Improvement of the Enzyme Performance of Trypsin via Adsorption in Mesoporous Silica SBA-15: Hydrolysis of BAPNA
Abstract
:1. Introduction
2. Results and Discussion
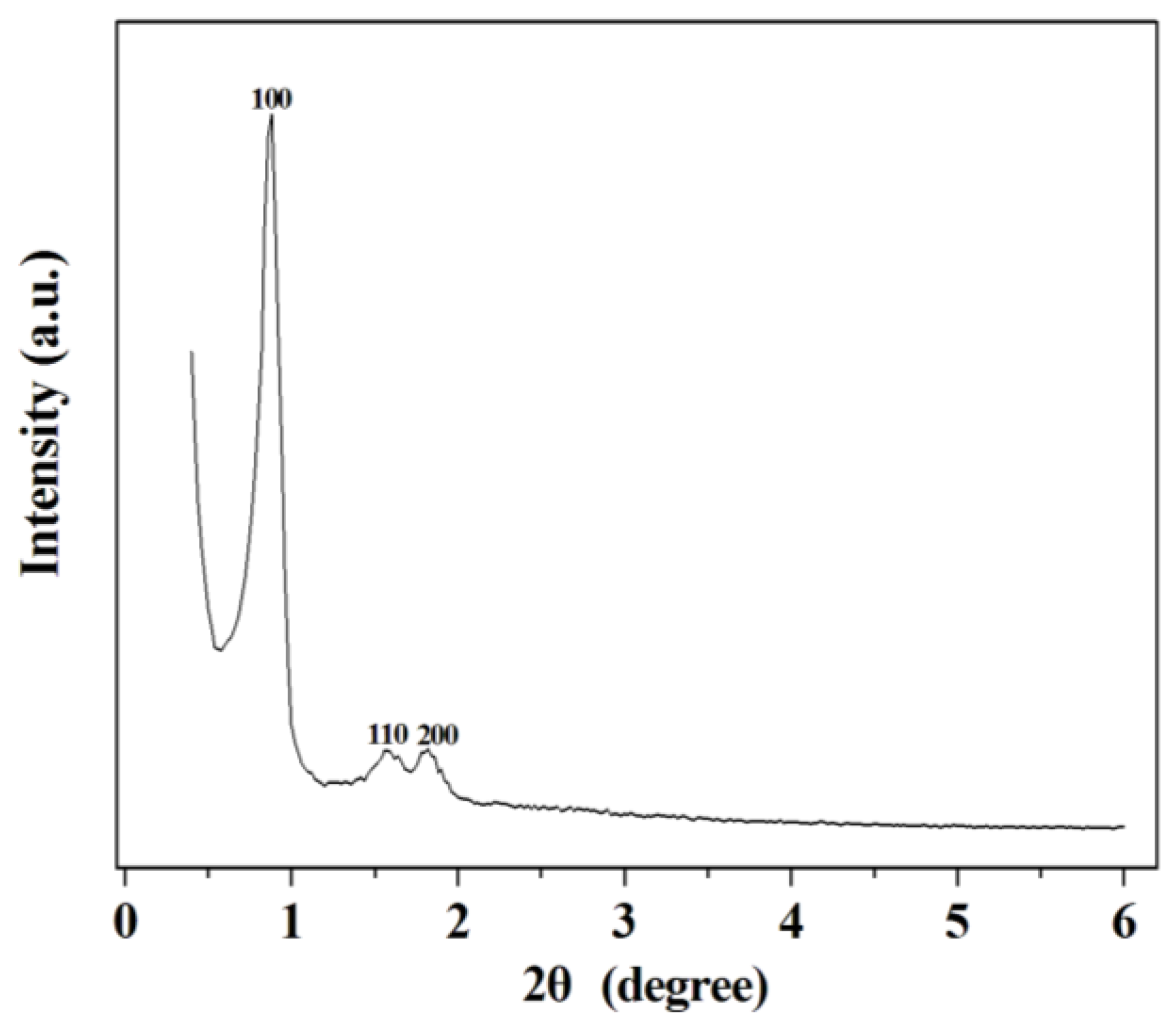
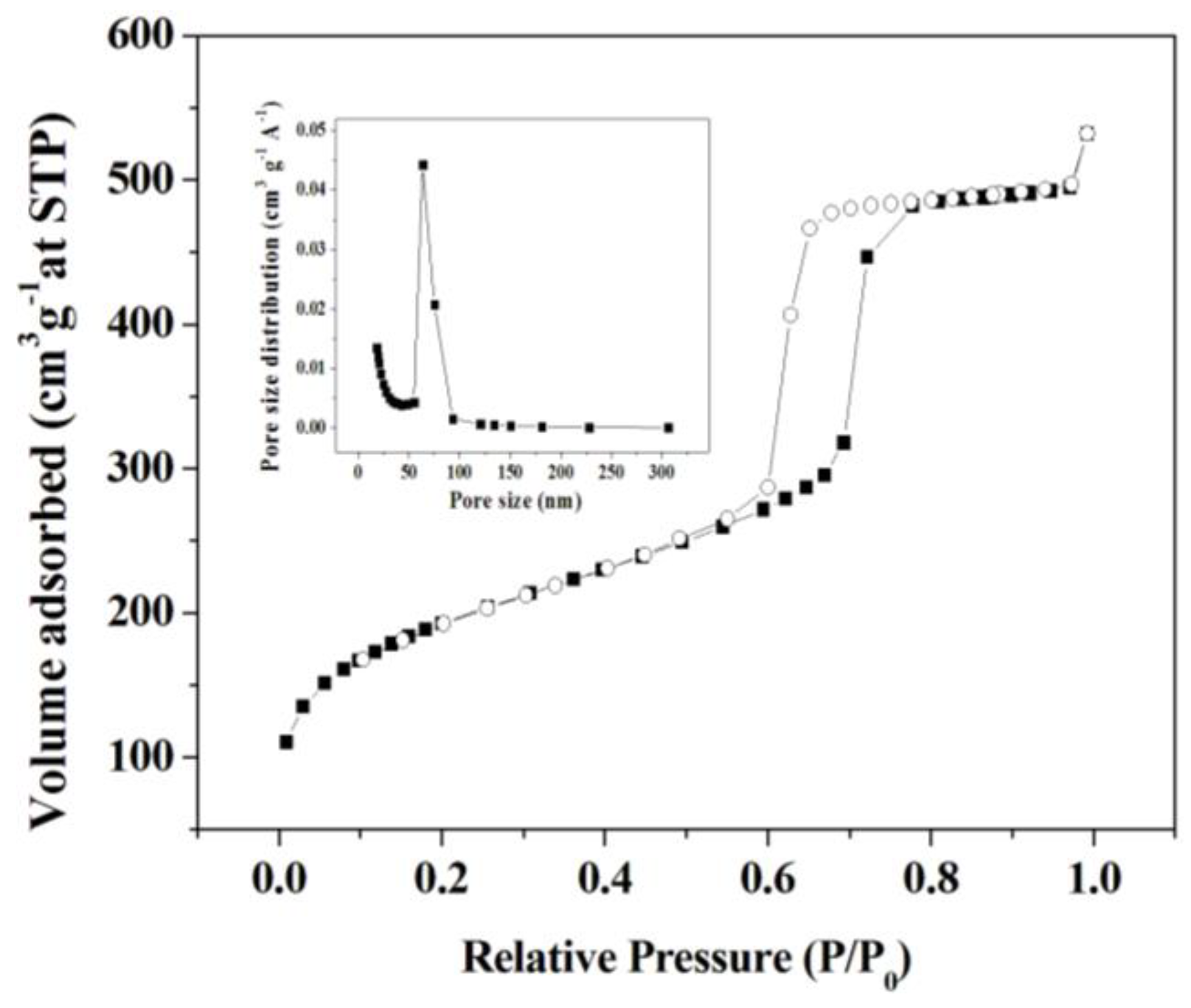
2.1. Synthesis and