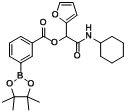
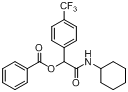
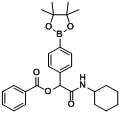
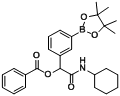
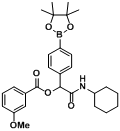
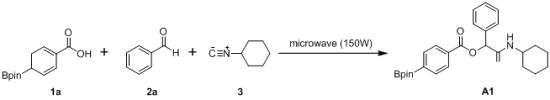
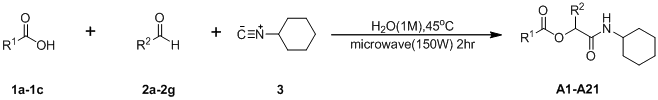
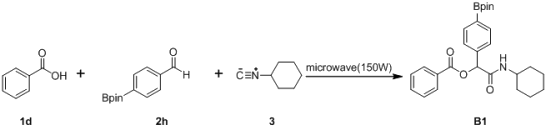
3.2. General Procedure A for the Synthesis of Boron-Containing α-Acyloxyl Amides A1–21
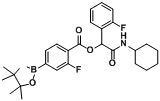
2-(Cyclohexylamino)-2-oxo-1-phenylethyl4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzoate (
A1). A 10 mL glass tube containing the 4-carboxyphenylboronic acid ester (248 mg, 1.00 mmol), benzaldehyde (0.10 mL, 1.00 mmol), and D.I. H
2O (1 mL) was first microwave irradiated for 6 min (45 °C, 150 W) under medium speed magnetic stirring. Cyclohexyl isocyanide (
3, 0.124 mL, 1.00 mmol) was then added to the reaction mixture. The additional microwave irradiation was applied for 120 min (45 °C, 150 W) under medium speed magnetic stirring. After being diluted in dichloromethane, the resulted reaction mixture was washed twice with a saturated aqueous solution of NaHCO
3 and with brine. The resulted organic layer was collected and dried over MgSO
4 and concentrated
in vacuo. The crude product was then dissolved in ethyl acetate (3 mL) prior the slow addition of
n-hexane. The resulting precipitate was formed and collected by filtration affording the desired product in 88% yield. mp = 198 °C.
1H-NMR (CDCl
3) δ: 8.06 (d,
J = 7.8 Hz, 2H), 7.90 (d,
J = 8.3 Hz, 2H), 7.53 (d,
J = 7.2 Hz, 2H), 7.42–7.33 (m, 3H), 6.31 (s, 1H), 6.03 (br, 1H), 3.87–3.79 (m, 1H), 1.94 (d,
J = 8.8 Hz, 1H), 1.91–1.85 (m, 1H), 1.72–1.61 (m, 2H), 1.61–1.56 (m, 3H), 1.41–1.31 (m, 14H), 1.23–1.08 (m, 3H).
13C-NMR (CDCl
3) δ: 167.3, 164.9, 135.7, 134.9, 131.4, 128.9, 128.8, 128.7, 127.4, 84.3, 76.0, 48.1, 32.8, 25.4, 24.9, 24.6.
11B-NMR (CDCl
3) δ: 31.0. HRMS (ESI, positive ion):
m/z [M+H]
+, found 464.2607. C
27H
34BNO
5 requires 464.2606.
2-(Cyclohexylamino)-2-oxo-1-phenylethyl3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzoate (
A2). The desired compound (384 mg, 83% yield) was prepared by General Procedure A using 3-carboxyphenyl boronic acid ester (248 mg, 1.00 mmol), benzaldehyde (0.102 mL, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 152 °C.
1H-NMR (CDCl
3) δ: 8.50 (s, 1H), 8.16 (d,
J = 7.8 Hz, 1H), 8.03 (d,
J = 7.4 Hz, 1H), 7.54 (d,
J = 7.2 Hz, 2H), 7.48 (t,
J = 7.5 Hz, 1H), 7.32–7.41 (m, 3H), 6.31 (s, 1H), 6.15 (br, 1H), 3.80–3.89 (m, 1H), 1.87–1.98 (m, 2H), 1.64–1.74 (m, 3H), 1.56–1.64 (m, 2H), 1.32–1.43 (m, 12H), 1.13–1.27 (m, 4H).
13C-NMR (CDCl
3) δ: 167.4, 164.9, 139.8, 136.0, 132.4, 128.9, 128.7, 128.1, 127.4, 84.2, 76.0, 48.1, 32.8, 25.5, 24.8, 24.6.
11B-NMR (CDCl
3) δ: 31.0. HRMS (ESI, positive ion):
m/z [M+H]
+, found 464.2583. C
27H
34BNO
5 requires 464.2606.
2-(Cyclohexylamino)-2-oxo-1-phenylethyl2-fluoro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzoate (
A3). The desired compound (311 mg, 80% yield) was prepared by General Procedure A using 4-carboxy-3-fluorophenylboronic acid ester (266 mg, 1.00 mmol), benzaldehyde (0.102 mL, 1.00 mmol) and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 181 °C.
1H-NMR (CDCl
3) δ: 7.93 (t,
J = 7.3 Hz, 1H), 7.67–7.59 (m, 2H), 7.51 (d,
J = 6.9 Hz, 2H), 7.38–7.28 (m, 3H), 6.82 (br, 1H), 6.31 (s, 1H), 3.87–3.79 (m, 1H), 1.96–1.88 (m, 2H), 1.70 (td,
J = 8.9, 4.3 Hz, 2H), 1.63–1.55 (m, 1H), 1.41–1.30 (m, 12H), 1.29–1.17 (m, 4H).
13C-NMR (CDCl
3) δ: 167.1, 162.5, 161.2(d), 135.7, 131.8, 130.3, 130.3, 128.8, 128.7, 128.6, 127.3, 122.7, 122.6, 119.7, 119.6, 84.5, 76.0, 47.9, 32.6, 25.3, 24.7, 24.4.
11B-NMR (CDCl
3) δ: 30.44. HRMS (ESI, positive ion):
m/z [M+H]
+, found 482.2515. C
27H
33BFNO
5 requires 482.2520.
2-(Cyclohexylamino)-2-oxo-1-(4-(trifluoromethyl)phenyl)ethyl4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzoate (
A4). The desired compound (405 mg, 76% yield) was prepared by General Procedure A using 4-carboxyphenylboronic acid ester (248 mg, 1.00 mmol), 4-(trifluoromethyl)benzaldehyde (0.13 mL, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol). mp = 249 °C.
