Synthesis and Evaluation of New Pyrazoline Derivatives as Potential Anticancer Agents
Abstract
:1. Introduction
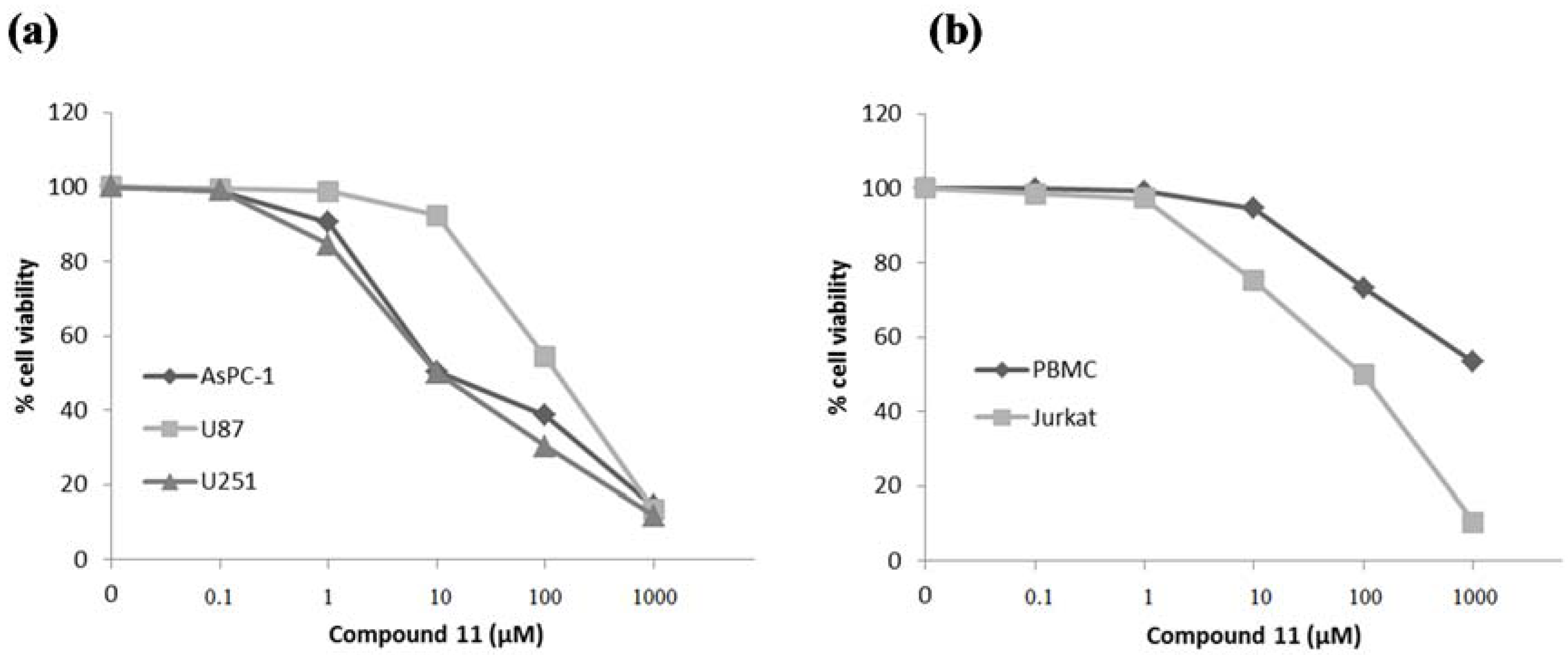
2. Results and Discussion
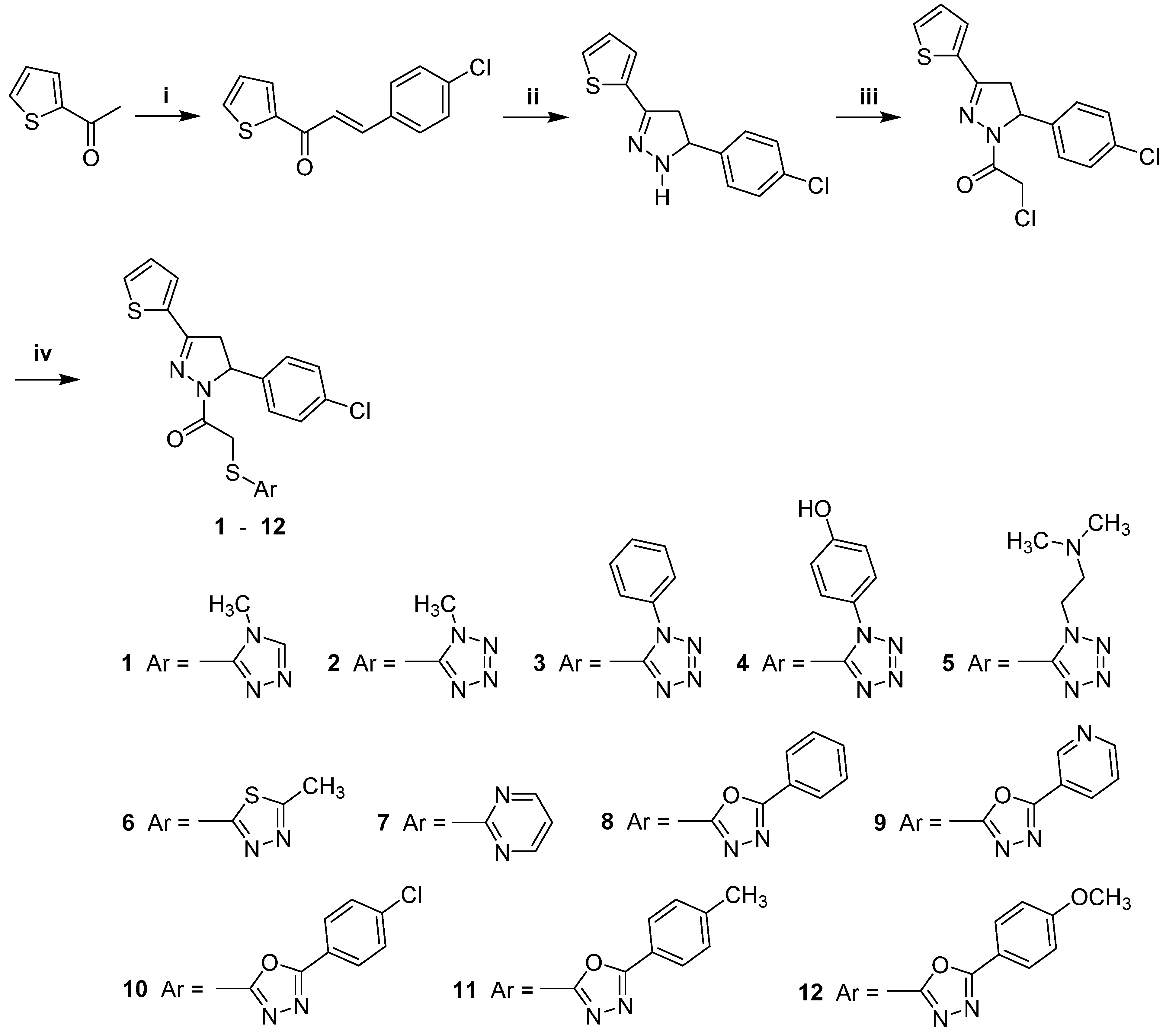
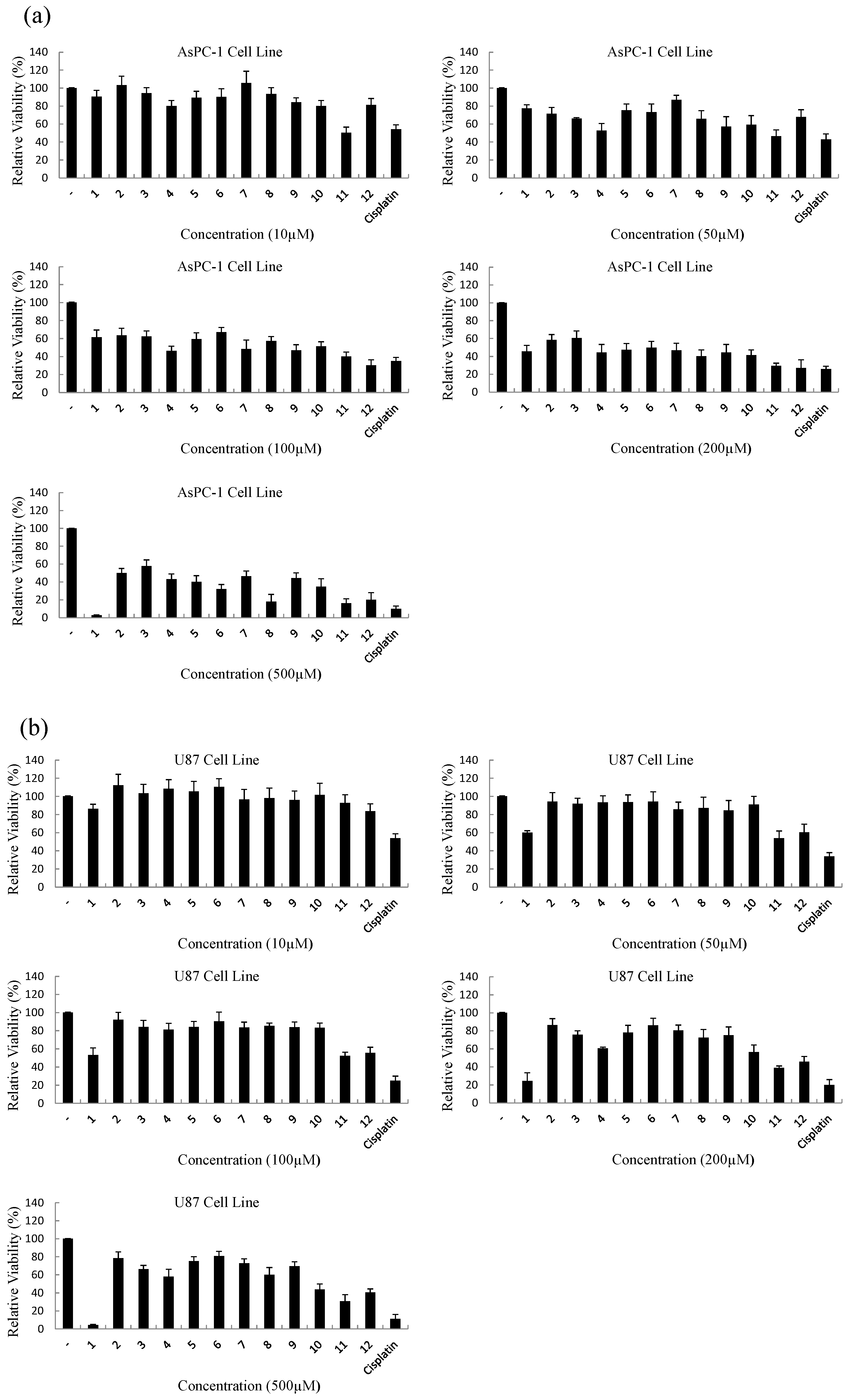
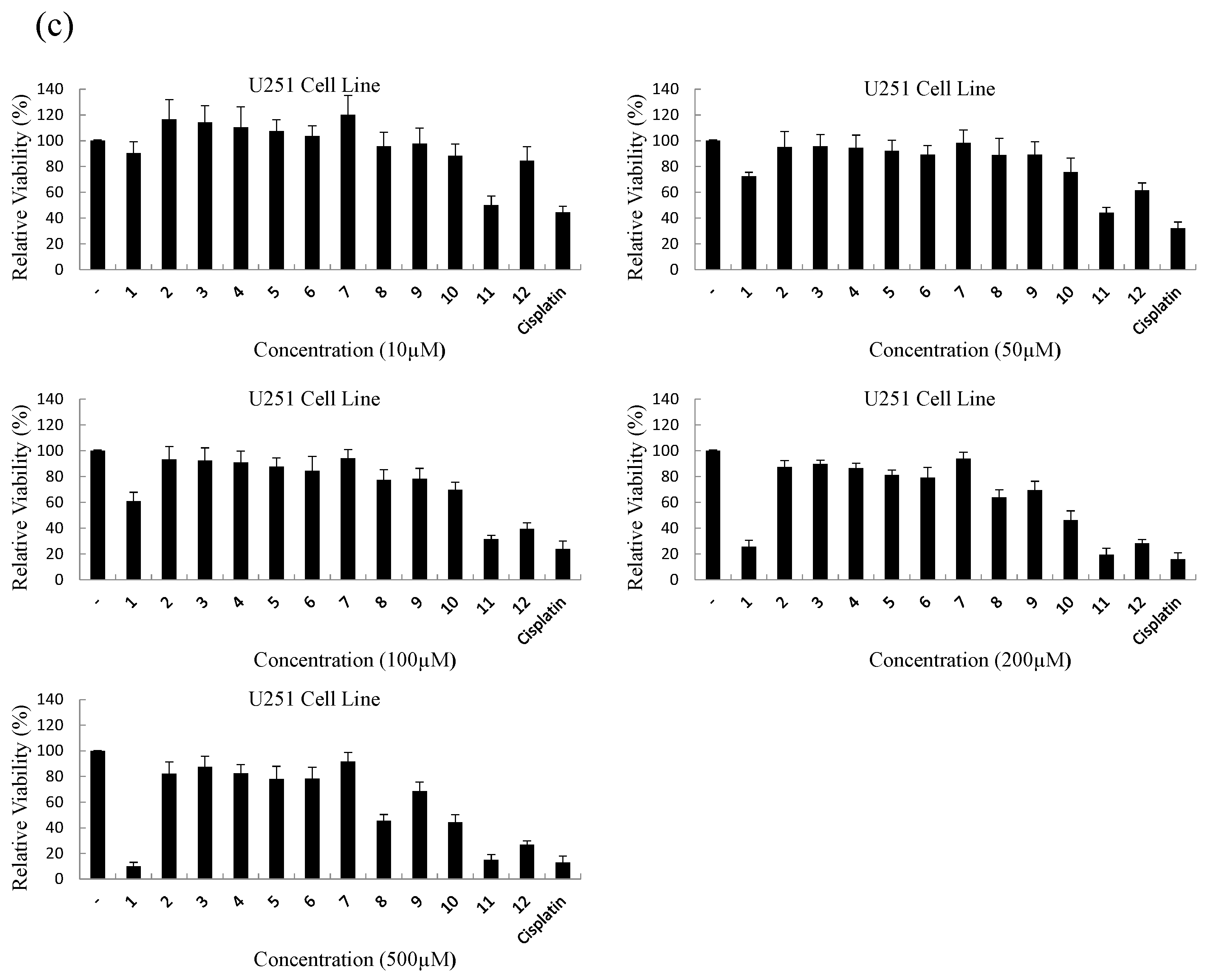
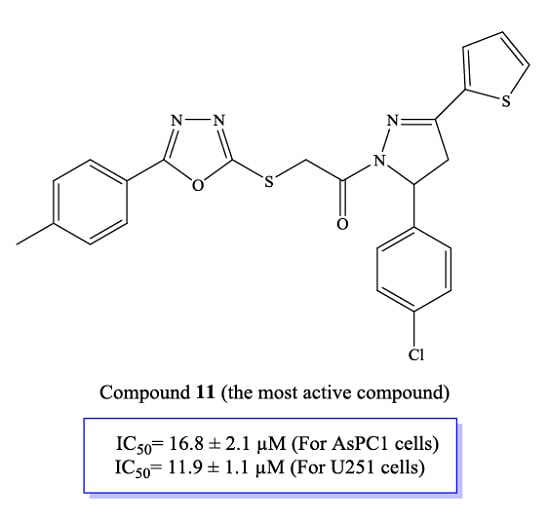
| Compound | IC50 (μM) | ||
|---|---|---|---|
| AsPC1 Cell Line | U87 Cell Line | U251 Cell Line | |
| 1 | 166.7 ± 10.6 | 112.2 ± 8.8 | 126.9 ± 15.6 |
| 2 | >500 | >500 | >500 |
| 3 | >500 | >500 | >500 |
| 4 | 65.0 ± 5.4 | >500 | >500 |
| 5 | 215.7 ± 29.7 | >500 | >500 |
| 6 | 199.3 ± 32.8 | >500 | >500 |
| 7 | 236.2 ± 36.4 | >500 | >500 |
| 8 | 139.3 ± 26.8 | >500 | 393.1 ± 60.4 |
| 9 | 108.1 ± 30.1 | >500 | >500 |
| 10 | 110.5±20.4 | 250.6 ± 30.4 | 166.6 ± 25.1 |
| 11 | 16.8 ± 2.1 | 127.4 ± 12.8 | 11.9 ± 1.1 |
| 12 | 62.1 ± 7.8 | 159.2 ± 20.7 | 70.1 ± 8.8 |
| Cisplatin | 22.5 ± 2.0 | 14.8 ± 1.4 | 4.9 ± 1.0 |
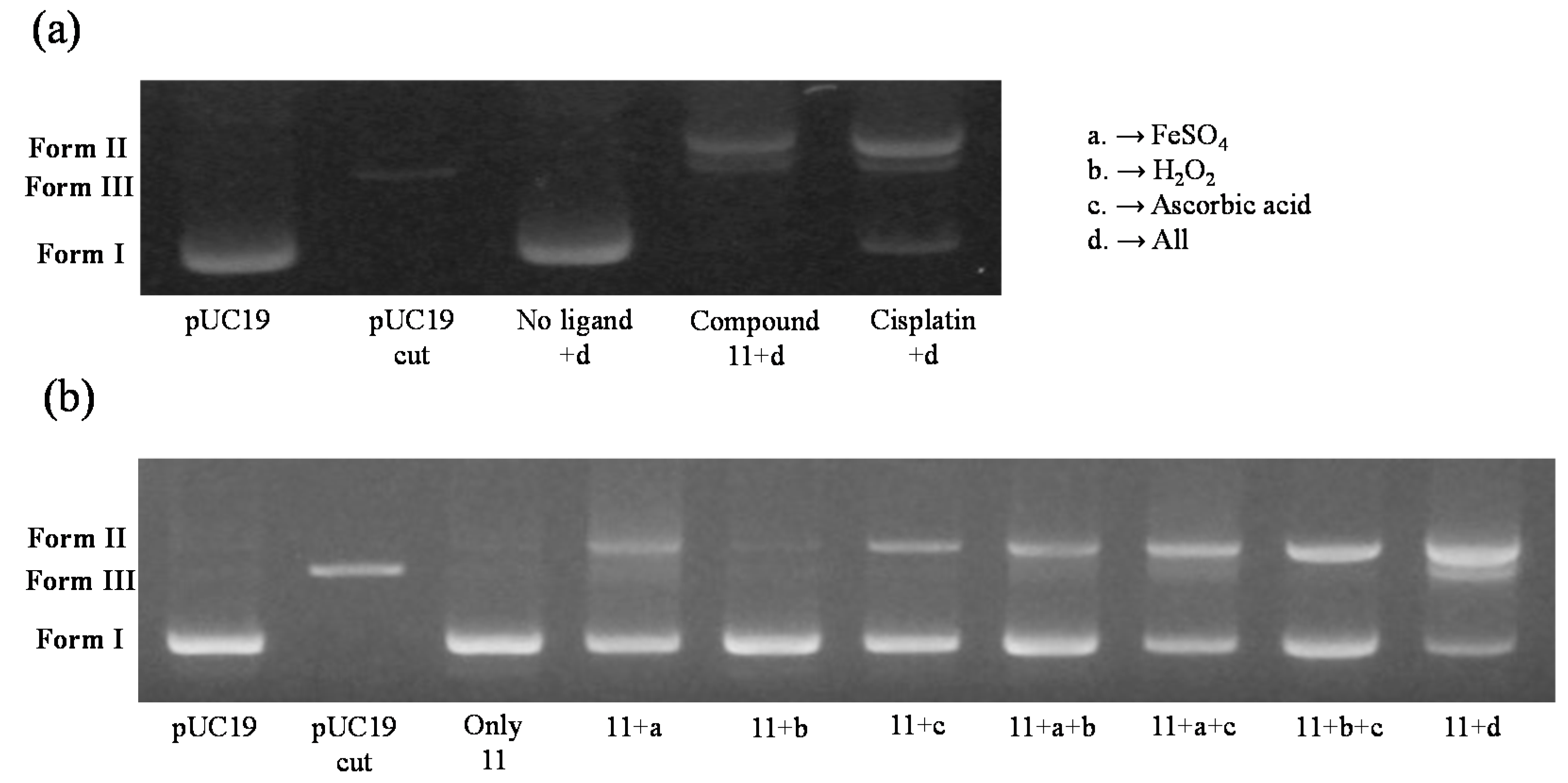
3. Experimental Section
3.1. Chemistry
General Procedures for the Synthesis of Compounds
3.2. Biochemistry
3.2.1. Cell Culture and Drug Treatment
3.2.2. MTT Assay for Cytotoxicity of Compounds
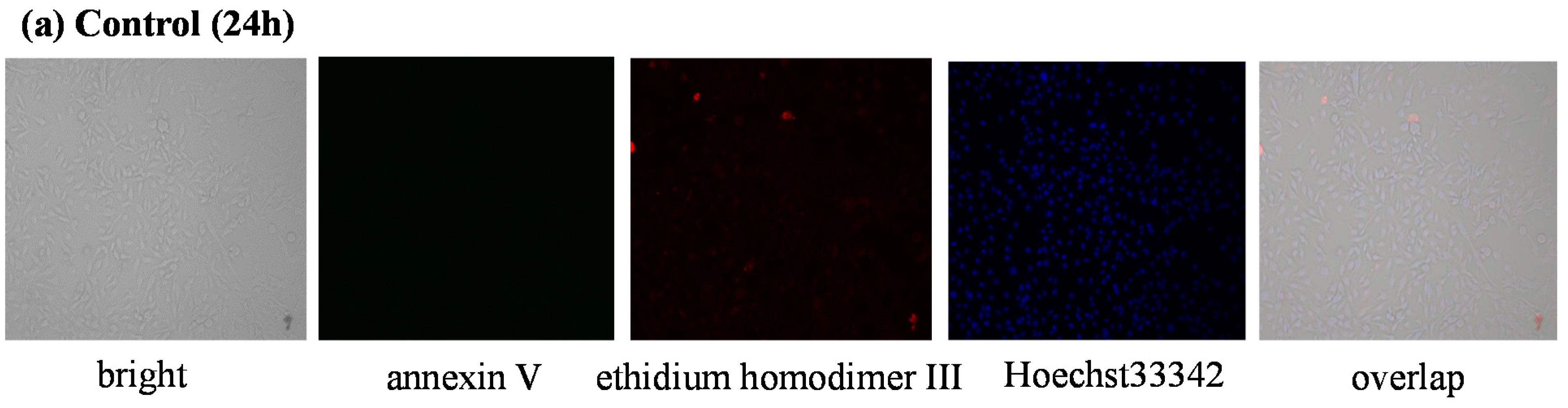
3.2.3. Detection of Apoptotic and Necrotic Cells
3.2.4. DNA Cleavage Assay
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mathur, G.; Nain, S.; Sharma, P.K. Cancer: An Overview. Acad. J. Cancer Res. 2015, 8. [Google Scholar] [CrossRef]