Characteristic of SBA-15 Mesoporous Silica
2.2. Influence of Immobilization Conditions on Trypsin Activity
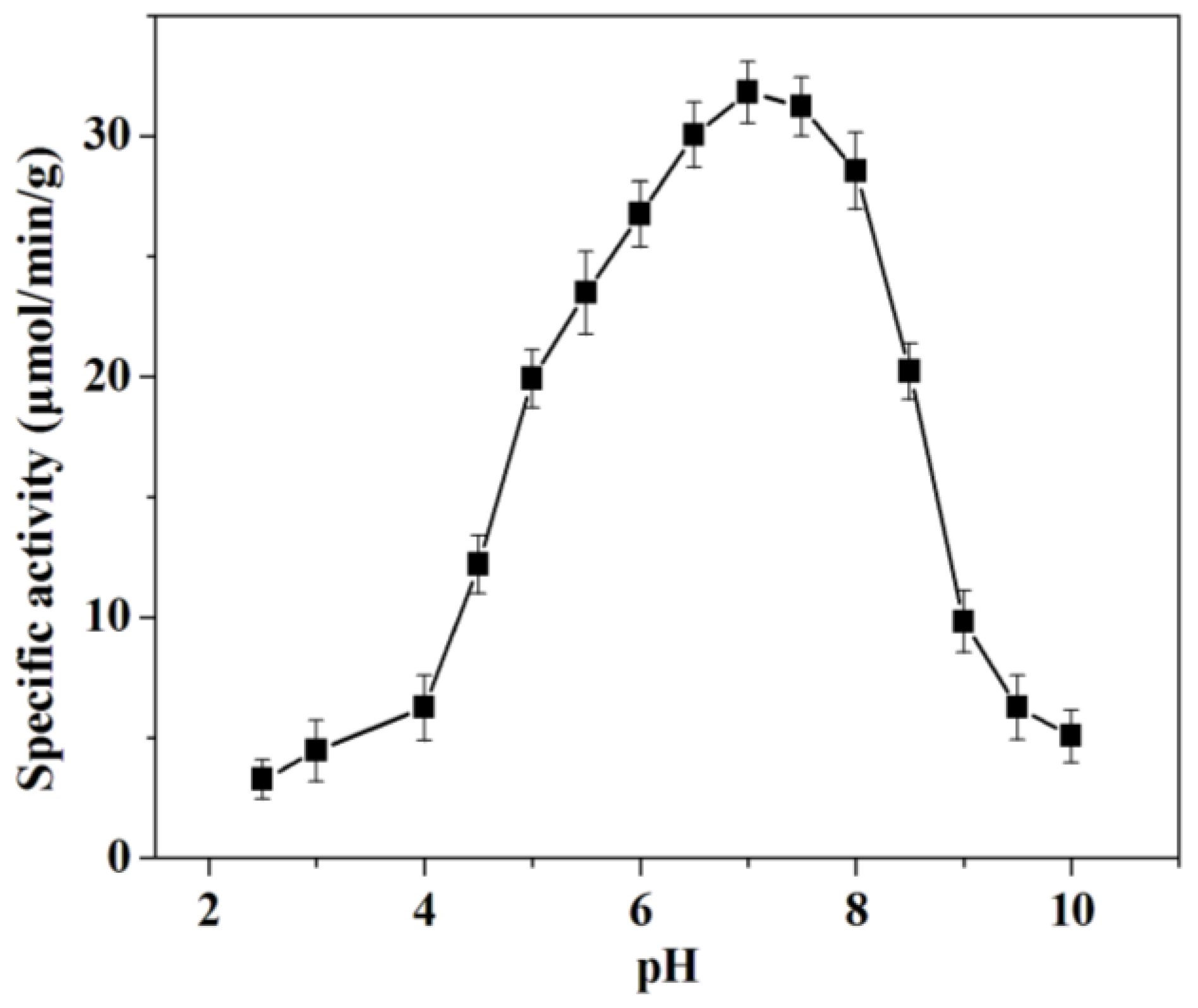
