A New Bioactive Metabolite Isolated from the Red Sea Marine Sponge Hyrtios erectus
Abstract
:1. Introduction
2. Results and Discussion
2.1. Purification of Compounds 1–4
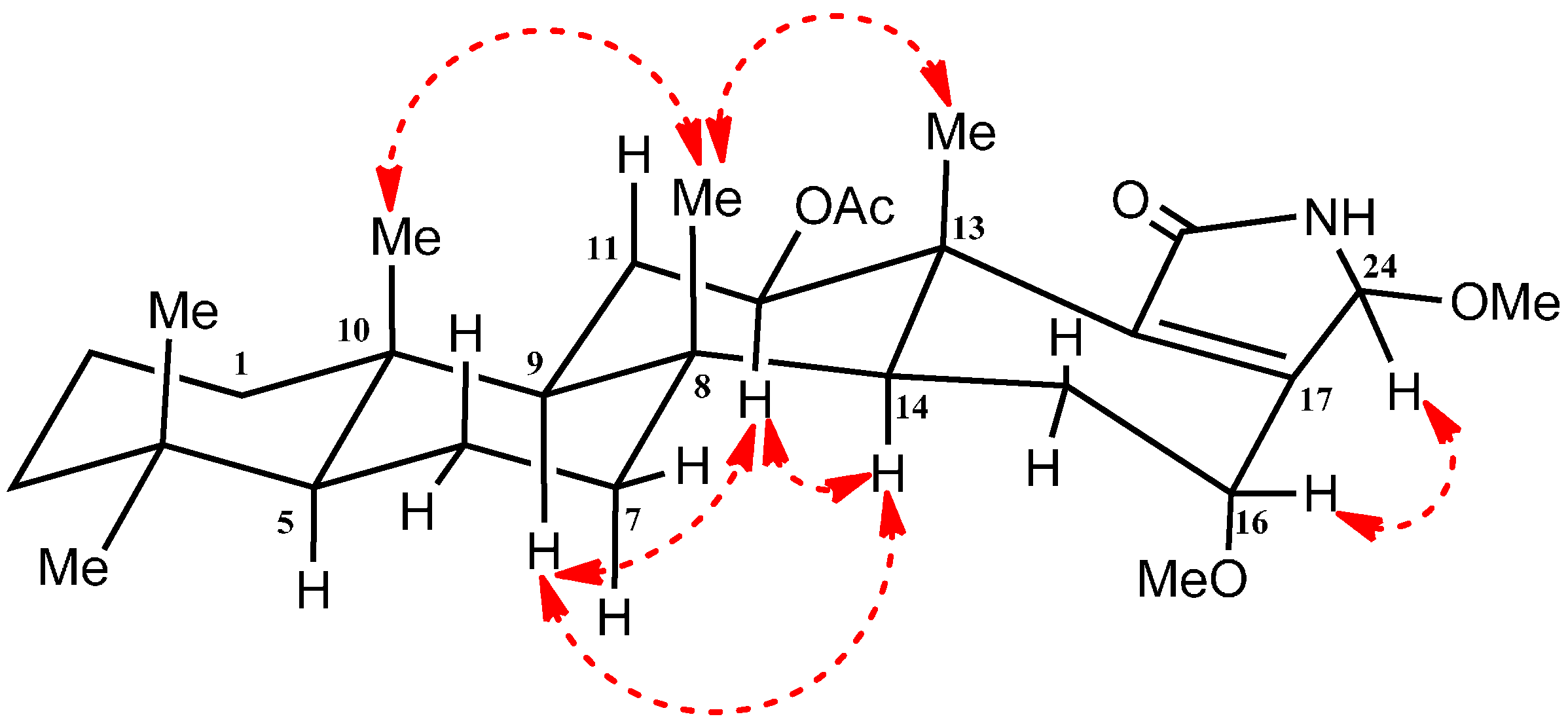
2.2. Structure Elucidation of Compound 1–4
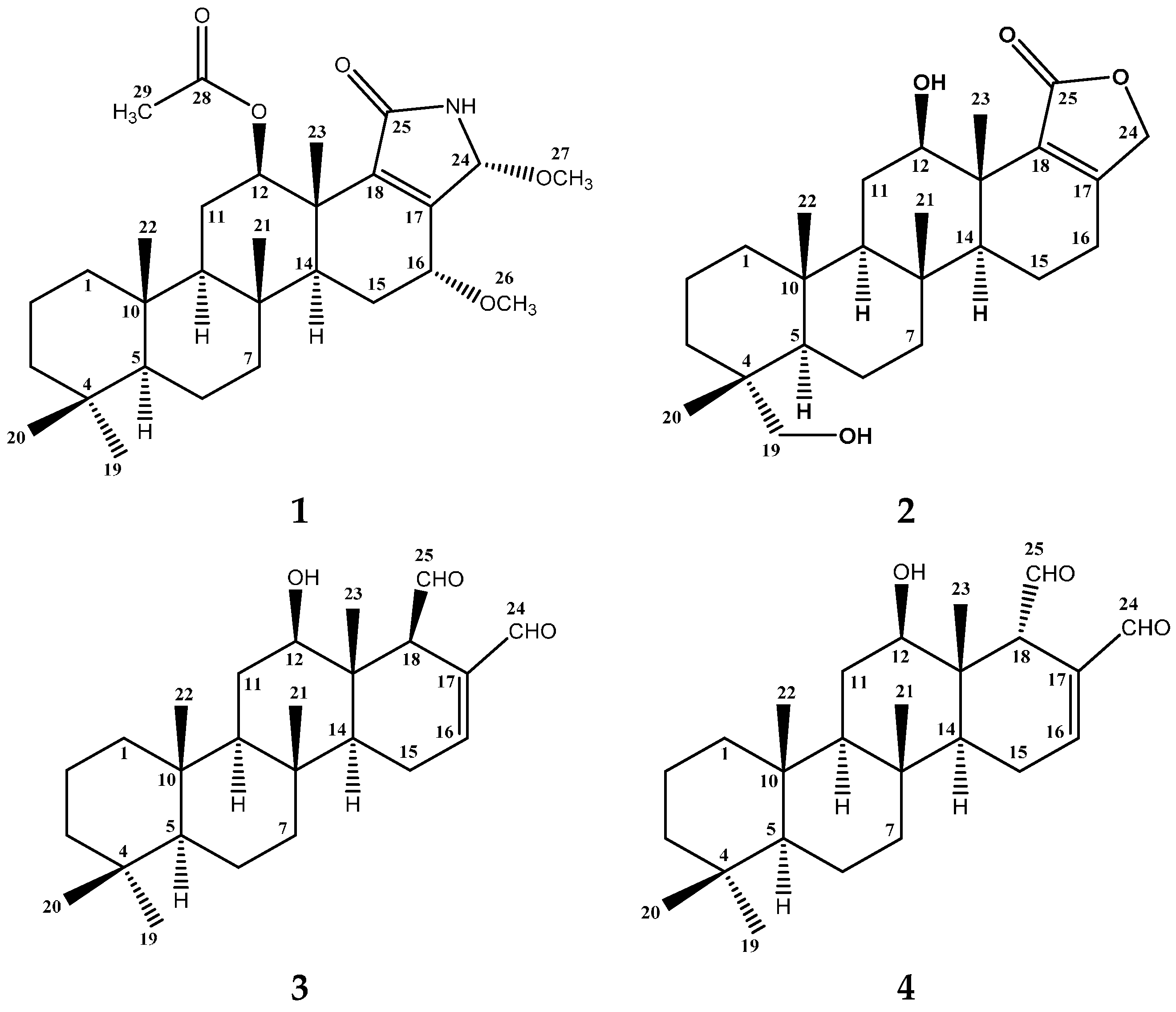
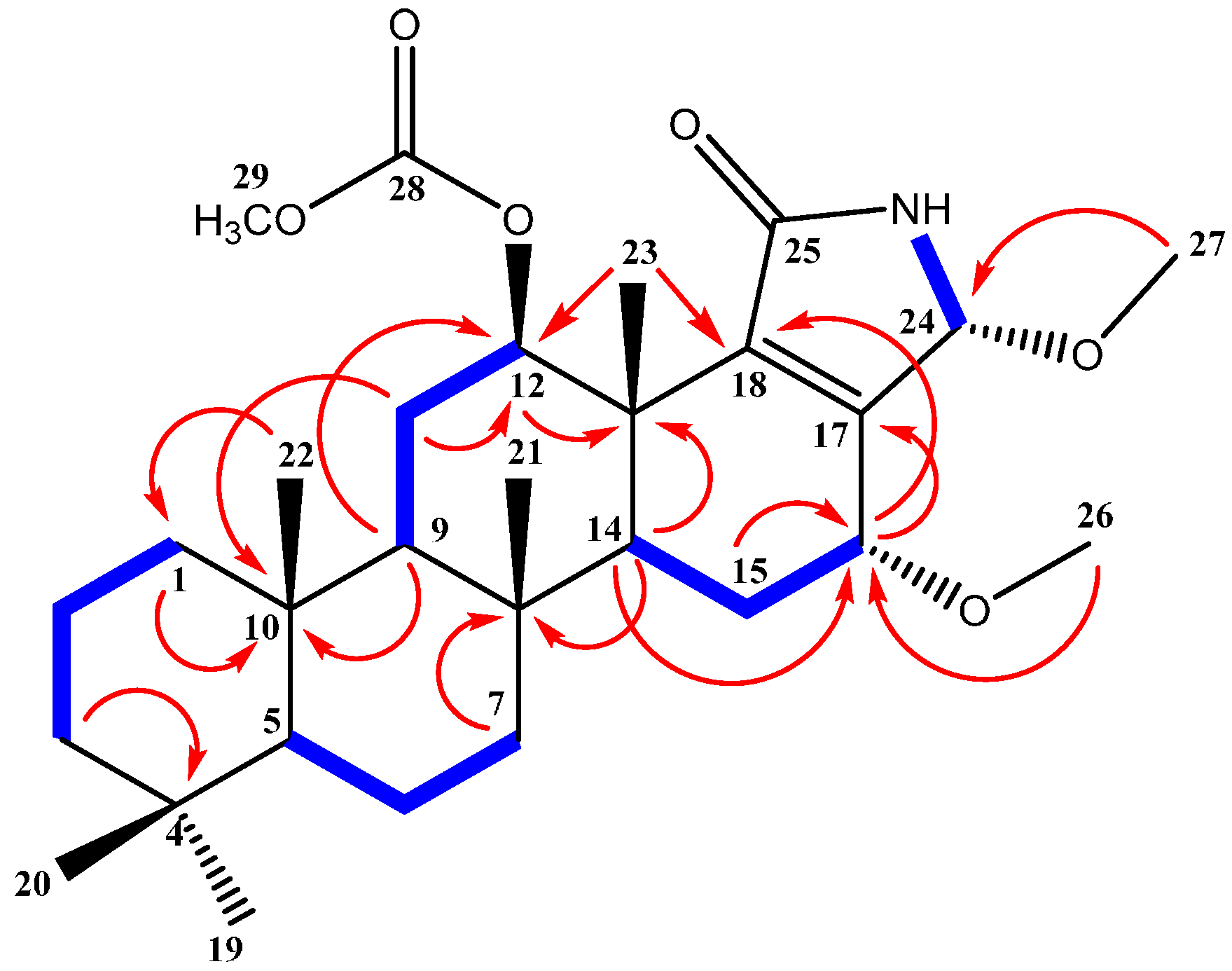
| Position | δC | δH (m, J in Hz) | HMBC (H→C) a |
|---|---|---|---|
| 1 | 39.6, CH2 | 1.62, 0.83 (m) | C-10 |
| 2 | 18.4, CH2 | 1.59, 1.41 (m) | C-4, C-10 |
| 3 | 42.0 CH2 | 1.35, 1.11 (m) | C-4 |
| 4 | 33.2 qC | ||
| 5 | 56.3 CH | 0.80 (m) | C-4 |
| 6 | 18.2 CH2 | 1.58, 1.42 (m) | |
| 7 | 41.3 CH2 | 1.81, 0.95 (m) | C-8 |
| 8 | 36.7 qC | ||
| 9 | 57.5 CH | 1.01 (m) | C-10, C-12 |
| 10 | 37.3 qC | ||
| 11 | 24.8 CH2 | 1.77, 1.54 (m) | C-10, C-12 |
| 12 | 75.3 CH | 4.95 (dd, 10.8, 4.8) | C-11, C-13, C-18, C-23, C-28 |
| 13 | 42.1 qC | ||
| 14 | 50.0 CH | 1.44 (m) | C-8, C-9, C-13, C-16, C-18 |
| 15 | 21.3 CH2 | 2.01, 1.55 (m) | |
| 16 | 69.8 CH | 3.85 (dd, 4.2, 1.2) | C-17, C-18, C-26, C-24 |
| 17 | 151.1 qC | ||
| 18 | 144.7 qC | ||
| 19 | 21.3 CH3 | 0.80 (s) | C-4 |
| 20 | 33.2 CH3 | 0.84 (s) | C-4 |
| 21 | 17.3 CH3 | 0.91 (s) | C-7, C-8, C-9, C-14 |
| 22 | 15.7 CH3 | 0.82 (s) | C-1, C-5, C-9, C-10 |
| 23 | 15.9 CH3 | 1.20 (s) | C-12, C-13, C-18, C-14 |
| 24 | 82.3 CH | 5.3 (s) | C-17, C-18, C-25, C-27 |
| 25 | 170.5 qC | ||
| 26 | 57.1 OCH3 | 3.39 (s) | C-16 |
| 27 | 52.1 OCH3 | 3.20 (s) | C-24 |
| 28 | 171.4 qC | ||
| 29 | 21.9 CH3 | 2.12 (s) | C-28 |
