Preventive Effects of Catechins on Cardiovascular Disease
Abstract
:1. Introduction
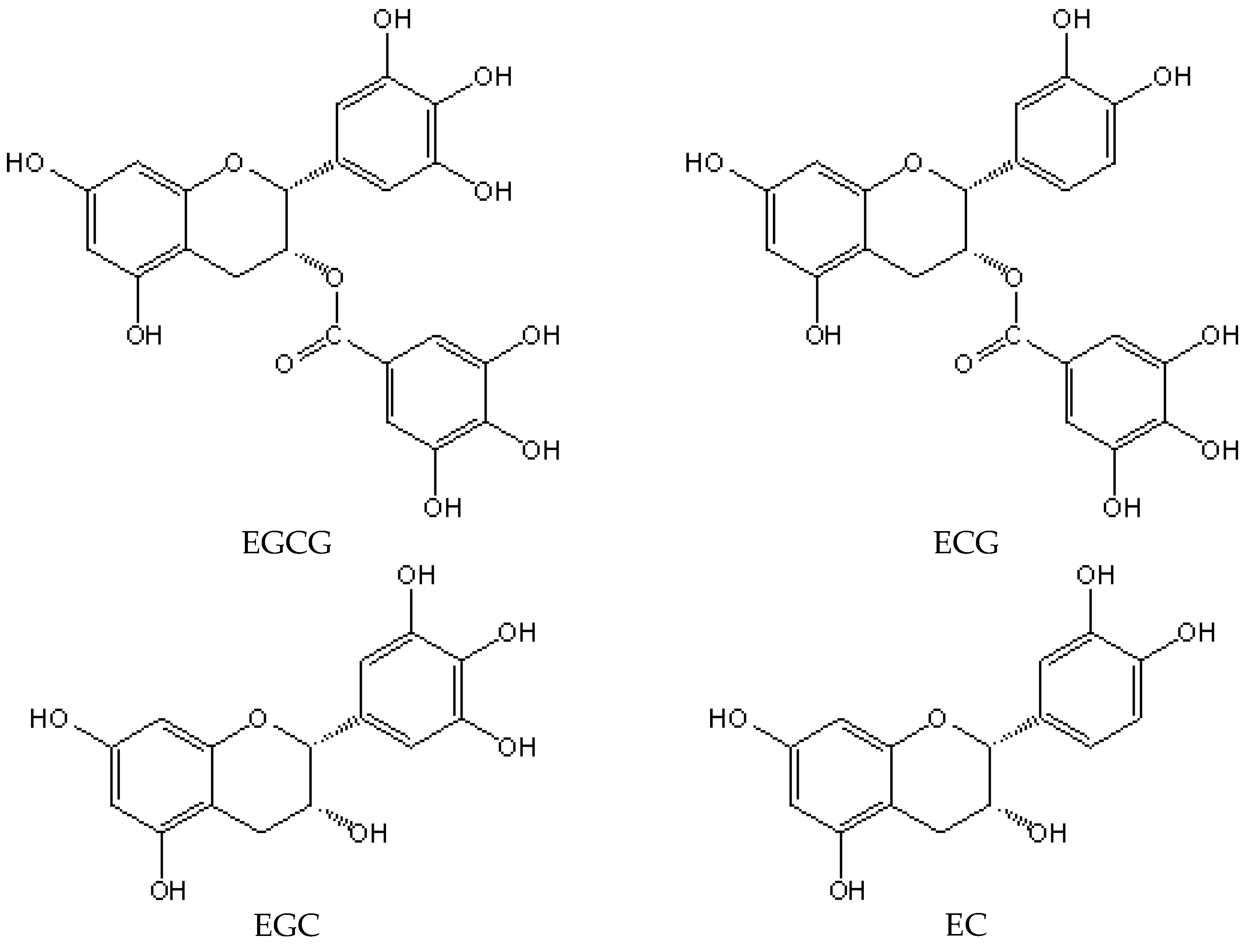
2. Catechins
3. Catechins Regulate Lipid Metabolism
4. Catechins Regulate Blood Lipid Metabolism
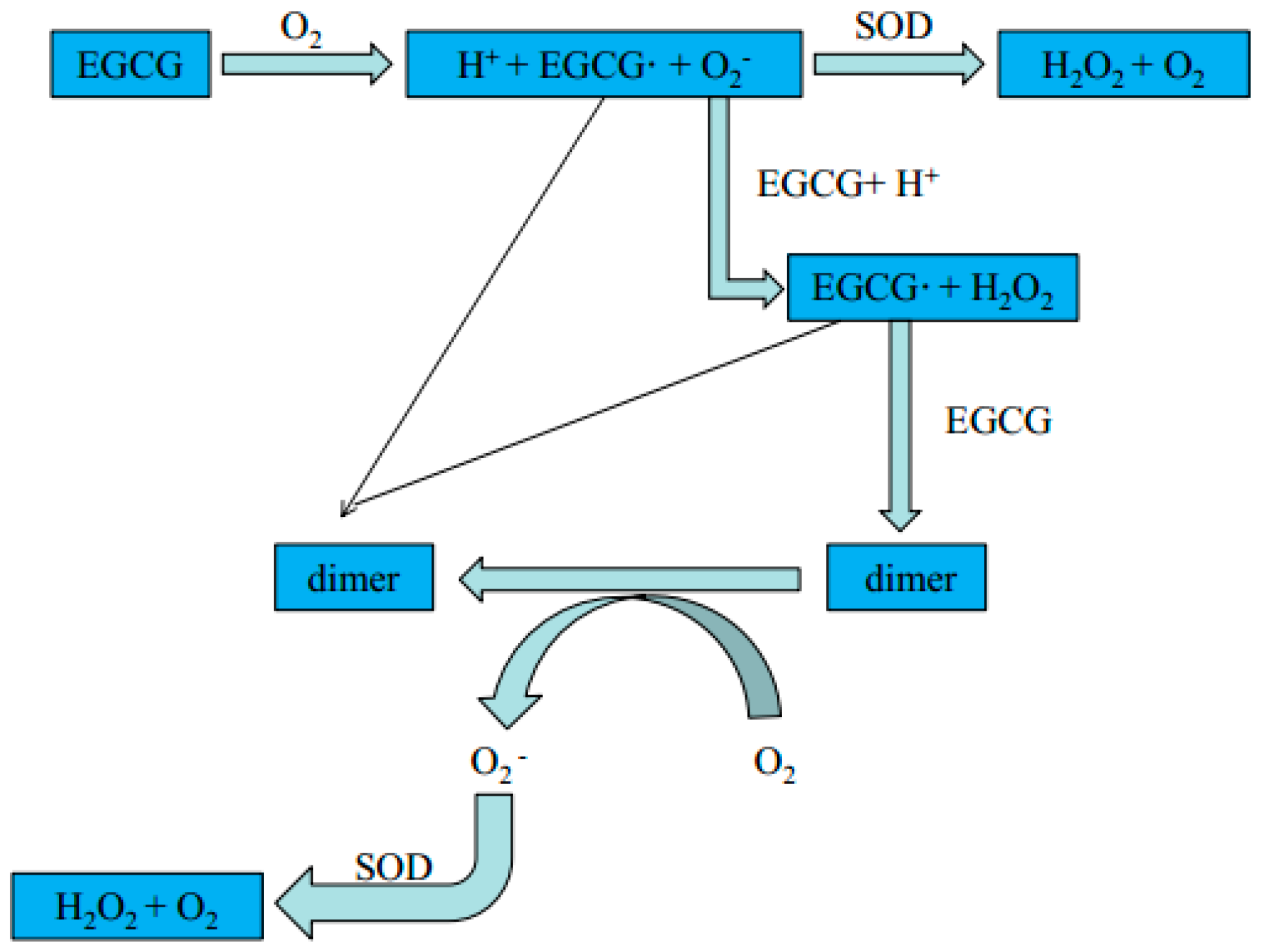
5. Protection of Vascular Endothelium from Catechins
6. Catechins Regulate Hypertension
7. Prospects
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Xiang, L.P.; Wang, A.; Ye, J.H.; Zheng, X.Q.; Polito, C.A.; Lu, J.L.; Li, Q.S.; Liang, Y.R. Suppressive effects of tea catechins on breast cancer. Nutrients 2016, 8, 458. [Google Scholar] [CrossRef] [PubMed]
- Matsui, T. Condensed catechins and their potential health-benefits. Eur. J. Pharmacol. 2015, 765, 495–502. [Google Scholar] [CrossRef] [PubMed]
- Assuncao, M.; Andrade, J.P. Protective action of green tea catechins in neuronal mitochondria during aging. Front. Biosci. Landmark Ed. 2015, 20, 247–262. [Google Scholar] [PubMed]
- Pasrija, D.; Anandharamakrishnan, C. Techniques for extraction of green tea polyphenols: A review. Food Bioprocess Technol. 2015, 8, 935–950. [Google Scholar] [CrossRef]
- Berry, J.D.; Dyer, A.; Cai, X.; Garside, D.B.; Ning, H.Y.; Thomas, A.; Greenland, P.; van Horn, L.; Tracy, R.P.; Lloyd-Jones, D.M. Lifetime Risks of Cardiovascular Disease. N. Engl. J. Med. 2012, 366, 321–329. [Google Scholar] [CrossRef] [PubMed]
- Vasan, R.S.; Larson, M.G.; Leip, E.P.; Evans, J.C.; O’Donnell, C.J.; Kannel, W.B.; Levy, D. Impact of high-normal blood pressure on the risk of cardiovascular disease. N. Engl. J. Med. 2001, 345, 1291–1297. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.K.; Siahpush, M.; Azuine, R.E.; Williams, S.D. Increasing area deprivation and socioeconomic inequalities in heart disease stroke and cardiovascular disease mortality among working age populations. Int. J. MCH AIDS 2016, 3, 1969–2011. [Google Scholar]
- Zhang, S.H.; Xuan, H.D.; Zhang, L.; Fu, S.C.; Wang, Y.J.; Yang, H.; Tai, Y.L.; Song, Y.H.; Zhang, J.S.; Ho, C.T.; et al. TBC2health: A database of experimentally validated health-beneficial effects of tea bioactive compounds. Brief. Bioinform. 2016. [Google Scholar] [CrossRef] [PubMed]