- Nepali, K.; Sharma, S.; Sharma, M.; Bedi, P.M.S.; Dhar, K.L. Rational approaches, design strategies, structure activity relationship and mechanistic insights for anticancer hybrids. Eur. J. Med. Chem. 2014, 77, 422–487. [Google Scholar] [CrossRef] [PubMed]
- Rebucci, M.; Michiels, C. Molecular aspects of cancer cell resistance to chemotherapy. Biochem. Pharmacol. 2013, 85, 1219–1226. [Google Scholar] [CrossRef] [PubMed]
- Nussbaumer, S.; Bonnabry, P.; Veuthey, J.-L.; Fleury-Souverain, S. Analysis of anticancer drugs: A review. Talanta 2011, 85, 2265–2289. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Bawa, S.; Drabu, S.; Kumar, R.; Gupta, H. Biological Activities of Pyrazoline Derivatives—A Recent Development. Recent Pat. Anti-Infect. Drug Discov. 2009, 4, 154–163. [Google Scholar] [CrossRef]
- Shaaban, M.R.; Mayhoub, A.S.; Farag, A.M. Recent advances in the therapeutic applications of pyrazolines. Expert Opin. Ther. Pat. 2012, 22, 253–291. [Google Scholar] [CrossRef] [PubMed]
- Marella, A.; Ali, R.; Alam, T.; Saha, R.; Tanwar, O.; Akhter, M.; Shaquiquzzaman, M.; Alam, M.M. Pyrazolines: A biological review. Mini-Rev. Med. Chem. 2013, 13, 921–931. [Google Scholar] [CrossRef] [PubMed]
- Alex, J.M.; Kumar, R. 4,5-Dihydro-1H-pyrazole: An indispensable scaffold. J. Enzyme Inhib. Med. Chem. 2014, 29, 427–442. [Google Scholar] [CrossRef] [PubMed]
- Havrylyuk, D.; Zimenkovsky, B.; Vasylenko, O.; Zaprutko, L.; Gzella, A.; Lesyk, R. Synthesis of novel thiazolone-based compounds containing pyrazoline moiety and evaluation of their anticancer activity. Eur. J. Med. Chem. 2009, 44, 1396–1404. [Google Scholar] [CrossRef] [PubMed]