| NH | 5.78 (br s) | C-17, C-18 |
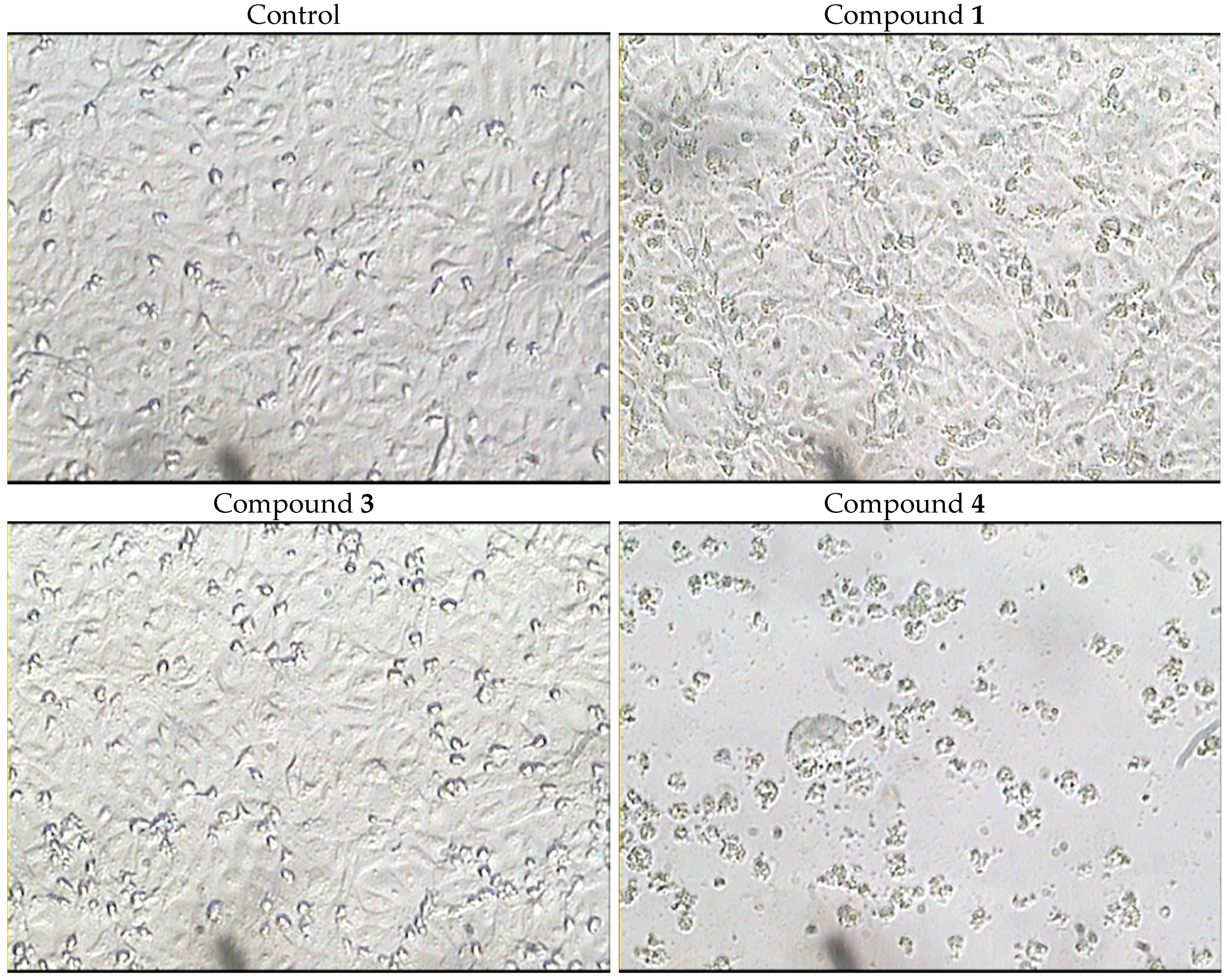
2.3. Biological Activity of the Isolated Compounds
| Cell Type | Cell Line | Doxorubicin a | 1 | 2 | 3 | 4 |
|---|---|---|---|---|---|---|
| Breast | MCF-7 | 0.41 | 55.4 | >100 | 36.0 | 3.3 |
| Hepatocellular | HepG2 | 0.85 | 25.4 | >100 | 23.4 | 1.7 |
| Colorectal | HCT-116 | 0.11 | 26.5 | >100 | 27.1 | 3.4 |
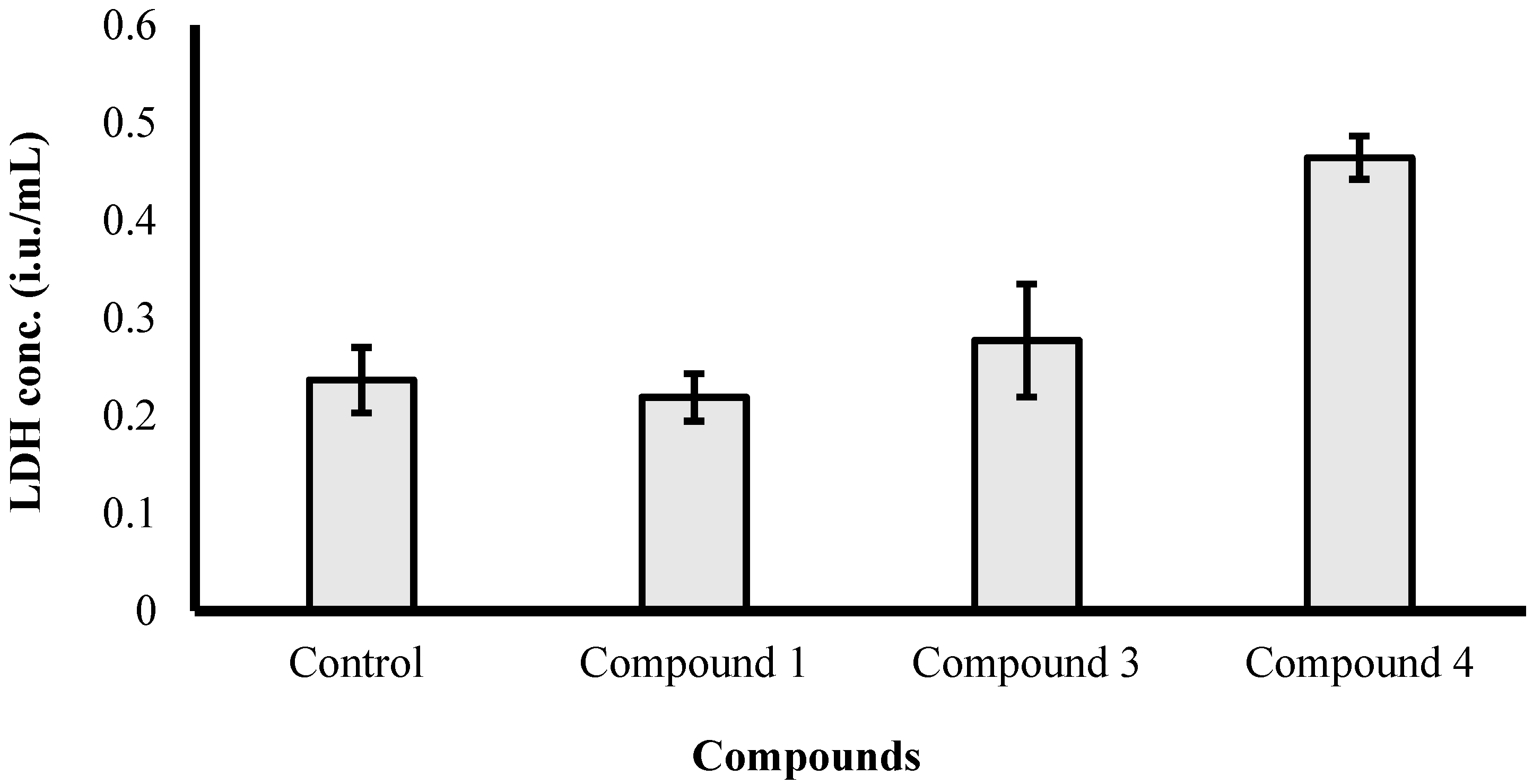
3. Experimental Section
3.1. General Procedures
3.2. Biological Materials

3.3. Purification of Compounds 1–4
3.4. Characterization of 24-Methoxy-petrosaspongia C (1)
3.5. Biological Activity of Compounds 1–4
3.5.1. Cell Culture
3.5.2. Cell Membrane Integrity Assessment
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Blunt, J.W.; Copp, B.R.; Munro, M.H.G.; Northcote, P.T.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2005, 22, 15–61. [Google Scholar] [CrossRef] [PubMed]
- Gomes, N.; Lefranc, F.; Kijjoa, A.; Kiss, R. Can Some Marine-Derived Fungal Metabolites Become Actual Anticancer Agents? Mar. Drugs 2015, 13, 3950–3991. [Google Scholar] [CrossRef] [PubMed]
- Youssef, D.T.A.; Yamaki, R.K.; Kelly, M.; Scheuer, P.J. Salmahyrtisol A, a novel cytotoxic sesterterpene from the Red Sea sponge Hyrtios erecta. J. Nat. Prod. 2002, 65, 2–6. [Google Scholar] [CrossRef] [PubMed]
- Pettit, G.R.; Cichacz, Z.A.; Tan, R.; Hoard, M.S.; Melody, N.; Pettit, R.K. Antineoplastic agents. 386. isolation of sesterstatins 1–3 from the marine sponge Hyrtios erecta. J. Nat. Prod. 1998, 61, 13–16. [Google Scholar] [CrossRef] [PubMed]
