. ex Schult. | Lf | A | - | - | - | - | - | - | - | - |
| M | 16 | 12 | 18 | - | - | 14 | 13 | 13 | ||
| St | A | - | - | - | - | - | - | 12 | - | |
| M | 14 | - | - | - | - | - | 14 | - | ||
| Asteraceae | ||||||||||
| Ageratum conyzoides (L.) L. | Wp | A | - | 11 | 12 | 12 | 11 | - | 12 | - |
| M | 10 | 16 | 10 | 12 | - | 13 | 12 | 10 | ||
| Blumea lacera (Burm.f.) DC. | Lf | A | - | - | - | - | - | - | - | - |
| M | - | 12 | - | - | - | - | - | - | ||
| Chrysanthellum americanum (L.) Vatke | Lf | A | 10 | 12 | - | - | - | - | - | - |
| M | 11 | 13 | - | - | - | - | - | - | ||
| Elephantopus scaber L. | Lf | A | 14 | 10 | - | - | 11 | - | 08 | - |
| M | 12 | 12 | - | - | 14 | 11 | 10 | 9 | ||
| Tridax procumbens (L.) L. | Lf | A | - | - | - | - | - | - | - | 13 |
| M | 13 | 14 | - | - | - | 11 | - | 12 | ||
| Vernonia aspera (Roxb.) Ham. | Lf | A | 09 | 12 | - | - | - | - | - | - |
| M | 11 | 14 | - | - | - | - | - | - | ||
| Vernonia squarrosa Dinter ex Merxm. | Lf | A | - | - | - | - | 12 | - | - | 10 |
| M | - | - | - | - | 11 | - | - | 12 | ||
| Baccharoides anthelmintica (L.) Moench | Lf | A | - | - | 10 | - | - | - | - | 10 |
| M | - | - | 14 | - | - | - | - | 12 | ||
| Bignoniaceae | ||||||||||
| Oroxylum indicum (L.) Kurz | Bk | A | 12 | 10 | - | - | - | 12 | 12 | - |
| M | 14 | 12 | - | - | 13 | 12 | 16 | 14 | ||
| Caesalpiniaceae | ||||||||||
| Bauhinia variegata L. | Lf | A | - | - | - | - | - | - | - | - |
| M | 11 | 10 | - | - | - | - | - | - | ||
| Cassia fistula L. | Lf | A | 13 | 12 | 10 | 09 | 11 | 12 | 08 | 12 |
| M | 12 | 14 | 12 | 12 | 10 | 14 | 12 | 13 | ||
| Cassia occidentalis L. | Lf | A | - | 12 | 10 | 11 | - | - | - | - |
| M | - | 11 | 12 | - | - | - | - | - | ||
| Cassia tora L. | Lf | A | - | - | - | - | - | - | - | - |
| M | - | 12 | 12 | - | 12 | - | - | - | ||
| Saraca asoca (Roxb.) Willd. | Lf | A | - | - | - | - | - | - | - | - |
| M | - | 10 | - | - | - | - | - | - | ||
| Tamarindus indica L. | Lf | A | 10 | 11 | - | 10 | - | - | - | 12 |
| M | 12 | 10 | 08 | 12 | - | - | - | 14 | ||
| Calophyllaceae | ||||||||||
| Mesua ferrea L. | Lf | A | 12 | 10 | 10 | 12 | 12 | - | 12 | - |
| M | 12 | 10 | 10 | 10 | 12 | - | 12 | - | ||
| Capparaceae | ||||||||||
| Capparis zeylanica L. | Lf | A | - | - | - | - | - | - | - | - |
| M | - | 10 | - | - | - | - | - | - | ||
| Cleome viscosa L. | Lf | A | 10 | 11 | - | - | 10 | - | - | - |
| M | 17 | 12 | 12 | - | 11 | 13 | 12 | 10 | ||
| Celastraceae | ||||||||||
| Celastrus paniculatus Willd. | Lf | A | - | 12 | - | - | - | 13 | - | 15 |
| M | 12 | 16 | 10 | 10 | - | 15 | - | 18 | ||
| Euonymus glaber Roxb. | Lf | A | - | 12 | 12 | 12 | - | - | - | 13 |
| M | 12 | 20 | 14 | 16 | - | 12 | - | 16 | ||
| Clusiaceae | ||||||||||
| Garcinia cowa Roxb. ex Choisy | Lf | A | 12 | 11 | 10 | 14 | - | 12 | - | - |
| M | 12 | 13 | 10 | 12 | 10 | 10 | 12 | 10 | ||
| Cochlospermaceae | ||||||||||
| Cochlospermum religiosum (L.) Alston | Lf | A | - | - | - | - | - | - | - | |
| M | - | 10 | - | - | - | - | - | |||
| Combretaceae | ||||||||||
| Anogeissus latifolia (Roxb. ex DC.) Wall. Ex Guillem. & Perr. | Lf | A | 12 | 11 | 10 | 12 | 12 | - | - | - |