- Feng, L.; Chong, M.S.; Lim, W.S.; Gao, Q.; Nyunt, M.S.Z.; Lee, T.S.; Collinson, S.L.; Tsoi, T.; Kua, E.H.; Ng, T.P. TEA consumption reduces the incidence of neurocognitive disorders: Findings from the Singapore Longitudinal Aging Study. J. Nutr. Health Aging 2016. [Google Scholar] [CrossRef] [PubMed]
- Conney, A.H.; Lu, Y.P.; Lou, Y.R.; Huang, M.T. Inhibitory effects of tea and caffeine on UV-induced carcinogenesis: Relationship to enhanced apoptosis and decreased tissue fat. Eur. J. Cancer Prev. 2002, 11, S28–S36. [Google Scholar] [PubMed]
- Legeay, S.; Rodier, M.; Fillon, L.; Faure, S.; Clere, N. Epigallocatechin gallate: A review of its beneficial properties to prevent metabolic syndrome. Nutrients 2015, 7, 5443–5468. [Google Scholar] [CrossRef] [PubMed]
- Vita, J.A. Polyphenols and cardiovascular disease: Effects on endothelial and platelet function. Am. J. Clin. Nutr. 2005, 81, 292S–297S. [Google Scholar] [PubMed]
- Islam, M.A. Cardiovascular effects of green tea catechins: Progress and promise. Recent Pat. Cardiovasc. Drug Discov. 2012, 7, 88–99. [Google Scholar] [CrossRef] [PubMed]
- Isozaki, T.; Tamura, H. Epigallocatechin gallate (EGCG) inhibits the sulfation of 1-naphthol in a human colon carcinoma cell line, Caco-2. Biol. Pharm. Bull. 2001, 24, 1076–1078. [Google Scholar] [CrossRef] [PubMed]
- Valcic, S.; Timmermann, B.N.; Alberts, D.S.; Wachter, G.A.; Krutzsch, M.; Wymer, J.; Guillen, J.M. Inhibitory effect of six green tea catechins and caffeine on the growth of four selected human tumor cell lines. Anticancer Drugs 1996, 7, 461–468. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.P.; Kim, Y.H.; Kang, M.H.; Roberts, C.; Shim, J.S.; Roh, J.K. Chemopreventive effect of green tea (Camellia sinensis) against cigarette smoke-induced mutations (SCE) in humans. J. Cell. Biochem. Suppl. 1997, 27, 68–75. [Google Scholar] [CrossRef]
- Rizvi, S.I.; Zaid, M.A. Intracellular reduced glutathione content in normal and type 2 diabetic erythrocytes: Effect of insulin and (−) epicatechin. J. Physiol. Pharmacol. 2001, 52, 483–488. [Google Scholar] [PubMed]
- Nagao, T.; Komine, Y.; Soga, S.; Meguro, S.; Hase, T.; Tanaka, Y.; Tokimitsu, I. Ingestion of a tea rich in catechins leads to a reduction in body fat and malondialdehyde-modified LDL in men. Am. J. Clin. Nutr. 2005, 81, 122–129. [Google Scholar] [PubMed]
- Chen, C.H.; Ho, M.L.; Chang, J.K.; Hung, S.H.; Wang, G.J. Green tea catechin enhances osteogenesis in a bone marrow mesenchymal stem cell line. Osteoporos. Int. 2005, 16, 2039–2045. [Google Scholar] [CrossRef] [PubMed]
