Evaluation of Selenite Effects on Selenoproteins and Cytokinome in Human Hepatoma Cell Lines
Abstract
:1. Introduction
2. Results and Discussion
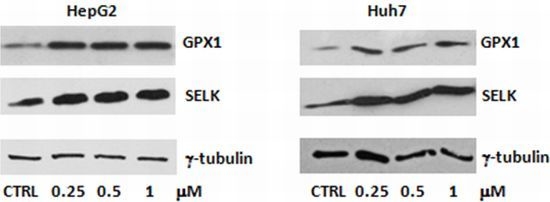
2.1. Evaluation of SELENBP1, SELK and GPX1 Expression in HepG2 and Huh7 Cells



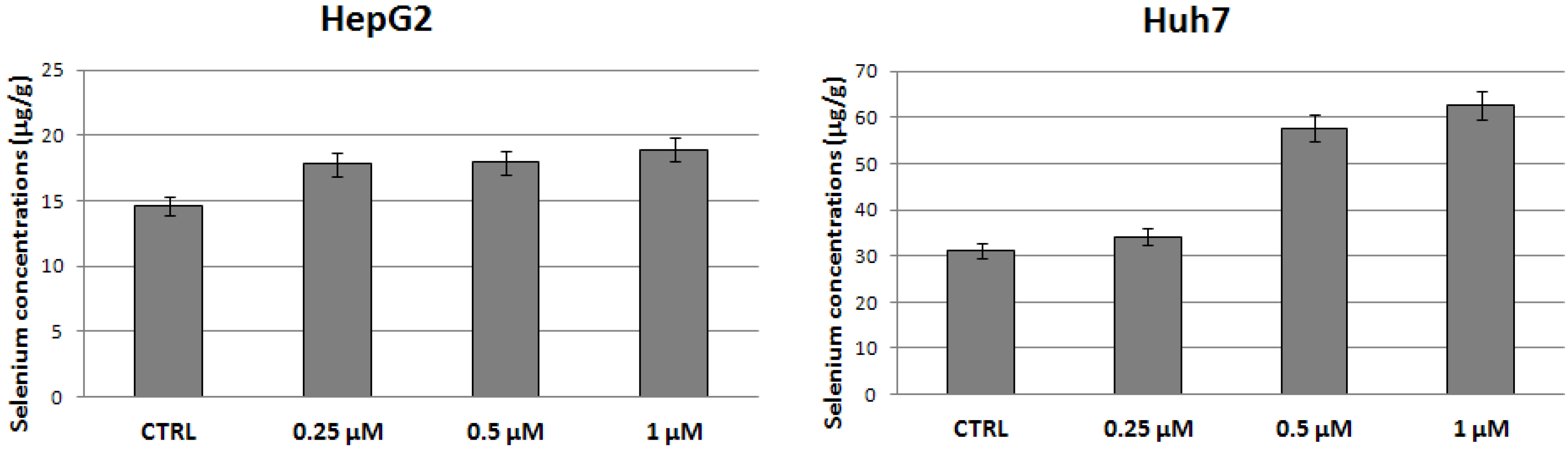
