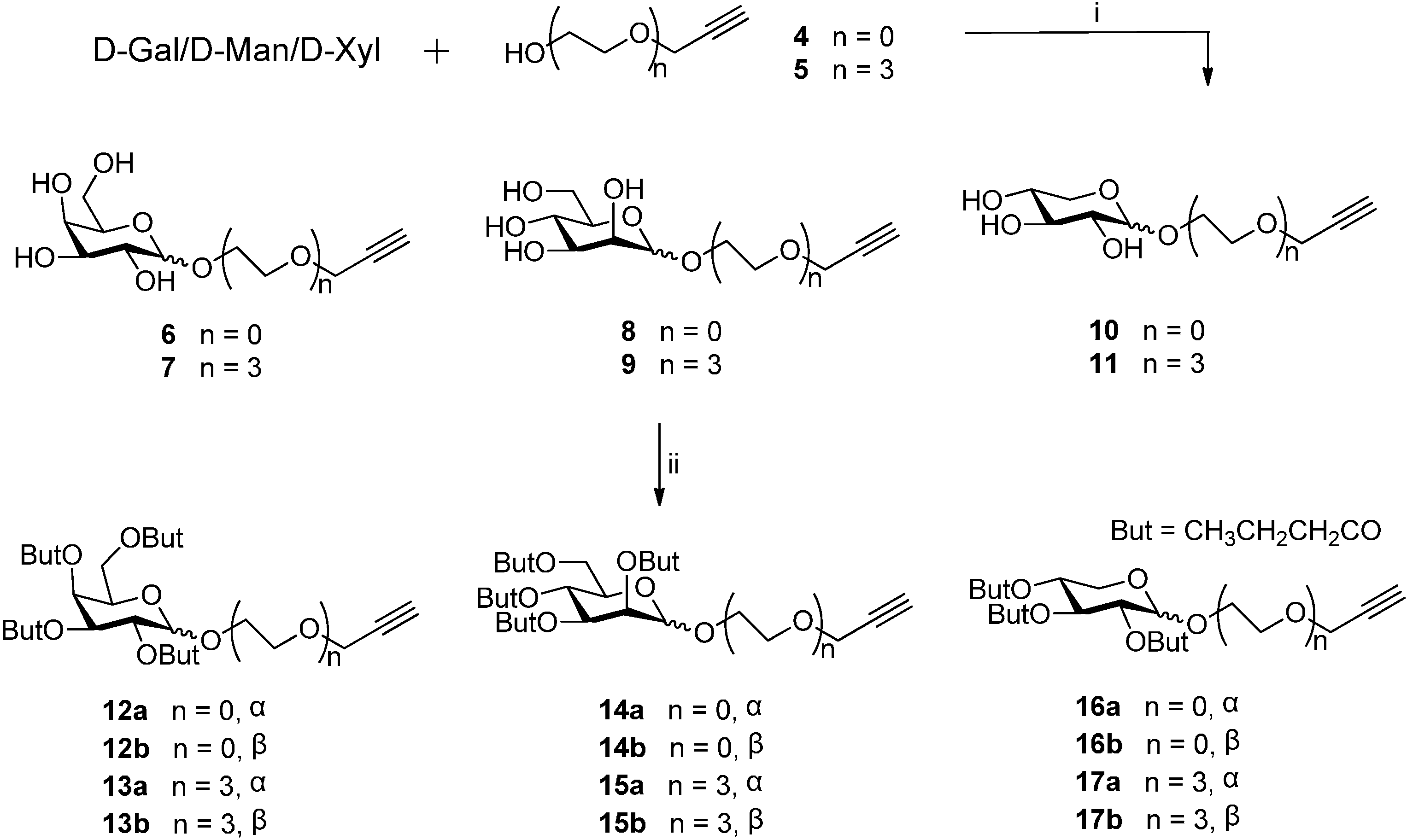
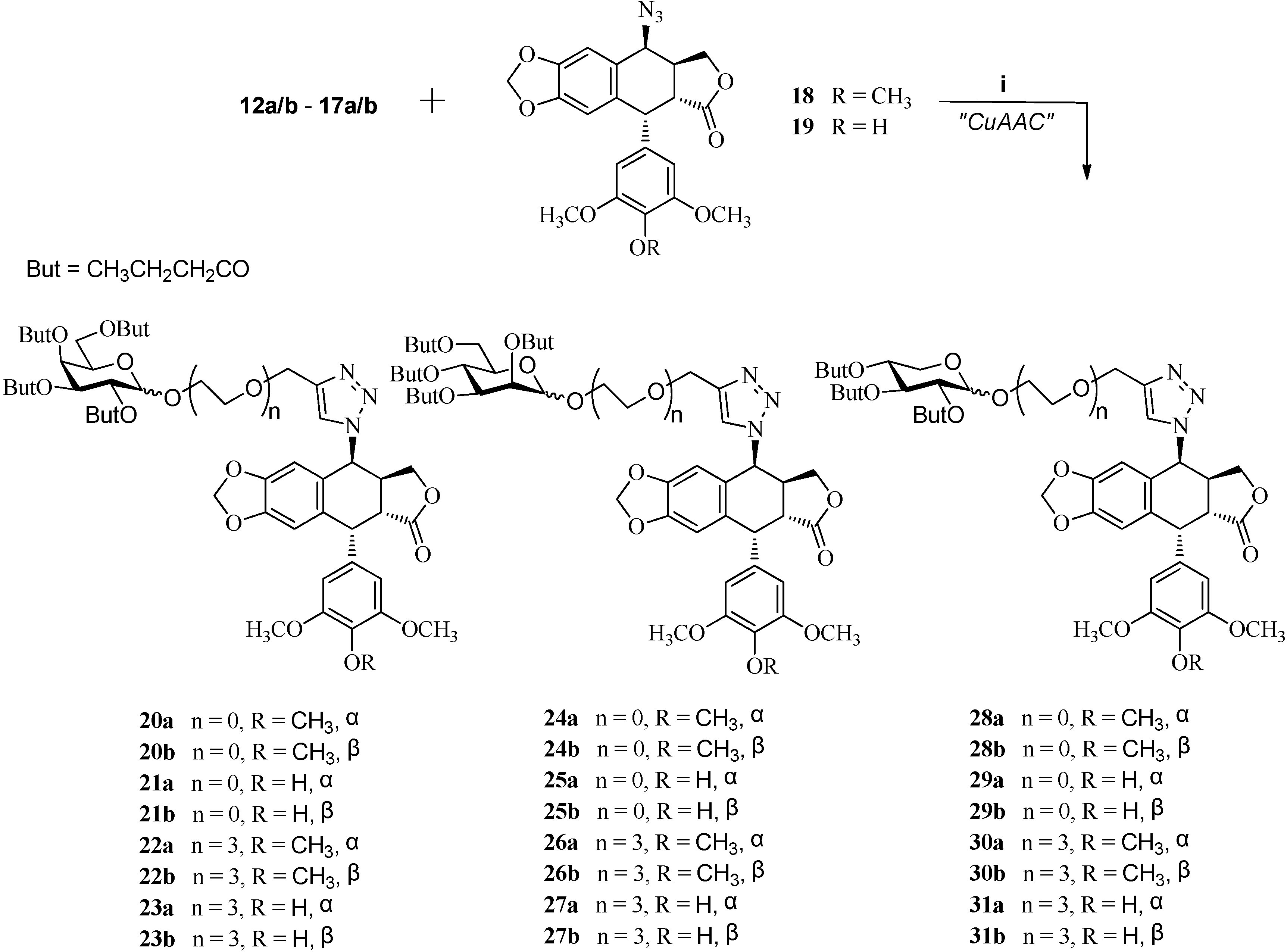3.2. General Procedure for the Synthesis of Compounds 12a/b–17a/b
D-sugar (5 mmol) was suspended in propargyl alcohol 4/5 (25 mmol) and stirred at 65 °C. H2SO4-silica (25 mg) was added and stirring was continued until all solids had dissolved (~2.5 h). After cooling to room temperature, the reaction mixture was transferred to a short silica gel column (CHCl3:CH3OH = 15:1→9:1) to afford the desired propargyl glycosides 6–11. Then, to a solution of a propargyl glycosides 6–11 (1 mmol) in pyridine (4 mL) at 0 °C butyryl anhydride (4 mL) was added. The reaction mixture was stirred overnight until the starting material disappeared as indicated by TLC. The reaction mixture was diluted with water (20 mL) and extracted with ethyl acetate (3 × 20 mL). The organic layer was washed with 10% aqueous hydrochloric acid (20 mL) and brine (20 mL). The organic layer was dried over magnesium sulfate and evaporated to give a residue, which was chromatographed on silica gel with petroleum ether-acetone = 4:1→2:1 to give the perbutyrylated product 12\a/b–17a/b.
3.2.1. 2-Propyn-1-yl-per-O-butyryl-α-d-galactopyranose (12a)
Yield: 56%. 1H-NMR (CDCl3, 400 MHz) δ 5.50 (d, 1H, J = 2.7 Hz, C4-H), 5.36 (dd, 1H, J = 4.0 Hz, 10.0 Hz, C3-H), 5.32 (d, 1H, J = 4.0 Hz, C1-H), 5.15 (dd, 1H, J = 4.0 Hz, 10.0 Hz, C2-H), 4.39–4.32 (m, 3H), 4.12 (d, 2H, J = 7.2 Hz, CH2-C≡CH), 2.92 (t, 1H, J = 2.2 Hz, C≡CH), 2.42 (t, 2H, J = 8.0 Hz, COCH2), 2.31 (m, 4H, 2 × COCH2), 2.20 (t, 2H, J = 8.0 Hz, COCH2), 1.68–1.58 (m, 8H, 4 × CH2CH3), 1.00–0.92 (m, 12H, 4 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) δ 174.3 (C=O), 174.2 (C=O), 174.1 (C=O), 173.7 (C=O), 96.1 (C-1), 79.6 (C≡CH), 77.0 (C≡CH), 69.2, 69.0, 68.7, 68.2, 62.5 (C-6), 56.0 (CH2-C≡C), 36.8 (CH2C=O), 36.8 (CH2C=O), 36.8 (CH2C=O), 36.7 (CH2C=O), 19.7 (CH2CH3), 19.6 (CH2CH3), 19.4 (CH2CH3), 19.2 (CH2CH3), 14.3 (CH2CH3), 14.2 (CH2CH3), 14.2 (CH2CH3), 14.2 (CH2CH3); ESIMS: m/z 521 [M+Na]+, HRESIMS: calcd for C25H38O10Na [M+Na]+ 521.2357, found 521.2366.
3.2.2. 2-Propyn-1-yl-per-O-butyryl-β-d-galactopyranose (12b)
Yield: 33%. 1H-NMR (CDCl3, 400 MHz) δ 5.42 (d, 1H, J = 2.8 Hz, C4-H), 5.19 (dd, 1H, J = 4.0 Hz, 10.0 Hz, C3-H), 5.14 (d, 1H, J = 8.0 Hz, C1-H), 4.86–4.84 (m, 2H), 4.35 (d, 2H, J = 1.9 Hz), 4.14 (s, 2H, CH2-C≡CH), 2.93 (t, 1H, J = 2.2 Hz, C≡CH), 2.42–2.18 (m, 8H, 4 × COCH2), 1.71–1.53 (m, 8H, 4 × CH2CH3), 0.99–0.89 (m, 12H, 4 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) δ 174.5 (C=O), 174.3 (C=O), 173.8 (C=O), 173.7 (C=O), 100.0 (C-1), 79.4 (C≡CH), 76.8 (C≡CH), 72.2, 72.0, 70.0, 68.6, 62.3 (C-6), 56.8 (CH2-C≡C), 36.9 (COCH2), 36.7 (COCH2), 36.7 (COCH2), 36.7 (COCH2), 19.6 (CH2CH3), 19.5 (CH2CH3), 19.3 (CH2CH3), 19.1 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3); ESIMS: m/z 521 [M+Na]+, HRESIMS: calcd for C25H38O10Na [M+Na]+ 521.2357, found 521.2360.
3.2.3. 2-[2-[2-(2-Propyn-1-yloxy)ethoxy]ethoxy-per-O-butyryl-α-d-galactopyranoside (13a)
Yield: 57%. 1H-NMR (CDCl3, 400 MHz) δ 5.45 (d, 1H, J = 2.4 Hz, C4-H), 5.36 (dd, 1H, J = 4.0 Hz, 10.0 Hz, C3-H), 5.14–5.10 (m, 2H, C1-H, C2-H), 4.14 (t, 1H, J = 8.0 Hz), 4.18 (d, 2H, J = 2.4 Hz, CH2-C≡CH), 4.09 (dd, 1H, J = 6.0 Hz, 10.0 Hz,), 3.84–3.80 (m, 1H), 3.66–3.63 (m, 12H, 3 × OCH2CH2O), 2.83 (t, 1H, J = 2.0 Hz, C≡CH), 2.39 (t, 2H, J = 8.0 Hz, COCH2), 2.30–2.28 (m, 4H, 2 × COCH2), 2.18 (t, 2H, J = 8.0 Hz, COCH2), 1.69–1.54 (m, 8H, 4 × CH2CH3), 0.98–0.90 (m, 12H, 4 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) 174.5 (C=O), 174.3 (C=O), 174.3 (C=O), 173.8 (C=O), 97.7 (C1-H), 82.7 (C≡CH), 76.0 (C≡CH), 71.6, 71.6, 71.4, 71.2, 70.1, 69.4, 69.2, 68.9, 68.6, 67.6, 62.6 (C-6), 59.0 (CH2-C≡C), 37.0 (COCH2), 36.8 (COCH2), 36.7 (COCH2), 36.6 (COCH2), 19.6 (CH2CH3), 19.5 (CH2CH3), 19.3 (CH2CH3), 19.2 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3); ESIMS: m/z 653 [M+Na]+, HRESIMS: calcd for C31H50O13Na [M+Na]+ 653.3144, found 653.3149.