- Pettit, G.R.; Cichaz, Z.A.; Tan, R.; Herald, D.L.; Melody, N.; Hoard, M.S.; Doubek, D.L.; Hooper, J.N.A. Antineoplastic agents. 385. The isolation and structure of a scalarane-type sesterterpene from the Indian Ocean porifera Hyrtios erecta. Collect. Czech. Chem. Commun. 1998, 63, 1671–1677. [Google Scholar] [CrossRef]
- Pettit, G.R.; Tan, R.; Melody, N.; Cichacz, Z.A.; Herald, D.L.; Hoard, M.S.; Pettit, R.K.; Chapuis, J.-C. Antineoplastic agents. 397: Isolation and structure of sesterstatins 4 and 5 from Hyrtios erecta (the Republic of Maldives). Bioorg. Med. Chem. Lett. 1998, 8, 2093–2098. [Google Scholar] [CrossRef]
- Pettit, G.R.; Tan, R.; Cichacz, Z.A. Antineoplastic agents. 542. Isolation and structure of sesterstatin 6 from the Indian Ocean sponge Hyrtios erecta. J. Nat. Prod. 2005, 68, 1253–1255. [Google Scholar] [CrossRef] [PubMed]
- Ryu, G.; Matsunaga, S.; Fusetani, N. Three new cytotoxic sesterterpenes from the marine sponge Hyrtios cf. erectus. J. Nat. Prod. 1996, 59, 515–517. [Google Scholar] [CrossRef] [PubMed]
- Nasu, S.S.; Yeung, B.K.S.; Hamann, M.T.; Scheuer, P.J.; Kelly-Borges, M.; Goins, K. Puupehenone-related metabolites from two Hawaiian sponges, Hyrtios spp. J. Organ. Chem. 1995, 60, 7290–7292. [Google Scholar] [CrossRef]
- Evidente, A.; Kornienko, A.; Lefranc, F.; Cimmino, A.; Dasari, R.; Evidente, M.; Mathieu, V.; Kiss, R. Sesterterpenoids with Anticancer Activity. Curr. Med. Chem. 2015, 22, 3502–3522. [Google Scholar] [CrossRef] [PubMed]
