Unexpected Behavior of Enaminones: Interesting New Routes to 1,6-Naphthyridines, 2-Oxopyrrolidines and Pyrano[4,3,2-de][1,6]naphthyridines
Abstract
:1. Introduction
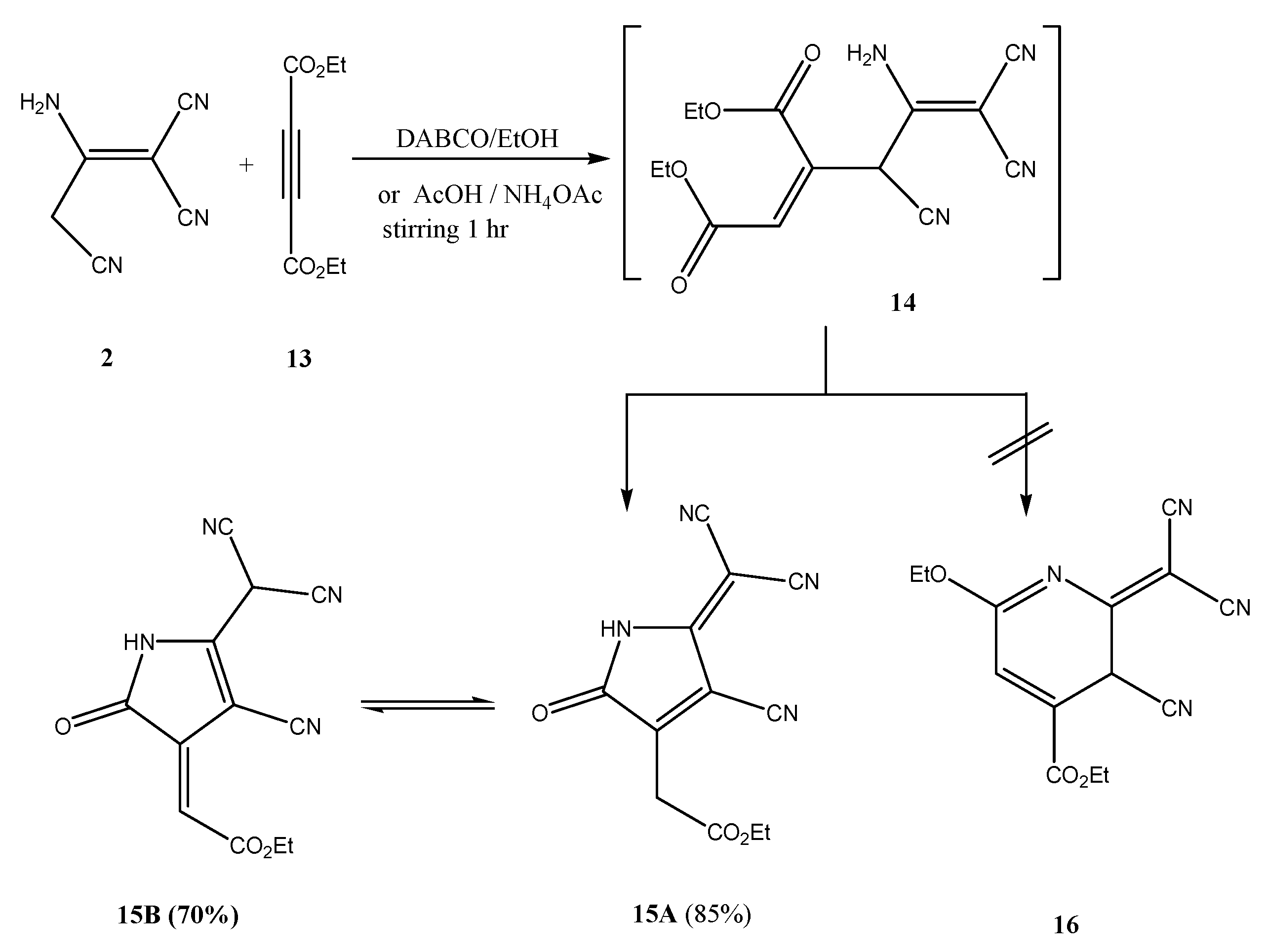
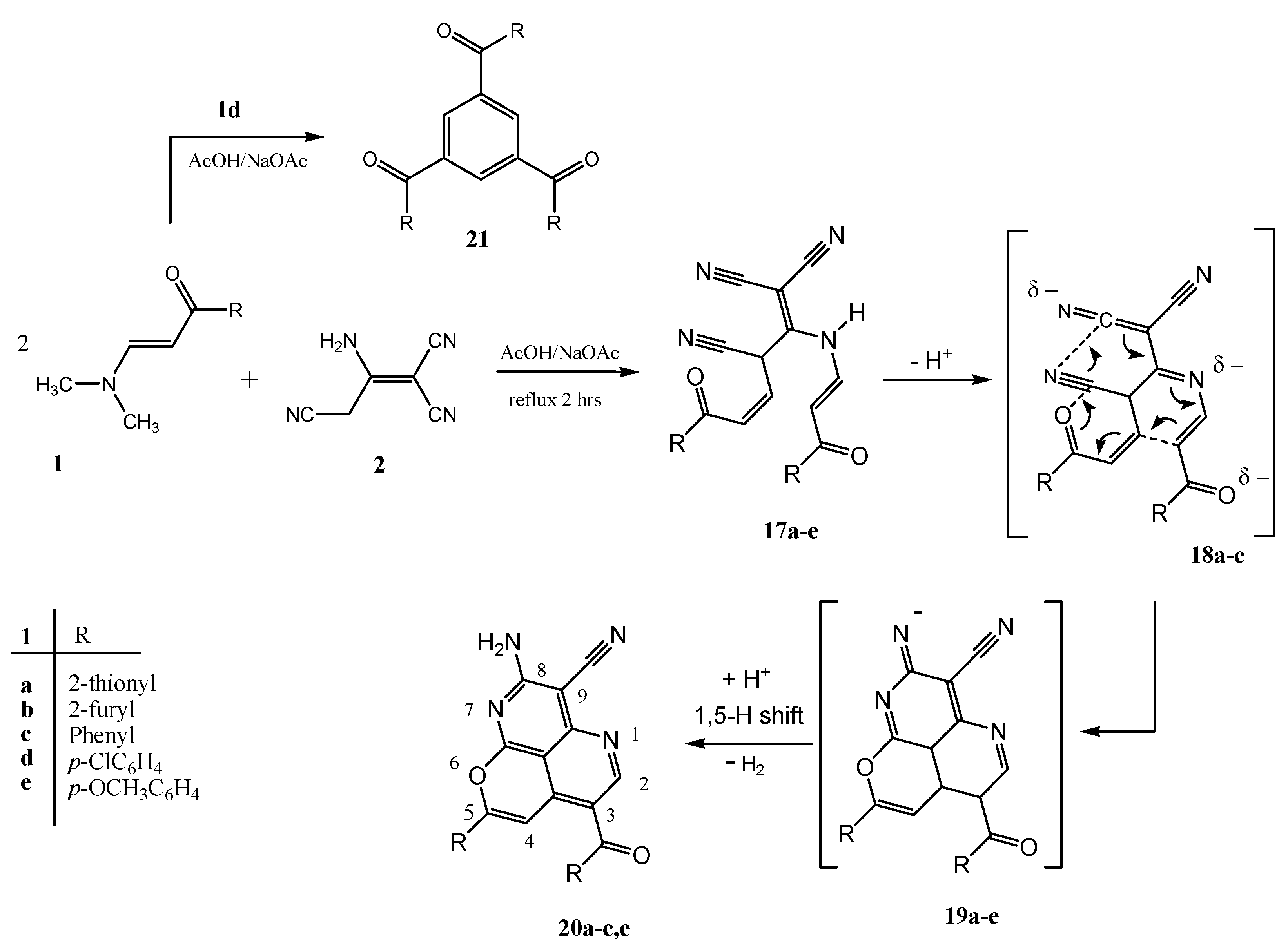
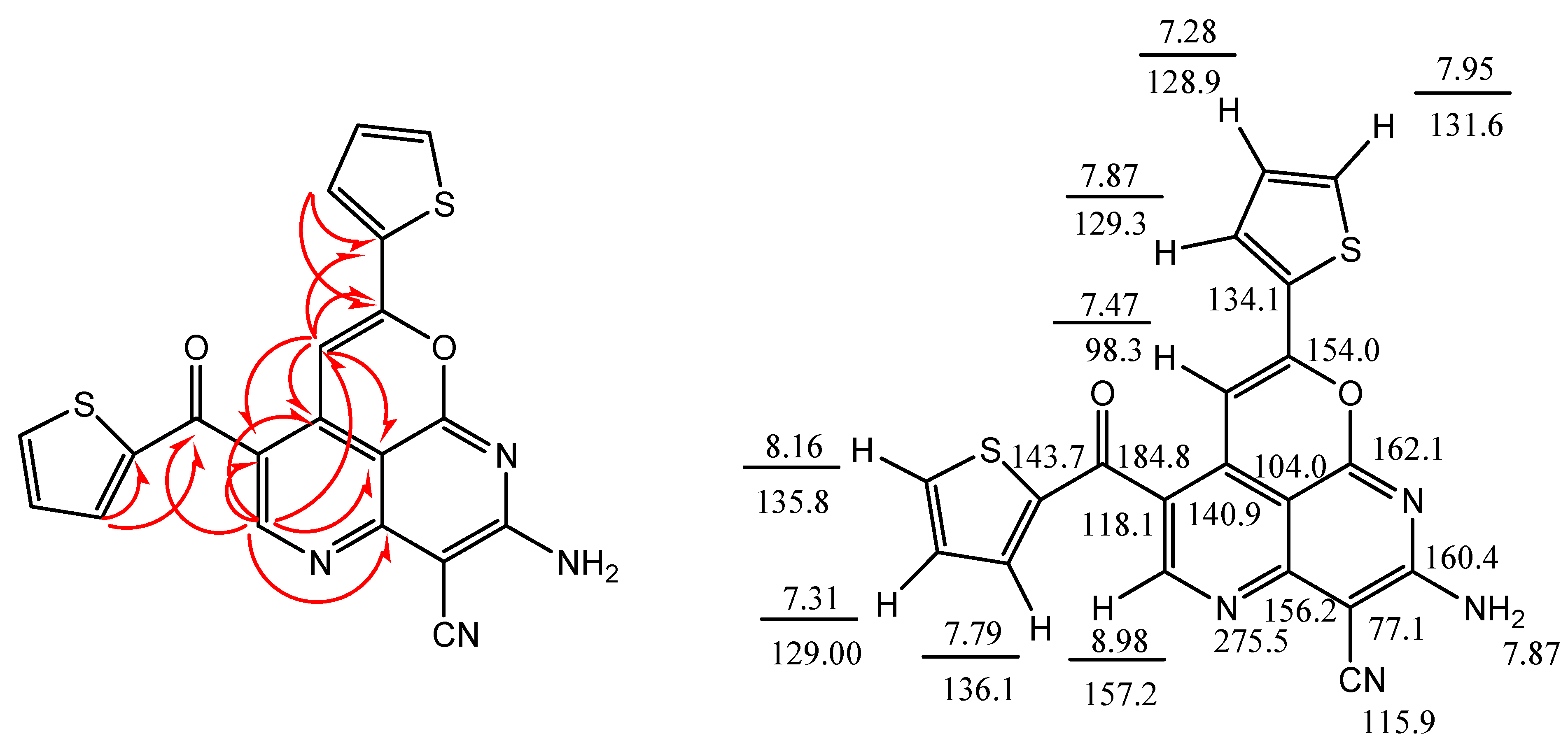
2. Results and Discussion
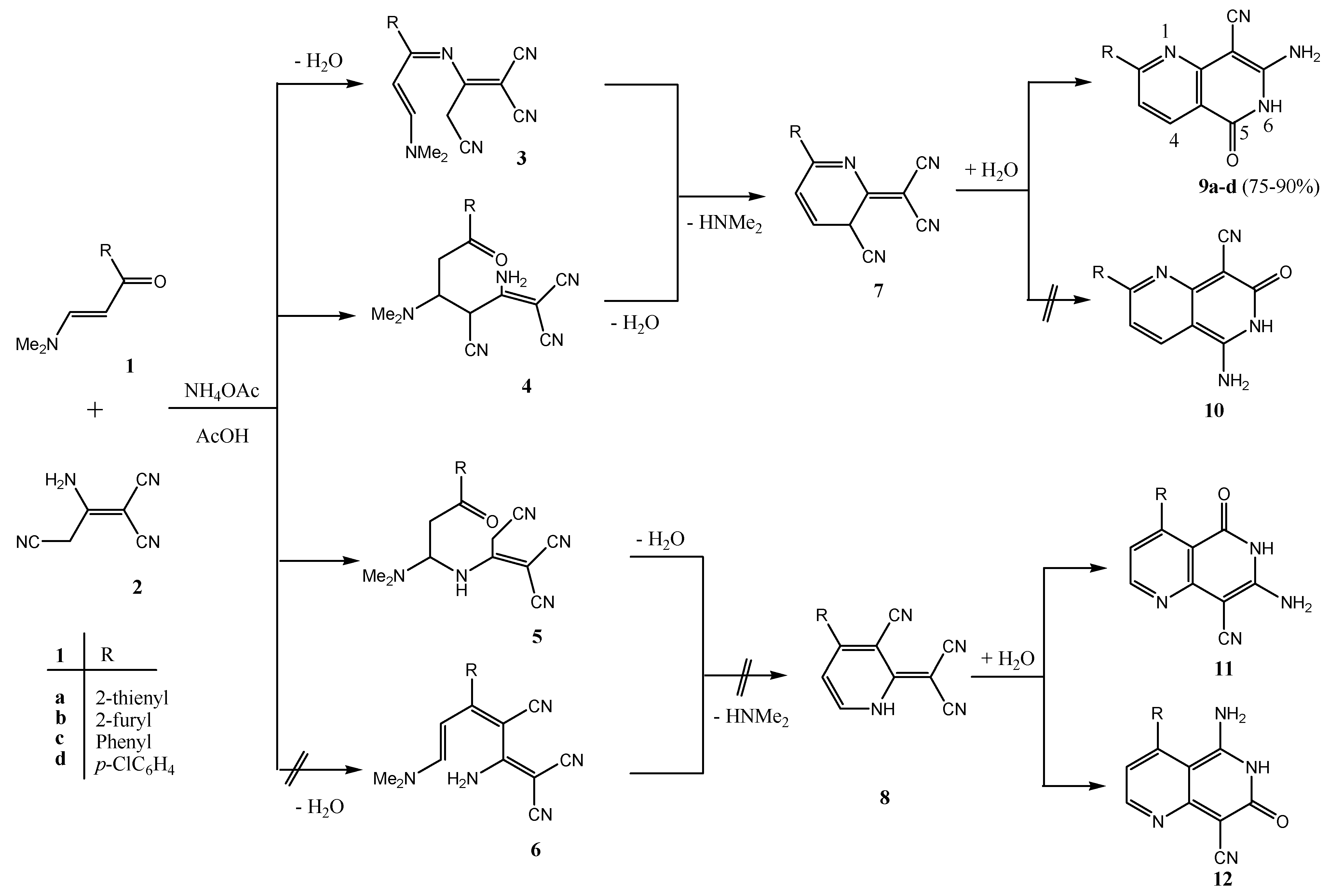
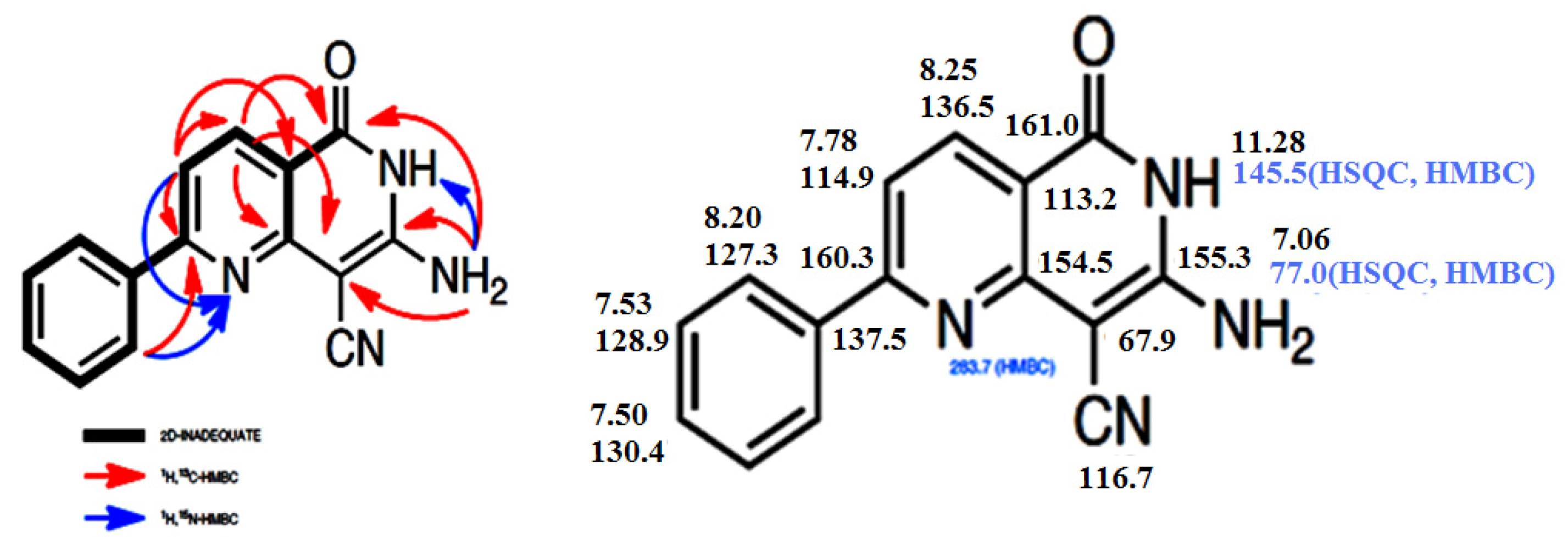
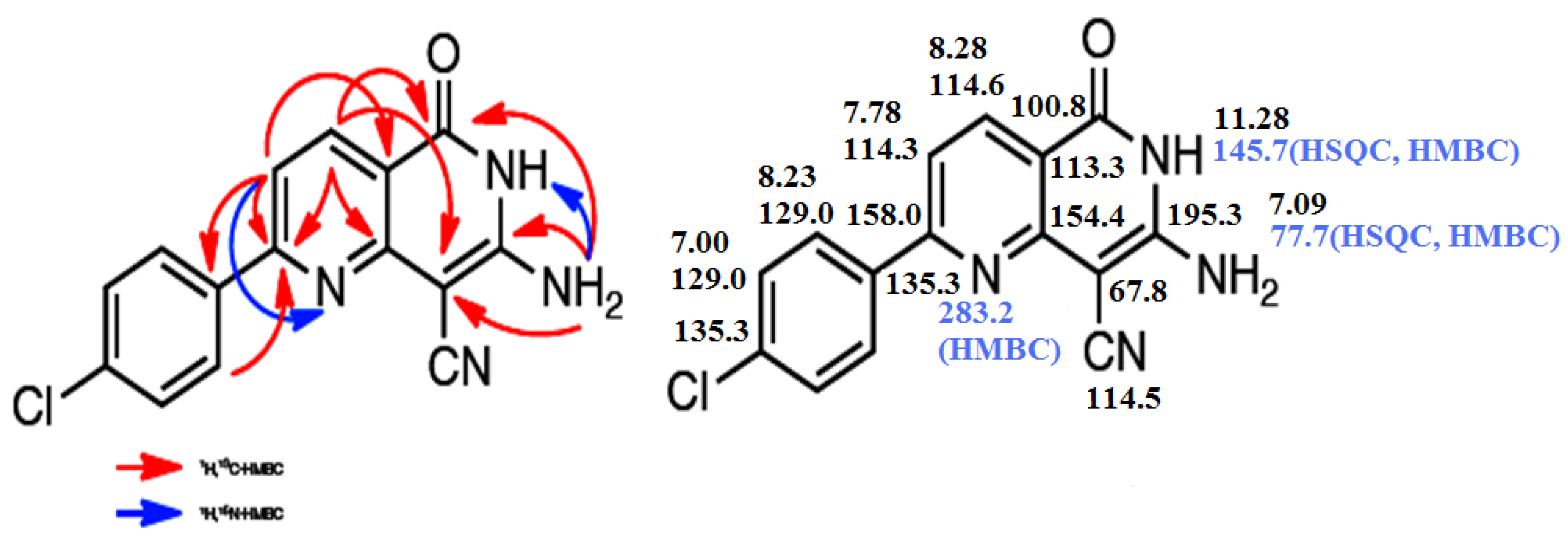
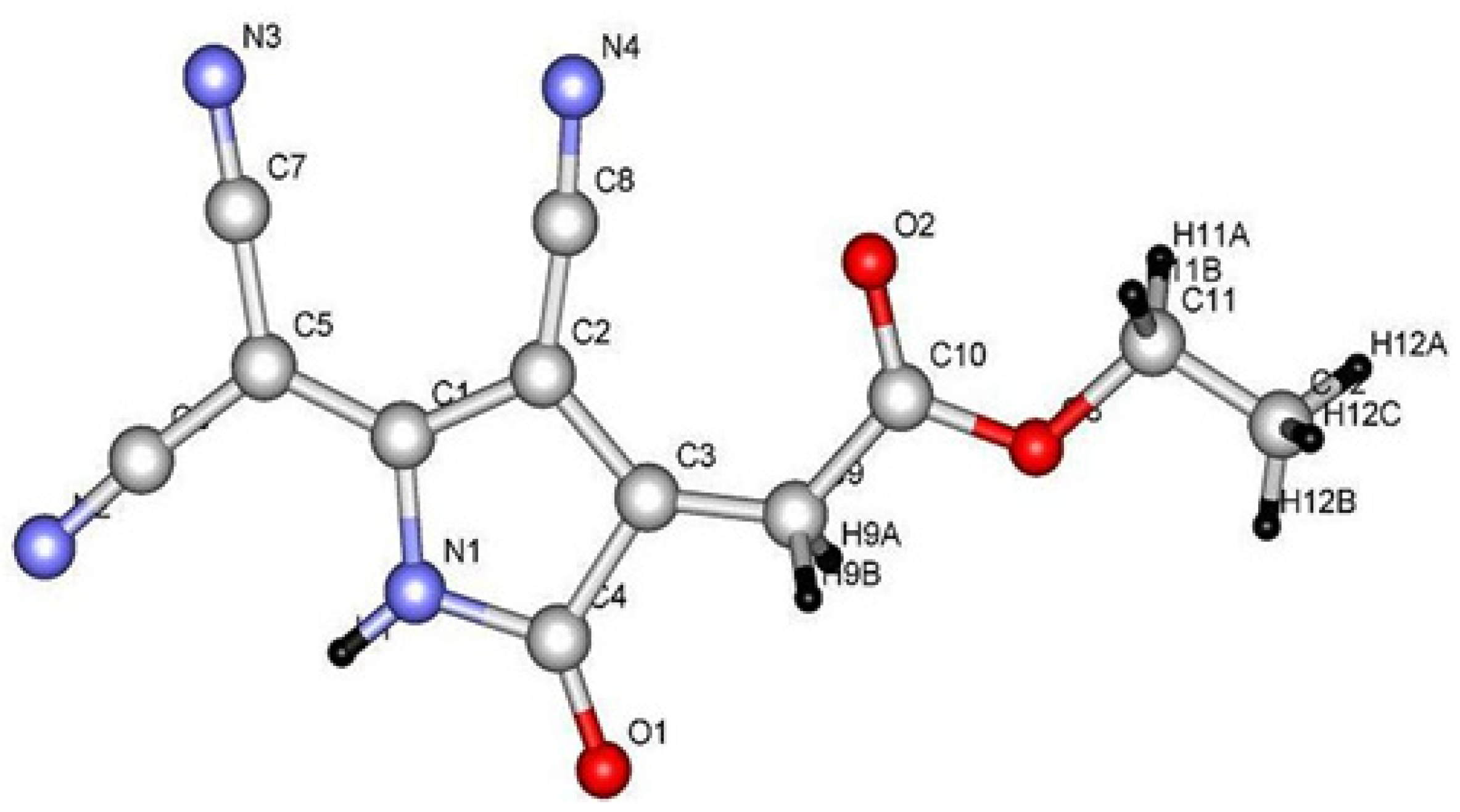
3. Experimental
3.1. General
3.2. General Procedure for the Synthesis of 9a–d
3.3. General Procedure to Syntheses of 20a–c,e
3.4. Synthesis of [3,5-bis-(4-chlorobenzoyl)phenyl]-(4-chlorophenyl)methanone (21)