3.2.4. 2-[2-[2-(2-Propyn-1-yloxy)ethoxy]ethoxy-per-O-butyryl-β-d-galactopyranoside (13b)
Yield: 39%. 1H-NMR (CDCl3, 400 MHz) δ 5.40 (d, 1H, J = 2.4 Hz, C4-H), 5.13–5.12 (m, 2H, C3-H, C2-H), 4.73 (d, 1H, J = 8.0 Hz, C1-H), 4.19 (d, 2H, J = 2.0 Hz), 4.12 (s, 2H, CH2-C≡CH), 3.66–3.60 (m, 12H, 3 × OCH2CH2O), 2.85 (t, 1H, J = 2.0 Hz, C≡CH), 2.41 (t, 2H, J = 8.0 Hz, COCH2), 2.30–2.29 (m, 4H, 2 × COCH2), 2.17 (t, 2H, J = 8.0 Hz, COCH2), 1.71–1.53 (m, 8H, 4 × CH2CH3), 0.99–0.89 (m, 12H, 4 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) δ 174.5 (C=O), 174.4 (C=O), 173.8 (C=O), 173.7 (C=O), 102.2 (C-1), 76.0 (C≡CH), 72.3, 71.8, 71.6, 71.5, 71.4, 70.2, 70.1, 70.0, 68.6, 62.3 (C-6), 59.0 (CH2-C≡C), 36.9 (COCH2), 36.8 (COCH2), 36.7 (COCH2), 36.7 (COCH2), 19.6 (CH2CH3), 19.5 (CH2CH3), 19.3 (CH2CH3), 19.1 (CH2CH3), 14.0 (CH2CH3), 13.9 (CH2CH3), 13.9 (CH2CH3), 13.9 (CH2CH3); ESIMS: m/z 653 [M+Na]+, HRESIMS: calcd for C31H50O13Na [M+Na]+ 653.3144, found 653.3150.
3.2.5. 2-Propyn-1-yl-per-O-butyryl-α-d-mannopyranoside (14a)
Yield: 60%. 1H-NMR (CDCl3, 400 MHz) δ 5.32 (t, 1H, J = 10.0 Hz, C4-H), 5.23–5.21 (m, 2H, C3-H, C2-H), 4.98 (s, 1H, C1-H), 4.30 (t, 2H, J = 2.4 Hz, CH2-C≡CH), 4.21 (dd, 1H, J = 4.0 Hz, 10.0 Hz), 4.12–4.08 (m, 1H), 4.03–4.00 (m, 1H,), 2.92 (t, 1H, J = 2.4 Hz, C≡CH), 2.37 (t, 2H, J = 8.0 Hz, COCH2), 2.30–2.24 (m, 4H, 2 × COCH2), 2.15 (t, 2H, J = 8.0 Hz, COCH2), 1.69–1.50 (m, 8H, 4 × CH2CH3), 0.97–0.86 (m, 12H, 4 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) δ 174.7 (C=O), 173.8 (C=O), 173.8 (C=O), 173.7 (C=O), 97.5 (C-1), 79.3 (C≡CH), 77.0 (C≡CH), 70.6, 70.4, 70.3, 66.6, 62.9 (C-6), 55.7 (CH2-C≡C), 36.9 (COCH2), 36.9 (COCH2), 36.8 (COCH2), 36.7 (COCH2), 19.6 (CH2CH3), 19.4 (CH2CH3), 19.3 (CH2CH3), 19.1 (CH2CH3), 14.1 (CH2CH3), 14.0 (CH2CH3), 13.9 (CH2CH3), 13.9 (CH2CH3); ESIMS: m/z 521 [M+Na]+, HRESIMS: calcd for C25H38O10Na [M+Na]+ 521.2357, found 521.2363.
3.2.6. 2-Propyn-1-yl-per-O-butyryl-β-d-mannopyranoside (14b)
Yield: 34%. 1H-NMR (CDCl3, 400 MHz) δ 5.44 (d, 1H, J = 3.2 Hz, C2-H), 5.28 (t, 1H, J = 10.0 Hz, C4-H), 5.20 (dd, 1H, J = 4.0 Hz, 10.0 Hz, C3-H), 5.06 (s, 1H, C1-H), 4.35 (d, 2H, J = 2.4 Hz, CH2-C≡CH), 4.26 (dd, 1H, J = 4.0 Hz. 10.0 Hz), 4.19–4.16 (m, 1H), 3.86–3.82 (m, 1H), 2.94 (t, 1H, J = 2.4 Hz, C≡CH), 2.40 (t, 2H, J = 8.0 Hz, COCH2), 2.34 (t, 2H, J = 8.0 Hz, COCH2), 2.28 (t, 2H, J = 8.0 Hz, COCH2), 2.18 (t, 2H, J = 8.0 Hz, COCH2), 1.72–1.64 (m, 4H, 2 × CH2CH3), 1.62–1.53 (m, 4H, 2 × CH2CH3), 1.00–0.89 (m, 12H, 4 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) δ 174.8 (C=O), 174.4 (C=O), 173.8 (C=O), 173.7 (C=O), 97.3 (C-1), 79.1 (C≡CH), 77.1 (C≡CH), 73.5, 82.5, 70.2, 66.8, 62.9 (C-6), 56.7 (CH2-C≡C), 36.9 (COCH2), 36.9 (COCH2), 36.8 (COCH2), 36.8 (COCH2), 19.6 (CH2CH3), 19.4 (CH2CH3), 19.3 (CH2CH3), 19.1 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3), 13.9 (CH2CH3); ESIMS: m/z 521 [M+Na]+, HRESIMS: calcd for C25H38O10Na [M+Na]+ 521.2357, found 521.2364.
3.2.7. 2-[2-[2-(2-Propyn-1-yloxy)ethoxy]ethoxy-per-O-butyryl-α-d-mannopyranoside (15a)
Yield: 62%. 1H-NMR (CDCl3, 400 MHz) δ 5.34 (t, 1H, J = 10.0 Hz, C4-H), 5.30 (d, 1H, J = 3.2 Hz, C2-H), 5.28–5.27 (m, 2H, C3-H, C1-H), 4.22–4.20 (m, 1H), 4.19 (d, 2H, J = 2.4 Hz, CH2-C≡CH), 4.15–4.12 (m, 1H), 3.88–3.84 (m, 1H), 3.71–3.66 (m, 12H, 3 × OCH2CH2O), 2.85 (t, 1H, J = 2.4 Hz, C≡CH), 2.41 (t, 2H, J = 8.0 Hz, COCH2), 2.34 (t, 2H, J = 8.0 Hz, COCH2), 2.32 (t, 2H, J = 8.0 Hz, COCH2), 2.19 (t, 2H, J = 8.0 Hz, COCH2), 1.73–1.65 (m, 4H, 2 × CH2CH3), 1.63–1.54 (m, 4H, 2 × CH2CH3), 1.01–0.90 (m, 12H, 4 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) δ 174.8 (C=O), 173.9 (C=O), 173.8 (C=O), 173.8 (C=O), 99.0 (C-1), 80.7 (C≡CH), 76.0 (C≡CH), 71.7, 71.6, 71.4, 71.2, 70.8, 70.5, 70.1, 69.9, 68.4, 66.8, 63.1 (C-6), 59.0 (CH2-C≡C), 36.9 (COCH2), 36.9 (COCH2), 36.9 (COCH2), 36.8 (COCH2), 19.6 (CH2CH3), 19.4 (CH2CH3), 19.4 (CH2CH3), 19.2 (CH2CH3), 14.1 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3); ESIMS: m/z 653 [M+Na]+, HRESIMS: calcd for C31H50O13Na [M+Na]+ 653.3144, found 653.3149.
3.2.8. 2-[2-[2-(2-Propyn-1-yloxy)ethoxy]ethoxy-per-O-butyryl-β-d-mannopyranoside (15b)
Yield: 34%. 1H-NMR (CDCl3, 400 MHz) δ 5.24–5.22 (m, 1H, C4-H), 5.13 (dd, 1H, J = 4.0 Hz, 10.0 Hz, C3-H), 4.86–4.84 (m, 1H, C2-H), 4.81 (d, 1H, J = 2.0 Hz, C1-H), 4.43 (dd, 1H, J = 2.0 Hz, 10.0 Hz), 4.29–4.25 (m, 1H), 4.19 (d, 2H, J = 2.4 Hz, CH2-C≡CH), 3.95–3.90 (m, 1H), 3.85–3.82 (m, 1H), 3.69–3.66 (m, 12H, 3 × OCH2CH2O), 2.85 (t, 1H, J = 2.4 Hz, C≡CH), 2.35–2.29 (m, 8H, 4 × COCH2), 1.70–1.60 (m, 8H, 4 × CH2CH3), 0.99–0.94 (m, 12H, 4 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) δ 175.1 (C=O), 174.4 (C=O), 174.4 (C=O), 174.0 (C=O), 99.0 (C-1), 80.7 (C≡CH), 76.0 (C≡CH), 73.0, 72.1, 71.7, 71.6, 71.4, 71.3, 70.8, 70.1, 68.2, 66.0, 64.2 (C-6), 59.0 (CH2-C≡C), 37.0 (COCH2), 36.9 (COCH2), 36.9 (COCH2), 36.9 (COCH2), 19.6 (CH2CH3), 19.5 (CH2CH3), 19.4 (CH2CH3), 19.2 (CH2CH3), 14.1 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3); ESIMS: m/z 653 [M+Na]+, HRESIMS: calcd for C31H50O13Na [M+Na]+ 653.3144, found 653.3144.
3.2.9. 2-Propyn-1-yl-per-O-butyryl-α-d-xylopyranoside (16a)
Yield: 61%. 1H-NMR (CDCl3, 400 MHz) δ 5.46 (t, 1H, J = 10.0 Hz, C3-H), 5.23 (d, 1H, J = 4.0 Hz, C1-H), 5.04–4.97 (m, 1H, C2-H), 4.89–4.85 (m, 1H, C4-H), 4.36–4.24 (m, 2H, CH2-C≡CH), 3.80 (dd, 1H, J = 6.0 Hz, 10.0 Hz), 3.63 (t, 1H, J = 10.0 Hz), 2.91 (t, 1H, J = 2.4 Hz, C≡CH), 2.30–2.25 (m, 6H, 3 × COCH2), 1.64–1.57 (m, 6H, 3 × CH2CH3) 0.94–0.92 (m, 9H, 3 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) δ 174.0 (C=O), 174.0 (C=O), 173.8 (C=O), 95.6 (C-1), 79.5 (C≡CH), 76.7 (C≡CH), 71.9, 70.5, 70.3, 59.8 (C-6), 55.8 (CH2-C≡C), 36.9 (COCH2), 36.7 (COCH2), 36.7 (COCH2), 19.4 (CH2CH3), 19.4 (CH2CH3), 19.3 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3); ESIMS: m/z 421 [M+Na]+, HRESIMS: calcd for C20H30O8Na [M+Na]+ 421.1833, found 421.1838.
3.2.10. 2-Propyn-1-yl-per-O-butyryl-β-d-xylopyranoside (16b)
Yield: 29%. 1H-NMR (CDCl3, 400 MHz) δ 5.26 (t, 1H, J = 9.0 Hz, C3-H), 4.96–4.88 (m, 2H, C4-H, C2-H), 4.79 (d, 1H, J = 8.0 Hz, C1-H), 4.33 (t, 2H, J = 1.6 Hz, CH2-C≡CH), 4.08 (dd, 1H, J = 5.0 Hz, 12.0 Hz), 3.47 (dd, 1H, J = 10.0 Hz, 12.0 Hz), 2.93 (t, 1H, J = 2.4 Hz, C≡CH), 2.29–2.23 (m, 6H, 3 × COCH2), 1.65–1.55 (m, 6H, 3 × CH2CH3), 0.94–0.91 (m, 9H, 3 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) δ 173.9 (C=O), 193.9 (C=O), 173.6 (C=O), 100.1 (C-1), 79.4 (C≡CH), 76.7 (C≡CH), 73.0, 72.0, 70.2, 63.3 (C-5), 56.7 (CH2-C≡C), 36.9 (COCH2), 36.8 (COCH2), 36.7 (COCH2), 19.4 (CH2CH3), 19.3 (CH2CH3), 19.3 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3), 13.9 (CH2CH3); ESIMS: m/z 421 [M+Na]+, HRESIMS: calcd for C20H30O8Na [M+Na]+ 421.1833, found 421.1836.