- Hochlowski, J.E.; Faulkner, D.J.; Bass, L.S.; Clardy, J. Metabolites of the dorid nudibranch Chromodoris sedna. J. Organ. Chem. 1983, 48, 1738–1740. [Google Scholar] [CrossRef]
- Bergquist, P.R.; Cambie, R.C.; Kernan, M.R. Scalarane sesterterpenes from Collospongia auris, a new thorectid sponge. Biochem. Syst. Ecol. 1990, 18, 349–357. [Google Scholar] [CrossRef]
- Walker, R.P.; Thompson, J.E.; Faulkner, D.J. Sesterterpenes from Spongia idia. J. Organ. Chem. 1980, 45, 4976–4979. [Google Scholar] [CrossRef]
- Braekman, J.C.; Daloze, D.; Kaisin, M.; Moussiaux, B. Ichthyotoxic sesterterpenoids from the neo guinean sponge carteriospongia foliascens. Tetrahedron 1985, 41, 4603–4614. [Google Scholar] [CrossRef]
- Kikuchi, H.; Tsukitani, Y.; Shimizu, I.; Kobayashi, M.; Kitagawa, I. Marine Natural Products. XI. An Antiinflammatory Scalarane-type Bishomosesterterpene, Foliaspongin, from the Okinawan Marine Sponge Phyllospongia foliascens (PALLAS). Chem. Pharm. Bull. 1983, 31, 552–556. [Google Scholar] [CrossRef]
- Crews, P.; Bescansa, P. Sesterterpenes from a common marine sponge, Hyrtios erecta. J. Nat. Prod. 1986, 49, 1041–1052. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa, M.; Hamamoto, Y.; Ishihama, M.; Hamasaki, S.; Endo, M. Pharmacologically active homosesterterpenes from palauan sponges. Tetrahedron Lett. 1987, 28, 431–434. [Google Scholar] [CrossRef]
