Cytotoxic Compounds Isolated from Murraya tetramera Huang
Abstract
:1. Introduction
2. Results and Discussion
2.1. Compounds Isolated from M. tetramera
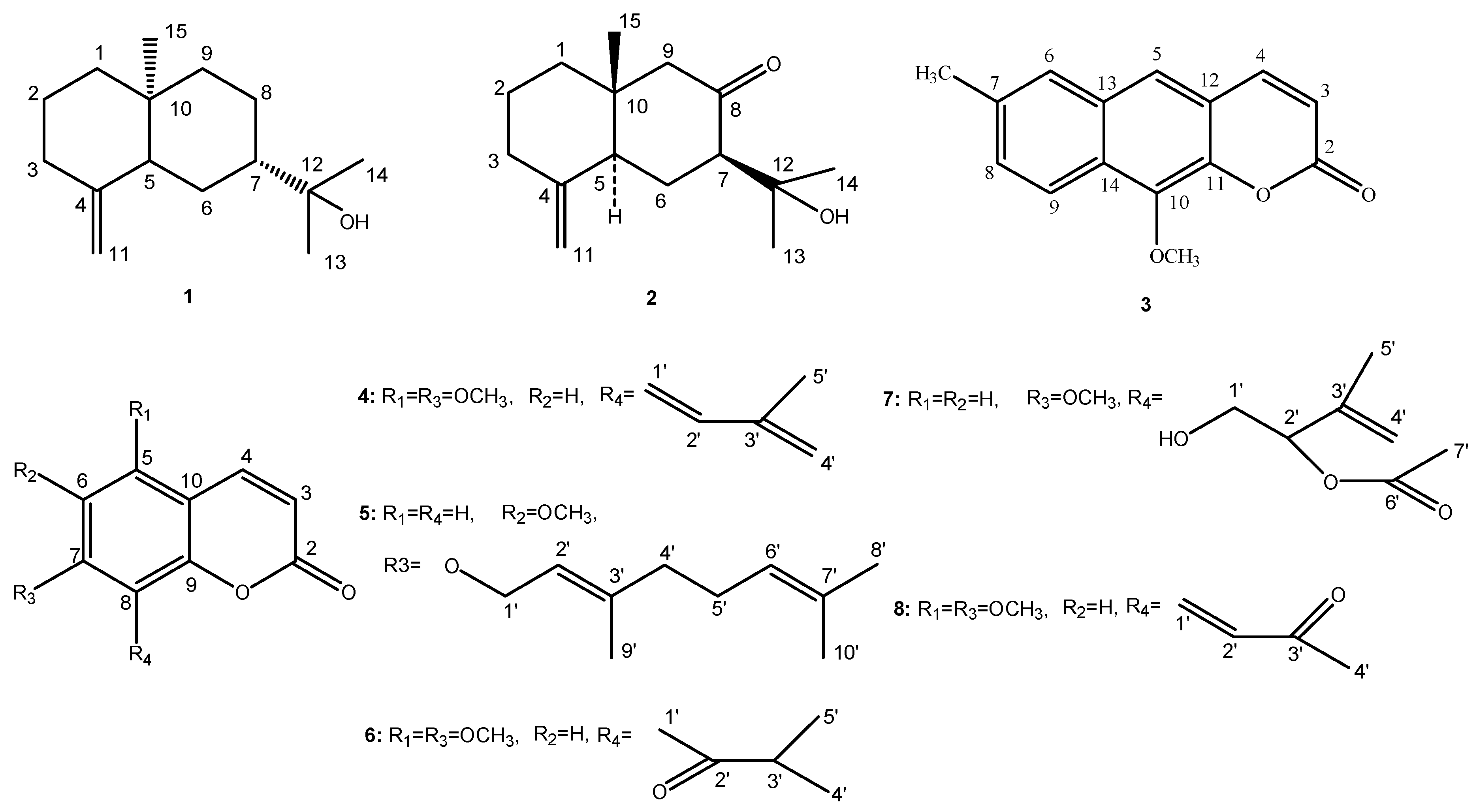
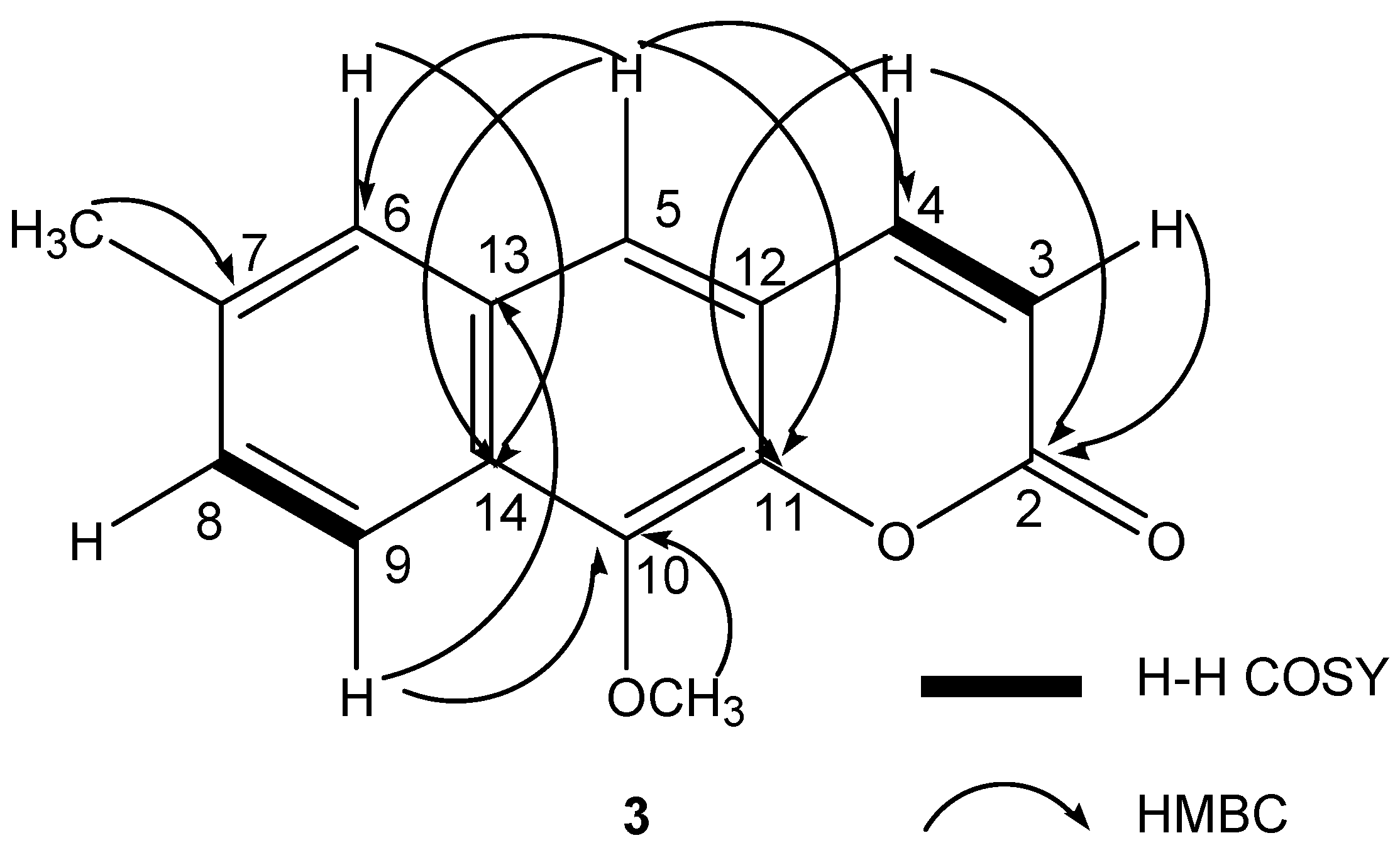
2.2. Chemical Structure Identification of the New Compound
2.3. Cytotoxic Activity of Isolated Compounds
| Compound | IC50 (µg/mL) a | ||||
|---|---|---|---|---|---|
| A549 | SMMC-7721 | EJ | Hela | BALL-1 | |
| 1 | 6.70 ± 1.05 | 5.17 ± 0.97 | 31.93 ± 2.84 | 17.82 ± 2.34 | 11.15 ± 1.62 |
| 2 | 31.67 ± 2.36 | 35.62 ± 2.73 | 47.45 ± 3.22 | 70.61 ± 3.95 | 33.91 ± 2.78 |
| 3 | >100 | >100 | >100 | >100 | 94.88 ± 3.25 |
| 4 | 17.04 ± 0.58 | >100 | 30.59 ± 2.73 | >100 | 22.54 ± 2.03 |
| 5 | 7.30 ± 0.46 | 9.09 ± 0.51 | 38.18 ± 2.23 | 46.63 ± 2.62 | 12.50 ± 1.47 |
| 6–8 | >100 | >100 | >100 | >100 | >100 |
| DOX b | 3.53 ± 0.25 | 1.35 ± 0.28 | 5.88 ± 0.18 | 2.11 ± 0.21 | 6.99 ± 0.37 |
3. Experimental Section
3.1. General Information
3.2. Plant Material
3.3. Extraction and Isolation
3.4. Characterization of Isolated Compounds
3.5. Cytotoxicity Assay
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pan, L.; Chai, H.; Kinghorn, A.D. The continuing search for antitumor agents from higher plants. Phytochem. Lett. 2010, 3, 1–8. [Google Scholar]
- Danhier, F.; Feron, O.; Préat, V. To exploit the tumor microenvironment: Passive and active tumor targeting of nanocarriers for anti-cancer drug delivery. J. Control. Release 2010, 148, 135–146. [Google Scholar] [CrossRef]
- Hsiao, W.; Liu, L. The role of traditional Chinese herbal medicines in cancer therapy-from TCM theory to mechanistic insights. Planta Med. 2010, 76, 1118–1131. [Google Scholar] [CrossRef]
- Hou, J.; Sun, T.; Hu, J.; Chen, S.; Cai, X.; Zou, G. Chemical composition, cytotoxic and antioxidant activity of the leaf essential oil of Photinia serrulata. Food Chem. 2007, 103, 355–358. [Google Scholar] [CrossRef]