- Dalluge, J.J.; Nelson, B.C. Determination of tea catechins. J. Chromatogr. A 2000, 881, 411–424. [Google Scholar] [CrossRef]
- Seenivasan, S.; Manikandan, N.; Muraleedharan, N.N.; Selvasundaram, R. Heavy metal content of black teas from south India. Food Control 2008, 19, 746–749. [Google Scholar] [CrossRef]
- Yang, Y.; Zeng, H.; Zhang, Q. Direct electron transfer and sensing performance for catechin of nano-gold particles-polymer nano-composite with immobilized Laccase. Chem. Phys. Lett. 2016, 658, 259–269. [Google Scholar] [CrossRef]
- Braicu, C.; Ladomery, M.R.; Chedea, V.S.; Irimie, A.; Berindan-Neagoe, L. The relationship between the structure and biological actions of green tea catechins. Food Chem. 2013, 141, 3282–3289. [Google Scholar] [CrossRef] [PubMed]
- Poirier, P.; Giles, T.D.; Bray, G.A.; Hong, Y.; Stern, J.S.; Pi-Sunyer, F.X.; Eckel, R.H. Obesity and cardiovascular disease: Pathophysiology, evaluation, and effect of weight loss. Circulation 2006, 26, 898–918. [Google Scholar] [CrossRef] [PubMed]
- Htay, H.H.; Macnaughton, L.E.; Kapoor, M.P.; Juneja, L.R. Functional behavior of tea polyphenols in cardiovascular disease. Econ. Crisis Tea Ind. 2008, 29, 256–273. [Google Scholar]
- Roos, C.J.; Quax, P.H.; Jukema, J.W. Cardiovascular metabolic syndrome: Mediators involved in the pathophysiology from obesity to coronary heart disease. Biomark. Med. 2015, 6, 35–52. [Google Scholar] [CrossRef] [PubMed]
- De Schutter, A.; Kachur, S.; Lavie, C.J.; Boddepalli, R.S.; Patel, D.A.; Milani, R.V. The impact of inflammation on the obesity paradox in coronary heart disease. Int. J. Obes. 2016, 40, 1730–1735. [Google Scholar] [CrossRef] [PubMed]
- Kashima, S.; Inoue, K.; Matsumoto, M.; Akimoto, K. Prevalence and characteristics of non-obese diabetes in Japanese men and women: The Yuport Medical Checkup Center Study. J. Diabetes 2015, 7, 523–530. [Google Scholar] [CrossRef] [PubMed]
- Ortega, F.B.; Lavie, C.J.; Blair, S.N. Obesity and Cardiovascular Disease. Circ. Res. 1996, 11, 490–495. [Google Scholar] [CrossRef] [PubMed]
- Matsuyama, T.; Tanaka, Y.; Kamimaki, I.; Nagao, T.; Tokimitsu, I. Catechin safely improved higher levels of fatness, blood pressure, and cholesterol in children. Obesity 2008, 16, 1338–1348. [Google Scholar] [CrossRef] [PubMed]