- Williams, D.E.; Tahir, A.; Andersen, R.J. A New Acyclic Diketotriterpenoid Isolated from the Indonesian Marine Sponge Hyrtios erectus. J. Nat. Prod. 1999, 62, 653–654. [Google Scholar] [CrossRef] [PubMed]
- Salmoun, M.; Devijver, C.; Daloze, D.; Braekman, J.C.; Gomez, R.; de Kluijver, M.; van Soest, R.W.M. New Sesquiterpene/Quinones from Two Sponges of the Genus Hyrtios. J. Nat. Prod. 2000, 63, 452–456. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, M.; Aoki, S.; Sakai, H.; Kawazoe, K.; Kihara, N.; Sasaki, T.; Kitagawa, I. Altohyrtin A, a potent anti-tumor macrolide from the Okinawan marine sponge Hyrtios altum. Tetrahedron Lett. 1993, 34, 2795–2798. [Google Scholar] [CrossRef]
- Kobayashi, M.; Aoki, S.; Sakai, H.; Kihara, N.; Sasaki, T.; Kitagawa, I. Altohyrtins B and C and 5-desacetylaltohyrtin A, potent cytotoxic macrolide congeners of Altohyrtin A, from the Okinawan marine sponge Hyrtios altum. Chem. Pharm. Bull. 1993, 41, 989–991. [Google Scholar] [CrossRef] [PubMed]
- Pettit, G.R.; Chicacz, Z.A.; Gao, F.; Herald, C.L.; Boyd, M.R.; Schmidt, J.M.; Hooper, J.N.A. Antineoplastic agents. 257. Isolation and structure of spongistatin 1. J. Organ. Chem. 1993, 58, 1302–1304. [Google Scholar] [CrossRef]
- Youssef, D.T.A. Hyrtioerectines A–C, cytotoxic alkaloids from the Red Sea sponge Hyrtios erectus. J. Nat. Prod. 2005, 68, 1416–1419. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, J.; Murayama, T.; Ishibashi, M.; Kosuge, S.; Takamatsu, M.; Ohizumi, Y.; Kobayashi, H.; Ohta, T.; Nozoe, S.; Sasaki, T. Hyrtiosins A and B, new indole alkaloids from the Okinawan marine sponge Hyrtios erecta. Tetrahedron 1990, 46, 7699–7702. [Google Scholar] [CrossRef]