3.2.11. 2-[2-[2-(2-Propyn-1-yloxy)ethoxy]ethoxy-per-O-butyryl-α-d-xylopyranoside (17a)
Yield: 62%. 1H-NMR (CDCl3, 400 MHz) δ 5.48 (t, 1H, J = 10.0 Hz, C3-H), 5.08 (d, 1H, J = 4.0 Hz, C1-H), 5.00–4.94 (m, 1H, C4-H), 4.83 (dd, 1H, J = 4.0 Hz, 10.0 Hz, C2-H), 4.19 (d, 2H, J = 2.4 Hz, CH2-C≡CH), 3.85–3.80 (m, 1H), 3.75–3.73 (m, 1H), 3.71–3.65 (m, 12H, 3 × OCH2CH2O), 2.86 (t, 1H, J = 2.0 Hz, C≡CH), 2.32–2.24 (m, 6H, 3 × COCH2), 1.64–1.56 (m, 6H, 3 × CH2CH3), 0.95–0.90 (m, 9H, 3 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) δ 174.1 (C=O), 174.0 (C=O), 173.9 (C=O), 97.3 (C-1), 80.7 (C≡CH), 76.0 (C≡CH), 72.2, 71.7, 71.6, 71.5, 71.3, 70.7, 70.5, 70.1, 68.6, 59.4 (C-5), 59.1 (CH2-C≡C), 36.9 (COCH2), 36.8 (COCH2), 36.7 (COCH2), 19.4 (CH2CH3), 19.3 (CH2CH3), 19.3 (CH2CH3), 14.0 (CH2CH3), 13.9 (CH2CH3), 13.9 (CH2CH3); ESIMS: m/z 553 [M+Na]+, HRESIMS: calcd for C26H42O11Na [M+Na]+ 553.2619, found 553.2625.
3.2.12. 2-[2-[2-(2-Propyn-1-yloxy)ethoxy]ethoxy-per-O-butyryl-β-d-xylopyranoside (17b)
Yield: 28%. 1H-NMR (CDCl3, 400 MHz) δ 5.23 (t, 1H, J = 9.0 Hz, C3-H), 4.96–4.86 (m, 2H, C2-H, C4-H), 4.65 (d, 1H, J = 8.0 Hz, C1-H), 4.19 (d, 2H, J = 2.4 Hz, CH2C≡CH), 4.06 (dd, 1H, J = 6.0 Hz, 12.0 Hz), 3.91–3.86 (m, 1H), 3.66–3.61 (m, 12H, 3 × OCH2CH2O), 2.86 (t, 1H, J = 2.4 Hz, C≡CH), 2.31–2.22 (m, 6H, 3 × COCH2), 1.63–1.54 (m, 6H, 3 × CH2CH3), 0.93–0.90 (m, 9H, 3 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) δ 173.9 (C=O), 173.9 (C=O), 173.6 (C=O), 102.3 (C-1), 80.7 (C≡CH), 76.0 (C≡CH), 73.1, 72.4, 71.6, 71.6, 71.4, 71.4, 70.3, 70.1, 69.9, 63.4 (C-5), 59.1 (CH2-C≡C), 36.9 (COCH2), 36.9 (COCH2), 36.7 (COCH2), 19.4 (CH2CH3), 19.4 (CH2CH3), 19.3 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3), 13.9 (CH2CH3); ESIMS: m/z 553 [M+Na]+, HRESIMS: calcd for C26H42O11Na [M+Na]+ 553.2619, found 553.2627.
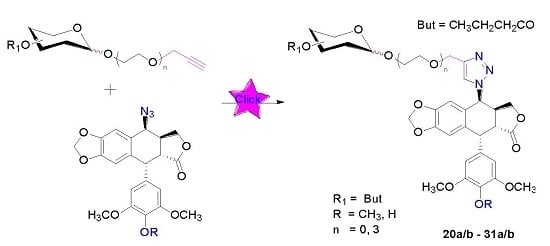
3.3. Click Chemistry-General Procedure
To a solution of a terminal-alkyne 12a/b–17a/b (0.1 mmol) and 4β-azidopodophyllotoxin analogues 18 or 19 (0.1 mmol) in t−BuOH-H2O (1:2, 1.0 mL) at room temperature were added copper (II) sulfate pentahydrate (0.01 mmol) and sodium ascorbate (1.0 M in H2O, 3 drops). The reaction mixture was stirred at room temperature for 2 h until the starting material disappeared as indicated by TLC. Then, the mixture was diluted with water (10 mL) and extracted with ethyl acetate (3 × 10 mL), and the combined organic layer was dried over sodium sulfate. The solvent was evaporated and the residue was purified by column chromatography to afford the cycloaddition product 20a/b–31a/b (82%–92%).
3.3.1. 4β-{4''-[1'''-(2''',3''',4''',6'''-Tetra-O-butyryl-α-d-galactopyranosyloxy)-1,2,3-triazol-1-yl]}-4-deoxy-podophyllotoxin (20a)
White amorphous powder, yield 90% (after chromatography with petroleum ether/acetone, 1:1); mp 87 °C; : +28.7 (c 0.27, CH3OH); 1H-NMR (CD3OD, 500 MHz) δ 7.87 (s, 1H, C5''-H), 6.71 (s, 1H, C5-H), 6.61 (s, 1H, C8-H), 6.43 (s, 2H, C2', C6'-H), 6.26 (d, 1H, J = 5.0 Hz, C4-H), 5.98 (d, 2H, J = 10.0 Hz, OCH2O), 5.50 (d, 1H, J = 4.0 Hz, C4'''-H), 5.36 (dd, 1H, J = 4.0 Hz, 10.0 Hz, C3'''-H), 5.26 (d, 1H, J = 4.0 Hz, C1'''-H), 5.00 (dd, 1H, J = 4.0 Hz, 10.0 Hz, C2'''-H), 4.74–4.72 (m, 2H), 4.31 (d, 1H, J = 5.5 Hz, C1-H), 4.10–4.05 (m, 1H), 4.01–3.99 (m, 1H), 3.74 (s, 2H), 3.75 (s, 6H, C3', C5'-OCH3), 3.72 (s, 3H, C4'-OCH3), 3.39 (dd, 1H, J = 4.0 Hz, 10.0 Hz, C2-H), 3.15–3.11 (m, 1H, C3-H), 2.34 (t, 2H, J = 9.0 Hz, COCH2), 2.23 (t, 2H, J = 9.0 Hz, COCH2), 2.16–2.14 (m, 4H, 2 × COCH2), 1.69–1.58 (m, 2H, CH2CH3), 1.55–1.48 (m, 6H, 3 × CH2CH3), 0.92–0.82 (m, 12H, 4 × CH2CH3); 13C-NMR (CD3OD, 125 MHz) δ 174.2 (C-12), 173.0 (C=O), 172.7 (C=O), 172.6 (C=O), 172.3 (C=O), 152.4 (C-3', C-5'), 149.0 (C-7), 147.7 (C-6), 143.1 (C-4''), 136.7 (C-1'), 135.2 (C-9), 133.2 (C-10), 125.4 (C-4'), 124.8 (C-5''), 109.7 (C-5), 108.3 (C-8), 107.8 (C-2', C-6'), 101.8 (OCH2O), 95.0 (C-1'''), 67.7, 67.6, 67.3 (C-11), 67.2, 66.3, 60.9 (C-6''), 59.9 (C-6'''), 59.5 (4'-OCH3), 58.3 (C-2), 55.1 (3', 5'-OCH3), 43.4 (C-4), 41.0 (C-1), 37.0 (C-3), 35.2 (COCH2), 35.2 (COCH2), 35.1 (COCH2), 35.1 (COCH2), 18.0 (CH2CH3), 17.9 (CH2CH3), 17.8 (CH2CH3), 17.6 (CH2CH3), 12.5 (CH2CH3), 12.5 (CH2CH3), 12.4 (CH2CH3), 12.4 (CH2CH3); ESIMS: m/z 960 [M+Na]+, HRESIMS: calcd for C47H59N3O17H [M+H]+ 938.3917, found 938.3915.
3.3.2. 4β-{4''-[1'''-(2''',3''',4''',6'''-Tetra-O-butyryl-β-d-galactopyranosyloxy)-1,2,3-triazol-1-yl]}-4-deoxy-podophyllotoxin (20b)
White amorphous powder, yield 90% (after chromatography with petroleum ether/acetone, 1:1); mp 92 °C; : −33.2 (c 0.16, CH3OH); 1H-NMR (CD3OD, 400 MHz) δ 7.72 (s,1H, C5''-H), 6.67 (s, 1H, C5-H), 6.58 (s, 1H, C8-H), 6.41 (s, 2H, C2', C6'-H), 6.24 (d, 1H, J = 4.3 Hz, C4-H), 5.94 (d, 2H, J = 7.4 Hz, OCH2O), 5.42 (d, 1H, J = 2.6 Hz, C4'''-H), 5.18–5.09 (m, 2H, C3'''-H, C2'''-H), 4.79 (d, 1H, J = 8.0 Hz, C1'''-H), 4.78–4.77 (m, 2H), 3.34–3.32 (m, 1H), 4.18–4.11 (m, 2H), 3.79 (s, 2H), 3.72 (s, 6H, C3', C5'-OCH3), 3.70 (s, 3H, C4'-OCH3), 3.39 (dd, 1H, J = 4.0 Hz, 10.0 Hz, C2-H), 3.15–3.11 (m, 1H, C3-H), 2.38 (t, 2H, J = 8.0 Hz, COCH2), 2.27 (t, 2H, J = 8.0 Hz, COCH2), 2.17–2.15 (m, 4H, 2 × COCH2), 1.67–1.47 (m, 8H, 4 × CH2CH3); 0.96–0.82 (m, 12H, 4 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) δ 175.6 (C-12), 174.4 (C=O), 174.3 (C=O), 173.7 (C=O), 173.7 (C=O), 154.0 (C-3', C-5'), 150.6 (C-7), 149.3 (C-6), 145.5 (C-4''), 138.6 (C-1'), 136.7 (C-9), 134.8 (C-10), 126.9 (C-4'), 126.0 (C-5''), 111.3 (C-5), 109.9 (C-8), 109.5 (C-2', C-6'), 103.3 (OCH2O), 101.5 (C-1'''), 72.1, 72.0, 70.1, 68.9 (C-11), 68.6, 63.3 (C-6''), 62.3 (C-6'''), 61.1 (4'-OCH3), 59.8 (C-2), 56.7 (3', 5'-OCH3), 44.9 (C-4), 42.5 (C-1), 38.6 (C-3), 36.8 (COCH2), 36.7 (COCH2), 36.7 (COCH2), 36.7 (COCH2), 19.6 (CH2CH3), 19.5 (CH2CH3), 19.3 (CH2CH3), 19.1 (CH2CH3), 14.1 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3); ESIMS: m/z 960 [M+Na]+, HRESIMS: calcd for C47H59N3O17Na [M+Na]+ 938.3917, found 938.3898.
3.3.3. 4β-{4''-[1'''-(2''',3''',4''',6'''-Tetra-O-butyryl-α-d-galactopyranosyloxy)-1,2,3-triazol-1-yl]}-4-deoxy-4'-demethylpodophyllotoxin (21a)
White amorphous powder, yield 89% (after chromatography with petroleum ether/acetone, 1:1); mp 89 °C; : +22.2 (c 0.22, CH3OH); 1H-NMR (CD3OD, 500 MHz) δ 8.24 (s, 1H, C5''-H), 6.61 (s, 3H, C5-H, C2', C6'-H), 6.24 (s, 1H, C8-H), 5.99–5.91 (m, 3H, C4-H, OCH2O), 5.52 (d, 1H, J = 4.0 Hz, C4'''-H), 5.42 (dd, 1H, J = 4.0 Hz, 10.0 Hz, C3'''-H), 5.32 (d. 1H, J = 4.0 Hz. C1'''-H), 5.12 (dd, 1H, J = 4.0 Hz, 10.0 Hz, C2'''-H), 4.80–4.77 (m, 2H), 4.67 (d, 1H, J = 5.0 Hz, C1-H), 4.43 (t, 1H, J = 8.0 Hz, C5'''-H), 4.25 (t, 1H, J = 8.0 Hz), 4.21–4.17 (m, 1H), 4.16–4.06 (m, 2H), 3.79 (s, 6H, C3', C5'-OCH3), 3.57–3.49 (m, 1H, C3-H), 3.19 (dd, 1H, J = 5.0 Hz, 10.0 Hz, C2-H), 2.42 (t, 2H, J = 8.0 Hz, COCH2), 2.29–2.20 (m, 6H, 3 × COCH2), 1.73–1.66 (m, 2H, CH2CH3), 1.61–1.52 (m, 6H, 3 × CH2CH3), 1.00–0.86 (m, 12H, 4 × CH2CH3); 13C-NMR (CD3OD, 125 MHz) δ 175.9 (C-12), 174.6 (C=O), 174.3 (C=O), 174.2 (C=O), 173.9 (C=O), 149.7 (C-7), 149.1 (C-6), 148.7 (C-3', C-5'), 144.9 (C-4''), 135.8 (C-1'), 134.3 (C-9), 131.7 (C-10), 129.0 (C-4'), 125.8 (C-5''), 110.0 (C-5), 109.5 (C-2', C-6'), 107.3 (C-8), 103.1 (OCH2O), 96.9 (C-1'''), 71.3 (C-11), 69.3, 69.3, 68.9, 68.0, 64.0 (C-4), 62.5 (C-6'''), 61.8 (C-6''), 57.0 (3', 5'-OCH3), 46.6 (C-1), 45.1 (C-2), 40.0 (C-3), 36.8 (COCH2), 36.8 (COCH2), 36.7 (COCH2), 36.7 (COCH2), 19.5 (CH2CH3), 19.4 (CH2CH3), 19.3 (CH2CH3), 19.1 (CH2CH3), 14.5 (CH2CH3), 14.0 (CH2CH3), 13.9 (CH2CH3), 13.9 (CH2CH3); ESIMS: m/z 946 [M+Na]+, HRESIMS: calcd for C46H57N3O17Na [M+Na]+ 946.3580, found 946.3555.