1H-NMR (CDCl
3) δ: 8.06 (d,
J = 8.3 Hz, 2H), 7.93 (d,
J = 8.3 Hz, 2H), 7.69–7.62 (m, 4H), 6.35 (s, 1H), 6.17 (br, 1H), 3.86–3.78 (m, 1H), 1.95–1.86 (m, 2H), 1.72–1.63 (m, 2H), 1.60 (td,
J = 12.9, 3.8 Hz, 1H), 1.41–1.31 (m, 14H), 1.23–1.11 (m, 3H).
13C-NMR (CDCl
3) δ: 166.6, 164.7, 139.5, 135.0, 130.9, 128.7, 127.6, 125.7, 125.7, 123.9(d), 84.3, 75.1, 48.3, 32.8, 25.4, 24.8, 24.6.
11B-NMR (CDCl
3) δ: 31.2. HRMS (ESI, positive ion):
m/z [M+H]
+, found 532.2483. C
28H
33BF
3NO
5 requires 532.2488.
2-(Cyclohexylamino)-2-oxo-1-(4-(trifluoromethyl)phenyl)ethyl3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzoate (A5). The desired compound (416 mg, 79% yield) was prepared by General Procedure A using 3-carboxyphenylboronic acid ester (248 mg, 1.00 mmol), 4-(trifluoromethyl)benzaldehyde (0.136 mL, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 150 °C.
1H-NMR (CDCl
3) δ: 8.51 (s, 1H), 8.16 (td,
J = 7.8, 1.5 Hz, 1H), 8.08–8.03 (m, 1H), 7.68 (d,
J = 8.1 Hz, 2H), 7.63 (d,
J = 8.3 Hz, 2H), 7.50 (t,
J = 7.5 Hz, 1H), 6.38 (br, 1H), 6.33 (s, 1H), 3.87–3.78 (m, 1H), 1.92 (dt,
J = 12.4, 3.1 Hz, 2H), 1.73–1.65 (m, 2H), 1.63–1.56 (m, 1H), 1.41–1.32 (m, 13H), 1.26–1.14 (m, 3H).
13C-NMR (CDCl
3) δ: 166.7, 164.7, 140.0, 139.7, 135.9, 132.3, 130.8, 128.3, 128.1, 127.6, 125.6, 125.5, 123.9(d), 84.2, 75.2, 48.2, 32.7, 25.4, 24.8, 24.5.
11B-NMR (CDCl
3) δ: 30.6. HRMS (ESI, positive ion):
m/z [M+H]
+, found 532.2487. C
28H
33BF
3NO
5 requires 532.2488.
2-(Cyclohexylamino)-2-oxo-1-(4-(trifluoromethyl)phenyl)ethyl2-fluoro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzoate (
A6). The desired compound (279 mg, 51% yield) was prepared by General Procedure A using 4-carboxy-3-fluorophenyl boronic acid ester (266 mg, 1.00 mmol), 4-(trifluoromethyl)benzaldehyde (0.136 mL, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 220 °C.
1H-NMR (CDCl
3) δ: 7.93 (t,
J = 7.3 Hz, 1H), 7.68–7.60 (m, 6H), 6.83 (br, 1H), 6.34 (s, 1H), 3.85–3.78 (m, 1H), 1.96–1.88 (m, 2H), 1.71 (td,
J = 13.4, 4.0 Hz, 2H), 1.60 (td,
J = 12.6, 3.7 Hz, 1H), 1.43–1.31 (m, 14H), 1.28–1.20 (m, 3H).
13C-NMR (CDCl
3) δ: 166.4, 162.5, 161.3, 139.7, 132.0, 130.9, 130.5, 127.6, 125.6, 125.6, 124.8, 122.8, 119.3, 119.2, 84.7, 75.6, 48.1, 32.7, 32.6, 25.4, 24.8, 24.4.
11B-NMR (CDCl
3) δ: 30.6. HRMS (ESI, positive ion):
m/z [M+H]
+, found 550.2376. C
28H
32BF
4NO
5 requires 550.2394.
2-(Cyclohexylamino)-1-(2-fluorophenyl)-2-oxoethyl4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzoate (
A7). The desired compound (418 mg, 87% yield) was prepared by General Procedure A using 4-carboxyphenylboronic acid ester (248 mg, 1.00 mmol), 2-fluorobenzaldehyde (0.105 mL, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 167 °C.
1H-NMR (CDCl
3) δ: 8.05 (d,
J = 8.30 Hz, 2H), 7.89 (d,
J = 8.1 Hz, 2H), 7.58 (dt,
J = 7.4, 1.5 Hz, 1H), 7.33 (ddt,
J = 7.7, 5.5, 1.7 Hz, 1H), 7.19–7.14 (m, 1H), 7.08 (t,
J = 9.1 Hz, 1H), 6.48 (s, 1H), 6.17 (br, 1H), 3.87–3.79 (m, 1H), 2.00–1.93 (m, 1H), 1.88–1.81 (m, 1H), 1.73–1.61 (m, 2H), 1.61–1.55 (m, 1H), 1.40–1.29 (m, 13H), 1.27–1.10 (m, 3H)
13C-NMR (CDCl
3) δ: 166.5, 164.9, 160.7(d), 134.8, 131.2, 130.7, 130.0, 128.7, 124.4, 124.4, 123.2, 115.7, 84.2, 70.8, 48.2, 32.6, 25.4, 24.8, 24.5.
11B-NMR (CDCl
3) δ: 31.1. HRMS (ESI, positive ion):
m/z [M+H]
+, found 482.2512. C
27H
33BFNO
5 requires 482.2520.
2-(Cyclohexylamino)-1-(2-fluorophenyl)-2-oxoethyl3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzoate (
A8). The desired compound (415 mg, 86% yield) was prepared by General Procedure A using 3-carboxyphenylboronic acid ester (248 mg, 1.00 mmol), 2-fluorobenzaldehyde (0.105 mL, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 173 °C.
1H-NMR (CDCl
3) δ: 8.50 (s, 1H), 8.13 (d,
J = 7.7 Hz, 1H), 8.01 (d,
J = 7.4 Hz, 1H), 7.60–7.55 (m, 1H), 7.45 (t,
J = 7.5 Hz, 1H), 7.34–7.28 (m, 1H), 7.15 (t,
J = 7.6 Hz, 1H), 7.06 (t,
J = 9.2 Hz, 1H), 6.32 (br, 1H), 3.87–3.80 (m, 1H), 1.96 (d,
J = 11.6 Hz, 1H), 1.85 (d,
J = 12.0 Hz, 1H), 1.72–1.61 (m, 2H), 1.59–1.53 (m, 1H), 1.40–1.29 (m, 14H), 1.29–1.12 (m, 3H).