2.2. Evaluation of Selenium Concentrations
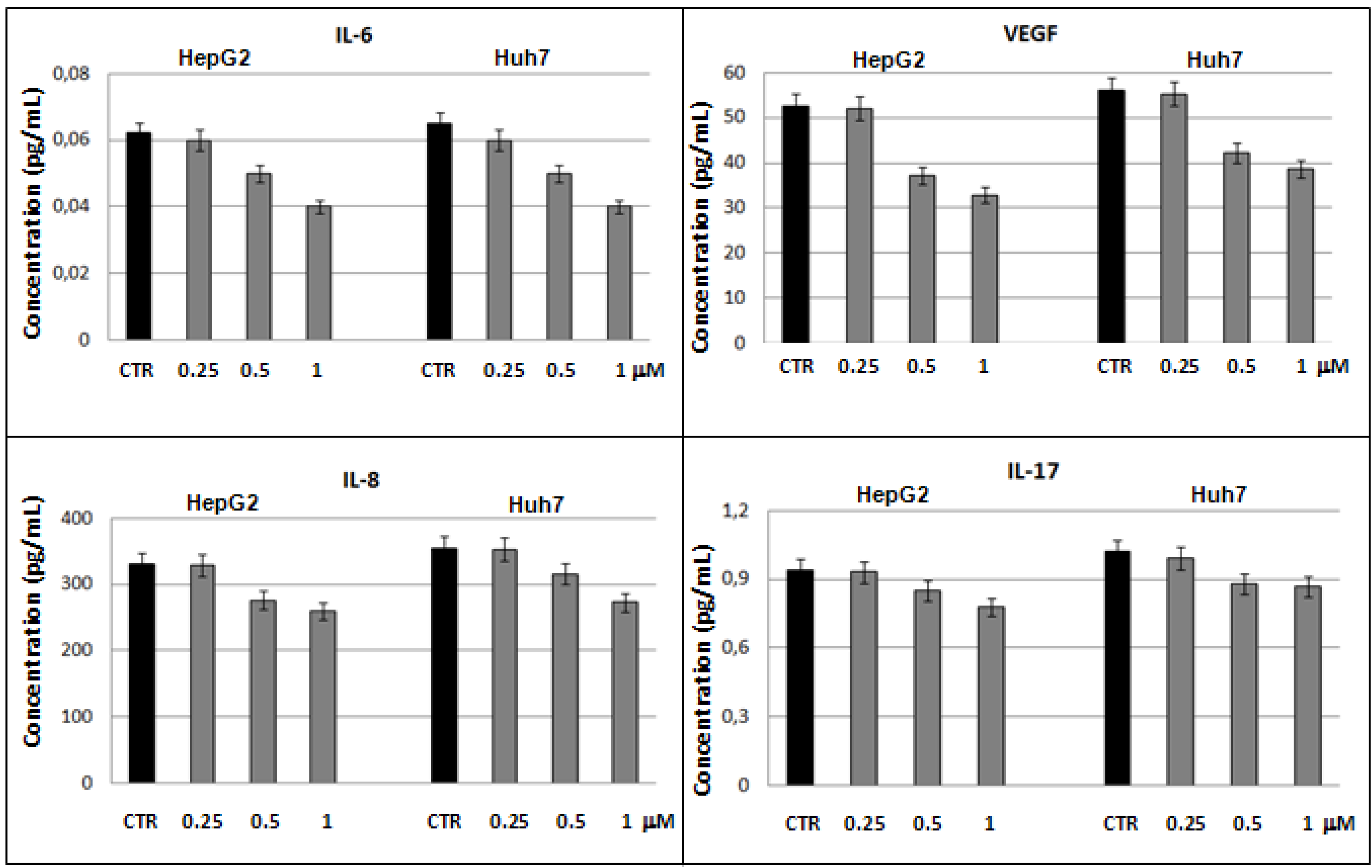
2.3. Bio-Plex assay on HepG2 and Huh7 cells
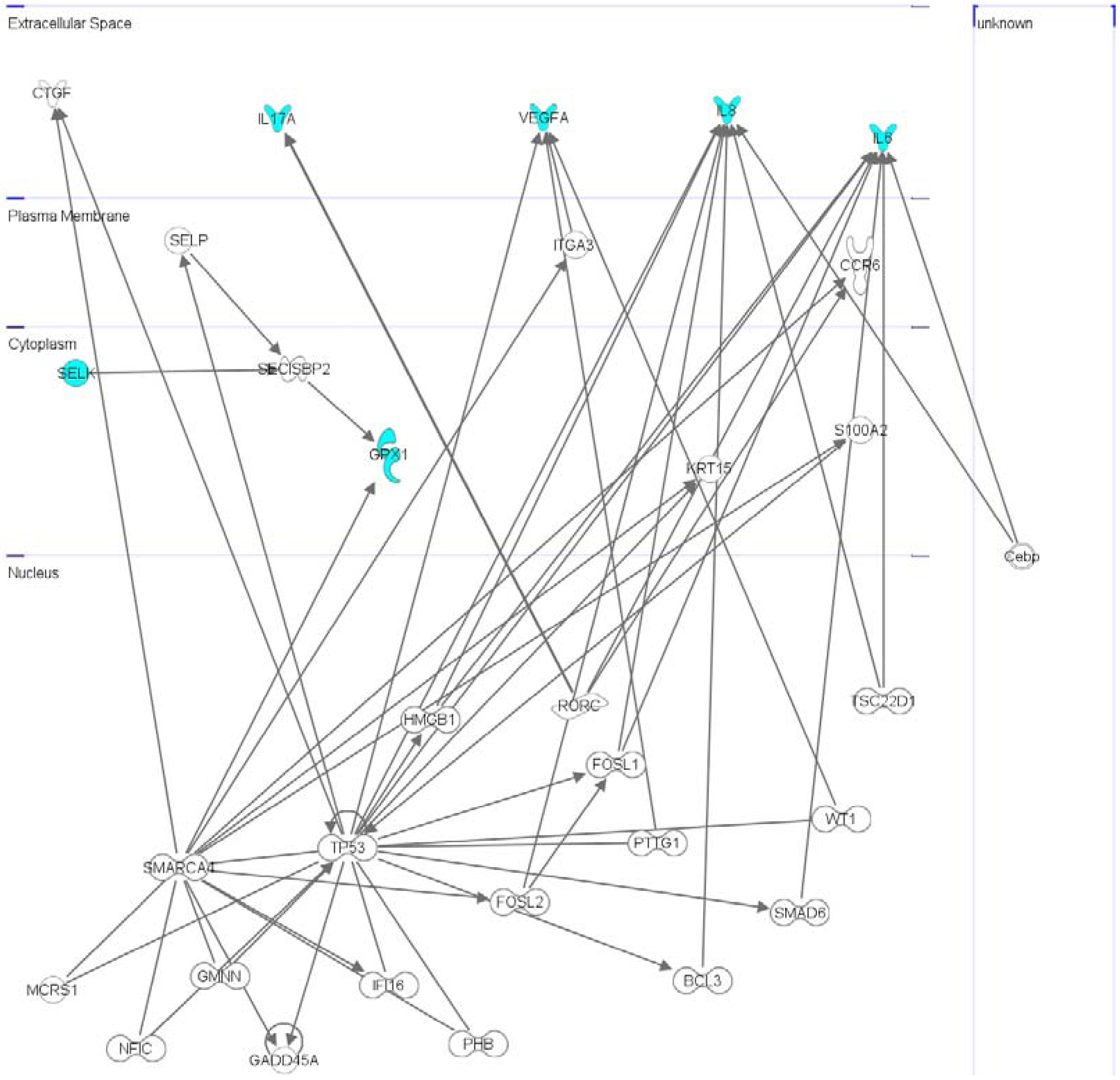
2.4. Interactomic Studies
3. Experimental
3.1. Cell Culture
3.2. Protein Extraction and Western Blot Analyses
3.3. Atomic Absorption Spectrometer Studies
3.4. Bio-Plex Assay
3.5. Statistical Analysis
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Keshan Disease Research Group. Epidemiologic studies on the ethiologic relationship of selenium and Keshan disease. Chin. Med. J. 1979, 92, 477–482.