| M | 14 | 08 | 11 | 12 | 12 | 10 | - | 11 | ||
| Combretum roxburghii Spreng. | Lf | A | - | - | - | - | - | - | - | - |
| M | - | 22 | - | - | 12 | 14 | - | 16 | ||
| Terminalia alata Heyne ex Roth | Bk | A | - | - | - | - | - | 12 | - | 14 |
| M | 14 | 12 | - | - | - | 11 | 12 | 12 | ||
| Terminalia arjuna (Roxb. ex DC.) Wight & Arn. | Bk | A | - | 12 | - | 12 | 14 | 11 | 11 | 12 |
| M | 11 | 12 | 10 | 12 | 15 | 12 | 14 | 14 | ||
| Terminalia bellirica (Gaertn.) Roxb. | Bk | A | 10 | 12 | 11 | 13 | - | - | 10 | - |
| M | - | 14 | - | 12 | - | - | - | - | ||
| Terminalia chebula Retz. | Bk | A | - | - | - | - | - | - | - | - |
| M | 12 | - | - | - | - | 12 | 10 | - | ||
| Terminalia tomentosa Wight & Arn. | Lf | A | - | - | - | - | - | - | - | 10 |
| M | 13 | 10 | 12 | 14 | - | 12 | - | 12 | ||
| Commelinaceae | ||||||||||
| Commelina paludosa Blume | Lf | A | 14 | - | 12 | - | - | - | - | |
| M | 11 | 10 | - | 13 | - | - | - | - | ||
| Convolvulaceae | ||||||||||
| Erycibe paniculata Roxb. | Lf | A | - | 10 | - | - | - | - | - | 10 |
| M | 10 | 10 | 12 | 12 | - | - | - | 14 | ||
| Crassulaceae | ||||||||||
| Kalanchoe pinnata (Lam.) Pers. | Lf | A | - | - | - | - | - | - | - | - |
| M | 12 | 12 | - | - | - | - | - | - | ||
| Cucurbitaceae | ||||||||||
| Coccinia grandis (L.) Voigt | Lf | A | - | 12 | - | 11 | - | - | - | - |
| M | 12 | 11 | - | 12 | - | 12 | - | - | ||
| Momordica charantia L. | Lf | A | 10 | - | - | - | - | - | - | - |
| M | 10 | - | - | - | 12 | 12 | - | - | ||
| Cyperaceae | ||||||||||
| Cyperus rotundus L. | Lf | A | 11 | 10 | - | 10 | - | - | - | - |
| M | 13 | 12 | - | 12 | 10 | - | - | - | ||
| Dilleniaceae | ||||||||||
| Dillenia pentogyna Roxb. | Lf | A | 12 | - | - | 12 | - | - | - | - |
| M | 10 | 12 | - | 12 | 12 | - | 10 | - | ||
| Dipterocarpaceae | ||||||||||
| Shorea robusta Gaertn. | Lf | A | 10 | - | - | - | - | 12 | - | 11 |
| M | 12 | - | - | 12 | - | 12 | - | 13 | ||
| Ebenaceae | ||||||||||
| Diospyros malabarica (Desr.) Kostel | Lf | A | - | - | - | - | - | - | - | - |
| M | 11 | - | - | 12 | - | - | - | - | ||
| Diospyros melanoxylon Roxb. | Lf | A | - | - | 10 | 11 | - | - | 12 | - |
| M | 10 | 15 | 18 | 12 | - | - | 14 | - | ||
| Bk | A | 14 | 10 | 10 | 12 | - | 12 | 13 | 11 | |
| M | 15 | 11 | 22 | 16 | - | 10 | 16 | 10 | ||
| Diospyros montana Roxb. | Lf | A | - | - | - | - | - | 10 | - | - |
| M | 12 | - | - | - | - | 10 | - | 10 | ||
| Diospyros sylvatica Roxb. | Lf | A | - | 12 | - | - | - | - | - | - |
| M | 14 | 14 | 10 | 20 | - | 14 | - | 18 | ||
| Euphorbiaceae | ||||||||||
| Antidesma ghaesembilla Gaertn. | Lf | A | - | - | - | - | - | - | - | - |
| M | 13 | 12 | - | - | - | - | - | - | ||
| Cleistanthus collinus (Roxb.) Benth ex Hook. f. | Lf | A | 12 | 10 | 12 | - | - | 12 | - | 12 |
| M | 10 | 12 | 14 | 14 | - | 12 | 10 | 12 | ||
| Croton caudatus Geiseler | Lf | A | - | - | - | - | - | - | - | - |
| M | 10 | - | - | - | - | - | - | - | ||
| Croton roxburghii Wall. | Lf | A | 10 | 16 | 10 | - | 12 | 12 | - | 13 |
| M | 12 | 14 | 17 | 15 | 15 | 13 | 12 | 10 | ||
| Croton roxburghii Wall. | Bk | A | - | 12 | 15 | 14 | - | - | 14 | - |