13C-NMR (CDCl
3) δ: 166.5, 164.8, 160.6, 139.7, 135.9, 132.2, 130.6, 129.9, 128.5, 127.9, 124.3, 123.2, 115.5, 84.0, 70.7, 48.1, 32.5, 25.3, 24.7, 24.4.
11B-NMR (CDCl
3) δ: 30.6. HRMS (ESI, positive ion):
m/z [M+H]
+, found 482.2514. C
27H
33BFNO
5 requires 482.2520.
2-(Cyclohexylamino)-1-(2-fluorophenyl)-2-oxoethyl2-fluoro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzoate (
A9). The desired compound (340 mg, 68% yield) was prepared by General Procedure A using 4-carboxy-3-fluorophenylboronic acid ester (266 mg, 1.00 mmol), 2-fluorobenzaldehyde (0.10 mL, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 69 °C.
1H-NMR (CDCl
3) δ: 7.89 (t,
J = 7.3 Hz, 1H), 7.63–7.55 (m, 2H), 7.50 (dt,
J = 7.4, 1.6 Hz, 1H), 7.31–7.26 (m, 1H), 7.11 (dt,
J = 7.5, 0.8 Hz, 1H), 7.06–7.01 (m, 1H), 6.78 (br, 1H), 6.45 (s, 1H), 3.87–3.79 (m, 1H), 1.94 (dd,
J = 12.0, 3.0 Hz, 1H), 1.91–1.85 (m, 1H), 1.68 (tdd,
J = 17.1, 13.1, 4.0 Hz, 3H), 1.60–1.52 (m, 1H), 1.38–1.19 (m, 18H).
13C-NMR (CDCl
3) δ: 166.3, 162.5, 161.8, 160.1, 131.7, 130.7, 130.0, 124.2, 123.2, 122.5, 119.4, 115.6, 84.4, 71.2, 47.9, 32.4, 25.3, 24.7, 24.3.
11B-NMR (CDCl
3) δ: 30.8. HRMS (ESI, positive ion):
m/z [M+H]
+, found 500.2425. C
27H
32BF
2NO
5 requires 500.2425.
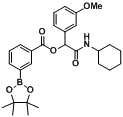
2-(Cyclohexylamino)-1-(3-methoxyphenyl)-2-oxoethyl4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzoate (
A10). The desired compound (347 mg, 70% yield) was prepared by General Procedure A using 4-carboxyphenylboronic acid ester (248 mg, 1.00 mmol), 3-methoxybenzaldehyde (0.12 mL, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 176 °C.
1H-NMR (CDCl
3) δ: 8.06 (d,
J = 8.1 Hz, 2H), 7.90 (d,
J = 8.1 Hz, 2H), 7.29 (t,
J = 8.0 Hz, 1H), 7.11 (d,
J = 7.7 Hz, 1H), 7.08 (s, 1H), 6.89 (dd,
J = 8.2, 2.3Hz, 1H), 6.27 (s, 1H), 6.08 (br, 1H), 3.84–3.77 (m, 4H), 1.92 (d,
J = 9.5 Hz, 1H), 1.89–1.84 (m, 1H), 1.66 (dt,
J = 14.4, 4.0 Hz, 2H), 1.61–1.55 (m, 1H), 1.40–1.30 (m, 14H), 1.21–1.08 (m, 3H).
13C-NMR (CDCl
3) δ: 167.1, 164.9, 159.7, 137.1, 134.8, 131.4, 129.7, 128.7, 119.5, 114.4, 113.0, 84.2, 75.8, 55.2, 48.1, 32.7, 25.4, 24.8, 24.6.
11B-NMR (CDCl
3) δ: 30.8. HRMS (ESI, positive ion):
m/z [M+H]
+, found 494.2717. C
28H
36BNO
6 requires 494.2720.
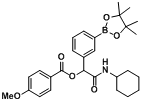
2-(Cyclohexylamino)-1-(3-methoxyphenyl)-2-oxoethyl3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzoate (
A11). The desired compound (377 mg, 77% yield) was prepared by General Procedure A using 3-carboxyphenylboronic acid ester (248 mg, 1.00 mmol), 3-methoxybenzaldehyde (0.121 mL, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 167 °C.
1H-NMR (CDCl
3) δ: 8.51 (s, 1H), 8.16 (td,
J = 7.8, 1.5 Hz, 1H), 8.03 (d,
J = 7.2 Hz, 1H), 7.48 (t,
J = 7.5 Hz, 1H), 7.29 (t,
J = 8.0 Hz, 1H), 7.13–7.08 (m, 2H), 6.89 (dd,
J = 8.2, 2.5 Hz, 1H), 6.27 (s, 1H), 6.15 (br, 1H), 3.87–3.80 (m, 4H), 1.97–1.87 (m, 2H), 1.74–1.64 (m, 3H), 1.59 (td,
J = 12.8, 3.6 Hz, 1H), 1.41–1.33 (m, 14H), 1.27–1.14 (m, 3H).
13C-NMR (CDCl
3) δ: 167.3, 164.8, 159.7, 139.8, 137.2, 136.0, 132.4, 129.7, 128.7, 128.0, 119.6, 114.5, 112.9, 84.2, 75.8, 55.3, 48.1, 32.7, 25.5, 24.8, 24.6.
11B-NMR (CDCl
3) δ: 30.5. HRMS (ESI, positive ion):
m/z [M+H]
+, found 494.2704. C
28H
36BNO
6 requires 494.2720.
2-(Cyclohexylamino)-1-(3-methoxyphenyl)-2-oxoethyl2-fluoro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzoate (
A12). The desired compound (320 mg, 63% yield) was prepared by General Procedure A using 4-carboxy-3-fluorophenylboronic acid ester (266 mg, 1.00 mmol), 3-methoxybenzaldehyde (0.12 mL, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 163 °C.
1H-NMR (CDCl
3) δ: 7.95 (t,
J = 7.2 Hz, 1H), 7.68–7.59 (m, 2H), 7.30–7.26 (m, 1H), 7.13–7.06 (m, 2H), 6.89–6.85 (m, 1H), 6.75 (br, 1H), 6.29 (s, 1H), 3.86–3.81 (m, 1H), 3.80 (s, 3H), 1.93 (br, s, 2H), 1.71 (td,
J = 8.6, 4.2 Hz, 2H), 1.63–1.57 (m, 1H), 1.43–1.38 (m, 2H), 1.36 (s, 13H), 1.28–1.19 (m, 3H).
13C-NMR (CDCl
3) δ: 166.9, 162.5, 161.2, 159.6, 137.1, 131.9, 130.3, 129.6, 122.6, 119.6, 119.4, 114.4, 112.9, 84.5, 76.2, 55.1, 47.9, 32.6, 25.4, 24.7, 24.4.