- Ma, X.; Zheng, C.; Hu, C.; Rahman, K.; Qin, L. The genus Desmodium (Fabaceae)-traditional uses in Chinese medicine, phytochemistry and pharmacology. J. Ethnopharmacol. 2011, 138, 314–332. [Google Scholar] [CrossRef]
- Rahman, S.; Hasnat, A.; Hasan, C.M.; Rashid, M.A.; Ilias, M. Pharmacological evaluation of Bangladeshi medicinal plants-a review. Pharm. Biol. 2001, 39, 1–6. [Google Scholar] [CrossRef]
- Yang, H.; Cho, H.; Sim, S.H.; Chung, Y.K.; Kim, D.; Sung, S.H.; Kim, J.; Kim, Y.C. Cytotoxic terpenoids from Juglans sinensis leaves and twigs. Bioorg. Med. Chem. Lett. 2012, 22, 2079–2083. [Google Scholar] [CrossRef]
- Znati, M.; Jannet, H.; Cazaux, S.; Bouajila, J. Chemical composition, biological and cytotoxic activities of plant extracts and compounds isolated from Ferula lutea. Molecules 2014, 19, 2733–2747. [Google Scholar] [CrossRef]
- Rasul, A.; Ma, T.H. In vitro cytotoxic screening of 300 selected Chinese medicinal herbs against human gastric adenocarcinoma SGC-7901 cells. Afr. J. Pharm. Pharmacol. 2012, 6, 592–600. [Google Scholar]
- Kong, Y.C.; Ng, K.H.; But, P.P.H.; Li, Q.; Yu, S.X.; Zhang, H.T.; Cheng, K.F.; Soejarto, D.D.; Kan, W.S.; Waterman, P.G. Sources of the anti-implantation alkaloid yuehchukene in the genus Murraya. J. Ethnopharmacol. 1986, 15, 195–200. [Google Scholar] [CrossRef]
- Editorial Committee of Flora of China. Flora of China; Science Press: Beijing, China, 1997; p. 145. [Google Scholar]
- Tantapakul, C.; Phakhodee, W.; Laphookhieo, S.; Ritthiwigrom, T.; Cheenpracha, S. Cytotoxic carbazole alkaloids from the stems of Murraya koenigii. Chem. Nat. Compd. 2014, 50, 186–188. [Google Scholar]
- Sukari, M.A.; Riyanto, S.; Ali, A.M.; Yusof, U.K.; Haron, M.J.; Ahmad, F.B.H. Isolation of flavonoids from Murraya paniculata L. Orient. J. Chem. 2001, 17, 27–30. [Google Scholar]