| M | 12 | 14 | 20 | 15 | - | - | 17 | - | ||
| Emblica officinalis Gaertn. | Lf | A | - | 12 | 10 | 10 | - | - | - | - |
| M | 11 | 10 | 12 | - | 12 | - | - | - | ||
| Euphorbia hirta L. | Lf | A | - | 10 | 12 | - | 10 | 12 | - | - |
| M | 10 | - | 14 | - | 12 | 10 | - | - | ||
| Jatropha gossypiifolia L. | Lf | A | - | - | - | - | - | - | - | - |
| M | - | - | 10 | 12 | - | - | - | - | ||
| Macaranga peltata (Roxb.) Mull. Arg. | Lf | A | - | - | - | - | - | - | - | - |
| M | - | - | 10 | - | - | - | - | - | ||
| Mallotus philippensis (Lam.) Mull. Arg. | Lf | A | - | - | - | - | - | - | - | - |
| M | 12 | 14 | - | - | - | - | - | - | ||
| Phyllanthus fraternus G. L. Webster | Wp | A | - | - | - | - | - | - | - | 10 |
| M | - | - | - | - | - | - | - | - | ||
| Ricinus communis L. | Lf | A | 12 | - | - | - | - | 10 | 10 | - |
| M | 10 | 14 | - | 12 | 10 | 12 | 12 | 10 | ||
| Flacourtiaceae | ||||||||||
| Flacourtia jangomas (Lour.) Raeusch. | Lf | A | - | 12 | 10 | - | - | 12 | - | 11 |
| M | - | 12 | 12 | - | - | - | - | - | ||
| Fabaceae | ||||||||||
| Butea monsperma (Lam.) Taub. | Lf | A | - | 10 | - | - | - | - | - | - |
| M | 12 | 10 | - | - | - | - | - | - | ||
| Butea superba Roxb. | Lf | A | - | 10 | 10 | 10 | - | - | - | - |
| M | 12 | 10 | 10 | - | - | - | - | - | ||
| Clitoria ternatea L. | Lf | A | - | - | - | - | - | - | - | - |
| M | - | 10 | - | - | - | - | - | - | ||
| Dalbergia latifolia Roxb. | Bk | A | - | - | - | - | - | - | - | - |
| M | - | 12 | 12 | - | - | - | - | - | ||
| Dalbergia volubilis Roxb. | Bk | A | - | - | - | - | - | - | - | - |
| M | - | 12 | - | - | - | - | - | - | ||
| Desmodium gangeticum (L.) DC. | Lf | A | 12 | - | 08 | - | 10 | - | - | - |
| M | 10 | 12 | 10 | 10 | 12 | - | - | - | ||
| Desmodium oojeinense (Roxb.) H. Ohashi | Lf | A | - | - | - | - | - | - | - | - |
| M | - | 10 | - | - | - | - | - | - | ||
| Desmodium pulchellum (L.) Benth. | Lf | A | - | - | 10 | - | - | - | - | - |
| M | - | 12 | 12 | - | - | - | - | - | ||
| Flemingia nana Roxb. | Rt | A | 15 | 11 | - | - | - | 12 | - | 10 |
| M | 14 | 12 | 10 | 10 | - | 12 | - | 12 | ||
| Glycyrrhiza glabra (L.) | Bk | A | - | 11 | - | - | - | - | - | 10 |
| M | 12 | 10 | - | - | - | - | - | 18 | ||
| Indigofera cassioides DC. | Lf | A | - | - | - | - | - | - | - | - |
| M | 14 | 12 | - | 10 | - | - | - | 10 | ||
| Indigofera glabra L. | Lf | A | - | 11 | - | 09 | - | - | - | - |
| M | - | - | - | - | - | - | - | - | ||
| Millettia extensa (Benth) Baker | Lf | A | - | 12 | - | - | - | - | - | - |
| M | 11 | 14 | 20 | - | 10 | 11 | - | - | ||
| Pterocarpus marsupium Roxb. | Bk | A | - | 12 | - | 10 | - | 10 | - | - |
| M | 12 | - | - | 12 | - | 14 | - | - | ||
| Tephrosia purpurea (L.) Pers. | Fr | A | - | - | - | 12 | - | - | - | - |
| M | - | - | - | 10 | - | - | - | - | ||
| Gentianaceae | ||||||||||
| Canscora decurrens Daizell | Wp | A | - | - | - | - | - | - | - | - |
| M | - | 12 | 09 | - | - | - | - | - | ||
| Iridaceae | ||||||||||
| Eleutherine bulbosa (Mill.) Urb. | Bl | A | 18 | 16 | 10 | 17 | - | 12 | - | - |
| M | 25 | 18 | 14 | 15 | 11 | 17 | - | - | ||