- Sagara, Y.; Tan, S.; Maher, P.; Schubert, D. Mechanisms of resistance to oxidative stress in Alzheimer’s diseases. Biofactors 1998, 8, 45–50. [Google Scholar] [CrossRef]
- Clark, L.C.; Combs, G.F., Jr.; Turnbull, B.W.; Slate, E.H.; Chalker, D.K.; Chow, J.; Davis, L.S.; Glover, R.A.; Graham, G.F.; Gross, E.G.; et al. Effects of Selenium supplementation for cancer prevention in patients with carcinoma of the skin. JAMA 1996, 276, 1957–1963. [Google Scholar]
- Rayman, M.P. Selenium in cancer prevention: A review of the evidence and mechanism of action. Proc. Nutr. Soc. 2005, 64, 527–542. [Google Scholar] [CrossRef]
- Peters, U.; Takata, Y. Selenium and the prevention of prostate and colorectal cancer. Mol. Nutr. Food Res. 2008, 52, 1261–1262. [Google Scholar] [CrossRef]
- Trottier, G.; Boström, P.J.; Lawrentschuk, N.; Fleshner, N.E. Nutraceuticals and prostate cancer prevention: A current review. Nat. Rev. Urol. 2009, 7, 21–30. [Google Scholar]
- Yoo, M.H.; Xu, X.M.; Carlson, B.A.; Patterson, A.D.; Gladyshev, V.N.; Hatfield, D.L. Targeting thioredoxin reductase 1 reduction in cancer cells inhibits self-sufficient growth and DNA replication. PLoS One 2007, 2, e1112. [Google Scholar]
- Lecomte, E.; Herberth, B.; Pirrollet, P.; Chancerelle, Y.; Arnaud, J.; Musse, N.; Paille, F.; Siest, G.; Artur, Y. Effect of alcohol comsumption on blood antioxidant nutrients and oxidative stress indicators. Am. J. Clin. Nutr. 1994, 60, 255–261. [Google Scholar]
- Loguercio, C.; de Girolamo, V.; Federico, A.; Feng, S.L.; Cataldi, V.; del Vecchio Blanco, C.; Gialanella, G. Trace elements and chronic liver diseases. J. Trace Elem. Med. Biol. 1997, 11, 158–161. [Google Scholar] [CrossRef]
- Guarini, P.; Stanzial, A.M.; Olivieri, O.; Casaril, M.; Galvani, S.; Pantalena, M.; Corrocher, R. Erythrocyte membrane lipids and serum selenium in post-viral and alcoholic cirrhosis. Clin. Chim. Acta 1998, 270, 139–150. [Google Scholar] [CrossRef]
- Navarro-Alarcòn, M.; Lòpez-Martìnez, M.C. Essentiality of selenium in the human body: Relationship with different diseases. Sci. Total Environ. 2000, 249, 347–371. [Google Scholar] [CrossRef]
- Navarro-Alarcòn, M.; Lòpez-G de la Serrana, H.; Pèrez-Valero, V.; López-Martínez, M.C. Selenium concentrations in serum of individuals with liver diseases (cirrhosis or hepatitis): Relationships with some nutritional and biochemical markers. Sci. Total Environ. 2002, 291, 135–141. [Google Scholar] [CrossRef]
- Tashiro, H.; Kawamoto, T.; Okubo, T.; Koide, O. Variation in the distribution of trace elements in hepatoma. Biol. Trace Elem. Res. 2003, 95, 49–63. [Google Scholar] [CrossRef]
- Marra, M.; Sordelli, I.M.; Lombardi, A.; Lamberti, M.; Tarantino, L.; Giudice, A.; Stiuso, P.; Abbruzzese, A.; Sperlongano, R.; Accardo, M.; et al. Molecular targets and oxidative stress biomarkers in hepatocellular carcinoma: An overview. J. Transl. Med. 2011, 9, 171. [Google Scholar] [CrossRef]
- Caraglia, M.; Giuberti, G.; Marra, M.; Addeo, R.; Montella, L.; Murolo, M.; Sperlongano, P.; Vincenzi, B.; Naviglio, S.; Prete, S.D.; et al. Oxidative stress and ERK1/2 phosphorylation as predictors of outcome in hepatocellular carcinoma patients treated with sorafenib plus octreotide LAR. Cell Death Dis. 2011, 2, e150. [Google Scholar] [CrossRef]