- Dulloo, A.G.; Duret, C.; Rohrer, D.; Girardier, L.; Mensi, N.; Fathi, M.; Chantre, P.; Vandermander, J. Efficacy of a green tea extract rich in catechin polyphenols and caffeine in increasing 24-h energy expenditure and fat oxidation in humans. Am. J. Clin. Nutr. 1999, 70, 1040–1045. [Google Scholar] [PubMed]
- Havel, R.J.; Eder, H.A.; Bragdon, J.H. The distribution and chemical composition of ultracentrifugally separated lipoproteins in human serum. J. Clin. Investig. 1955, 34, 1345–1353. [Google Scholar] [CrossRef] [PubMed]
- Cybulsky, M.I.; Cheong, C.; Robbins, C.S. Macrophages and dendritic cells: Partners in atherogenesis. Circ. Res. 2016, 118, 637–652. [Google Scholar] [CrossRef] [PubMed]
- Oikonomou, E.; Siasos, G.; Tousoulis, D. Atherogenesis and hyperlipidemia a not straightforward association: We really need a novel biomarker? Int. J. Cardiol. 2016, 202, 586–588. [Google Scholar] [CrossRef] [PubMed]
- Herrington, D.M.; Reboussin, D.M.; Brosnihan, K.B.; Sharp, P.C.; Shumaker, S.A.; Snyder, T.E.; Furberg, C.D.; Kowalchuk, G.J.; Stuckey, T.D.; Rogers, W.J.; et al. Effects of estrogen replacement on the progression of coronary-artery atherosclerosis. N. Engl. J. Med. 2000, 343, 522–529. [Google Scholar] [CrossRef] [PubMed]
- Sugaya, K.; Igarashi, M.; Kojima, Y.; Tsubata, M.; Nagaoka, I. Evaluation of the effect of flavangenol on serum lipid peroxide levels and development of atherosclerosis in spontaneously hyperlipidemic B6.KOR-Apoe(shl) mice. Int. J. Mol. Med. 2011, 27, 33–38. [Google Scholar] [PubMed]
- Ruidavets, J.B.; Teissedre, P.L.; Ferrières, J.; Carando, S.; Bougard, G.; Cabanis, J.C. Catechin in the Mediterranean diet: Vegetable, fruit or wine? Atherosclerosis 2000, 153, 107–117. [Google Scholar] [CrossRef]
- Hofmann, C.S.; Sonenshein, G.E. Green tea polyphenol epigallocatechin-3 gallate induces apoptosis of proliferating vascular smooth muscle cells via activation of p53. FASEB J. 2003, 17, 702–704. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.X.; Lin, Q.L.; Shi, Z.P. Effect of catechin and epicatechin on serum lipid level in mice. J. Hunan Agric. Univ. 2002, 28, 232–233. [Google Scholar]