| Lamiaceae | ||||||||||
| Hyptis suaveolens (L.) Poit. | Lf | A | 12 | - | - | - | - | - | - | - |
| M | 14 | - | - | - | - | - | - | - | ||
| Ocimum americanum L. | Lf | A | - | 09 | - | - | 10 | - | - | - |
| M | - | 10 | - | - | 12 | - | - | 12 | ||
| Ocimum sanctum L. | Lf | A | - | - | - | 10 | 10 | - | - | - |
| M | - | 12 | - | 10 | 10 | - | 10 | - | ||
| Lauraceae | ||||||||||
| Litsea glutinosa (Lour.) C.B. Rob. | Lf | A | - | - | - | - | - | - | - | - |
| M | - | - | 10 | 11 | - | - | - | - | ||
| Leguminosae | ||||||||||
| Abrus precatorius L. | Lf | A | - | - | - | - | - | - | - | - |
| M | - | 12 | - | - | - | - | - | - | ||
| Fr | A | - | - | - | - | - | - | - | - | |
| M | - | - | - | - | - | - | 12 | - | ||
| Linaceae | ||||||||||
| Linum usitatissimum L. | Lf | A | - | - | - | - | - | - | - | - |
| M | - | 10 | - | - | - | - | - | - | ||
| Loranthaceae | ||||||||||
| Dendrophthoe falcata (L.f.) Ettingsh. | Lf | A | - | - | - | - | - | - | - | - |
| M | - | 10 | - | - | - | - | - | - | ||
| Lythraceae | ||||||||||
| Lagerstroemia speciosa (L.) Pers. | Lf | A | - | - | 10 | 12 | - | - | - | - |
| M | - | 12 | 12 | - | - | - | - | - | ||
| Malvaceae | ||||||||||
| Sida acuta Burm. f. | Lf | A | - | 10 | - | - | - | - | - | - |
| M | - | 14 | 14 | - | - | - | - | - | ||
| Sida cordata (Burm.f.) Borss.Waalk. | Wp | A | - | - | - | - | - | - | - | - |
| M | 12 | 10 | - | - | - | - | - | - | ||
| Marattiaceae | ||||||||||
| Angiopteris evecta (G. Forst.) Hoffm. | Lf | A | - | - | - | - | - | - | - | - |
| M | - | 12 | - | - | - | 14 | - | 13 | ||
| Melastomataceae | ||||||||||
| Melastoma malabathricum L. | Bk | A | - | 10 | - | - | - | - | - | 10 |
| M | - | 16 | - | - | 16 | - | - | 20 | ||
| Meliaceae | ||||||||||
| Azadirachta indica A. Juss. | Bk | A | 15 | - | 10 | - | - | 10 | - | - |
| M | 16 | 11 | 12 | - | 12 | 15 | - | 12 | ||
| Menispermaceae | ||||||||||
| Cissampelos pareira L. | Rt | A | - | - | - | 12 | - | 10 | - | - |
| M | 12 | 12 | 12 | 14 | 10 | 12 | - | 10 | ||
| Mimosoideae | ||||||||||
| Acacia leucophloea (Roxb.) Willd. | Lf | A | - | - | - | 09 | - | - | - | - |
| M | 14 | - | 10 | 14 | 12 | - | - | 10 | ||
| Moraceae | ||||||||||
| Ficus racemosa L. | Bk | A | - | - | 12 | - | - | - | - | - |
| M | 16 | - | 14 | 12 | - | - | 10 | 14 | ||
| Moringaceae | ||||||||||
| Moringa oleafera Lam. | Lf | A | - | 19 | 18 | - | - | 15 | - | 08 |
| M | 11 | 16 | 12 | 12 | 10 | 14 | 12 | 12 | ||
| Myrsinaceae | ||||||||||
| Ardisia solanacea (Poir.) Roxb. | Lf | A | - | 10 | 10 | - | - | - | - | - |
| M | 10 | 12 | 12 | 10 | - | 14 | - | - | ||
| Myrtaceae | ||||||||||
| Eucalyptus citriodora Hook. | Bk | A | - | - | - | - | - | - | - | - |
| M | - | - | - | - | 11 | 10 | - | - | ||
| Psidium guajava L. | Lf | A | - | 11 | - | 12 | - | - | - | - |
| M | - | 12 | - | 14 | - | - | - | - | ||
| Syzygium cumini (L.) Skeels | Lf | A | - | 10 | - | - | 09 | - | - | 10 |
| M | 14 | 11 | - | - | 12 | - | - | 11 | ||
| Syzygium jambos (L.) Alston | Lf | A | - | 12 | - | - | - | - | - | 10 |
| M | - | 10 | - | - | - | - | 10 | 12 | ||
| Oleaceae | ||||||||||