- Prete, S.D.; Montella, L.; Caraglia, M.; Maiorino, L.; Cennamo, G.; Montesarchio, V.; Piai, G.; Febbraro, A.; Tarantino, L.; Capasso, E.; et al. Sorafenib plus octreotide is an effective and safe treatment in advanced hepatocellular carcinoma: Multicenter phase II So.LAR. study. Cancer Chemother. Pharmacol. 2010, 66, 837–844. [Google Scholar] [CrossRef]
- Castello, G.; Scala, S.; Palmieri, G.; Curley, S.A.; Izzo, F. HCV-related hepatocellular carcinoma: From chronic inflammation to cancer. Clin. Immunol. 2010, 134, 237–250. [Google Scholar] [CrossRef]
- Fusco, M.; Girardi, E.; Piselli, P.; Palombino, R.; Polesel, J.; Maione, C.; Scognamiglio, P.; Pisanti, F.A.; Solmone, M.; di Cicco, P.; et al. Collaborating Study Group, Epidemiology of viral hepatitis infections in an area of southern Italy with high incidence rates of liver cancer. Eur. J. Cancer 2008, 44, 847–853. [Google Scholar]
- Colombino, M.; Sperlongano, P.; Izzo, F.; Tatangelo, F.; et al. BRAF and PIK3CA genes are somatically mutated in hepatocellular carcinoma among patients from South Italy. Cell Death Dis. 2012, 3, e259. [Google Scholar] [CrossRef]
- Bansal, M.P.; Mukhopadhyay, T.; Scott, J.; Cook, R.G.; Mukhopadhyay, R.; Medina, D. DNA sequencing of a mouse liver protein that binds selenium: Implications for selenium’s mechanism of action in cancer prevention. Carcinogenesis 1990, 11, 2071–2073. [Google Scholar] [CrossRef]
- Chang, P.W.; Tsui, S.K.; Liew, C.; Lee, C.C.; Waye, M.M.; Fung, K.P. Isolation characterization and chromosomal mapping of a novel cDNA clone encoding human selenium binding protein. J. Cell. Biochem. 1997, 64, 217–224. [Google Scholar]
- Raucci, R.; Colonna, G.; Guerriero, E.; Capone, F.; Accardo, M.; Castello, G.; Costantini, S. Structural and functional studies of the human selenium binding protein-1 and its involvment in hepatocellular carcinoma. Biochim. Biophys. Acta 2011, 1814, 513–522. [Google Scholar]
- Di Stasio, M.; Volpe, M.G.; Colonna, G.; Nazzaro, M.; Polimeno, M.; Scala, S.; Castello, G.; Costantini, S. A possible predictive marker of progression for hepatocellular carcinoma. Oncol. Lett. 2011, 2, 1247–1251. [Google Scholar]
- Lim, S.O.; Gu, J.M.; Kim, M.S.; Kim, H.S.; Park, Y.N.; Park, C.K.; Cho, J.W.; Park, Y.M.; Jung, G. Epigenetic changes induced by reactive oxygen species in hepatocellular carcinoma: Methylation of the E-cadherin promoter. Gastroenterology 2008, 135, 2128–2140. [Google Scholar] [CrossRef]
- Kałuża, J.; Fijałkowski, P.; Krawczyk, P.; Błaszczyk, J.; Dworniak, D. Assessment of selected oxygen metabolism indexes in patients with chronic hepatitis C. Exp. Clin. Hepatol. 2008, 4, 7–11. [Google Scholar]
- Du, S.; Zhou, J.; Jia, Y.; Huang, K. SelK is a novel ER stress-regulated protein and protects HepG2 cells from ER stress agent-induced apoptosis. Arch. Biochem. Biophys. 2010, 502, 137–143. [Google Scholar] [CrossRef]
- Huang, C.; Ding, G.; Gu, C.; Zhou, J.; Kuang, M.; Ji, Y.; He, Y.; Kondo, T.; Fan, J. Decreased Selenium-Binding Protein 1 Enhances Glutathione Peroxidase 1 Activity and Downregulates HIF-1α to Promote Hepatocellular Carcinoma Invasiveness. Clin. Cancer Res. 2012, 18, 3042–3053. [Google Scholar] [CrossRef]