- Shaharyar, M.; Abdullah, M.M.; Bakht, M.A.; Majeed, J. Pyrazoline bearing benzimidazoles: Search for anticancer agent. Eur. J. Med. Chem. 2010, 45, 114–119. [Google Scholar] [CrossRef] [PubMed]
- Congiu, C.; Onnis, V.; Vesci, L.; Castorina, M.; Pisano, C. Synthesis and in vitro antitumor activity of new 4,5-dihydropyrazole derivatives. Bioorg. Med. Chem. 2010, 18, 6238–6248. [Google Scholar] [CrossRef] [PubMed]
- Al-Abdullah, E.S. Synthesis and Anticancer Activity of Some Novel Tetralin-6-yl-pyrazoline, 2-Thioxopyrimidine, 2-Oxopyridine, 2-Thioxo-pyridine and 2-Iminopyridine Derivatives. Molecules 2011, 16, 3410–3419. [Google Scholar] [CrossRef] [PubMed]
- Bano, S.; Javed, K.; Ahmad, S.; Rathish, I.G.; Singh, S.; Alam, M.S. Synthesis and biological evaluation of some new 2-pyrazolines bearing benzene sulfonamide moiety as potential anti-inflammatory and anti-cancer agents. Eur. J. Med. Chem. 2011, 46, 5763–5768. [Google Scholar] [CrossRef] [PubMed]
- Bashir, R.; Ovais, S.; Yaseen, S.; Hamid, H.; Alam, M.S.; Samim, M.; Singh, S.; Javed, K. Synthesis of some new 1,3,5-trisubstituted pyrazolines bearing benzene sulfonamide as anticancer and anti-inflammatory agents. Bioorg. Med. Chem. Lett. 2011, 21, 4301–4305. [Google Scholar] [CrossRef] [PubMed]
- Amin, K.M.; Eissa, A.A.M.; Abou-Seri, S.M.; Awadallah, F.M.; Hassan, G.S. Synthesis and biological evaluation of novel coumarine-pyrazoline hybrids endowed with phenylsulfonyl moiety as antitumor agents. Eur. J. Med. Chem. 2013, 60, 187–198. [Google Scholar] [CrossRef] [PubMed]