4. Conclusions
Acknowledgments
- Sample Availability: Samples of the all compounds are available from the authors.
References and Notes
- Al-Mousawi, S.M.; Moustafa, M.S.; Elnagdi, M.H. Green synthetic approaches: solventless synthesis of polyfunctionally substituted aromatics as potential versatile building blocks in organic synthesis utilizing enaminones and enaminonitriles as precursors. Green Chem. Lett. Rev. 2011, 4, 185–193. [Google Scholar] [CrossRef]
- Al-Zaydi, K.M.; Borik, R.M.; Mekheimer, R.A.; Elnagdi, M.H. Green chemistry: A facile synthesis of polyfunctionally substituted thieno[3,4-c]pyridinones and thieno[3,4-d]pyridazinones under neat reaction conditions. Ultrasonics Sonochem. 2010, 17, 909–915. [Google Scholar] [CrossRef]
- Al-Mousawi, S.M.; Moustafa, M.S.; Elnagdi, M.H. A novel synthesis of 2-arylhydrazono-6-amino-4-arylbenzene-1,3-dicarbonitriles and their conversion into phthalazines. Tetrahedron Lett. 2009, 50, 6411–6413. [Google Scholar] [CrossRef]
- Al-Awadi, N.A.; Abdelkhalik, M.M.; Abdelhamid, I.A.; Elnagdi, M.H. Pyrolytic methods in organic synthesis: Novel routes for the synthesis of 3-oxoalkanenitriles, 2-acyl anilines, and 2-aroyl anilines. Synlett 2007, 19, 2979–2982. [Google Scholar]