- Pan, S.; Wang, Y.; Feng, L.; Fan, C.; Guo, D.; Liu, X.; Fan, J. Study on proteomics of Hela cell apoptosis in bufalin-induced human cervical carcinoma. Zhong Guo Zhong Yao Za Zhi 2012, 37, 1998–2004. [Google Scholar]
- Baker, R.D.; Baker, S.S.; LaRosa, K.; Whitney, C.; Newburger, P.E. Selenium regulation of glutathione peroxidase in human hepatoma cell line Hep3B. Arch. Biochem. Biophys. 1993, 304, 53–57. [Google Scholar] [CrossRef]
- Fang, W.; Goldberg, M.L.; Pohl, N.M.; Bi, X.; Tong, C.; Xiong, B.; Koh, T.J.; Diamond, A.M.; Yang, W. Functional and physical interaction between the selenium-binding protein 1 (SBP1) and the glutathione peroxidase 1 selenoprotein. Carcinogenesis 2010, 31, 1360–1366. [Google Scholar] [CrossRef]
- Jabari, S.; Meissnitzer, M.; Quint, K.; Gahr, S.; Wissniowski, T.; Hahn, E.G.; Neureiter, D.; Ocker, M. Cellular plasticity of trans- and dedifferentiation markers in human hepatoma cellsin in vitro and in vivo. Int. J. Oncol. 2009, 35, 69–80. [Google Scholar]
- Hussain, S.P.; Schwank, J.; Staib, F.; Wang, X.W.; Harris, C.C. TP53 mutations and hepatocellular carcinoma: Insights into the etiology and pathogenesis of liver cancer. Oncogene 2007, 26, 2166–2176. [Google Scholar] [CrossRef]
- Costantini, S.; Capone, F.; Guerriero, E.; Maio, P.; Colonna, G.; Castello, G. Serum cytokine levels as putative prognostic markers in the progression of chronic HCV hepatitis to cirrhosis. Eur. Cytokine Netw. 2010, 21, 251–256. [Google Scholar]
- Capone, F.; Costantini, S.; Guerriero, E.; Calemma, R.; Napolitano, M.; Scala, S.; Izzo, F.; Castello, G. Serum cytokine levels in patients with hepatocellular carcinoma. Eur. Cytokine Netw. 2010, 21, 99–104. [Google Scholar]
- Akiba, J.; Yano, H.; Ogasawara, S.; Higaki, K.; Kojiro, M. Expression and function of interleukin-8 in human hepatocellular carcinoma. Int. J. Oncol. 2001, 18, 257–264. [Google Scholar]
- Kubo, F.; Ueno, S.; Hiwatashi, K.; Sakoda, M.; Kawaida, K.; Nuruki, K.; Aikou, T. Interleukin 8 in human hepatocellular carcinoma correlates with cancer cell invasion of vessels but not with tumor angiogenesis. Ann. Surg. Oncol. 2005, 12, 800–807. [Google Scholar] [CrossRef]
- Kolls, J.K.; Linden, A. Interleukin-17 family members and infiammation. Immunity 2004, 21, 467–476. [Google Scholar] [CrossRef]
- Feurino, L.W.; Zhang, Y.; Bharadwaj, U.; Zhang, R.; Li, F.; Fisher, W.E.; Brunicardi, F.C.; Chen, C.; Yao, Q.; Min, L. IL-6 stimulates Th2 type cytokine secretion and upregulates VEGF and NRP-1 expression in pancreatic cancer cells. Cancer Biol. Ther. 2007, 6, 1096–1100. [Google Scholar] [CrossRef]
- Pei, Z.; Li, H.; Guo, Y.; Jin, Y.; Lin, D. Sodium selenite inhibits the expression of VEGF TGFbeta(1) and IL-6 induced by LPS in human PC3 cells via TLR4-NF-(K)B signaling blockage. Int. Immunopharmacol. 2010, 10, 50–56. [Google Scholar] [CrossRef]
- Guerriero, E.; Sorice, A.; Capone, F.; Costantini, S.; Palladino, P.; D’ischia, M.; Castello, G. Effects of Lipoic Acid Caffeic Acid and a Synthesized Lipoyl-Caffeic Conjugate on Human Hepatoma Cell Lines. Molecules 2011, 16, 6365–6377. [Google Scholar] [CrossRef]