- Riyanto, S.; Sukari, M.A.; Rahmani, M.; Ali, A.M.; Aimi, D.N. Isolation and identification of compounds in petroleum extract of Murraya paniculata (L.) brands cortex. Maj. Farm. Indones. 1999, 10, 95–103. [Google Scholar]
- Itoigawa, M.; Kashiwada, Y.; Ito, C.; Furukawa, H.; Tachibana, Y.; Bastow, K.F.; Lee, K.H. Antitumor agents. 203. Carbazole alkaloid murrayaquinone A and related synthetic carbazolequinones as cytotoxic agents. J. Nat. Prod. 2000, 63, 893–897. [Google Scholar]
- Bishay, D.W.; El-Sayyad, S.M.; Abdel-Hafiz, M.A.; Achenbach, H.; Desoky, E.K. Phytochemical study of Murraya exotica L. cultivated in Egypt. III. Coumarins and cycloartenols of the leaves. Bull. Pharm. Sci. Assiut Univ. 1988, 11, 105–121. [Google Scholar]
- Ya, Q.K.; Lu, W.J.; Chen, J.Y.; Tan, X. Study on the chemical constituent from Murraya tetramera Huang. Guangxi Sci. 2010, 17, 347–348. [Google Scholar]
- Wang, X.F.; Ohlin, C.A.; Lu, Q.H.; Fei, Z.F.; Hu, J.; Dyson, P.J. Cytotoxicity of ionic liquids and precursor compounds towards human cell line HeLa. Green Chem. 2007, 9, 1191–1197. [Google Scholar]
- Duan, J.; Wang, L.; Qian, S.; Su, S.; Tang, Y. A new cytotoxic prenylated dihydrobenzofuran derivative and other chemical constituents from the rhizomes of Atractylodes lancea DC. Arch. Pharm. Res. 2008, 31, 965–969. [Google Scholar]
- Torii, S.; Inlkuchi, T. Functionalization of trans-Decalin. IV. A stereoselective synthesis of dl-β-costol, dl-arctiol, and the related eudesmane type sesquiterpenes. Chem. Soc. Jpn. 1980, 53, 2642–2646. [Google Scholar]
- Kinoshita, T.; Firman, K. Prenylcoumarin derivatives from the leaves of an indonesian medicinal plant Murraya paniculata (Rutaceae). Chem. Pharm. Bull. 1996, 44, 1261–1262. [Google Scholar]
- Maes, D.; van Syngel, K.; de Kimpe, N. Synthesis of artekeiskeanin A: A new coumarin monoterpene ether from Artemisia keiskeana. Heterocycles 2007, 74, 927–930. [Google Scholar] [CrossRef]