3.3.4. 4β-{4''-[1'''-(2''',3''',4''',6'''-Tetra-O-butyryl-β-d-galactopyranosyloxy)-1,2,3-triazol-1-yl]}-4-deoxy-4'-demethylpodophyllotoxin (21b)
White amorphous powder, yield 91% (after chromatography with petroleum ether/acetone, 1:1); mp 103–105 °C; : −45.1 (c 0.27, CH3OH); 1H-NMR (CD3OD, 400 MHz) δ 7.72 (s, 1H, C5''-H), 6.66 (s, 1H, C5-H), 6.61 (s, 1H, C8-H), 6.38 (s, 2H, C2', C6'-H), 6.23 (d, 1H, J = 3.9 Hz, C4-H), 5.95 (d, 2H, J = 8.2 Hz, OCH2O), 5.42 (d, 1H, J = 2.4 Hz, C4'''-H), 5.14–5.11 (m, 2H, C3'''-H, C2'''-H), 4.81 (d, 1H, J = 8.0 Hz, C1'''-H), 4.79–4.75 (m, 3H), 4.36 (m, 1H), 4.17–4.09 (m, 4H), 3.73 (s, 6H, C3', C5'-OCH3), 3.35–3.34 (m, 1H, C2-H), 3.15–3.11 (m, 1H, C3-H), 2.38 (t, 2H, J = 7.2 Hz, COCH2), 2.27 (t, 2H, J = 7.2 Hz, COCH2), 2.17 (t, 2H, J = 7.2 Hz, COCH2), 2.15 (t, 2H, J = 7.2 Hz, COCH2), 1.68–1.48 (m, 8H, 4 × CH2CH3), 0.97–0.83 (m, 12H, 4 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) δ 175.8 (C-12), 174.5 (C=O), 174.3 (C=O), 173.8 (C=O), 173.8 (C=O), 150.5 (C-7), 149.2 (C-6), 148.7 (C-3', C-5'), 145.4 (C-4''), 136.1 (C-1'), 135.1 (C-9), 131.3 (C-10), 126.9 (C-4'), 126.0 (C-5''), 111.3 (C-5), 109.8 (C-8), 109.4 (C-2', C-6'), 103.3 (OCH2O), 101.5 (C-1'''), 72.1, 72.0, 70.1, 62.2, 68.5 (C-11), 63.2 (C-6''), 62.2 (C-6'''), 59.9 (C-2), 56.8 (3', 5'-OCH3), 44.8 (C-4), 42.7 (C-1), 38.5 (C-3), 36.8 (COCH2), 36.7 (COCH2), 36.7 (COCH2), 36.6 (COCH2), 19.6 (CH2CH3), 19.4 (CH2CH3), 19.3 (CH2CH3), 19.1 (CH2CH3), 14.0 (CH2CH3), 13.9 (CH2CH3), 13.9 (CH2CH3), 13.9 (CH2CH3); ESIMS: m/z 946 [M+Na]+, HRESIMS: calcd for C46H57N3O17H [M+H]+ 924.3761, found 924.3745.
3.3.5. 4β-{4''-[1'''-(2''',3''',4''',6'''-Tetra-O-butyryl-α-d-galactopyranosyloxy)-3,6,9-trioxadec-10-yl]-1,2,3-triazol-1-yl}-4-deoxypodophyllotoxin (22a)
White amorphous powder, yield 82% (after chromatography with petroleum ether/acetone, 1:1); mp 82 °C; : −26.2 (c 0.18, CH3OH); 1H-NMR (CD3OD, 400 MHz) δ 7.81 (s, 1H, C5''-H), 6.70 (s, 1H, C5-H), 6.63 (s, 1H, C8-H), 6.42 (s, 2H, C2', C6'-H), 6.27 (d, 1H, J = 4.8 Hz, C4-H), 5.98 (d, 2H, J = 8.4 Hz, OCH2O), 5.41 (d, 1H, J = 1.2 Hz, C4'''-H), 5.16–5.10 (m, 3H, C1'''-H, C3'''-H, C2'''-H), 4.81 (d, 1H, J = 5.2 Hz, C1-H), 4.74–4.72 (m, 1H), 4.65–4.63 (m, 2H), 4.41–4.36 (m, 1H), 4.13–4.12 (m, 2H), 3.92–3.86 (m, 1H), 3.81–3.80 (m, 1H), 3.74 (s, 6H, C3', C5'-OCH3), 3.72 (s, 3H, C4'-OCH3), 3.66–3.58 (m, 12H, 3 × OCH2CH2O), 3.43 (dd, 1H, J = 1.2 Hz, 10.0 Hz, C2-H), 3.19–3.14 (m, 1H, C3-H), 3.36 (t, 2H, J = 8.0 Hz, COCH2), 2.28–2.26 (m, 4H, 2 × COCH2), 1.64–1.52 (m, 8H, 4 × CH2CH3), 0.96–0.89 (m, 12H, 4 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) δ 175.8 (C-12), 174.5 (C=O), 174.3 (C=O), 173.8 (C=O), 173.8 (C=O), 154.0 (C-3', C-5'), 150.6 (C-7), 149.3 (C-6), 146.1 (C-4''), 138.3 (C-1'), 136.8 (C-9), 134.8 (C-10), 127.0 (C-4'), 125.9 (C-5''), 111.2 (C-5), 109.9 (C-8), 109.4 (C-2', C-6'), 103.3 (OCH2O), 102.3 (C-1'''), 72.3, 71.8, 71.6, 71.5, 71.4, 70.9, 70.2, 70.1, 68.9 (C-11), 68.6, 65.0 (C-6''), 63.3 (C-6'''), 61.1 (4'-OCH3), 59.8 (C-2), 56.6 (3', 5'-OCH3), 44.9 (C-4), 42.5 (C-1), 38.6 (C-3), 36.9 (COCH2), 36.7 (COCH2), 36.7 (COCH2), 36.7 (COCH2), 19.6 (CH2CH3), 19.5 (CH2CH3), 19.3 (CH2CH3), 19.1 (CH2CH3), 14.0 (CH2CH3), 13.9 (CH2CH3), 13.9 (CH2CH3), 13.9 (CH2CH3); ESIMS: m/z 1092 [M+Na]+, HRESIMS: calcd for C53H71N3O20Na [M+Na]+ 1092.4523, found 1092.4484.
3.3.6. 4β-{4''-[1'''-(2''',3''',4''',6'''-Tetra-O-butyryl-β-d-galactopyranosyloxy)-3,6,9-trioxadec-10-yl]-1,2,3-triazol-1-yl}-4-deoxypodophyllotoxin (22b)
White amorphous powder, yield 88% (after chromatography with petroleum ether/acetone, 1:1); mp 75 °C; : −25.5 (c 0.14, CH3OH); 1H-NMR (CD3OD, 400 MHz) δ 7.81 (s,1H, C5''- H), 6.70 (s, 1H, C5-H), 6.62 (s, 1H, C8-H), 6.42 (s, 2H, C2', C6'-H), 6.26 (d, 1H, J = 4.8 Hz, C4-H), 5.96 (d, 2H, J = 9.2 Hz, OCH2O), 5.41 (d, 1H, J = 2.8 Hz, C4'''-H), 5.16–5.12 (m, 2H, C3'''-H, C2'''-H), 4.80 (d, 1H, J = 5.2 Hz, C1-H), 4.74 (d, 1H, J = 7.2 Hz, C1'''-H), 4.62 (s, 2H), 4.40–4.34 (m, 1H), 4.13-4.12 (m, 2H), 3.91–3.86 (m, 1H), 3.74 (s, 6H, C3', C5'-OCH3), 3.72 (s, 3H, C4'-OCH3), 3.66–3.57 (m, 12H, 3 × OCH2CH2O), 3.44 (dd, 1H, J = 5.2 Hz, 10.8 Hz, C2-H), 3.18–3.13 (m, 1H, C3-H), 3.35 (t, 2H, J = 8.0 Hz, COCH2), 2.29–2.27 (m, 4H, 2 × COCH2), 2.18 (t, 2H, J = 7.6 Hz, COCH2), 1.67–1.52 (m, 8H, 4 × CH2CH3), 0.95–0.89 (m, 12H, 4 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) δ 175.7 (C-12), 174.5 (C=O), 174.3 (C=O), 173.8 (C=O), 173.8 (C=O), 154.0 (C-3', C-5'), 150.5 (C-7), 149.3 (C-6), 146.1 (C-4''), 138.3 (C-1'), 136.8 (C-9), 134.8 (C-10), 127.0 (C-4'), 125.9 (C-5''), 111.2 (C-5), 109.9 (C-8), 109.5 (C-2', C-6'), 103.3 (OCH2O), 102.3 (C-1'''), 72.3, 71.8, 71.6, 71.5, 71.4, 70.9, 70.2, 70.1, 68.9 (C-11), 68.7, 65.0 (C-6''), 62.4 (C-6'''), 61.1 (4'-OCH3), 59.8 (C-2), 56.7 (3', 5'-OCH3), 44.8 (C-4), 42.5 (C-1), 38.6 (C-3), 36.9 (COCH2), 36.7 (COCH2), 36.7 (COCH2), 36.7 (COCH2), 19.6 (CH2CH3), 19.5 (CH2CH3), 19.3 (CH2CH3), 19.1 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3), 13.9 (CH2CH3), 13.9 (CH2CH3); ESIMS: m/z 1078 [M+Na]+, HRESIMS: calcd for C52H69N3O20Na [M+Na]+ 1078.4367, found 1078.4345.
3.3.7. 4β-{4''-[1'''-(2''',3''',4''',6'''-Tetra-O-butyryl-α-d-galactopyranosyloxy)-3,6,9-trioxadec-10-yl]-1,2,3-triazol-1-yl}-4-deoxy-4'-demethylpodophyllotoxin (23a)
White amorphous powder, yield 87% (after chromatography with petroleum ether/acetone, 1:1); mp 84–85 °C; : +6.7 (c 0.23, CH3OH); 1H-NMR (CD3OD, 400 MHz) δ 7.79 (s,1H, C5''-H), 6.69 (s, 1H, C5-H), 6.65 (s, 1H, C8-H), 6.38 (s, 2H, C2', C6'-H), 6.26 (d, 1H, J = 4.8 Hz, C4-H), 5.98 (d, 2H, J = 5.6 Hz, OCH2O), 5.47 (d, 1H, J = 2.4 Hz, C4'''-H), 5.37 (dd, 1H, J = 3.2 Hz, 10.8 Hz, C2'''-H), 5.15–5.08 (m, 2H, C1'''-H, C3'''-H), 4.77 (d, 1H, J = 4.4 Hz, C1-H), 4.63 (s, 2H), 4.44–4.37 (m, 2H), 4.12–4.06 (m, 1H), 3.84–3.80 (m, 1H), 3.74 (s, 6H, C3', C5'-OCH3), 3.67–3.60 (m, 12H, 3 × OCH2CH2CO), 3.40 (dd, 1H, J = 4.4 Hz, 10.8 Hz, C2-H), 3.19–3.13 (m, 1H, C3-H), 2.39 (t, 2H, J = 8.0 Hz, COCH2), 2.29–2.27 (m, 4H, 2 × COCH2), 2.19 (t, 2H, J = 8.0 Hz, COCH2), 1.69–1.53 (m, 8H, 2 × CH2CH3), 0.98–0.90 (m, 12H, 4 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) δ 174.4 (C-12), 173.0 (C=O), 172.8 (C=O), 172.8 (C=O), 172.4 (C=O), 149.0 (C-7), 147.7 (C-6), 147.2 (C-3', C-5'), 144.6 (C-4''), 134.5 (C-1'), 133.6 (C-9), 129.8 (C-10), 125.3 (C-4'), 124.2 (C-5''), 109.7 (C-5), 108.2 (C-8), 107.8 (C-2', C-6'), 101.7 (OCH2O), 96.1 (C-1'''), 70.0, 69.6, 69.6, 69.4, 67.8, 67.7, 67.3 (C-11), 67.0, 63.5 (C-6''), 60.0 (C-6'''), 58.3 (C-2), 55.2 (3', 5'-OCH3), 43.2 (C-4), 41.1 (C-1), 37.0 (C-3), 35.2 (COCH2), 35.1 (COCH2), 35.1 (COCH2), 35.1 (COCH2), 18.0 (CH2CH3), 17.9 (CH2CH3), 17.7 (CH2CH3), 17.6 (CH2CH3), 12.4 (CH2CH3), 12.4 (CH2CH3), 12.4 (CH2CH3), 12.4 (CH2CH3); ESIMS: m/z 1078 [M+Na]+, HRESIMS: calcd for C52H69N3O20Na [M+Na]+ 1078.4367, found 1078.4345.