- Al-Saleh, B.; Abdelkhalik, M.M.; Eltoukhy, A.M.; Elnagdi, M.H. Enaminones in heterocyclic synthesis: A new regioselective synthesis of 2,3,6-trisubstituted pyridines, 6-substituted-3-aroylpyridines and 1,3,5-triaroylbenzenes. J. Heterocycl. Chem. 2002, 39, 1035–1038. [Google Scholar] [CrossRef]
- Al-Mousawi, S.M.; Moustafa, M.S.; Abdelhamid, I.A.; Elnagdi, M.H. Reassignment of the structures of condensation products of α-keto α'-formylarylhydrazones with ethyl cyanoacetate: A novel route to ethyl 5-arylazo-2-hydroxynicotinates. Tetrahedron Lett. 2011, 52, 202–204. [Google Scholar]
- Riyadh, S.M.; Abdelhamid, I.A.; Al-Matar, H.M.; Hilmy, N.M.; Elnagdi, M.H. Enamines as precursors to polyfunctional heteroaromatic compounds; A decade of development. Heterocycles 2008, 75, 1849–1905. [Google Scholar] [CrossRef]
- VanAllan, J.A.; Petropoulos, C.C.; George, A.; Maier, D.P. Reactions of some 4-dicyanomethylenepyran derivatives with malononitrile and hydrazines. J. Heterocycl. Chem. 1970, 7, 1363–1368. [Google Scholar] [CrossRef]
- Helmy, N.M.; El-Baih, F.E.M.; Al-Alshaikh, M.A.; Moustafa, M.S. A route to dicyanomethylene pyridines and substituted benzonitriles utilizingmalononitrile dimer as a precursor. Molecules 2011, 16, 298–306. [Google Scholar] [CrossRef]