- Yu, C.G.; Li, R.Y.; Liu, Z.T.; Gao, H.; Liang, H.; Liu, K. Effects of catechin on blood fat level in experimental atherosclerosis model. Acta Acad. Med. Qingdao Univ. 2012, 48, 324–326. [Google Scholar]
- Arts, I.C.W.; Hollman, P.C.H.; Feskens, E.J.M.; de Mesquita, H.B.B.; Kromhout, D. Catechin intake might explain the inverse relation between tea consumption and ischemic heart disease: The Zutphen Elderly Study. Am. J. Clin. Nutr. 2001, 74, 227–232. [Google Scholar] [PubMed]
- Trnkova, L.; Bousova, I.; Stankova, V.; Drsata, J. Study on the interaction of catechins with human serum albumin using spectroscopic and electrophoretic techniques. J. Mol. Struct. 2011, 985, 243–250. [Google Scholar] [CrossRef]
- Miura, Y.; Chiba, T.; Miura, S.; Tomita, I.; Umegaki, K.; Ikeda, M.; Tomita, T. Green tea polyphenols (flavan 3-ols) prevent oxidative modification of low density lipoproteins: An ex vivo study in humans. J. Nutr. Biochem. 2000, 11, 216–222. [Google Scholar] [CrossRef]
- Aird, W.C. Phenotypic heterogeneity of the endothelium I. Structure, function, and mechanisms. Circ. Res. 2007, 100, 158–173. [Google Scholar] [CrossRef] [PubMed]
- Forrester, J.S.; Fishbein, M.; Helfant, R. A paradigm for restenosis based on cell biology: Clues for the development of new preventive therapies. J. Am. Coll. Cardiol. 1991, 17, 758–769. [Google Scholar] [CrossRef]
- Bersi, M.R.; Bellini, C.; Wu, J.; Montaniel, K.R.C.; Harrison, D.G.; Humphrey, J.D. Excessive adventitial remodelling leads to early aortic maladaptation in angiotensin-induced hypertension. Hypertension 2016, 67, 890–896. [Google Scholar] [CrossRef] [PubMed]
- Barton, M.; Husmann, M.; Meyer, M.R. Accelerated Vascular Aging as a Paradigm for Hypertensive Vascular Disease: Prevention and Therapy. Can. J. Cardiol. 2016, 32, 680–686. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.Q.; Wang, X.B.; Guan, R.F.; Tu, J.; Gong, Z.H.; Zheng, N.; Yang, J.H.; Zhang, Y.Y.; Ying, M.M. Blood anticoagulation and antiplatelet activity of green tea (−)-epigallocatechin (EGC) in mice. Food Funct. 2013, 4, 1521–1525. [Google Scholar] [CrossRef] [PubMed]
- Kipshidze, N.; Dangas, G.; Tsapenko, M.; Moses, J.; Leon, M.B.; Kutryk, M.; Serruys, P. Role of the endothelium in modulating neointimal formation-Vasculoprotective approaches to attenuate restenosis after percutaneous coronary interventions. J. Am. Coll. Cardiol. 2004, 44, 733–739. [Google Scholar] [PubMed]