| Nyctanthes arbor-tristis L. | Lf | A | - | 14 | 10 | 12 | - | - | 10 | 10 |
| M | 20 | 22 | 15 | 10 | - | - | 18 | 13 | ||
| Bk | A | 10 | 10 | 10 | 14 | - | - | 10 | 10 | |
| M | 22 | 14 | 22 | 11 | - | 15 | 18 | |||
| Onagraceae | ||||||||||
| Ludwigia octovalvis (Jacq.) P.H. Raven | Lf | A | - | 09 | - | - | - | - | - | - |
| M | - | 12 | - | - | - | - | - | - | ||
| Papaveraceae | ||||||||||
| Argemone mexicana L. | Lf | A | - | - | - | - | - | - | - | - |
| M | - | - | - | - | - | - | - | 12 | ||
| Peripiocaceae | ||||||||||
| Hemidesmus indicus (L.) R.Br. ex Schult. | Lf | A | 11 | - | 12 | - | 10 | - | - | - |
| M | 12 | 10 | 13 | - | 10 | - | - | - | ||
| Polypodiaceae | ||||||||||
| Drynaria quercifolia (L.) J. Sm. | St | A | - | - | - | - | - | - | - | - |
| M | 12 | 15 | - | - | - | - | - | - | ||
| Punicaceae | ||||||||||
| Punica granatum L. | Lf | A | 10 | 12 | 10 | 12 | - | 12 | - | 14 |
| M | 17 | 12 | - | 10 | - | 10 | - | 12 | ||
| Rhamnaceae | ||||||||||
| Ziziphus mauritiana Lam. | Lf | A | - | 10 | - | - | - | - | - | - |
| M | - | 12 | - | - | 10 | - | - | - | ||
| Rubiaceae | ||||||||||
| Anthocephalus chinensis (Lam.) Hassk. | Lf | A | - | 10 | 12 | - | - | - | - | - |
| M | - | 12 | 12 | - | 10 | - | - | - | ||
| Canthium dicoccum (Gaertn.) Merr. | Lf | A | 10 | - | - | - | - | - | - | - |
| M | 14 | - | - | - | - | - | - | - | ||
| Ixora pavetta Andr. | Lf | A | 10 | - | - | - | - | - | - | - |
| M | 10 | 10 | - | - | - | 10 | - | - | ||
| Mitragyna parvifolia (Roxb.) Korth. | Lf | A | - | - | - | - | - | - | - | 08 |
| M | 11 | 08 | - | - | - | 10 | - | 12 | ||
| Paederia foetida L. | Lf | A | - | - | - | - | - | - | - | 08 |
| M | 12 | 12 | - | - | - | 12 | - | 12 | ||
| Wendlandia tinctoria (Roxb.) DC | Lf | A | 12 | - | - | 10 | - | 10 | - | - |
| M | - | - | - | 10 | 10 | 12 | - | - | ||
| Rutaceae | ||||||||||
| Acronychia pedunculata (L.) Miq. | Lf | A | - | 10 | - | - | - | - | - | - |
| M | - | 12 | - | - | 12 | - | - | - | ||
| Aegle marmelos (L.) Correa | Lf | A | - | 10 | - | - | - | - | - | - |
| M | - | 12 | - | - | - | 12 | - | 10 | ||
| Citrus aurantium L. | Lf | A | - | - | - | - | - | - | - | - |
| M | 10 | 12 | - | - | - | - | - | - | ||
| Clausena excavate Burm. f. | Lf | A | 11 | 09 | - | 14 | - | - | - | - |
| M | 13 | 12 | 14 | 12 | - | - | - | 12 | ||
| Murraya koenigii (L.) Spreng. | Lf | A | 12 | 10 | - | - | - | 12 | - | - |
| M | 12 | - | - | - | - | 10 | - | - | ||
| Sapindaceae | ||||||||||
| Schleichera oleosa (Lour.) Merr. | Lf | A | - | 10 | - | - | - | 12 | - | - |
| M | - | - | - | - | - | 10 | - | - | ||
| Sapotaceae | ||||||||||
| Madhuca longifolia (J.Koenig ex L.) J.F.Macbr. | Lf | A | 12 | 10 | - | - | - | 12 | - | - |
| M | - | - | - | - | - | 10 | - | - | ||
| Mimusops elengi L. | Lf | A | - | 10 | - | - | - | 12 | - | - |
| M | 11 | - | - | - | - | 10 | - | - | ||
| Scrophulariaceae | ||||||||||
| Scoparia dulcis L. | Lf | A | 12 | 10 | - | - | 09 | - | - | - |
| M | 14 | 12 | - | 10 | 11 | - | - | - | ||
| Solanaceae | ||||||||||
| Datura metel L. | Lf | A | - | - | - | - | - | - | - | - |