11B-NMR (CDCl
3) δ: 30.2. HRMS (ESI, positive ion):
m/z [M+H]
+, found 512.2615. C
28H
35BFNO
6 requires 512.2625.
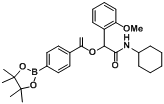
2-(Cyclohexylamino)-1-(2-methoxyphenyl)-2-oxoethyl4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzoate (
A13). The desired compound (428 mg, 87% yield) was prepared by General Procedure A using 4-carboxyphenylboronic acid ester (248 mg, 1.00 mmol), 2-methoxybenzaldehyde (0.121 mL, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 182 °C.
1H-NMR (CDCl
3) δ: 8.08 (d,
J = 8.1 Hz, 2H), 7.88 (d,
J = 8.1 Hz, 2H), 7.58 (dd,
J = 7.5, 1.4 Hz, 1H), 7.34–7.29 (m, 1H), 7.00 (t,
J = 7.5 Hz, 1H), 6.92 (d,
J = 8.3 Hz, 1H), 6.57 (s, 1H), 6.17 (br, 1H), 3.89–3.84 (m, 3H), 3.82–3.74 (m, 1H), 1.98–1.92 (m, 1H), 1.79–1.72 (m, 1H), 1.69–1.63 (m, 1H), 1.60–1.51 (m, 2H), 1.38–1.26 (m, 14H), 1.25–1.12 (m, 2H), 1.10–1.03 (m, 1H).
13C-NMR (CDCl
3) δ: 167.3, 165.3, 156.5, 134.6, 131.8, 129.9, 128.7, 128.5, 124.2, 121.0, 110.9, 84.1, 77.2, 76.8, 70.8, 55.5, 47.8, 32.5, 25.4, 24.7, 24.3.
11B-NMR (CDCl
3) δ: 31.0. HRMS (ESI, positive ion):
m/z [M+H]
+, found 494.2713. C
28H
36BNO
6 requires 494.2720.
2-(Cyclohexylamino)-1-(2-methoxyphenyl)-2-oxoethyl3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzoate (
A14). The desired compound (392 mg, 79% yield) was prepared by General Procedure A using 3-carboxyphenylboronic acid ester (248 mg, 1.00 mmol), 2-methoxybenzaldehyde (0.121 mL, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 164 °C.
1H-NMR (CDCl
3) δ: 8.54 (s, 1H), 8.18 (d,
J = 7.8 Hz, 1H), 7.99 (d,
J = 7.2 Hz, 1H), 7.58 (d,
J = 7.4 Hz, 1H), 7.44 (t,
J = 7.5 Hz, 1H), 7.30 (t,
J = 7.7 Hz, 1H), 6.99 (t,
J = 7.5 Hz, 1H), 6.91 (d,
J = 8.1 Hz, 1H), 6.57 (s, 1H), 6.24 (br, 1H), 3.87 (s, 3H), 3.82–3.76 (m, 1H), 1.95 (d,
J = 9.4 Hz, 1H), 1.77 (d,
J = 11.6 Hz, 1H), 1.70–1.63 (m, 1H), 1.61–1.51 (m, 2H), 1.38–1.27 (m, 13H), 1.26–1.06 (m, 4H).
13C-NMR (CDCl
3) δ: 167.4, 165.3, 156.6, 139.3, 136.0, 132.3, 129.9, 129.0, 128.6, 127.7, 124.2, 120.9, 110.9, 83.9, 70.9, 55.5, 47.7, 32.5, 25.3, 24.7, 24.3.
11B-NMR (CDCl
3) δ: 30.6. HRMS (ESI, positive ion):
m/z [M+H]
+, found 494.2715. C
28H
36BNO
6 requires 494.2720.
2-(Cyclohexylamino)-1-(2-methoxyphenyl)-2-oxoethyl2-fluoro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzoate (
A15). The desired compound (324 mg, 63% yield) was prepared by General Procedure A using 4-carboxy-3-fluorophenylboronic acid ester (266 mg, 1.00 mmol), 2-methoxybenzaldehyde (0.121 mL, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 75 °C.
1H-NMR (CDCl
3) δ: 7.93 (t,
J = 7.2 Hz, 1H), 7.60 (d,
J = 7.6 Hz, 1H), 7.56 (d,
J = 11.1 Hz, 1H), 7.51 (dd,
J = 7.6, 1.5 Hz, 1H), 7.30–7.26 (m, 2H), 6.95 (t,
J = 7.4 Hz, 1H), 6.88 (d,
J = 8.3 Hz, 1H), 6.60 (br, 1H), 6.56 (s, 1H), 3.86–3.76 (m, 4H), 1.95 (dd,
J = 12.0, 3.0 Hz, 1H), 1.84–1.78 (m, 1H), 1.72–1.65 (m, 1H), 1.65–1.58 (m, 1H), 1.58–1.52 (m, 1H), 1.39–1.28 (m, 14H), 1.27–1.11 (m, 3H).
13C-NMR (CDCl
3) δ: 167.2, 162.8, 162.0, 160.3, 156.8, 131.6, 130.0, 129.1, 124.1, 122.5, 122.4, 120.8, 120.1, 111.0, 84.3, 71.7, 55.5, 47.6, 32.4, 25.3, 24.6, 24.3.
11B-NMR (CDCl
3) δ: 30.1. HRMS (ESI, positive ion):
m/z [M+H]
+, found 512.2608. C
28H
35BFNO
6 requires 512.2625.
2-(Cyclohexylamino)-2-oxo-1-(pyridin-3-yl)ethyl4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzoate (
A16). The desired compound (260 mg, 56% yield) was prepared by General Procedure A using 4-carboxyphenylboronic acid ester (248 mg, 1.00 mmol), pyridine-3-aldehyde (0.09 mL, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 230 °C.
1H-NMR (CDCl
3) δ: 8.77 (s, 1H), 8.60 (d,
J = 4.5 Hz, 1H), 8.04 (d,
J = 8.3 Hz, 2H), 7.93-7.86 (m, 3H), 7.32 (dd,
J = 7.9, 4.8 Hz, 1H), 6.32 (s, 1H), 6.25 (br, 1H), 3.84–3.78 (m, 1H), 1.95–1.85 (m, 2H), 1.71–1.62 (m, 2H), 1.61–1.56 (m, 1H), 1.39–1.30 (m, 13H), 1.25–1.10 (m, 4H).
13C-NMR (CDCl
3) δ: 166.5, 164.8, 150.1, 148.5, 135.3, 135.0, 131.7, 130.9, 128.7, 123.5, 84.3, 73.8, 48.3, 32.7, 25.3, 24.8, 24.6.