- Kim, Y.; Keogh, J.B.; Clifton, P.M. Polyphenols and glycemic control. Nutrients 2016, 8, 17. [Google Scholar] [CrossRef] [PubMed]
- Nagle, D.G.; Ferreira, D.; Zhou, Y.D. Epigallocatechin-3-gallate (EGCG): Chemical and biomedical perspectives. Phytochemistry 2006, 17, 1849–1855. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.N.; Shankar, S.; Srivastava, R.K. Green tea catechin, epigallocatechin-3-gallate (EGCG): Mechanisms, perspectives and clinical applications. Biochem. Pharmacol. 2011, 82, 1807–1821. [Google Scholar] [CrossRef] [PubMed]
- Forstermann, U.; Munzel, T. Endothelial nitric oxide synthase in vascular disease: From marvel to menace. Circulation 2006, 113, 1708–1714. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.D.; Li, X.; McCracken, B.; Shao, Y.; Ward, K.; Fu, J.P. A miniaturized hemoretractometer for blood clot retraction testing. Small 2016, 12, 3926–3934. [Google Scholar] [CrossRef] [PubMed]
- Wahlgren, N.; Ahmed, N.; Dávalos, A.; Ford, G.A.; Grond, M.; Hacke, W.; Hennerici, M.G.; Kaste, M.; Kuelkens, S.; Larrue, V.; et al. Thrombolysis with alteplase for acute ischaemic stroke in the Safe Implementation of Thrombolysis in Stroke-Monitoring Study (SITS-MOST): An observational study. Lancet 2007, 369, 275–282. [Google Scholar] [CrossRef]
- Ngo-Metzger, Q.; Blitz, J. Screening for High Blood Pressure in Adults. Am. Fam. Phys. 2016, 93, 511–512. [Google Scholar]
- Zhang, X.E.; Cheng, B.; Wang, Q. Relationship between high blood pressure and cardiovascular outcomes in elderly frail patients: A systematic review and meta-analysis. Geriatr. Nurs. 2016. [Google Scholar] [CrossRef] [PubMed]
- Bogdanski, P.; Suliburska, J.; Szulinska, M.; Stepien, M.; Pupek-Musialik, D.; Jablecka, A. Green tea extract reduces blood pressure, inflammary biomarkers, and oxidative stress and improves, parameters associated with insulin resistance in obese, hypertensive patients. Nutr. Res. 2012, 32, 421–427. [Google Scholar] [CrossRef] [PubMed]
- Ademiluyi, A.O.; Oboh, G.; Ogunsuyi, O.B.; Oloruntoba, F.M. A comparative study on antihypertensive and antioxidant properties of phenolic extracts from fruit and leaf of some guava (Psidium guajava L.) varieties. Comp. Clin. Pathol. 2016, 25, 363–374. [Google Scholar] [CrossRef]