| M | - | - | - | 12 | 10 | - | - | - | ||
| Solanum virginianum L. | Lf | A | - | - | - | 10 | - | - | - | - |
| M | - | 10 | - | 11 | - | - | - | - | ||
| Sterculiaceae | ||||||||||
| Helicteres isora L. | Lf | A | - | - | - | 12 | - | - | - | - |
| M | 11 | 10 | - | 10 | - | - | - | - | ||
| Rt | A | - | - | - | 10 | - | - | - | - | |
| M | 12 | 11 | 12 | 12 | - | - | 12 | 13 | ||
| Pterospermum acerifolium (L.) Willd. | Lf | A | - | - | - | 12 | - | - | 12 | - |
| M | 15 | 11 | 10 | 15 | - | 14 | 10 | - | ||
| Pterospermum xylocarpum (Gaertn.) Sant. & Wagh | Lf | A | - | - | - | - | - | - | - | - |
| M | - | 12 | - | - | - | - | - | - | ||
| Tilliaceae | ||||||||||
| Grewia elastica Royle | Lf | A | - | - | - | - | - | - | - | - |
| M | - | - | - | - | 13 | - | 10 | - | ||
| Ulmaceae | ||||||||||
| Trema orientalis (L.) Blume | Lf | A | - | 10 | - | - | - | - | - | - |
| M | 15 | 12 | - | - | - | - | - | - | ||
| Verbenaceae | ||||||||||
| Clerodendrum indicum (L.) Kuntze | Lf | A | - | 10 | 10 | - | 14 | - | 10 | - |
| M | 12 | 14 | 12 | - | 12 | 11 | 10 | 09 | ||
| Clerodendrum viscosum Vent. | Lf | A | 14 | - | - | - | - | - | - | - |
| M | 13 | - | - | - | - | - | 10 | - | ||
| Lantana camara L. | Lf | A | - | - | 12 | - | - | - | - | - |
| M | - | - | - | - | - | - | 10 | - | ||
| Vitex negundo L. | Lf | A | 10 | 12 | 10 | - | - | - | - | - |
| M | 18 | 16 | 12 | 10 | - | - | 18 | 14 | ||
| Bk | A | 12 | 12 | 10 | 12 | - | - | 10 | 10 | |
| M | 14 | 13 | 18 | 17 | - | - | 12 | 16 | ||
| Vitaceae | ||||||||||
| Leea indica (Burm. f.) Merr. | Lf | A | - | - | - | - | - | - | - | - |
| M | - | - | 12 | - | - | 10 | - | - | ||
| Cissus quadrangularis L. | Wp | A | - | - | - | - | - | - | - | - |
| M | - | - | 10 | - | - | - | - | 10 | ||
| Zingiberaceae | ||||||||||
| Curcuma anguistifolia Roxb. | Lf | A | - | - | - | - | - | 10 | - | - |
| M | - | - | - | 10 | - | 08 | - | 08 | ||
| Curcuma aromatic Salisb. | Rh | A | - | - | - | - | - | - | - | - |
| M | 11 | - | - | - | - | - | - | 12 | ||
| Kaempferia rotunda L. | Lf | A | - | - | - | - | - | - | - | - |
| M | 13 | - | - | - | - | - | 11 | - | ||
| Antibiotic-Ciprofloxacin | 22 | 16 | 16 | 24 | 20 | 26 | 23 | R | ||
| Antibiotic-Gentamicin | 27 | 24 | 26 | 18 | 22 | 24 | 21 | 20 | ||
| Scrutiny | No. of Extracts Reported as Antibacterial (%) | ||
|---|---|---|---|
| Organism | Methanol Extract | Aqueous Extract | |
| Total number of plant species tested—222 | Gram positive | 146 (56.58%) | 89 (34.49%) |
| Total number of Genus tested—177 | Gram negative | 137 (53.10%) | 102 (39.53%) |
| Total number of family tested—83 | B. cereus | 108 (41.86%) | 50 (19.37%) |
| Total number of parts tested = 258 | S. aureus | 124 (48.06%) | 76 (29.45%) |
| Leaves-125; Bark-19; Whole part-08; Stem-04 | E. coli | 68 (26.35%) | 45 (17.44%) |
| Root-04; Rhizome-03; Fruit-03 and Bulb-01 | S. typhimurium | 65 (25.19%) | 41 (15.89%) |
| Total number of methanol extracts active—165 | S. dysentriae | 50 (19.37%) | 22 (8.52%) |
| Total number of aqueous extracts active—127 | S. flexneri | 66 (25.58%) | 28 (10.85%) |