11B-NMR (CDCl
3) δ: 30.6. HRMS (ESI, positive ion):
m/z [M+H]
+, found 465.2553. C
26H
33BN
2O
5 requires 465.2566.
2-(Cyclohexylamino)-2-oxo-1-(pyridin-3-yl)ethyl3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzoate (
A17). The desired compound (240 mg, 52% yield) was prepared by General Procedure A using 3-carboxyphenylboronic acid ester (248 mg, 1.00 mmol), pyridine-3-aldehyde (0.094 mL, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 127 °C.
1H-NMR (CDCl
3) δ: 8.76 (s, 1H), 8.56 (s, 1H), 8.46 (s, 1H), 8.10 (d,
J = 7.8 Hz, 1H), 8.00 (d,
J = 7.4 Hz, 1H), 7.88 (d,
J = 8.1 Hz, 1H), 7.43 (t,
J = 7.5 Hz, 1H), 7.32–7.27 (m, 1H), 6.60 (br, 1H), 6.28 (s, 1H), 3.82–3.74 (m, 1H), 1.93–1.81 (m, 2H), 1.67–1.59 (m, 2H), 1.57–1.50 (m, 1H), 1.35–1.27 (m, 13H), 1.22–1.09 (m, 4H).
13C-NMR (CDCl
3) δ: 166.5, 164.7, 149.8, 148.5, 140.0, 135.9, 135.3, 132.3, 128.1, 123.5, 84.1, 73.7, 48.2, 32.6, 25.3, 24.7, 24.5.
11B-NMR (CDCl
3) δ: 30.9. HRMS (ESI, positive ion):
m/z [M+H]
+, found 465.2548. C
26H
33BN
2O
5 requires 465.2566.
2-(Cyclohexylamino)-2-oxo-1-(pyridin-3-yl)ethyl2-fluoro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzoate (
A18). The desired compound (270 mg, 52% yield) was prepared by General Procedure A using 4-carboxy-3-fluorophenylboronic acid ester (266 mg, 1.00 mmol), pyridine-3-aldehyde (0.094 mL, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 203 °C.
1H-NMR (CDCl
3) δ: 8.75 (s, 1H), 8.57 (dd,
J = 4.7, 1.1 Hz, 1H), 7.91 (t,
J = 7.2 Hz, 1H), 7.83 (td,
J = 7.9, 1.7 Hz, 1H), 7.66–7.58 (m, 2H), 6.83 (br, 1H), 6.32 (s, 1H), 3.86–3.78 (m, 1H), 1.95–1.89 (m, 2H), 1.70 (td,
J = 8.9, 4.1 Hz, 2H), 1.62–1.55 (m, 1H), 1.41–1.31 (m, 14H), 1.26–1.19 (m, 3H).
13C-NMR (CDCl
3) δ: 166.2, 162.6, 161.3, 150.0, 148.6, 135.2, 131.9, 131.8, 130.5, 123.4, 122.7, 119.2, 84.6, 74.1, 48.1, 32.6, 25.4, 24.8, 24.4.
11B-NMR (CDCl
3) δ: 30.0. HRMS (ESI, positive ion):
m/z [M+H]
+, found 483.2455. C
26H
32BFN
2O
5 requires 483.3012.
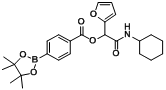
2-(Cyclohexylamino)-1-(furan-2-yl)-2-oxoethyl4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzoate (
A19). The desired compound (260 mg, 57% yield) was prepared by General Procedure A using 4-carboxyphenyl boronic acid ester (248 mg, 1.00 mmol), furan-2-carbaldehyde (0.08 mL, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 182.5 °C.
1H-NMR (CDCl
3) δ: 8.02 (d,
J = 8.3 Hz, 2H), 7.87 (d,
J = 8.3 Hz, 2H), 7.41 (s, 1H), 6.55 (d,
J = 3.2 Hz, 1H), 6.39 (s, 1H), 6.36 (dd,
J = 3.1, 1.8 Hz, 1H), 6.20 (br, 1H), 3.87–3.80 (m, 1H), 1.99–1.93 (m, 1H), 1.90–1.83 (m, 1H), 1.71–1.61 (m, 2H), 1.57 (td,
J = 12.9, 3.7 Hz, 1H), 1.38–1.29 (m, 14H), 1.25–1.09 (m, 3H).
13C-NMR (CDCl
3) δ: 164.9, 164.8, 148.1, 143.5, 134.7, 131.1, 128.7, 111.2, 110.6, 84.2, 69.2, 48.2, 32.6, 25.3, 24.7, 24.5.
11B-NMR (CDCl
3) δ: 30.6. HRMS (ESI, positive ion):
m/z [M+H]
+, found 454.2399. C
25H
32BNO
6 requires 454.2406.
2-(Cyclohexylamino)-1-(furan-2-yl)-2-oxoethyl3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzoate (
A20). The desired compound (280 mg, 63% yield) was prepared by General Procedure A using 3-carboxyphenylboronic acid ester (248 mg, 1.00 mmol), furan-2-carbaldehyde (0.082 mL, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 161.5 °C.
1H-NMR (CDCl
3) δ: 8.46 (s, 1H), 8.12 (d,
J = 7.8 Hz, 1H), 8.00 (d,
J = 7.3 Hz, 1H), 7.44 (t,
J = 7.4 Hz, 1H), 7.40 (s, 1H), 6.55 (d,
J = 3.3 Hz, 1H), 6.38 (s, 1H), 6.35 (dd,
J = 3.0, 1.9 Hz, 1H), 6.30 (br, 1H), 3.89–3.81 (m, 1H), 1.97 (d,
J = 9.2 Hz, 1H), 1.91–1.85 (m, 1H), 1.73–1.62 (m, 2H), 1.54–1.60 (m, 1H), 1.29–1.40 (m, 14H), 1.12–1.28 (m, 3H).
13C-NMR (CDCl
3) δ: 165.0, 164.7, 148.2, 143.4, 139.7, 135.9, 132.4, 128.4, 127.9, 111.2, 110.5, 84.0, 69.2, 48.2, 32.5, 25.3, 24.7, 24.4.
11B-NMR (CDCl
3) δ: 30.7. HRMS (ESI, positive ion):
m/z [M+H]
+, found 454.2394. C
25H
32BNO
6 requires 454.2406.
2-(Cyclohexylamino)-2-oxo-1-(4-(trifluoromethyl)phenyl)ethyl benzoate (
A21). The desired compound (360 mg, 89% yield) was prepared by General Procedure A using benzoic acid (122.12 mg, 1.00 mmol), 4-(trifluoromethyl)benzaldehyde (0.13 mL, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 201 °C.