- Bourguet-Kondracki, M.L.; Martin, M.T.; Guyot, M. A new β-carboline alkaloid isolated from the marine sponge Hyrtios erecta. Tetrahedron Lett. 1996, 37, 3457–3460. [Google Scholar] [CrossRef]
- Festa, C.; Cassiano, C.; D′Auria, M.V.; Debitus, C.; Monti, M.C.; de Marino, S. Scalarane sesterterpenes from Thorectidae sponges as inhibitors of TDP-43 nuclear factor. Organ. Biomol. Chem. 2014, 12, 8646–8655. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Guerrero, C.J.; Zubía, E.; Ortega, M.J.; Carballo, J.L. Sesterterpene metabolites from the sponge Hyatella intestinalis. Tetrahedron 2006, 62, 5392–5400. [Google Scholar] [CrossRef]
- Cafieri, F.; de Napoli, L.; Fattorusso, E.; Santacroce, C.; Sica, D. Molliorin-A: A unique scalarin-like pyrroloterpene from the sponge Cacospongia mollior. Tetrahedron Lett. 1977, 18, 477–480. [Google Scholar] [CrossRef]
- Cafieri, F.; de Napoli, L.; Fattorusso, E.; Santacroce, C. Molliorin-B, a second scalarin-like pyrroloterpene from the sponge Cacospongia mollior. Experientia 1977, 33, 994–995. [Google Scholar] [CrossRef] [PubMed]
- Cafieri, F.; de Napoli, L.; Iengo, A.; Santacroce, C. Molliorin-C, a further pyrroloterpene present in the sponge Cacospongia mollior. Experientia 1978, 34, 300–301. [Google Scholar] [CrossRef]