- CCDC 861196 contains the supplementary crystallographic data for compound 15A. These data can be obtained free of charge from the Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk.
- CCDC 838314 contains the supplementary crystallographic data for compound 20c. These data can be obtained free of charge from the Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk
- Al-Enezi, A.; Al-Saleh, B.; Elnagdi, M.H. Studies with heteroaromatic amines: The reaction of some heteroaromatic amines with 1-substituted 3-dimethylaminopropanones, Enaminones and cinnamonitriles. J. Chem. Res. (S) 1997, 28, 116–138. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Moustafa, M.S.; Al-Mousawi, S.M.; Hilmy, N.M.; Ibrahim, Y.A.; Liermann, J.C.; Meier, H.; Elnagdi, M.H. Unexpected Behavior of Enaminones: Interesting New Routes to 1,6-Naphthyridines, 2-Oxopyrrolidines and Pyrano[4,3,2-de][1,6]naphthyridines. Molecules 2013, 18, 276-286. https://doi.org/10.3390/molecules18010276
Moustafa MS, Al-Mousawi SM, Hilmy NM, Ibrahim YA, Liermann JC, Meier H, Elnagdi MH. Unexpected Behavior of Enaminones: Interesting New Routes to 1,6-Naphthyridines, 2-Oxopyrrolidines and Pyrano[4,3,2-de][1,6]naphthyridines. Molecules. 2013; 18(1):276-286. https://doi.org/10.3390/molecules18010276
Chicago/Turabian StyleMoustafa, Moustafa Sherief, Saleh Mohammed Al-Mousawi, Noha Mohamed Hilmy, Yehia A. Ibrahim, Johannes C. Liermann, Herbert Meier, and Mohamed Hilmy Elnagdi. 2013. "Unexpected Behavior of Enaminones: Interesting New Routes to 1,6-Naphthyridines, 2-Oxopyrrolidines and Pyrano[4,3,2-de][1,6]naphthyridines" Molecules 18, no. 1: 276-286. https://doi.org/10.3390/molecules18010276
APA StyleMoustafa, M. S., Al-Mousawi, S. M., Hilmy, N. M., Ibrahim, Y. A., Liermann, J. C., Meier, H., & Elnagdi, M. H. (2013). Unexpected Behavior of Enaminones: Interesting New Routes to 1,6-Naphthyridines, 2-Oxopyrrolidines and Pyrano[4,3,2-de][1,6]naphthyridines. Molecules, 18(1), 276-286. https://doi.org/10.3390/molecules18010276