- Macias, F.A.; Massanet, G.M.; Rodriguez-luis, F.; Salva, J. I3C-NMR of coumarios III*-simple coumarins. Magn. Reson. Chem. 1989, 27, 892–904. [Google Scholar] [CrossRef]
- Mesquita, S.G.; Martinez, M.F.; Romoff, P.; Fávero, O.A.; Lieber, S.R.; Lago, J.H.G. Constituintes químicos das folhas de Murraya paniculata(Rutaceae). Rev. Bras. Farmacogn.Braz. J. Pharmacogn. 2008, 18, 563–568. [Google Scholar] [CrossRef]
- Kinoshita, T.; Wu, J.B.; Ho, F.C. The isolation of a prenylcoumarin of chemotaxonomic significance from Murraya paniculata var omphalocarpa. Phytochemistry 1996, 43, 125–128. [Google Scholar] [CrossRef]
- Wu, S.; Ji, Y.; Zhu, J.; Zhao, Y.; Xia, G.; Hu, Y.; Hu, J. Steroids from the leaves of Chinese Melia azedarach and their cytotoxic effects on human cancer cell lines. Steroids 2009, 74, 761–765. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are not available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
You, C.-X.; Yang, K.; Wang, C.-F.; Zhang, W.-J.; Wang, Y.; Han, J.; Fan, L.; Du, S.-S.; Geng, Z.-F.; Deng, Z.-W. Cytotoxic Compounds Isolated from Murraya tetramera Huang. Molecules 2014, 19, 13225-13234. https://doi.org/10.3390/molecules190913225
You C-X, Yang K, Wang C-F, Zhang W-J, Wang Y, Han J, Fan L, Du S-S, Geng Z-F, Deng Z-W. Cytotoxic Compounds Isolated from Murraya tetramera Huang. Molecules. 2014; 19(9):13225-13234. https://doi.org/10.3390/molecules190913225
Chicago/Turabian StyleYou, Chun-Xue, Kai Yang, Cheng-Fang Wang, Wen-Juan Zhang, Ying Wang, Jiao Han, Li Fan, Shu-Shan Du, Zhu-Feng Geng, and Zhi-Wei Deng. 2014. "Cytotoxic Compounds Isolated from Murraya tetramera Huang" Molecules 19, no. 9: 13225-13234. https://doi.org/10.3390/molecules190913225
APA StyleYou, C.-X., Yang, K., Wang, C.-F., Zhang, W.-J., Wang, Y., Han, J., Fan, L., Du, S.-S., Geng, Z.-F., & Deng, Z.-W. (2014). Cytotoxic Compounds Isolated from Murraya tetramera Huang. Molecules, 19(9), 13225-13234. https://doi.org/10.3390/molecules190913225