3.3.8. 4β-{4''-[1'''-(2''',3''',4''',6'''-Tetra-O-butyryl-β-d-galactopyranosyloxy)-3,6,9-trioxadec-10-yl]-1,2,3-triazol-1-yl}-4-deoxy-4'-demethylpodophyllotoxin (23b)
White amorphous powder, yield 85% (after chromatography with petroleum ether/acetone, 1:1); mp 77°C; : −21.5 (c 0.29, CH3OH); 1H-NMR (CD3OD, 400 MHz) δ 7.81 (s, 1H, C5''-H), 6.69 (s, 1H, C5-H), 6.63 (s, 1H, C8-H), 6.39 (s, 2H, C2', C6'-H), 6.25 (d, 1H, J = 4.8 Hz, C4-H), 5.96 (d, 2H, J = 8.8 Hz, OCH2O), 5.41 (d, 1H, J = 2.8 Hz, C4'''-H), 5.16–5.10 (m, 2H, C3'''-H, C2'''-H), 4.77 (d, 1H, J = 4.8 Hz, C1-H), 4.73 (d, 1H, J = 7.2 Hz, C1'''-H), 4.62 (s, 2H), 4.39–4.36 (m, 1H), 4.13 (s, 2H), 3.91–3.86 (m, 1H), 3.74 (s, 6H, C3', C5'-OCH3), 3.66–3.57 (m, 12H, 3 × OCH2CH2CO), 3.40 (dd, 1H, J = 4.8Hz, 10.8 Hz, C2-H), 3.17–3.13 (m, 1H, C3-H), 2.36 (t, 2H, J = 7.6 Hz, COCH2), 2.29–2.27 (m, 4H, 2 × COCH2), 2.18 (t, 2H, J = 7.6 Hz, COCH2), 1.67–1.52 (m, 8H, 4 × CH2CH3) 0.95–0.89 (m, 12H, 4 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) δ 174.8 (C-12), 173.4 (C=O), 174.2 (C=O), 172.7 (C=O), 172.7 (C=O), 149.4 (C-7), 148.1 (C-6), 147.6 (C-3', C-5'), 145.0 (C-4''), 134.9 (C-1'), 134.0 (C-9), 130.2 (C-10), 125.8 (C-4'), 124.8 (C-5''), 110.2 (C-5), 108.7 (C-8), 108.3 (C-2', C-6'), 102.1 (OCH2O), 101.1 (C-1'''), 72.3, 71.8, 71.6, 71.5, 71.4, 70.9, 70.2, 70.1, 68.9 (C-11), 68.6, 65.0 (C-6''), 62.4 (C-6'''), 59.9 (C-2), 55.7 (3', 5'-OCH3), 43.6 (C-4), 41.5 (C-1), 37.4 (C-3), 35.8 (COCH2), 35.6 (COCH2), 35.6 (COCH2), 35.5 (COCH2), 18.4 (CH2CH3), 18.4 (CH2CH3), 18.2 (CH2CH3), 18.0 (CH2CH3), 12.9 (CH2CH3), 12.8 (CH2CH3), 12.8 (CH2CH3), 12.8 (CH2CH3); ESIMS: m/z 1078 [M+Na]+, HRESIMS: calcd for C52H69N3O20H [M+H]+ 1056.4547, found 1056.4528.
3.3.9. 4β-{4''-[1'''-(2''',3''',4''',6'''-Tetra-O-butyryl-α-d-mannopyranosyloxy)-1,2,3-triazol-1-yl]}-4-deoxy-podophyllotoxin (24a)
White amorphous powder, yield 90% (after chromatography with petroleum ether/acetone, 1:1); mp 80 °C; : −3.8 (c 0.27, CH3OH); 1H-NMR (CD3OD, 400 MHz) δ 7.85 (s, 1H, C5''-H), 6.68 (s, 1H, C5-H), 6.57 (s, 1H, C8-H), 6.40 (s, 2H, C2', C6'-H), 6.24 (d, 1H, J = 4.4 Hz, C4-H), 5.93 (d, 2H, J = 9.2 Hz, OCH2O), 5.35 (t, 1H, J = 10.0 Hz, C4'''-H), 5.25 (dd, 1H, J = 2.8 Hz, 10.0 Hz, C3'''-H), 5.19–5.18 (m, 1H, C2'''-H), 4.93 (d, 1H, J = 2.8 Hz, C1'''-H), 4.78–4.76 (m, 2H), 4.68 (s, 1H, C1-H), 4.35 (t, 1H, J = 6.8 Hz), 4.22–4.20 (m, 1H), 4.10–4.06 (m, 2H), 3.73 (s, 6H, C3', C5'-OCH3), 3.71 (s, 3H, C4'-OCH3), 3.30 (dd, 1H, J = 4.8 Hz, 10.4 Hz, C2-H), 3.20–3.15 (m, 1H, C3-H), 3.38–3.34 (m, 8H, 4 × COCH2), 1.69–1.62 (m, 4H, 2 × CH2CH3), 1.60–1.51 (m, 4H, 2 × CH2CH3), 0.97–0.87 (m, 12H, 4 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) δ 175.7 (C-12), 174.7 (C=O), 174.0 (C=O), 173.9 (C=O), 173.7 (C=O), 154.0 (C-3', C-5'), 150.6 (C-7), 149.3 (C-6), 144.9 (C-4''), 138.3 (C-1'), 136.8 (C-9), 134.8 (C-10), 126.9 (C-4'), 126.2 (C-5''), 111.2 (C-5), 109.9 (C-8), 109.5 (C-2', C-6'), 103.3 (OCH2O), 98.2 (C-1'''), 70.6, 70.6, 70.2, 68.9 (C-11), 66.6, 62.9 (C-6''), 61.7 (C-6'''), 61.0 (4'-OCH3), 59.9 (C-2), 56.6 (3', 5'-OCH3), 44.9 (C-4), 42.50 (C-1), 38.6 (C-3), 36.8 (COCH2), 36.8 (COCH2), 36.8 (COCH2), 36.7(COCH2), 19.6 (CH2CH3), 19.4 (CH2CH3), 19.3 (CH2CH3), 19.2 (CH2CH3), 14.1 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3); ESIMS: m/z 960 [M+Na]+, HRESIMS: calcd for C47H59N3O17H [M+H]+ 938.3917, found 938.3906.
3.3.10. 4β-{4''-[1'''-(2''',3''',4''',6'''-Tetra-O-butyryl-β-d-mannopyranosyloxy)-1,2,3-triazol-1-yl]}-4-deoxy-podophyllotoxin (24b)
White amorphous powder, yield 86% (after chromatography with petroleum ether/acetone, 1:1); mp 92–93 °C; : −52.1 (c 0.17, CH3OH); 1H-NMR (CD3OD, 400 MHz) δ 7.74 (s, 1H, C5''-H), 6.66 (s, 1H, C5-H), 6.59 (s, 1H, C8-H), 6.40 (s, 2H, C2', C6'-H), 6.22 (d, 1H, J = 4.4 Hz, C4-H), 5.94 (d, 2H, J = 10.0 Hz, OCH2O), 5.42 (d, 1H, J = 2.8 Hz, C2'''-H), 5.26 (t, 1H, J = 10.0 Hz, C4'''-H), 5.16 (dd, 1H, J = 2.8 Hz, 10.0 Hz, C3'''-H), 4.99 (s, 1H, C1'''-H), 4.84 (s, 1H, C1-H), 4.77–4.72 (m, 2H), 4.36–4.32 (m, 1H), 4.25 (dd, 1H, J = 4.4 Hz, 10.4 Hz), 4.17–4.12 (m, 1H,), 3.85–3.82 (m, 1H), 3.72 (s, 6H, C3', C5'-OCH3), 3.71 (s, 3H, C4'-OCH3), 3.39 (dd, 1H, J = 4.8 Hz, 10.0 Hz, C2-H), 3.17–3.13 (m, 1H, C3-H), 2.34–2.25 (m, 8H, 4 × COCH2), 1.66–1.51 (m, 8H, 4 × CH2CH3) 0.94–0.88 (t, 12H, 4 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) δ 175.7 (C-12), 174.7 (C=O), 174.3 (C=O), 173.8 (C=O), 173.7 (C=O), 154.0 (C-3', C-5'), 150.5 (C-7), 149.3 (C-6), 145.3 (C-4''), 138.3 (C-1'), 136.7 (C-9), 134.8 (C-10), 126.9 (C-5''), 126.2 (C-4'), 111.2 (C-5), 109.4 (C-8), 109.9 (C-2', C-6'), 103.3 (OCH2O), 99.3 (C-1'''), 73.5, 72.5, 70.3, 69.3 (C-11), 66.8, 63.5 (C-6''), 62.9 (C-6'''), 61.1 (4'-OCH3), 59.9 (C-2), 56.6 (3', 5'-OCH3), 44.9 (C-4), 42.5 (C-1), 38.9 (COCH2), 36.9 (COCH2), 36.8 (COCH2), 36.8 (COCH2), 19.7 (CH2CH3), 19.4 (CH2CH3), 19.2 (CH2CH3), 19.2 (CH2CH3), 14.1 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3); ESIMS: m/z 960 [M+Na]+, HRESIMS: calcd for C47H59N3O17H [M+H]+ 938.3917, found 938.3902.
3.3.11. 4β-{4''-[1'''-(2''',3''',4''',6'''-Tetra-O-butyryl-α-d-mannopyranosyloxy)-1,2,3-triazol-1-yl]}-4-deoxy-4'-demethylpodophyllotoxin (25a)
White amorphous powder, yield 92% (after chromatography with CHCl3/CH3OH, 9:1); mp 94–96 °C; : −46.3 (c 0.17, Pyridine); 1H-NMR (C5D5N, 400 MHz) δ 8.32 (s,1H, C5''-H), 6.82 (s, 1H, C5-H), 6.83 (s, 1H, C8-H), 6.78 (s, 2H, C2', C6'-H), 6.55 (d, 1H, J = 4.8 Hz, C4-H), 5.97 (d, 2H, J = 4.4 Hz, OCH2O), 5.83 (t, 1H, J = 10.0 Hz, C4'''-H), 5.72 (dd, 1H, J = 3.2 Hz, 10.0 Hz, C3'''-H), 5.68–5.67 (m, 1H, C2'''-H), 5.38 (s, 1H, C1'''-H), 5.18–5.15 (m, 2H), 5.02 (s, 1H, C1-H), 4.97 (t, 1H, J = 5.2 Hz), 4.55 (dd, 1H, J = 4.8 Hz, 10.0 Hz), 4.48–4.45 (m, 2H), 3.77 (dd, 1H, J = 5.2 Hz, 10.8 Hz, C2-H), 3.72 (s, 6H, C3', C5'-OCH3), 3.50–3.45 (m, 1H, C3-H), 2.42–2.38 (m, 4H, 2 × COCH2), 2.31–2.26 (m, 6H, 3 × COCH2), 1.70–1.56 (m, 8H, 4 × CH2CH3), 0.87–0.80 (m, 12H, 4 × CH2CH3); 13C-NMR (C5D5N, 100 MHz) δ 174.1 (C-12), 173.2 (C=O), 172.7 (C=O), 172.5 (C=O), 172.5 (C=O), 149.5 (C-7), 148.7 (C-3', C-5'), 148.2 (C-6), 144.2 (C-4''), 137.3 (C-1'), 134.6 (C-9), 130.1 (C-10), 126.3 (C-5''), 125.2 (C-4'), 110.8 (C-5), 109.7 (C-2', C-6'), 109.3 (C-8), 102.4 (OCH2O), 97.4 (C-1'''), 69.9, 69.8, 69.6, 67.9 (C-11), 66.1, 62.3 (C-6''), 61.4 (C-6'''), 59.0 (C-2), 56.5 (3', 5'-OCH3), 44.1 (C-4), 42.0 (C-1), 38.0 (C-3), 36.2 (COCH2), 36.0 (COCH2), 36.0 (COCH2), 36.0 (COCH2), 18.7 (CH2CH3), 18.7 (CH2CH3), 18.7 (CH2CH3), 18.5 (CH2CH3), 13.8 (CH2CH3), 13.7 (CH2CH3), 13.7 (CH2CH3), 13.6 (CH2CH3); ESIMS: m/z 946 [M+Na]+, HRESIMS: calcd for C46H57N3O17H [M+H]+ 924.3761, found 924.3752.