- Lee, D.; Kim, J.W.; Seo, T.; Hwang, S.G.; Choi, E.J.; Choe, J. SWI/SNF complex interacts with tumor suppressor p53 and is necessary for the activation of p53-mediated transcription. J. Biol. Chem. 2002, 277, 22330–22337. [Google Scholar]
- Copeland, P.R.; Fletcher, J.E.; Carlson, B.A.; Hatfield, D.L.; Driscoll, D.M. A novel RNA binding protein, SBP2, is required for the translation of mammalian selenoprotein mRNAs. EMBO J. 2000, 19, 306–314. [Google Scholar] [CrossRef]
- Burgler, S.; Mantel, P.Y.; Bassin, C.; Ouaked, N.; Akdis, C.A.; Schmidt-Weber, C.B. RORC2 is Involved in T Cell Polarization through Interaction with the FOXP3 Promoter. J. Immunol. 2010, 184, 6161–6169. [Google Scholar] [CrossRef]
- Sharma, A.; Gautam, V.K.; Costantini, S.; Paladino, A.; Colonna, G. Interactomic and pharmacological insights on human Sirt-1. Front. Pharmacol. 2012, 2012, 40. [Google Scholar]
- Chatterjee, A.; Mir, S.A.; Dutta, D.; Mitra, A.; Pathak, K.; Sarkar, S. Analysis of p53 and NF-κB signaling in modulating the cardiomyocyte fate during hypertrophy. J. Cell. Physiol. 2011, 226, 2543–2554. [Google Scholar] [CrossRef]
- Yoon, S.O.; Kim, M.M.; Park, S.J.; Kim, D.; Chung, J.; Chung, A.S. Selenite suppresses hydrogen peroxide-induced cell apoptosis through inhibition of ASK1/JNK and activation of PI3-K/Akt pathways. FASEB J. 2002, 16, 111–113. [Google Scholar]
- O’Neill, C.F.; Hunakova, L.; Kelland, L.R. Cellular pharmacology of cis and trans pairs of platinum complexes in cisplatin-sensitive and -resistant human ovarian carcinoma cells. Chem. Biol. Interact. 1999, 123, 11–29. [Google Scholar] [CrossRef]
- Halliwell, B. Free radicals and antioxidants—quo vadis? Trends Pharmacol. Sci. 2011, 32, 125–130. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the gels obtained by Western Blot are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Rusolo, F.; Pucci, B.; Colonna, G.; Capone, F.; Guerriero, E.; Milone, M.R.; Nazzaro, M.; Volpe, M.G.; Bernardo, G.D.; Castello, G.; et al. Evaluation of Selenite Effects on Selenoproteins and Cytokinome in Human Hepatoma Cell Lines. Molecules 2013, 18, 2549-2562. https://doi.org/10.3390/molecules18032549
Rusolo F, Pucci B, Colonna G, Capone F, Guerriero E, Milone MR, Nazzaro M, Volpe MG, Bernardo GD, Castello G, et al. Evaluation of Selenite Effects on Selenoproteins and Cytokinome in Human Hepatoma Cell Lines. Molecules. 2013; 18(3):2549-2562. https://doi.org/10.3390/molecules18032549
Chicago/Turabian StyleRusolo, Fabiola, Biagio Pucci, Giovanni Colonna, Francesca Capone, Eliana Guerriero, Maria Rita Milone, Melissa Nazzaro, Maria Grazia Volpe, Gianni Di Bernardo, Giuseppe Castello, and et al. 2013. "Evaluation of Selenite Effects on Selenoproteins and Cytokinome in Human Hepatoma Cell Lines" Molecules 18, no. 3: 2549-2562. https://doi.org/10.3390/molecules18032549
APA StyleRusolo, F., Pucci, B., Colonna, G., Capone, F., Guerriero, E., Milone, M. R., Nazzaro, M., Volpe, M. G., Bernardo, G. D., Castello, G., & Costantini, S. (2013). Evaluation of Selenite Effects on Selenoproteins and Cytokinome in Human Hepatoma Cell Lines. Molecules, 18(3), 2549-2562. https://doi.org/10.3390/molecules18032549