1H-NMR (CDCl
3) δ: 8.10 (d,
J = 8.4 Hz, 2H), 7.67–7.63 (m, 5H), 7.52 (t,
J = 7.5 Hz, 2H), 6.34 (s, 1H), 6.14 (br, 1H), 3.84–3.79 (m, 1H), 1.94–1.90 (m, 2H), 1.72–1.63 (m, 2H), 1.90–1.60 (m, 4H), 1.39–1.33 (m, 2H), 1.23–1.16 (m, 3H).
13C-NMR (CDCl
3) δ: 166.6, 164.6, 139.6, 133.9, 131.1, 130.9, 129.7, 128.9, 128.7, 127.58, 125.7, 124.7, 122.9, 75.1, 48.3, 32.9, 32.8, 25.3, 24.6.
3.3. General Procedure B for the Synthesis of Boron-Containing α-Acyloxyl Amide B1–10
2-(Cyclohexylamino)-2-oxo-1-(4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl)ethyl benzoate (
B1). A 10 mL glass tube containing the benzoic acid (122 mg, 1.00 mmol),
p-formylphenylboronic acid ester (232 mg, 1.00 mmol), and D.I. H
2O (1 mL) was first microwave irradiated for 6 min (45 °C, 150 W) under medium speed magnetic stirring. The cyclohexyl isocyanide (
3, 0.124 mL, 1.00 mmol) was then added to the reaction mixture. The additional microwave irradiation was applied for 150 min (45 °C, 150 W) under medium speed magnetic stirring. After being diluted in dichloromethane, the resulted reaction mixture was washed twice with a saturated aqueous solution of NaHCO
3 and with brine. The resulted organic layer was collected and dried over MgSO
4 and concentrated
in vacuo. The crude product was then dissolved in ethyl acetate (3.0 mL) prior the slow addition of
n-hexane. The resulting precipitate was formed and collected by filtration affording the desired product in 75% yield, mp = 166 °C.
1H-NMR (CDCl
3) δ: 8.08 (d,
J = 7.7 Hz, 2H), 7.83 (d,
J = 7.9 Hz, 2H), 7.61–7.56 (m, 1H), 7.54 (d,
J = 7.9 Hz, 2H), 7.45 (t,
J = 7.7 Hz, 2H), 6.29 (s, 1H), 6.11 (br, 1H), 3.83–3.75 (m, 1H), 1.93–1.82 (m, 2H), 1.69–1.60 (m, 2H), 1.59–1.53 (m, 1H), 1.36–1.27 (m, 15H), 1.19–1.04 (m, 3H).
13C-NMR (CDCl
3) δ: 167.0, 164.9, 138.5, 135.1, 133.5, 129.7, 129.2, 128.5, 126.5, 83.8, 75.9, 48.2, 32.7, 25.3, 24.7, 24.6.
11B-NMR (CDCl
3) δ: 31.5. HRMS (ESI, positive ion):
m/z [M+H]
+, found 464.2616. C
27H
34BNO
5 requires 464.2614.
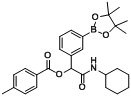
2-(Cyclohexylamino)-2-oxo-1-(3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl)ethyl benzoate (
B2). The desired compound (320 mg, 69% yield) was prepared by General Procedure B using benzoic acid (122.12 mg, 1.00 mmol),
m-formylphenylboronic acid ester (232 mg, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 165 °C.
1H-NMR (CDCl
3) δ: 8.08 (d,
J = 7.4 Hz, 2H), 7.96 (s, 1H), 7.80 (d,
J = 7.0 Hz, 1H), 7.65 (d,
J = 7.8 Hz, 1H), 7.57 (t,
J = 7.4 Hz, 1H), 7.45 (t,
J = 7.6 Hz, 2H), 7.39 (t,
J = 7.6 Hz, 1H), 6.29 (s, 1H), 6.11 (br, 1H), 3.85–3.78 (m, 1H), 1.93 (d,
J = 9.6 Hz, 1H), 1.86 (d,
J = 9.6 Hz, 1H), 1.70–1.60 (m, 2H), 1.60–1.54 (m, 1H), 1.40–1.25 (m, 14H), 1.22–1.06 (m, 3H).
13C-NMR (CDCl
3) δ: 167.2, 164.9, 135.3, 135.0, 134.0, 133.4, 130.2, 129.7, 129.3, 128.4, 128.0, 83.8, 76.0, 48.1, 32.7, 25.3, 24.7, 24.6.
11B-NMR (CDCl
3) δ: 30.6. HRMS (ESI, positive ion):
m/z [M+H]
+, found 464.2607. C
27H
34BNO
5 requires 464.2614.
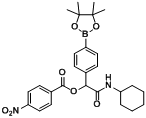
2-(Cyclohexylamino)-2-oxo-1-(4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl)ethyl-4-nitro-benzoate (
B3). The desired compound (290 mg, 57% yield) was prepared by General Procedure B using 4-nitrobenzoic acid (167.19 mg, 1.00 mmol),
p-formylphenylboronic acid ester (232 mg, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 108 °C.
1H-NMR (CDCl
3) δ: 8.30–8.20 (m, 4H), 7.84 (d,
J = 8.1 Hz, 2H), 7.53 (d,
J = 7.6 Hz, 2H), 6.23 (s, 1H), 5.93 (br, 1H), 3.80–3.73 (m, 1H), 1.92–1.86 (m, 1H), 1.80 (d,
J = 11.4 Hz, 1H), 1.69–1.53 (m, 3H), 1.37–1.26 (m, 14H), 1.17–1.00 (m, 3H).
13C-NMR (CDCl
3) δ: 166.4, 163.5, 150.7, 137.6, 135.3, 134.7, 130.9, 126.8, 123.6, 83.9, 76.7, 48.5, 32.6, 25.3, 24.7, 24.6.
11B-NMR (CDCl
3) δ: 31.0. HRMS (ESI, positive ion):
m/z [M+H]
+, found 509.2448. C
27H
33BN
2O
7 requires 509.2465.
2-(Cyclohexylamino)-2-oxo-1-(3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl)ethyl-4-nitro-benzoate (
B4). The desired compound (299 mg, 59% yield) was prepared by General Procedure B using 4-nitrobenzoic acid (167 mg, 1.00 mmol),
m-formylphenylboronic acid ester (232 mg, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 94 °C.
1H-NMR (CDCl
3) δ: 8.21 (s, 4H), 7.94 (s, 1H), 7.79 (d,
J = 7.1 Hz, 1H), 7.63 (d,
J = 8.0 Hz, 1H), 7.38 (t,
J = 7.6 Hz, 1H), 6.22 (s, 1H), 6.16 (br, 1H), 3.78–3.70 (m, 1H), 1.87 (d,
J = 10.4 Hz, 1H), 1.80–1.75 (m, 1H), 1.66–1.55 (m, 2H), 1.55–1.50 (m, 1H), 1.34–1.23 (m, 14H), 1.17–0.99 (m, 3H).