| Number of species do not show activity-90 species | S. sonnei | 47 (18.21%) | 24 (9.30%) |
| Number of extracts do not show activity | V. cholerae | 72 (27.90%) | 38 (14.72%) |
| (93 methanol + 131 aqueous = 224) | Zone ≥ 20 mm | 10 (3.87%) | 0 |
| Total number of family show activity—68 | Zone (15–20) mm | 34 (13.17%) | 9 (3.48%) |
| Total number of family do not show activity—15 | Zone < 15 | 160 (62.01%) | 121 (46.89%) |
| Plant Species | Plant Part | Test Bacteria | |||
|---|---|---|---|---|---|
| Sa | Bc | Sf | Vc | ||
| Achyranthes aspera | Rt | >4000 | >4000 | >4000 | 2000 |
| Acorus calamus | Rh | >5000 | >5000 | >5000 | >5000 |
| Adhatoda vasica | Lf | 500 | 500 | 1000 | 2000 |
| Aegle marmelos | Lf | >4000 | >4000 | 4000 | >4000 |
| Ageratum conyzoides | Wp | 500 | >4000 | 500 | 4000 |
| Alangium salvifolium | Lf | >5000 | >5000 | >5000 | >5000 |
| Alpinia galanga | Lf | 1000 | 1000 | 2000 | 500 |
| Alstonia scholaris | Lf | >2000 | >2000 | 1000 | 500 |
| Andrographis paniculata | Lf | 1000 | 1000 | 2000 | 500 |
| A. paniculata | St | 500 | 1000 | 2000 | 1000 |
| Angiopteris evecta | Lf | >4000 | >4000 | 2000 | >5000 |
| Anogeissus latifolia | Lf | 1000 | 4000 | 1000 | 1000 |
| Annona squamosa | Lf | 1000 | 2000 | 1000 | 1000 |
| Annona reticulata | Lf | 1000 | 2000 | 1000 | 1000 |
| Ardisia solanacea | Lf | 1000 | 2000 | 1000 | 4000 |
| Azadirachta indica | Lf | 250 | 250 | 250 | 250 |
| Buchanania lanzan | Bk | 187 | 312 | 625 | 625 |
| Cassia fistula | Lf | 94 | 312 | 625 | 625 |
| Celastrus paniculatus | Lf | 1000 | 500 | 1000 | 500 |
| Centella asiatica | Wp | 1000 | 1000 | 1000 | 2000 |
| Cissampelos pareira | Lf | >4000 | 500 | 500 | 1000 |
| Clausena excavata | Lf | 1250 | 625 | 1250 | 1250 |
| Cleome viscosa | Lf | 1000 | 500 | 500 | 1000 |
| Cleistanthus collinus | Lf | 1250 | 1250 | 1250 | 2500 |
| Clerodendrum indicum | Lf | 250 | 2000 | 250 | 500 |
| Combretum roxburghii | Bk | 1250 | 1250 | 2500 | 2500 |
| Croton roxburghii | Lf | 625 | 625 | 625 | 156 |
| C. roxburghii | Bk | 312 | 312 | >5000 | 5000 |
| Diospyros melanoxylon | Lf | >5000 | >5000 | >5000 | 2500 |
| D. melanoxylon | Bk | 1000 | 250 | 500 | 250 |
| Diospyros sylvatica | Bk | 1250 | 625 | 625 | 1250 |
| Elephantopus scaber | Lf | 2000 | 250 | 2000 | 250 |
| Eleutherine bulbosa | Bl | 62 | 22 | 125 | 125 |
| Erycibe paniculata | Lf | 500 | 500 | 1250 | 1250 |
| Eryngium foetidum | Lf | 2500 | 2500 | 2500 | 2500 |
| E. foetidum | St | 1250 | 1250 | 5000 | 1250 |
| Euonymus glaber | Lf | 250 | 500 | 1000 | 2000 |
| Flemingia nana | Rt | 4000 | 1000 | >4000 | 4000 |
| Garcinia cowa | Lf | 625 | 1250 | 1250 | 1250 |
| Helicteres isora | Rt | 1250 | 1250 | 1250 | 1250 |
| Hemidesmus indicus | Lf | 4000 | 1000 | 4000 | 4000 |
| Holarrhena antidysenterica | Lf | 1250 | 312 | 625 | 2500 |
| Lannea coromandelica | Lf | 625 | 312 | 2500 | 2500 |
| Millettia extensa | Lf | 2500 | >5000 | >5000 | >5000 |
| Mimusops elengi | Lf | 5000 | >5000 | 2500 | >5000 |
| Momordica dioica | Lf | >5000 | >5000 | >5000 | >5000 |
| Mimusops elengi | Lf | 1000 | 4000 | 2000 | 4000 |
| Moringa oleafera | Lf | 625 | 312 | 2500 | 2500 |
| Nyctanthes arbor-tristis | Lf | 312 | 312 | 1250 | 312 |