- Montoya, A.; Quiroga, J.; Abonia, R.; Nogueras, M.; Cobo, J.; Insuasty, B. Synthesis and in Vitro Antitumor Activity of a Novel Series of 2-Pyrazoline Derivatives Bearing the 4-Aryloxy-7-chloroquinoline Fragment. Molecules 2014, 19, 18656–18675. [Google Scholar] [CrossRef] [PubMed]
- Insuasty, B.; Montoya, A.; Becerra, D.; Quiroga, J.; Abonia, R.; Robledo, S.; Darío Vélez, I.; Upegui, Y.; Nogueras, M.; Cobo, J. Synthesis of novel analogs of 2-pyrazoline obtained from [(7-chloroquinolin-4-yl)amino]chalcones and hydrazine as potential antitumor and antimalarial agents. Eur. J. Med. Chem. 2013, 67, 252–262. [Google Scholar] [CrossRef] [PubMed]
- Rathore, P.; Yaseen, S.; Ovais, S.; Bashir, R.; Yaseen, R.; Hameed, A.D.; Samim, M.; Gupta, R.; Hussain, F.; Javed, K. Synthesis and evaluation of some new pyrazoline substituted benzenesulfonylureas as potential antiproliferative agents. Bioorg. Med. Chem. Lett. 2014, 24, 1685–1691. [Google Scholar] [CrossRef] [PubMed]
- Lv, P.-C.; Li, D.-D.; Li, Q.-S.; Lu, X.; Xiao, Z.-P.; Zhu, H.-L. Synthesis, molecular docking and evaluation of thiazolyl-pyrazoline derivatives as EGFR TK inhibitors and potential anticancer agents. Bioorg. Med. Chem. Lett. 2011, 21, 5374–5377. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.Y.; Yoon, H.; Hwang, D.; Ahn, S.; Kim, D.-W.; Koh, D.; Lee, Y.H.; Lim, Y. Benzochalcones bearing pyrazoline moieties show anti-colorectal cancer activities and selective inhibitory effects on aurora kinases. Bioorg. Med. Chem. 2013, 21, 7018–7024. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Yang, H.; Wu, K.; Ji, Y.; Ju, L.; Lu, X. Novel pyrazoline derivatives as bi-inhibitor of COX-2 and B-Raf in treating cervical carcinoma. Bioorg. Med. Chem. 2014, 22, 4109–4118. [Google Scholar] [CrossRef] [PubMed]