13C-NMR (CDCl
3) δ: 166.6, 163.4, 150.5, 135.6, 134.7, 134.1, 134.0, 130.8, 130.3, 128.2, 123.4, 83.8, 76.7, 48.4, 32.5, 25.2, 24.6, 24.5.
11B-NMR (CDCl
3) δ: 30.8. HRMS (ESI, positive ion):
m/z [M+H]
+, found 509.2442. C
27H
33BN
2O
7 requires 509.2465.
2-(Cyclohexylamino)-2-oxo-1-(4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl)ethyl-4-methyl-benzoate (
B5). The desired compound (344 mg, 72% yield) was prepared
by General Procedure B using 4-methylbenzoic acid (163.38 mg, 1.00 mmol),
p-formylphenylboronic acid ester (232 mg, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 90 °C.
1H-NMR (CDCl
3) δ: 7.97 (d, J = 7.8 Hz, 2H), 7.83 (d, J = 7.9 Hz, 2H), 7.53 (d, J = 7.9 Hz, 2H), 7.26 (d, J = 7.8 Hz, 2H), 6.28 (s, 1H), 6.07 (br, 1H), 3.83–3.75 (m, 1H), 2.41 (s, 3H), 1.92–1.84 (m, 3H), 1.69–1.61 (m, 2H), 1.60–1.55 (m, 1H), 1.37–1.30 (m, 14H), 1.18–1.07 (m, 3H).
13C-NMR (CDCl
3) δ: 167.2, 164.9, 144.4, 138.6, 135.1, 129.7, 129.3, 126.5, 83.8, 75.7, 48.1, 32.7, 25.3, 24.8, 24.6, 21.6.
11B-NMR (CDCl
3) δ: 30.9. HRMS (ESI, positive ion): m/z [M+H]
+, found 478.2752. C
28H
36BNO
5 requires 478.2771.
2-(Cyclohexylamino)-2-oxo-1-(3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl)ethyl-4-methyl-benzoate (
B6). The desired compound (302 mg, 63% yield) was prepared by General Procedure B using 4-methylbenzoic acid (163.38 mg, 1.00 mmol),
m-formylphenylboronic acid ester (232 mg, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 81 °C.
1H-NMR (CDCl
3) δ: 8.00–7.93 (m, 3H), 7.79 (d,
J = 7.4 Hz, 1H), 7.64 (d,
J = 7.8 Hz, 1H), 7.38 (t,
J = 7.6 Hz, 1H), 7.24 (d,
J = 7.8 Hz, 2H), 6.28 (s, 1H), 6.14 (br, 1H), 3.85–3.77 (m, 1H), 2.39 (s, 3H), 1.92 (d,
J = 9.6 Hz, 1H), 1.86 (d,
J = 11.4 Hz, 1H), 1.70–1.61 (m, 2H), 1.59–1.53 (m, 1H), 1.37–1.28 (m, 14H), 1.23–1.07 (m, 3H).
13C-NMR (CDCl
3) δ: 167.4, 164.9, 144.2, 135.2, 135.1, 133.9, 130.2, 129.7, 129.1, 128.0, 126.5, 83.8, 75.8, 48.1, 32.6, 25.3, 24.7, 24.5, 21.6.
11B-NMR (CDCl
3) δ: 30.7. HRMS (ESI, positive ion):
m/z [M+H]
+, found 478.2752. C
28H
36BNO
5 requires 478.2771.
2-(Cyclohexylamino)-2-oxo-1-(4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl)ethyl-4-methoxybenzoate (
B7). The desired compound (393 mg, 80% yield) was prepared by General Procedure B using 4-methoxybenzoic acid (182.58 mg, 1.00 mmol),
p-formylphenylboronic acid ester (232 mg, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 94 °C.
1H-NMR (CDCl
3) δ: 8.02 (d,
J = 8.7 Hz, 2H), 7.81 (d,
J = 8.1 Hz, 2H), 7.53 (d,
J = 8.1 Hz, 2H), 6.90 (d,
J = 8.7 Hz, 2H), 6.28–6.23 (m, 2H), 3.82–3.72 (m, 4H), 1.89–1.78 (m, 2H), 1.66–1.57 (m, 2H), 1.56–1.50 (m, 1H), 1.33–1.24 (m, 14H), 1.14–1.02 (m, 3H).
13C-NMR (CDCl
3) δ: 167.2, 164.5, 163.7, 138.7, 134.9, 131.7, 126.3, 121.4, 113.7, 83.6, 75.5, 55.2, 48.0, 32.5, 25.2, 24.6, 24.5.
11B-NMR (CDCl
3) δ: 30.8. HRMS (ESI, positive ion):
m/z [M+H]
+, found 494.2717. C
28H
36BNO
6 requires 494.2720.
2-(Cyclohexylamino)-2-oxo-1-(3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl)ethyl-4-methoxybenzoate (
B8). The desired compound (404 mg, 82% yield) was prepared by General Procedure B using 4-methoxybenzoic acid (182.58 mg, 1.00 mmol),
m-formylphenylboronic acid ester (232 mg, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 80 °C.
1H-NMR (CDCl
3) δ: 8.01 (d,
J = 8.56 Hz, 2H), 7.95 (s, 1H), 7.77 (d,
J = 7.6 Hz, 1H), 7.62 (d,
J = 7.6 Hz, 1H), 7.35 (t,
J = 7.6 Hz, 1H), 6.87 (d,
J = 9.0 Hz, 2H), 6.29 (br, 1H), 6.25 (s, 1H), 3.81–3.72 (m, 4H), 1.87 (d,
J = 10.4 Hz, 1H), 1.80 (d,
J = 10.4 Hz, 1H), 1.66–1.56 (m, 2H), 1.55–1.48 (m, 1H), 1.34–1.22 (m, 14H), 1.18–1.02 (m, 3H).
13C-NMR (CDCl
3) δ: 167.4, 164.5, 163.5, 135.1, 135.0, 133.8, 131.6, 130.0, 127.8, 121.4, 113.6, 83.6, 75.5, 55.2, 48.0, 32.4, 25.1, 24.6, 24.4.
11B-NMR (CDCl
3) δ: 31.6. HRMS (ESI, positive ion):
m/z [M+H]
+, found 494.2707. C
28H
36BNO
6 requires 494.2720.