3.3.12. 4β-{4''-[1'''-(2''',3''',4''',6'''-Tetra-O-butyryl-β-d-mannopyranosyloxy)-1,2,3-triazol-1-yl]}-4-deoxy-4'-demethylpodophyllotoxin (25b)
White amorphous powder, yield 87% (after chromatography with petroleum ether/acetone, 1:1); mp 99–100 °C; : −63.6 (c 0.13, CH3OH); 1H-NMR (CD3OD, 400 MHz) δ 7.73 (s, 1H, C5''-H), 6.66 (s, 1H, C5-H), 6.61 (s, 1H, C8-H), 6.38 (s, 2H, C2', C6'-H), 6.22 (d, 1H, J = 4.4 Hz, C4-H), 5.95 (d, 2H, J = 9.2 Hz, OCH2O), 5.42 (d, 1H, J = 2.8 Hz, C2'''-H), 5.26 (t, 1H, J = 10.0 Hz, C4'''-H), 5.15 (dd, 1H, J = 2.8 Hz, 10.0 Hz, C3'''-H), 4.99 (s, 1H, C1'''-H), 4.86 (d, 1H, J = 4.4 Hz, C1-H), 4.76–4.73 (m, 2H), 4.37–4.34 (m, 1H), 4.26 (dd, 1H, J = 4.0 Hz, 10.0 Hz), 4.14 (dd, 1H, J = 2.0 Hz, 10.0 Hz), 3.86–3.81 (m, 1H), 3.73 (s, 6H, C3', C5'-OCH3), 3.34–3.33 (m, 1H, C2-H), 3.18–3.13 (m, 1H, C3-H), 2.35–2.26 (m, 8H, 4 × COCH2), 1.67–1.52 (m, 8H, 4 × CH2CH3), 0.96–0.88 (m, 12H, 4 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) δ 175.9 (C-12), 174.7 (C=O), 174.4 (C=O), 173.4 (C=O), 173.7 (C=O), 150.5 (C-7), 149.2 (C-6), 148.7 (C-3', C-5'), 145.3 (C-4''), 136.0 (C-1'), 135.1 (C-9), 131.3 (C-10), 126.8 (C-4'), 126.2 (C-5''), 111.3 (C-5), 109.8 (C-8), 109.3 (C-2', C-6'), 103.3 (OCH2O), 99.3 (C-1'''), 73.5, 72.4, 70.3, 66.8, 68.9 (C-11), 63.5 (C-6''), 62.9 (C-6'''), 60.0 (C-2), 56.8 (3', 5'-OCH3), 44.89 (C-4), 42.7 (C-1), 38.5 (C-3), 36.9 (COCH2), 36.8 (COCH2), 36.8 (COCH2), 36.7 (COCH2), 19.7 (CH2CH3), 19.4 (CH2CH3), 19.3 (CH2CH3), 19.2 (CH2CH3), 14.1 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3), 13.9 (CH2CH3); ESIMS: m/z 946 [M+Na]+, HRESIMS: calcd for C46H57N3O17H [M+H]+ 924.3761, found 924.3753.
3.3.13. 4β-{4''-[1'''-(2''',3''',4''',6'''-Tetra-O-butyryl-α-d-mannopyranosyloxy)-3,6,9-trioxadec-10-yl]-1,2,3-triazol-1-yl}-4-deoxypodophyllotoxin (26a)
White amorphous powder, yield 84% (after chromatography with petroleum ether/acetone, 1:1); mp 90 °C; : −9.5 (c 0.26, CH3OH); 1H-NMR (CD3OD, 400 MHz) δ 7.79 (s, 1H, C5''-H), 6.69 (s, 1H, C5-H), 6.62 (s, 1H, C8-H), 6.40 (s, 2H, C2', C6'-CH), 6.26 (d, 1H, J = 4.8 Hz, C4-H), 5.97 (d, 2H, J = 4.4 Hz, OCH2O), 5.34 (d, 1H, J = 8.0 Hz, C4'''-H), 5.29–5.26 (m, 3H, C1'''-H, C3'''-H, C2'''-H), 4.88–4.87 (m, 2H), 4.79 (d, 1H, J = 4.8 Hz, C1-H), 4.40–4.36 (m, 1H), 4.23 (dd, 1H, J = 4.8 Hz, 10.8 Hz), 4.13–4.10 (m, 2H), 3.73 (s, 6H, C3', C5'-OCH3), 3.72 (s, 3H, C4'-OCH3), 3.65–3.60 (m, 12H, 3 × OCH2CH2O), 3.42 (dd, 1H, J = 4.8 Hz, 10.0 Hz, C2-H), 3.18–3.13 (m, 1H, C3-H), 2.39 (t, 2H, J = 7.2 Hz, COCH2), 2.32 (t, 2H, J = 7.2 Hz, COCH2), 2.26 (t, 2H, J = 7.2 Hz, COCH2), 2.18 (t, 2H, J = 7.2 Hz, COCH2), 1.71–1.62 (m, 4H, 2 × CH2CH3), 1.61–1.51 (m, 4H, 2 × CH2CH3), 0.99–0.88 (m, 12H, 4 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) δ 175.7 (C-12), 174.7 (C=O), 174.0 (C=O), 173.9 (C=O), 173.8 (C=O), 154.0 (C-3', C-5'), 150.6 (C-7), 149.3 (C-6), 146.1 (C-4''), 140.6 (C-1'), 138.3 (C-9), 134.8 (C-10), 127.0 (C-4'), 125.8 (C-5''), 111.2 (C-5), 109.9 (C-8), 109.4 (C-2', C-6'), 103.3 (OCH2O), 99.0 (C-1'''), 71.6, 71.5, 71.4, 71.2, 70.9, 70.8, 70.5, 69.8, 68.9 (C-11), 68.3, 66.8, 65.1 (C-6''), 63.1 (C-6'''), 61.1 (4'-OCH3), 59.8 (C-2), 56.6 (3', 5'-OCH3), 44.9 (C-4), 42.5 (C-1), 38.6 (C-3), 36.9 (COCH2), 36.8 (COCH2), 36.8 (COCH2), 36.8 (COCH2), 19.6 (CH2CH3), 19.4 (CH2CH3), 19.4 (CH2CH3), 19.2 (CH2CH3), 14.1 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3); ESIMS: m/z 1092 [M+Na]+, HRESIMS: calcd for C53H71N3O20H [M+H]+ 1070.4704, found 1070.4677.
3.3.14. 4β-{4''-[1'''-(2''',3''',4''',6'''-Tetra-O-butyryl-β-d-mannopyranosyloxy)-3,6,9-trioxadec-10-yl]-1,2,3-triazol-1-yl}-4-deoxypodophyllotoxin (26b)
White amorphous powder, yield 85% (after chromatography with petroleum ether/acetone, 1:1); mp 74–75 °C; : −16.1 (c 0.21, CH3OH); 1H-NMR (CD3OD, 400 MHz) δ 7.80 (s, 1H, C5''-H), 6.69 (s, 1H, C5-H), 6.63 (s, 1H, C8-H), 6.41 (s, 2H, C2', C6'-H), 6.26 (d, 1H, J = 4.8 Hz, C4-H), 5.97 (d, 2H, J = 4.8 Hz, OCH2O), 5.22 (d, 1H, J = 1.6 Hz, C2'''-H), 5.12 (dd, 1H, J = 4.0 Hz, 10.0 Hz, C3'''-H), 4.80–4.79 (m, 2H), 4.66 (s, 1H), 4.43–4.36 (m, 3H), 4.26 (dd, 1H, J = 4.0 Hz, 10.0 Hz), 3.94–3.90 (m, 1H), 3.73 (s, 6H, C3', C5'-OCH3), 3.72 (s, 3H, C4'-OCH3), 3.65–3.60 (m, 12H, 3 × OCH2CH2O), 3.43 (dd, 1H, J = 4.0 Hz, 10.0 Hz, C2-H), 3.18–3.13 (m, 1H, C3-H), 2.35–2.25 (m, 8H, 4 × COCH2), 1.68–1.58 (m, 8H, 4 × CH2CH3), 0.97–0.92 (m, 12H, 4 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) δ 179.0 (C-12), 175.8 (C=O), 175.0 (C=O), 174.4 (C=O), 174.0 (C=O), 154.7 (C-4''), 154.0 (C-3', C-5'), 150.6 (C-7), 149.3 (C-6), 138.3 (C-1'), 136.8 (C-9), 134.8 (C-10), 127.0 (C-4'), 125.9 (C-5''), 110.2 (C-5), 109.9 (C-8), 109.4 (C-2', C-6'), 103.3 (OCH2O), 99.0 (C-1'''), 73.1, 72.1, 71.6, 71.5, 71.4, 71.2, 70.9, 70.7, 68.2 (C-11), 66.0, 65.0 (C-6''), 64.2 (C-6'''), 61.1 (4'-OCH3), 59.8 (C-2), 56.6 (3', 5'-OCH3), 44.9 (C-4), 42.5 (C-1), 38.6 (C-3), 37.0 (COCH2), 36.9 (COCH2), 36.9 (COCH2), 36.8 (COCH2), 19.6 (CH2CH3), 19.5 (CH2CH3), 19.4 (CH2CH3), 19.2 (CH2CH3), 14.1 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3); ESIMS: m/z 1092 [M+Na]+, HRESIMS: calcd for C53H71N3O20H [M+H]+ 1070.4704, found 1070.4703.
3.3.15. 4β-{4''-[1'''-(2''',3''',4''',6'''-Tetra-O-butyryl-α-d-mannopyranosyloxy)-3,6,9-trioxadec-10-yl]-1,2,3-triazol-1-yl}-4-deoxy-4'-demethylpodophyllotoxin (27a)
White amorphous powder, yield 89% (after chromatography with petroleum ether/acetone, 1:1); mp 95–97 °C; : −12.6 (c 0.29, CH3OH); 1H-NMR (CD3OD, 400 MHz) δ 7.79 (s, 1H, C5''-H), 6.69 (s, 1H, C5-H), 6.65 (s, 1H, C8-H), 6.38 (s, 2H, C2', C6'-H), 6.26 (d, 1H, J = 4.8 Hz, C4-H), 5.98 (d, 2H, J = 5.2 Hz, OCH2O), 5.34 (d, 1H, J = 10.0 Hz, C4'''-H), 5.28–5.26 (m, 2H, C3'''-H, C2'''-H), 4.88–4.87 (m, 2H), 4.76 (d, 1H, J = 4.4 Hz, C1-H), 4.41–4.37 (m, H), 4.23 (dd, 1H, J = 4.8 Hz, 10.8 Hz), 4.23–4.10 (m, 2H), 3.83–3.78 (m, 1H), 3.74 (s, 6H, C3'-OCH3, C5'-OCH3), 3.66–3.61 (m, 12H, 3 × OCH2CH2O), 3.39 (dd, 1H, J = 4.8 Hz, 10.0 Hz, C2-H), 3.18–3.13 (m, 1H, C3-H), 2.39 (t, 2H, J = 7.6 Hz, COCH2), 2.32 (t, 2H, J = 7.6 Hz, COCH2), 2.26 (t, 2H, J = 7.6 Hz, COCH2), 2.18 (t, 2H, J = 7.6 Hz, COCH2), 1.71–1.62 (m, 4H, 2 × CH2CH3), 1.61–1.51 (m, 4H, 2 × CH2CH3), 0.99–0.88 (t, 12H, 4 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) δ 176.0 (C-12), 174.8 (C=O), 174.0 (C=O), 173.9 (C=O), 173.8 (C=O), 150.5 (C-7), 149.2 (C-6), 148.7 (C-3', C-5'), 146.1 (C-4''), 136.0 (C-1'), 135.2 (C-9), 131.3 (C-10), 127.0 (C-4'), 125.8 (C-5''), 111.3 (C-5), 109.8 (C-8), 109.3 (C-2', C-6'), 103.3 (OCH2O), 99.0 (C-1'''), 71.6, 71.5, 71.4, 71.2, 70.9, 70.8, 70.5, 69.8, 68.9 (C-11), 68.3, 66.8, 65.1 (C-6''), 63.1 (C-6'''), 59.9 (C-2), 56.8 (3', 5'-OCH3), 44.8 (C-4), 42.8 (C-1), 38.5 (C-3), 36.9 (COCH2), 36.9 (COCH2), 36.8 (COCH2), 36.8 (COCH2), 19.6 (CH2CH3), 19.3 (CH2CH3), 19.3 (CH2CH3), 19.2 (CH2CH3), 14.1 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3), 13.9 (CH2CH3); ESIMS: m/z 1078 [M+Na]+, HRESIMS: calcd for C52H69N3O20H [M+H]+ 1056.4547, found 1056.4533.