- Amin, K.M.; Abou-Seri, S.M.; Awadallah, F.M.; Eissa, A.A.M.; Hassan, G.S.; Abdulla, M.M. Synthesis and anticancer activity of some 8-substituted-7-methoxy-2H-chromen-2-one derivatives toward hepatocellular carcinoma HepG2 cells. Eur. J. Med. Chem. 2015, 90, 221–231. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.-J.; Li, Y.-J.; Jiang, A.-Q.; Yang, M.-R.; Zhu, Q.-Z.; Dong, H.; Zhu, H.-L. Design, synthesis and biological evaluation of novel pyrazoline-containing derivatives as potential tubulin assembling inhibitors. Eur. J. Med. Chem. 2015, 94, 447–457. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, C.S.; Lira, B.F.; Barbosa-Filho, J.M.; Lorenzo, J.G.F.; de Athayde-Filho, P.F. Synthetic Approaches and Pharmacological Activity of 1,3,4-Oxadiazoles: A Review of the Literature from 2000–2012. Molecules 2012, 17, 10192–10231. [Google Scholar] [CrossRef] [PubMed]
- Khan, I.; Ibrar, A.; Abbas, N. Oxadiazoles as Privileged Motifs for Promising Anticancer Leads: Recent Advances and Future Prospects. Arch. Pharm. Chem. Life Sci. 2014, 347. [Google Scholar] [CrossRef] [PubMed]
- Bajaj, S.; Asati, V.; Singh, J.; Roy, P.P. 1,3,4-Oxadiazoles: An emerging scaffold to target growth factors, enzymes and kinases as anticancer agents. Eur. J. Med. Chem. 2015, 97, 124–141. [Google Scholar] [CrossRef] [PubMed]
- Küçükgüzel, Ş.G.; Çıkla-Süzgün, P. Recent advances bioactive 1,2,4-triazole-3-thiones. Eur. J. Med. Chem. 2015, 97, 830–870. [Google Scholar] [CrossRef] [PubMed]