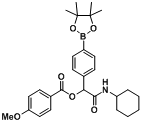
2-(Cyclohexylamino)-2-oxo-1-(4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl)ethyl-3-methoxybenzoate (
B9). The desired compound (419 mg, 85% yield) was prepared by General Procedure B using 3-methoxybenzoic acid (182.58 mg, 1.00 mmol),
p-formylphenylboronic acid ester (232 mg, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 74 °C.
1H-NMR (CDCl
3) δ: 7.81 (d,
J = 7.4 Hz, 2H), 7.65 (d,
J = 7.8 Hz, 1H), 7.58–7.50 (m, 3H), 7.32 (dt,
J = 7.8, 3.5 Hz, 1H), 7.10–7.05 (m, 1H), 6.33 (br, 1H), 6.24 (s, 1H), 3.803.71 (m, 4H), 1.86 (d,
J = 11.8 Hz, 1H), 1.83–1.77 (m, 1H), 1.61 (t,
J = 13.8 Hz, 2H), 1.52 (d,
J = 9.6 Hz, 1H), 1.33–1.23 (m, 14H), 1.16–1.01 (m, 3H).
13C-NMR (CDCl
3) δ: 167.0, 164.7, 159.4, 138.4, 134.9, 130.4, 129.4, 126.3, 121.8, 119.5, 114.3, 83.6, 75.9, 55.2, 48.1, 32.5, 25.2, 24.6, 24.5.
11B-NMR (CDCl
3) δ: 31.5. HRMS (ESI, positive ion):
m/z [M+H]
+, found 494.2702. C
28H
36BNO
6 requires 494.2720.
2-(Cyclohexylamino)-2-oxo-1-(3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl)ethyl-3-methoxybenzoate (
B10). The desired compound (437 mg, 89% yield) was prepared by General Procedure B using 3-methoxybenzoic acid (182.58 mg, 1.00 mmol),
m-formylphenylboronic acid ester (232 mg, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 67 °C.
1H-NMR (CDCl
3) δ: 7.96 (s, 1H), 7.78 (d,
J = 7.4 Hz, 1H), 7.63 (d,
J = 7.4 Hz, 1H), 7.66 (d,
J = 7.8 Hz, 1H), 7.57 (s, 1H), 7.36 (t,
J = 7.4 Hz, 1H), 7.32 (t,
J = 7.8 Hz, 1H), 7.08 (dd,
J = 8.3, 2.6 Hz, 1H), 6.29–6.22 (m, 2H), 3.82–3.76 (m, 4H), 1.89 (d,
J = 9.6 Hz, 1H), 1.82 (d,
J = 11.8 Hz, 1H), 1.67–1.58 (m, 2H), 1.56–1.51 (m, 1H), 1.34–1.26 (m, 14H), 1.19–1.13 (m, 1H), 1.13–1.05 (m, 2H).
13C-NMR (CDCl
3) δ: 167.2, 164.8, 159.4, 135.2, 134.9, 133.8, 130.4, 130.0, 129.4, 127.9, 121.9, 119.6, 114.3, 83.7, 75.9, 55.2, 48.1, 32.5, 25.2, 24.6, 24.5.
11B-NMR (CDCl
3) δ: 31.5. HRMS (ESI, positive ion):
m/z [M+H]
+, found 494.2713. C
28H
36BNO
6 requires 494.2720.
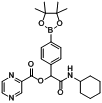
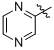
2-(Cyclohexylamino)-2-oxo-1-(4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl)ethyl pyrazine-2-carboxylate (
B11). The desired compound (237 mg, 51% yield) was prepared by General Procedure B using pyrazine-2-carboxylic acid (124 mg, 1.00 mmol),
p-formylphenylboronic acid ester (232 mg, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 184 °C.
1H-NMR (CDCl
3) δ 9.30 (s, 1H), 8.78 (s, 1H), 8.72 (s, 1H), 7.82(d,
J= 7.8 Hz, 2H), 7.53 (d,
J = 7.8 Hz, 2H), 6.46 (br.), 6.32 (s, 1H), 3.80–3.79 (m, 1H), 1.91–1.84 (m, 2H), 1.69–1.64 (m, 2H), 1.58–1.56 (m, 1H), 1.34–1.32 (m, 14H), 1.25–1.20 (m, 3H).
13C-NMR (CDCl
3) δ 166.5, 162.4, 148.0, 146.4, 144.4, 142.8, 137.8, 135.2, 126.7, 83.87, 83.8, 48.2, 32.7, 32.6, 25.3, 24.7, 24.5.
11B-NMR (CDCl
3) δ 30.9. HRMS (ESI, positive ion):
m/z [M+H]
+, found 466.2503. C
25H
32BN
3O
5 requires 466.2519.
2-(Cyclohexylamino)-2-oxo-1-(3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl)ethyl pyrazine-2-carboxylate (
B12). The desired compound (195 mg, 42% yield) was prepared by General Procedure B using pyrazine-2-carboxylic acid (124 mg, 1.00 mmol),
m-formylphenylboronic acid ester (232 mg, 1.00 mmol), and cyclohexyl isocyanide (0.124 mL, 1.00 mmol); mp = 140 °C.
1H-NMR (CDCl
3) δ 9.32 (s, 1H), 8.78 (s, 1H), 8.73 (s, 1H), 7.95 (s, 1H), 7.80 (d,
J = 7.2 Hz, 1H), 7.65 (d,
J = 7.8 Hz, 1H), 7.40 (t,
J = 7.2, 15 Hz, 1H), 6.42 (br.), 6.34 (s, 1H), 3.84–3.77 (m, 1H), 1.88–1.87 (m, 2H), 1.73–1.66 (m, 4H), 1.61–1.58 (m, 1H), 1.38–1.33 (m, 14H), 1.26–1.16 (m, 3H).
13C-NMR (CDCl
3) δ 166.7, 162.5, 148.0, 146.5, 144.4, 143.0, 135.7, 134.4, 134.2, 130.5, 128.3, 83.9, 48.3, 32.8, 32.7, 25.4, 24.9, 24.8, 24.61.
11B-NMR (CDCl
3) δ 30.4. HRMS (ESI, positive ion):
m/z [M+H]
+, found 466.2512. C
25H
32BN
3O
5 requires 466.2519.

 (1a)
(1a) (2a)
(2a) (1b)
(1b)
 (1c)
(1c)

 (2b)
(2b)




 (2c)
(2c)




 (2d)
(2d)




 (e)
(e)




 (f)
(f)




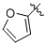 (g)
(g)

 (1d)
(1d) (2h)
(2h)
 (2i)
(2i)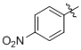 (1e)
(1e)


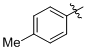 (1f)
(1f)


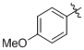 (1g)
(1g)


 (1h)
(1h)


 (1i)
(1i)