3.3.16. 4β-{4''-[1'''-(2''',3''',4''',6'''-Tetra-O-butyryl-β-d-mannopyranosyloxy)-3,6,9-trioxadec-10-yl]-1,2,3-triazol-1-yl}-4-deoxy-4'-demethylpodophyllotoxin (27b)
White amorphous powder, yield 88% (after chromatography with petroleum ether/acetone, 1:1); mp 80–82 °C; : −26.6 (c 0.25, CH3OH); 1H-NMR (CD3OD, 400 MHz) δ 7.80 (s, 1H, C5''-H), 6.67 (s, 1H, C5-H), 6.62 (s, 1H, C8-H), 6.41 (s, 2H, C2', C6'-H), 6.24 (d, 1H, J = 4.8 Hz, C4-H), 5.97 (d, 2H, J = 5.6 Hz, OCH2O), 5.23 (d, 1H, J = 1.6 Hz), 5.12 (dd, 1H, J = 4.0 Hz, 10.0 Hz, C3'''-H), 4.80 (d, 1H, J = 1.6 Hz), 4.76 (d, 1H, J = 4.0 Hz), 4.43–4.35 (m, 3H), 4.26 (dd, 1H, J = 1.2 Hz, 5.2 Hz), 3.94–3.90 (m, 1H), 3.82–3.80 (m, 1H), 3.74 (s, 6H, C3', C5'-OCH3), 3.65–3.60 (m, 12H, 3 × OCH2CH2O), 3.39 (dd, 1H, J = 4.0 Hz, 10.0 Hz, C2-H), 3.16–3.12 (m, 1H, C3-H), 2.35–2.24 (m, 8H, 4 × COCH2), 1.68–1.58 (m, 8H, 4 × CH2CH3), 0.97–0.91 (m, 12H, 4 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) δ 179.0 (C-12), 175.9 (C=O), 175.1 (C=O), 174.5 (C=O), 174.0 (C=O), 150.5 (C-7), 149.2 (C-6), 148.7 (C-3', C-5'), 146.1 (C-4''), 136.0 (C-1'), 135.1 (C-9), 131.4 (C-10), 126.9 (C-4'), 125.9 (C-5''), 111.3 (C-5), 109.8 (C-8), 109.4 (C-2', C-6'), 103.3 (OCH2O), 99.0 (C-1'''), 73.1, 72.1, 71.6, 71.5, 71.5, 71.2, 70.9, 70.7, 68.9 (C-11), 68.2, 66.0, 65.0, (C-6''), 64.2 (C-6'''), 59.9 (C-2), 56.8 (3', 5'-OCH3), 44.8 (C-4), 42.7 (C-1), 38.5 (C-3), 37.0 (COCH2), 36.9 (COCH2), 36.9 (COCH2), 36.9 (COCH2), 19.6 (CH2CH3), 19.5 (CH2CH3), 19.4 (CH2CH3), 19.2 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3); ESIMS: m/z 1078 [M+Na]+, HRESIMS: calcd for C52H69N3O20H [M+H]+ 1056.4547, found 1056.4509.
3.3.17. 4β-{4''-[1"'-(2"',3"',4"'-Tri-O-butyryl-α-d-xylopyranosyloxy)-1,2,3-triazol-1-yl]}-4-deoxy-podophyllotoxin (28a)
White amorphous powder, yield 83% (after chromatography with petroleum ether/acetone, 1:1); mp 98–99 °C; : +20.5 (c 0.26, CH3OH); 1H-NMR (CD3OD, 400 MHz) δ 7.86 (s, 1H, C5''-H), 6.68 (s, 1H, C5-H), 6.59 (s, 1H, C8-H), 6.41 (s, 2H, C2', C6'-H), 6.24 (d, 1H, J = 4.4 Hz, C4-H), 5.96 (d, 2H, J = 8.0 Hz, OCH2O), 5.44 (t, 1H, J = 10.0 Hz, C3"'-H), 5.13 (d, 1H, J = 3.2 Hz, C1"'-H), 5.00–5.96 (m, 1H, C2"'-H), 4.77–4.73 (m, 3H), 4.65 (d, 1H, J = 4.8 Hz, C1-H), 4.38–4.35 (m, 1H), 3.80–3.78 (m, 3H), 3.73 (s, 6H, C3', C5'-OCH3), 3.71 (s, 3H, C4'-OCH3), 3.43 (dd, 1H, J = 4.8 Hz, 10.4 Hz, C2-H), 3.19–3.14 (m, 1H, C3-H), 2.24–2.17 (m, 6H, 3 × COCH2), 1.60–1.49 (m, 6H, 3 × CH2CH3), 0.90–0.88 (m, 9H, 3 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) δ 175.7 (C-12), 174.1 (C=O), 174.0 (C=O), 173.9 (C=O), 154.0 (C-3', C-5'), 150.6 (C-7), 149.3 (C-6), 144.6 (C-4''), 138.3 (C-1'), 136.7 (C-9), 134.7 (C-10), 127.0 (C-4'), 126.4 (C-5''), 111.2 (C-5), 109.9 (C-8), 109.4 (C-2', C-6'), 103.3 (OCH2O), 96.1 (C-1'''), 72.2, 70.5, 70.3, 68.9 (C-11), 61.2 (C-5'''), 61.1 (4'-OCH3), 59.8 (C-2), 59.6 (C-6''), 56.6 (3', 5'-OCH3), 44.9 (C-4), 42.5 (C-1), 38.6 (C-3), 36.8 (COCH2), 36.7 (COCH2), 36.7 (COCH2), 19.4 (CH2CH3), 19.4 (CH2CH3), 19.3 (CH2CH3), 14.0 (CH2CH3), 13.9 (CH2CH3), 13.9 (CH2CH3); ESIMS: m/z 861 [M+Na]+, HRESIMS: calcd for C42H51N3O25H [M+H]+ 838.3393, found 838.3367.
3.3.18. 4β-{4''-[1'''-(2''',3''',4'''-Tri-O-butyryl-β-d-xylopyranosyloxy)-1,2,3-triazol-1-yl]}-4-deoxy-podophyllotoxin (28b)
White amorphous powder, yield 83% (after chromatography with petroleum ether/acetone, 1:1); mp 97–99 °C; : −99.9 (c 0.25, Pyridine); 1H-NMR (C5D5N, 500 MHz) δ 8.14 (s, 1H, C5''-H), 6.86 (s, 1H, C5-H), 6.86 (s, 1H, C8-H), 6.76 (s, 2H, C2', C6'-H), 6.57 (d, 1H, J = 5.0 Hz, C4-H), 6.00 (d, 2H, J = 10.0 Hz, OCH2O), 5.67 (d, 1H, J = 9.0 Hz), 5.42 (t, 1H, J = 9.0 Hz, C3'''-H), 5.33–5.29 (m, 1H), 5.14–5.12 (m, 2H), 5.10 (d, 1H, J = 8.0 Hz, C1'''-H), 5.01 (d, 1H, J = 5.0 Hz, C1-H), 5.06–5.04 (m, 2H), 4.42 (t, 1H, J = 8.0 Hz), 4.28 (dd, 1H, J = 5.0 Hz, 10.0 Hz), 3.82 (s, 6H, C3', C5'-OCH3), 3.78 (s, 3H, C4'-OCH3), 3.60–3.58 (m, 1H, C2-H), 3.45–3.42 (m, 1H, C3-H), 2.30–2.24 (m, 6H, 3 × COCH2) 1.60–1.54 (m, 6H, 3 × CH2CH3), 0.83–0.78 (m, 9H, 3 × CH2CH3); 13C-NMR (C5D5N, 100 MHz) δ 174.0 (C-12), 172.6 (C=O), 172.6 (C=O), 172.3 (C=O), 153.5 (C-3', C-5'), 149.5 (C-7), 148.3 (C-6), 144.6 (C-4''), 138.3 (C-1'), 136.7 (C-9), 134.0 (C-10), 126.4 (C-4'), 124.9 (C-5''), 110.7 (C-5), 109.4 (C-8), 109.2 (C-2', C-6'), 102.5 (OCH2O), 100.5 (C-1'''), 72.1, 71.4, 69.4, 67.9 (C-11), 62.7 (C-5'''), 62.6 (C-6''), 60.6 (4'-OCH3), 58.8 (C-2), 56.2 (3', 5'-OCH3), 44.3 (C-4), 41.9 (C-1), 38.0 (C-3), 36.0 (COCH2), 36.0 (COCH2), 35.9 (COCH2), 18.7 (CH2CH3), 18.6 (CH2CH3), 18.6 (CH2CH3), 13.6 (CH2CH3), 13.6 (CH2CH3), 13.6 (CH2CH3); ESIMS: m/z 860 [M+Na]+, HRESIMS: calcd for C42H51N3O15H [M+H]+ 838.3393, found 838.3369.
3.3.19. 4β-{4''-[1'''-(2''',3''',4'''-Tri-O-butyryl-α-d-xylopyranosyloxy)-1,2,3-triazol-1-yl]}-4-deoxy-4'-demethylpodophyllotoxin (29a)
White amorphous powder, yield 84% (after chromatography with petroleum ether/acetone, 1:1); mp 200–203 °C; : −27.3 (c 0.25, Pyridine); 1H-NMR (C5D5N, 400 MHz) δ 8.30 (s, 1H, C5''-H), 6.87 (s, 1H, C5-H), 6.85 (s, 1H, C8-H), 6.81 (s, 2H, C2', C6'-H), 6.56 (d, 1H, J = 4.8 Hz, C4-H), 6.03–5.96 (m, 3H, OCH2O, C4'''-H), 5.64 (d, 1H, J = 4.0 Hz, C1'''-H), 5.41–5.35 (m, 1H, C3'''-H), 5.23 (dd, 1H, J = 4.0 Hz, 10.0 Hz, C2'''-H), 5.08–5.05 (m, 2H), 5.02 (d, 1H, J = 5.0 Hz, C1-H), 4.45 (t, 1H, J = 8.0 Hz), 4.01–3.94 (m, 3H), 3.80 (dd, 1H, J = 5.0 Hz, 10.0 Hz, C2-H), 3.72 (s, 6H, C3', C5'-OCH3), 3.46 (t, 1H, J = 10.0 Hz, C3-H), 2.32–2.20 (m, 6H, 3 × COCH2), 1.62–1.50 (m, 6H, 3 × CH2CH3), 0.83–0.77 (m, 9H, 3 × CH2CH3); 13C-NMR (C5D5N, 100 MHz) δ 174.1 (C-12), 172.8 (C=O), 172.7 (C=O), 172.6 (C=O), 149.4 (C-7), 148.8 (C-3', C-5'), 148.2 (C-6), 144.0 (C-4''), 137.4 (C-1'), 134.5 (C-9), 130.0 (C-10), 126.4 (C-5''), 125.4 (C-4'), 110.7 (C-5), 109.7 (C-2', C-6'), 109.3 (C-8), 102.4 (OCH2O), 95.4 (C-1'''), 71.5, 69.7, 69.6, 67.9 (C-11), 60.9 (C-5'''), 59.1 (C-2), 58.9 (C-6''), 56.5 (3', 5'-OCH3), 44.2 (C-4), 42.1 (C-1), 37.9 (C-3), 36.1 (COCH2), 36.0 (COCH2), 35.9 (COCH2), 18.7 (CH2CH3), 18.6 (CH2CH3), 18.6 (CH2CH3), 13.6 (CH2CH3), 13.6 (CH2CH3), 13.6 (CH2CH3); ESIMS: m/z 846 [M+Na]+, HRESIMS: calcd for C41H49N3O15H [M+H]+ 824.3236, found 824.3226.
3.3.20. 4β-{4''-[1'''-(2''',3''',4'''-Tri-O-butyryl-β-d-xylopyranosyloxy)-1,2,3-triazol-1-yl]}-4-deoxy-4'-demethylpodophyllotoxin (29b)
White amorphous powder, yield 82% (after chromatography with petroleum ether/acetone, 1:1); mp 100–101 °C; : −121.4 (c 0.19, Pyridine); 1H-NMR (C5D5N, 400 MHz) δ 8.14 (s, 1H, C5''-H), 6.87 (s, 1H, C5-H), 6.83 (s, 1H, C8-H), 6.80 (s, 2H, C2', C6'-H), 6.56 (d, 1H, J = 4.8 Hz, C4-H), 6.00–5.97 (m, 2H, OCH2O), 5.68 (t, 1H, J = 8.8 Hz, C3'''-H), 5.46–5.42 (m, 1H, C4'''-H), 5.35–5.30 (m, 1H, C2'''-H), 5.15–5.14 (m, 2H), 5.11 (d, 1H, J = 7.2 Hz, C1'''-H), 5.00–4.99 (m, 3H), 4.46 (t, 1H, J = 8.0 Hz), 4.29 (dd, 1H, J = 5.0 Hz, 10.0 Hz), 3.72 (s, 6H, C3', C5'-OCH3), 3.67–3.58 (m, 1H, C2-H), 3.44–3.42 (m, 1H, C3-H), 2.31–2.21 (m, 6H, 3 × COCH2), 1.61–1.52 (m, 6H, 3 × CH2CH3), 0.85–0.78 (m, 9H, 3 × CH2CH3); 13C-NMR (C5D5N, 100 MHz) δ 174.1 (C-12), 172.7 (C=O), 172.6 (C=O), 172.3 (C=O), 149.5 (C-7), 148.8 (C-3', C-5'), 148.2 (C-6), 144.6 (C-4''), 137.4 (C-1'), 134.5 (C-9), 130.0 (C-10), 126.3 (C-5''), 124.9 (C-4'), 110.8 (C-5), 109.7 (C-2', C-6'), 109.3 (C-8), 102.4 (OCH2O), 100.5 (C-1'''), 72.1, 71.4, 69.4, 67.9 (C-11), 62.7 (C-5'''), 62.6 (C-6''), 58.9 (C-2), 56.5 (3', 5'-OCH3), 44.1 (C-4), 42.1 (C-1), 38.0 (C-3), 36.1 (COCH2), 36.0 (COCH2), 35.9 (COCH2), 18.7 (CH2CH3), 18.6 (CH2CH3), 18.6 (CH2CH3), 13.6 (CH2CH3), 13.6 (CH2CH3), 13.6 (CH2CH3); ESIMS: m/z 860 [M+Na]+, HRESIMS: calcd for C42H51N3O15H [M+H]+ 838.3393, found 838.3369.