- Kumar, C.N.S.S.P.; Parida, D.K.; Santhoshi, A.; Kota, A.K.; Sridhar, B.; Rao, V.J. Synthesis and biological evaluation of tetrazole containing compounds as possible anticancer agents. Med. Chem. Commun. 2011, 2, 486–492. [Google Scholar] [CrossRef]
- Haider, S.; Alam, M.S.; Hamid, H. 1,3,4-Thiadiazoles: A potent multi targeted pharmacological scaffold. Eur. J. Med. Chem. 2015, 92, 156–177. [Google Scholar] [CrossRef] [PubMed]
- Kaplancıklı, Z.A.; Özdemir, A.; Turan-Zitouni, G.; Altintop, M.D.; Can, Ö.D. New pyrazoline derivatives and their antidepressant activity. Eur. J. Med. Chem. 2010, 45, 4383–4387. [Google Scholar]
- Altintop, M.D.; Özdemir, A.; Kaplancikli, Z.A.; Turan-Zitouni, G.; Temel, H.E.; Çiftçi Gülşen, A. Synthesis and Biological Evaluation of Some Pyrazoline Derivatives Bearing a Dithiocarbamate Moiety as New Cholinesterase Inhibitors. Arch. Pharm. Chem. Life Sci. 2013, 346, 189–199. [Google Scholar] [CrossRef] [PubMed]
- Özdemir, A.; Altintop, M.D.; Kaplancıklı, Z.A.; Turan-Zitouni, G.; Karaca, H.; Tunalı, Y. Synthesis and Biological Evaluation of Pyrazoline Derivatives Bearing an Indole Moiety as New Antimicrobial Agents. Arch. Pharm. Chem. Life Sci. 2013, 346, 463–469. [Google Scholar] [CrossRef] [PubMed]
- Özdemir, A.; Altıntop, M.D.; Kaplancıklı, Z.A.; Turan-Zitouni, G.; Çiftçi, G.A.; Ulusoylar Yıldırım, Ş. Synthesis of 1-acetyl-3-(2-thienyl)-5-aryl-2-pyrazoline derivatives and evaluation of their anticancer activity. J. Enzyme Inhib. Med. Chem. 2013, 28, 1221–1227. [Google Scholar] [CrossRef] [PubMed]