- Tounekti, T.; Joubert, E.; Hernandez, I.; Munne-Bosch, S. Improving the polyphenol content of tea. Crit. Rev. Plant Sci. 2013, 32, 192–215. [Google Scholar] [CrossRef]
- Gómez-Guzmán, M.; Jiménez, R.; Sánchez, M.; Zarzuelo, M.J.; Galindo, P.; Quintela, A.M.; Lopez-Sepulveda, R.; Romero, M.; Tamargo, J.; Vargas, F.; et al. Epicatechin lowers blood pressure, restores endothelial function, and decreases oxidative stress and endothelin-1 and NADPH oxidase activity in DOCA-salt hypertension. Free Radic. Biol. Med. 2012, 52, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, N.; Dhawan, V.; Sharma, G.; Jain, S.; Kaul, D. Induction of inflammatory gene expression by THP-1 macrophages cultured in normocholesterolaemic hypertensive sera and modulatory effects of green tea polyphenols. J. Hum. Hypertens. 2008, 22, 141–143. [Google Scholar] [CrossRef] [PubMed]
- Namkung, W.; Thiagarajah, J.R.; Phuan, P.W.; Verkman, A.S. Inhibition of Ca2+-activated Cl− channels by gallotannins as a possible molecular basis for health benefits of red wine and green tea. FASEB J. 2010, 24, 4178–4186. [Google Scholar] [CrossRef] [PubMed]
- Ohno, S.; Yokoi, H.; Mori, K.; Kasahara, M.; Kuwahara, K.; Fujikura, J.; Naito, M.; Kuwabara, T.; Imamaki, H.; Ishii, A.; et al. Ablation of the N-type calcium channel ameliorates diabetic nephropathy with improved glycemic control and reduced blood pressure. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, X.-Q.; Hu, T.; Han, Y.; Huang, W.; Yuan, H.-B.; Zhang, Y.-T.; Du, Y.; Jiang, Y.-W. Preventive Effects of Catechins on Cardiovascular Disease. Molecules 2016, 21, 1759. https://doi.org/10.3390/molecules21121759
Chen X-Q, Hu T, Han Y, Huang W, Yuan H-B, Zhang Y-T, Du Y, Jiang Y-W. Preventive Effects of Catechins on Cardiovascular Disease. Molecules. 2016; 21(12):1759. https://doi.org/10.3390/molecules21121759
Chicago/Turabian StyleChen, Xiao-Qiang, Ting Hu, Yu Han, Wei Huang, Hai-Bo Yuan, Yun-Tian Zhang, Yu Du, and Yong-Wen Jiang. 2016. "Preventive Effects of Catechins on Cardiovascular Disease" Molecules 21, no. 12: 1759. https://doi.org/10.3390/molecules21121759
APA StyleChen, X.-Q., Hu, T., Han, Y., Huang, W., Yuan, H.-B., Zhang, Y.-T., Du, Y., & Jiang, Y.-W. (2016). Preventive Effects of Catechins on Cardiovascular Disease. Molecules, 21(12), 1759. https://doi.org/10.3390/molecules21121759