- Dumont, P.; Ingrassia, L.; Rouzeau, S.; Ribaucour, F.; Thomas, S.; Roland, I.; Darro, F.; Lefranc, F.; Kiss, R. The Amaryllidaceae isocarbostyril narciclasine induces apoptosis by activation of the death receptor and/or mitochondrial pathways in cancer cells but not in normal fibroblasts. Neoplasia 2007, 9, 766–776. [Google Scholar] [CrossRef] [PubMed]
- De Hauwer, C.; Camby, I.; Darro, F.; Migeotte, I.; Decaestecker, C.; Verbeek, C.; Danguy, A.; Pasteels, J.L.; Brotchi, J.; Salmon, I.; et al. Gastrin inhibits motility, decreases cell death levels and increases proliferation in human glioblastoma cell lines. J. Neurobiol. 1998, 37, 373–382. [Google Scholar] [CrossRef]
- Delbrouck, C.; Doyen, I.; Belot, N.; Decaestecker, C.; Ghanooni, R.; de Lavareille, A.; Kaltner, H.; Choufani, G.; Danguy, A.; Vandenhoven, G.; et al. Galectin-1 is overexpressed in nasal polyps under budesonide and inhibits eosinophil migration. Lab. Investig. J. Technol. Methods Pathol. 2002, 82, 147–158. [Google Scholar] [CrossRef]
- Skehan, P.; Storeng, R.; Scudiero, D.; Monks, A.; McMahon, J.; Vistica, D.; Warren, J.T.; Bokesch, H.; Kenney, S.; Boyd, M.R. New colorimetric cytotoxicity assay for anticancer-drug screening. J. Natl. Cancer Instit. 1990, 82, 1107–1112. [Google Scholar] [CrossRef]
- Mahmoud, A.M.; Al-Abd, A.M.; Lightfoot, D.A.; El-Shemy, H.A. Anti-cancer characteristics of mevinolin against three different solid tumor cell lines was not solely p53-dependent. J. Enzyme Inhib. Med. Chem. 2012, 27, 673–679. [Google Scholar] [CrossRef] [PubMed]