- Altıntop, M.D.; Özdemir, A.; Turan-Zitouni, G.; Ilgın, S.; Atlı, Ö.; Demirel, R.; Kaplancıklı, Z.A. A novel series of thiazolyl-pyrazoline derivatives: Synthesis and evaluation of antifungal activity, cytotoxicity and genotoxicity. Eur. J. Med. Chem. 2015, 92, 342–352. [Google Scholar] [CrossRef] [PubMed]
- Özdemir, A.; Altıntop, M.D.; Kaplancıklı, Z.A.; Can, Ö.D.; Demir-Özkay, Ü.; Turan-Zitouni, G. Synthesis and Evaluation of New 1,5-Diaryl-3-[4-(methylsulfonyl)phenyl]-4,5-dihydro-1H-pyrazole Derivatives as Potential Antidepressant Agents. Molecules 2015, 20, 2668–2684. [Google Scholar] [CrossRef] [PubMed]
- Turan-Zitouni, G.; Özdemir, A.; Güven, K. Synthesis of some 1-[(N,N-disubstitutedthiocarbamoylthio)acetyl]-3-(2-thienyl)-5-aryl-2-pyrazoline derivatives and investigation of their antibacterial and antifungal activities. Arch. Pharm. Chem. Life Sci. 2005, 338, 96–104. [Google Scholar] [CrossRef] [PubMed]
- Özdemir, A.; Turan-Zitouni, G.; Kaplancıklı, Z.A.; Revial, G.; Demirci, F.; İşcan, G. Preparation of some pyrazoline derivatives and evaluation of their antifungal activities. J. Enzyme Inhib. Med. Chem. 2010, 25, 565–571. [Google Scholar] [CrossRef] [PubMed]
- Mossman, T. Rapid calorimetric assay for cellular growth and survival: Application to proliferation and cytotoxic assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Boersma, A.; Nooter, K.; Oostrum, R.; Stoter, G. Quantification of apoptotic cells with fluorescein isothiocyanate-labeled annexin V in Chinese hamster ovary cell cultures treated with cisplatin. Cytometry 1996, 24, 123–130. [Google Scholar] [CrossRef]