| N. arbor-tristis | Bk | 156 | 156 | 156 | 625 |
| Oroxylum indicum | Bk | 250 | 250 | 500 | 125 |
| Paederia foetida | Lf | 1000 | 1000 | 2000 | 1000 |
| Pterospermum acerifolium | Bk | 312 | 312 | 1250 | >5000 |
| Punica granatum | Lf | 625 | 1250 | 2500 | 2500 |
| Ricinus communis | Lf | 1000 | 1000 | >5000 | 1000 |
| Semecarpus anacardium | Fr | 500 | 2000 | 500 | 2000 |
| Shorea robusta | Lf | 4000 | 2000 | >4000 | >4000 |
| Spondias pinnata | Lf | 500 | 500 | 500 | 500 |
| Tamarindus indica | Lf | 2000 | 2000 | >4000 | >4000 |
| Terminalia alata | Bk | 625 | 312 | 2500 | 2500 |
| Terminalia arjuna | Bk | 1000 | 2000 | >4000 | 4000 |
| Terminalia tomentosa | Bk | 2500 | 2500 | 2500 | 2500 |
| Tridax procumbens | Lf | 3000 | >6000 | >6000 | >6000 |
| Vitex negundo | Lf | >5000 | 2500 | 1250 | 5000 |
| V. negundo | Bk | >5000 | >5000 | >5000 | >5000 |
| Antibiotic(s) | Bacterial Strains (Zone of Inhibition in mm) | |||||||
|---|---|---|---|---|---|---|---|---|
| Bs | Sa | Ec | St | Sd | Sf | Ss | Vc | |
| Amikacin | R | R | R | R | R | R | R | R |
| Ampicillin | R | 18 | R | R | 12 | 14 | R | R |
| Ciprofloxacin | 22 | 16 | 16 | 24 | 20 | 26 | 23 | R |
| Erythromycin | 20 | 23 | R | R | R | R | 16 | 18 |
| Gatifloxacin | 22 | 22 | 18 | 19 | 14 | 18 | R | R |
| Gentamicin | 27 | 24 | 26 | 18 | 22 | 24 | 21 | 20 |
| Vancomycin | 20 | 16 | 19 | 15 | 14 | 17 | 23 | 14 |
| Streptomycin | 18 | 26 | 22 | 14 | 18 | 14 | 25 | R |
| Tetracycline | 22 | 14 | 23 | 18 | 14 | 13 | 17 | 16 |
| Amoxicillin | 14 | R | R | R | R | 12 | 14 | R |
| Cefoxitin | R | R | R | R | R | 15 | 26 | 21 |
| Cephotaxime | R | R | 14 | R | 26 | 22 | 20 | 17 |
| Ceftriaxone | 14 | 17 | 16 | 18 | 22 | 28 | 32 | 18 |
| Ofloxacin | 23 | 21 | 18 | 19 | 14 | 23 | 24 | 15 |
| Levofloxacin | 19 | 22 | 2R | 18 | 18 | 2R | 18 | 16 |
| Chloramphencol | 17 | 19 | 29 | 23 | R | 14 | 12 | R |
| Nalidaxic acid | R | R | R | R | 25 | 28 | R | R |
| Polymyxin B | 14 | R | 12 | R | 14 | 12 | R | R |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Panda, S.K.; Mohanta, Y.K.; Padhi, L.; Park, Y.-H.; Mohanta, T.K.; Bae, H. Large Scale Screening of Ethnomedicinal Plants for Identification of Potential Antibacterial Compounds. Molecules 2016, 21, 293. https://doi.org/10.3390/molecules21030293
Panda SK, Mohanta YK, Padhi L, Park Y-H, Mohanta TK, Bae H. Large Scale Screening of Ethnomedicinal Plants for Identification of Potential Antibacterial Compounds. Molecules. 2016; 21(3):293. https://doi.org/10.3390/molecules21030293
Chicago/Turabian StylePanda, Sujogya Kumar, Yugal Kishore Mohanta, Laxmipriya Padhi, Young-Hwan Park, Tapan Kumar Mohanta, and Hanhong Bae. 2016. "Large Scale Screening of Ethnomedicinal Plants for Identification of Potential Antibacterial Compounds" Molecules 21, no. 3: 293. https://doi.org/10.3390/molecules21030293
APA StylePanda, S. K., Mohanta, Y. K., Padhi, L., Park, Y.-H., Mohanta, T. K., & Bae, H. (2016). Large Scale Screening of Ethnomedicinal Plants for Identification of Potential Antibacterial Compounds. Molecules, 21(3), 293. https://doi.org/10.3390/molecules21030293