- Phillips, C.A.; Hughes, D.R.; Huja, S.S. Modifications of the lactate dehydrogenase assay, a histochemical determinant of osteocyte viability—A qualitative study. Acta Histochem. 2009, 111, 166–171. [Google Scholar] [CrossRef] [PubMed]
- Abdallah, H.M.; Al-Abd, A.M.; Asaad, G.F.; Abdel-Naim, A.B.; El-halawany, A.M. Isolation of antiosteoporotic compounds from seeds of Sophora japonica. PLoS ONE 2014, 9, e98559. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds are not available from the authors.
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elhady, S.S.; El-Halawany, A.M.; Alahdal, A.M.; Hassanean, H.A.; Ahmed, S.A. A New Bioactive Metabolite Isolated from the Red Sea Marine Sponge Hyrtios erectus. Molecules 2016, 21, 82. https://doi.org/10.3390/molecules21010082
Elhady SS, El-Halawany AM, Alahdal AM, Hassanean HA, Ahmed SA. A New Bioactive Metabolite Isolated from the Red Sea Marine Sponge Hyrtios erectus. Molecules. 2016; 21(1):82. https://doi.org/10.3390/molecules21010082
Chicago/Turabian StyleElhady, Sameh S., Ali M. El-Halawany, Abdulrahman M. Alahdal, Hashim A. Hassanean, and Safwat A. Ahmed. 2016. "A New Bioactive Metabolite Isolated from the Red Sea Marine Sponge Hyrtios erectus" Molecules 21, no. 1: 82. https://doi.org/10.3390/molecules21010082
APA StyleElhady, S. S., El-Halawany, A. M., Alahdal, A. M., Hassanean, H. A., & Ahmed, S. A. (2016). A New Bioactive Metabolite Isolated from the Red Sea Marine Sponge Hyrtios erectus. Molecules, 21(1), 82. https://doi.org/10.3390/molecules21010082