- Martin, S.; Reutelingsperger, C.; Mcgahon, A.; Rader, J.; Vanschie, R.; Laface, D.; Green, D. Early redistribution of plasma-membrane phosphatidylserine is a general feature of apoptosis regardless of the initiating stimulus—Inhibition by overexpression of bcl-2 and abl. J. Exp. Med. 1995, 182, 1545–1556. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds 1–12 are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karabacak, M.; Altıntop, M.D.; İbrahim Çiftçi, H.; Koga, R.; Otsuka, M.; Fujita, M.; Özdemir, A. Synthesis and Evaluation of New Pyrazoline Derivatives as Potential Anticancer Agents. Molecules 2015, 20, 19066-19084. https://doi.org/10.3390/molecules201019066
Karabacak M, Altıntop MD, İbrahim Çiftçi H, Koga R, Otsuka M, Fujita M, Özdemir A. Synthesis and Evaluation of New Pyrazoline Derivatives as Potential Anticancer Agents. Molecules. 2015; 20(10):19066-19084. https://doi.org/10.3390/molecules201019066
Chicago/Turabian StyleKarabacak, Muhammed, Mehlika Dilek Altıntop, Halil İbrahim Çiftçi, Ryoko Koga, Masami Otsuka, Mikako Fujita, and Ahmet Özdemir. 2015. "Synthesis and Evaluation of New Pyrazoline Derivatives as Potential Anticancer Agents" Molecules 20, no. 10: 19066-19084. https://doi.org/10.3390/molecules201019066
APA StyleKarabacak, M., Altıntop, M. D., İbrahim Çiftçi, H., Koga, R., Otsuka, M., Fujita, M., & Özdemir, A. (2015). Synthesis and Evaluation of New Pyrazoline Derivatives as Potential Anticancer Agents. Molecules, 20(10), 19066-19084. https://doi.org/10.3390/molecules201019066