3.3.21. 4β-{4''-[1'''-(2''',3''',4'''-Tri-O-butyryl-α-d-xylopyranosyloxy)-3,6,9-trioxadec-10-yl]-1,2,3-triazol-1-yl}-4-deoxypodophyllotoxin (30a)
White amorphous power, yield 83% (after chromatography with petroleum ether/acetone, 1:1); mp 84 °C; : +12.2 (c 0.28, CH3OH); 1H-NMR (CD3OD, 400 MHz) δ 7.78 (s, 1H, C5''-H), 6.68 (s, 1H, C5-H), 6.60 (s, 1H, C8-H), 6.41 (s, 2H, C2', C6'-H), 6.25 (d, 1H, J = 4.8 Hz, C4-H), 5.97 (d, 2H, J = 5.2 Hz, OCH2O), 5.47 (t, 1H, J = 10.0 Hz, C3'''-H), 5.06–5.04 (m, 1H, C4'''-H), 4.96–4.94 (m, 1H, C2'''-H), 4.84 (d, 1H, J = 4.0 Hz, C1'''-H), 4.81 (d, 1H, J = 4.0 Hz, C1-H), 4.79–4.78 (m, 2H), 4.39–4.34 (m, 1H), 3.80–3.78 (m, 3H), 3.73 (s, 6H, C3', C5'-OCH3), 3.71 (s, 3H, C4'-OCH3), 3.65–3.59 (m, 12H, 3 × OCH2CH2O), 3.41 (dd, 1H, J = 4.0 Hz, 10.8 Hz, C2-H), 3.17–3.12 (m, 1H, C3-H), 2.27–2.23 (m, 6H, 3 × COCH2), 1.60–1.53 (m, 6H, 3 × CH2CH3), 0.92–0.87 (m, 9H, 3 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) δ 175.7 (C-12), 174.1 (C=O), 174.1 (C=O), 173.9 (C=O), 154.0 (C-3', C-5'), 150.5 (C-7), 149.3 (C-6), 146.1 (C-4''), 138.3 (C-1'), 136.7 (C-9), 134.8 (C-10), 127.0 (C-4'), 125.8 (C-5''), 111.2 (C-5), 109.9 (C-8), 109.4 (C-2', C-6'), 103.3 (OCH2O), 97.3 (C-1'''), 72.2, 71.7, 71.6, 71.5, 71.3, 71.0, 70.7, 70.5, 68.9 (C-11), 68.5, 65.1 (C-5'''), 61.2 (4'-OCH3), 59.8 (C-2), 59.4 (C-6''), 56.7 (3', 5'-OCH3), 44.9 (C-4), 42.5 (C-1), 38.6 (C-3), 36.9 (COCH2), 36.8 (COCH2), 36.6 (COCH2), 19.4 (CH2CH3), 19.4 (CH2CH3), 19.3 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3); ESIMS: m/z 992 [M+Na]+, HRESIMS: calcd for C48H63N3O18H [M+H]+ 970.4179, found 970.4167.
3.3.22. 4β-{4''-[1"'-(2"',3"',4"'-Tri-O-butyryl-β-d-xylopyranosyloxy)-3,6,9-trioxadec-10-yl]-1,2,3-triazol-1-yl}-4-deoxypodophyllotoxin (30b)
White amorphous powder, yield 85% (after chromatography with petroleum ether/acetone, 1:1); mp 88–90 °C; : −45.9 (c 0.22, CH3OH); 1H-NMR (CD3OD, 400 MHz) δ 7.80 (s, 1H, C5''-H), 6.69 (s, 1H, C5-H), 6.62 (s, 1H, C8-H), 6.42 (s, 2H, C2', C6'-H), 6.26 (d, 1H, J = 4.8 Hz, C4-H), 5.97 (d, 2H, J = 5.6 Hz, OCH2O), 5.24 (t, 1H, J = 9.2 Hz, C3"'-H), 4.89–4.86 (m, 2H, C2"'-H, C4"'-H), 4.80 (d, 1H, J = 5.2 Hz, C1-H), 4.64 (d, 1H, J = 8.0 Hz, C1"'-H), 4.63–4.62 (m, 2H), 4.41–4.36 (m, 1H), 4.06–4.02 (m, 1H), 3.88–3.83 (m, 1H), 3.81–3.79 (s, 2H, C5'''-CH2), 3.74 (s, 6H, C3', C5'-OCH3), 3.72 (s, 3H, C4'-OCH3), 3.66–3.3.60 (m, 12H, 3 × OCH2CH2O), 3.48–3.43 (m, 1H, C2-H), 3.18–3.14 (m, 1H, C3-H), 2.30–2.20 (m, 6H, 3 × COCH2), 1.61–1.53 (m, 6H, 3 × CH2CH3), 0.91–0.89 (m, 9H, 3 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) δ 175.8 (C-12), 174.0 (C=O), 174.0 (C=O), 173.7 (C=O), 154.0 (C-3', C-5'), 150.6 (C-7), 149.3 (C-6),146.1 (C-4''), 138.3 (C-1'), 136.7 (C-9), 134.8 (C-10), 127.0 (C-4'), 125.8 (C-5''), 111.2 (C-5), 109.9 (C-8), 109.4 (C-2', C-6'), 103.3 (OCH2O), 102.3 (C-1'''), 73.1, 72.3, 71.6, 71.5, 71.4, 70.9, 70.3, 69.9 (C-11), 65.1 (C-5'''), 63.3 (C-6''), 61.6 (4'-OCH3), 59.8 (C-2), 56.6 (3', 5'-OCH3), 44.9 (C-4), 42.5 (C-1), 38.6 (C-3), 36.9 (COCH2), 36.8 (COCH2), 36.7 (COCH2), 19.4 (CH2CH3), 19.4 (CH2CH3), 19.3 (CH2CH3), 14.0 (CH2CH3), 14.0 (CH2CH3), 13.9 (CH2CH3); ESIMS: m/z 992 [M+Na]+, HRESIMS: calcd for C48H63N3O18H [M+H]+ 970.4179, found 970.4162.
3.3.23. 4β-{4''-[1'''-(2''',3''',4'''-Tri-O-butyryl-α-d-xylopyranosyloxy)-3,6,9-trioxadec-10-yl]-1,2,3-triazol-1-yl}-4-deoxy-4'-demethylpodophyllotoxin (31a)
White amorphous powder, yield 86% (after chromatography with petroleum ether/acetone, 1:1); mp 87–88 °C; : +7.1 (c 0.22, CH3OH); 1H-NMR (CD3OD, 400 MHz) δ 7.77 (s, 1H, C5''-H), 6.67 (s, 1H, C5-H), 6.63 (s, 1H, C8-H), 6.38 (s, 2H, C2', C6'-H), 6.24 (d, 1H, J = 4.4 Hz, C4-H), 5.97 (d, 2H, J = 5.6 Hz, OCH2O), 5.47 (t, 1H, J = 10.0 Hz, C3'''-H), 5.06 (d, 1H, J = 3.2 Hz, C1'''-H), 4.97–4.94 (m, 1H, C2'''-H), 4.85–4.84 (m, 1H, C4'''-H), 4.81 (d, 1H, J = 4.0 Hz, C1-H), 4.76–4.73 (m, 2H), 4.37 (t, 1H, J = 7.2 Hz), 3.74 (s, 6H, C3', C5'-OCH3), 3.66–3.60 (m, 12H, 3 × OCH2CH2O), 3.39 (dd, 1H, J = 4.0 Hz, 10.0 Hz, C2-H), 3.15 (t, 1H, J = 10.0 Hz, C3-H), 2.29–2.22 (m, 6H, 3 × COCH2), 1.61–1.54 (m, 6H, 3 × CH2CH3), 0.91–0.87 (m, 9H, 3 × CH2CH3); 13C-NMR (CD3OD, 100 MHz) δ 174.4 (C-12), 172.6 (C=O), 172.5 (C=O), 172.4 (C=O), 149.0 (C-7), 147.7 (C-6), 147.2 (C-3', C-5'), 144.6 (C-4''), 134.5 (C-1'), 133.6 (C-9), 129.8 (C-10), 125.3 (C-4'), 124.2 (C-5''), 109.7 (C-5), 108.3 (C-8), 107.8 (C-2', C-6'), 101.7 (OCH2O), 95.7 (C-1'''), 70.6, 70.1, 70.0, 69.7, 69.4, 69.1, 69.0, 67.4 (C-11), 67.0, 63.5 (C-5'''), 58.3 (C-2), 57.8 (C-6''), 55.2 (3', 5'-OCH3), 43.2 (C-4), 41.2 (C-1), 37.0 (C-3), 35.3 (COCH2), 35.2 (COCH2), 35.1 (COCH2), 17.9 (CH2CH3), 17.8 (CH2CH3), 17.8 (CH2CH3), 12.4 (CH2CH3), 12.4 (CH2CH3), 12.4 (CH2CH3); ESIMS: m/z 978 [M+Na]+, HRESIMS: calcd for C47H61N3O18H [M+H]+ 956.4023, found 956.4015.
3.3.24. 4β-{4''-[1'''-(2''',3''',4'''-Tri-O-butyryl-β-d-xylopyranosyloxy)-3,6,9-trioxadec-10-yl]-1,2,3-triazol-1-yl}-4-deoxy-4'-demethylpodophyllotoxin (31b)
White amorphous powder, yield 87% (after chromatography with petroleum ether/acetone, 1:1); mp 79–80 °C; : −93.4 (c 0.29, Pyridine); 1H-NMR (C5D5N, 400 MHz) δ 8.11 (s, 1H, C5''-H), 6.86 (s, 1H, C5-H), 6.85 (s, 1H, C8-H), 6.80 (s, 2H, C2', C6'-H), 6.53 (d, 1H, J = 4.8 Hz, C4-H), 5.97 (d, 2H, J = 6.8 Hz, OCH2O), 5.69 (t, 1H, J = 9.2 Hz, C3'''-H), 5.44–5.40 (m, 1H, C4'''-H), 5.35–5.30 (m, 1H, C2'''-H), 4.98 (d, 1H, J = 4.8 Hz, C1-H), 4.87 (d, 1H, J = 7.2 Hz, C1'''-H), 4.84 (s, 2H), 4.44 (t, 1H, J = 8.0 Hz), 4.28 (dd, 1H, J = 6.0 Hz, 10.0 Hz), 4.01–3.96 (m, 1H), 3.78–3.75 (m, 1H), 3.72 (s, 6H, C3', C5'-OCH3), 3.63–3.57 (m, 12H, 3 × OCH2CH2O), 3.45–3.41 (m, 1H, C3-H), 2.38–2.26 (m, 6H, 3 × COCH2), 1.66–1.52 (m, 6H, 3 × CH2CH3), 0.87–0.80 (m, 9H, 3 × CH2CH3); 13C-NMR (C5D5N, 100 MHz) δ 174.1 (C-12), 172.7 (C=O), 172.6 (C=O), 172.3 (C=O), 148.7 (C-7), 148.2 (C-3', C-5'), 148.2 (C-6), 145.7 (C-4''), 137.4 (C-1'), 134.5 (C-9), 130.0 (C-10), 126.4 (C-4'), 124.5 (C-5''), 110.7 (C-5), 109.7 (C-2', C-6'), 109.3 (C-8), 102.4 (OCH2O), 101.5 (C-1'''), 72.2, 71.5, 70.8, 70.7, 70.5, 70.4, 69.5, 69.0, 67.9 (C-11), 65.0 (C-5'''), 62.6 (C-2), 58.8 (C-6''), 56.5 (3', 5'-OCH3), 44.1 (C-4), 42.1 (C-1), 37.9 (C-3), 36.1 (COCH2), 36.1 (COCH2), 36.0 (COCH2), 18.7 (CH2CH3), 18.7 (CH2CH3), 18.6 (CH2CH3), 13.7 (CH2CH3), 13.7 (CH2CH3), 13.6 (CH2CH3); ESIMS: m/z 978 [M+Na]+, HRESIMS: calcd for C47H61N3O18H [M+H]+ 956.4023, found 956.4007.