2.2.1. Effect of Immobilization pH
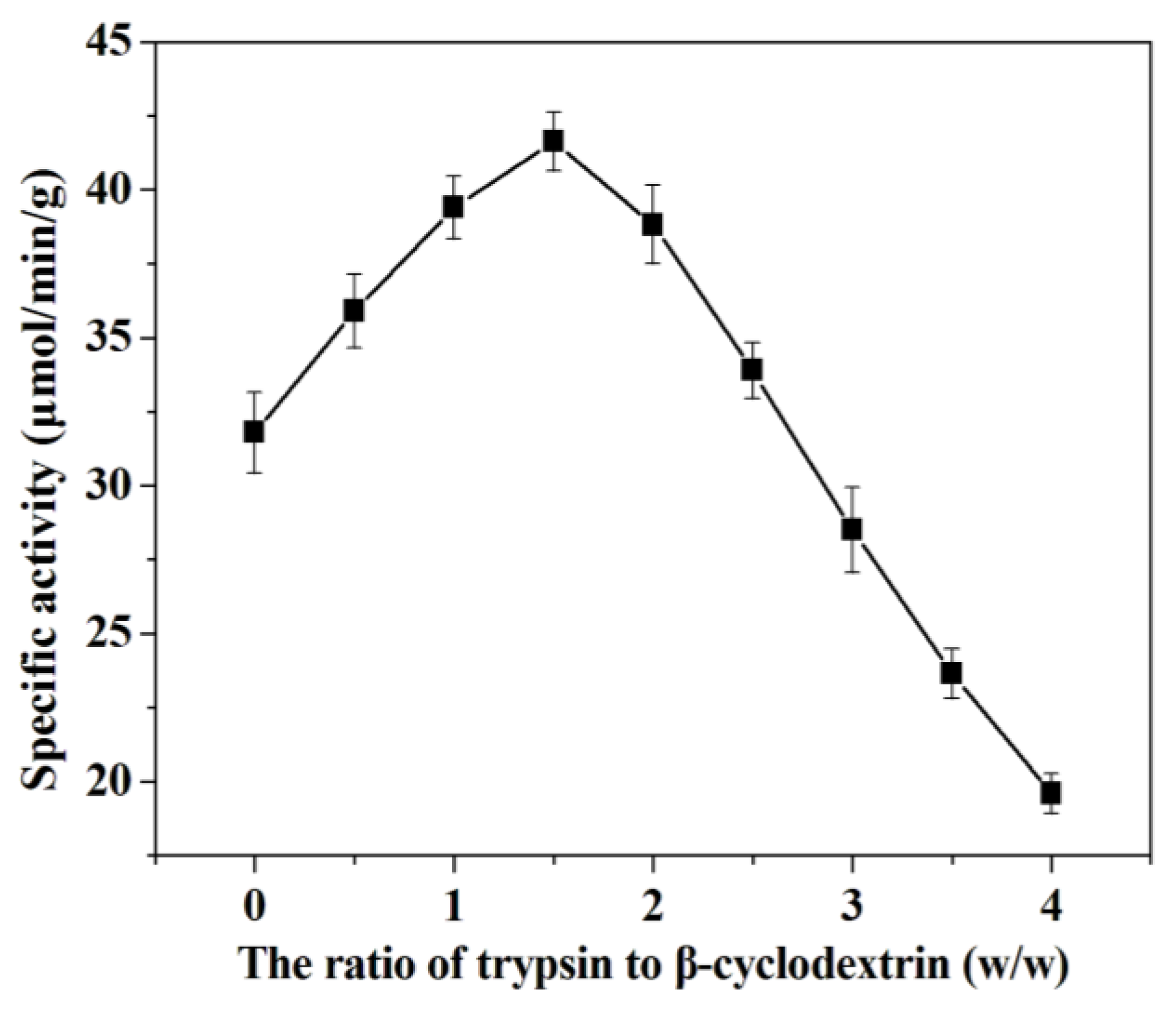
2.2.2. Effect of the Ratio of Trypsin to β-Cyclodextrin
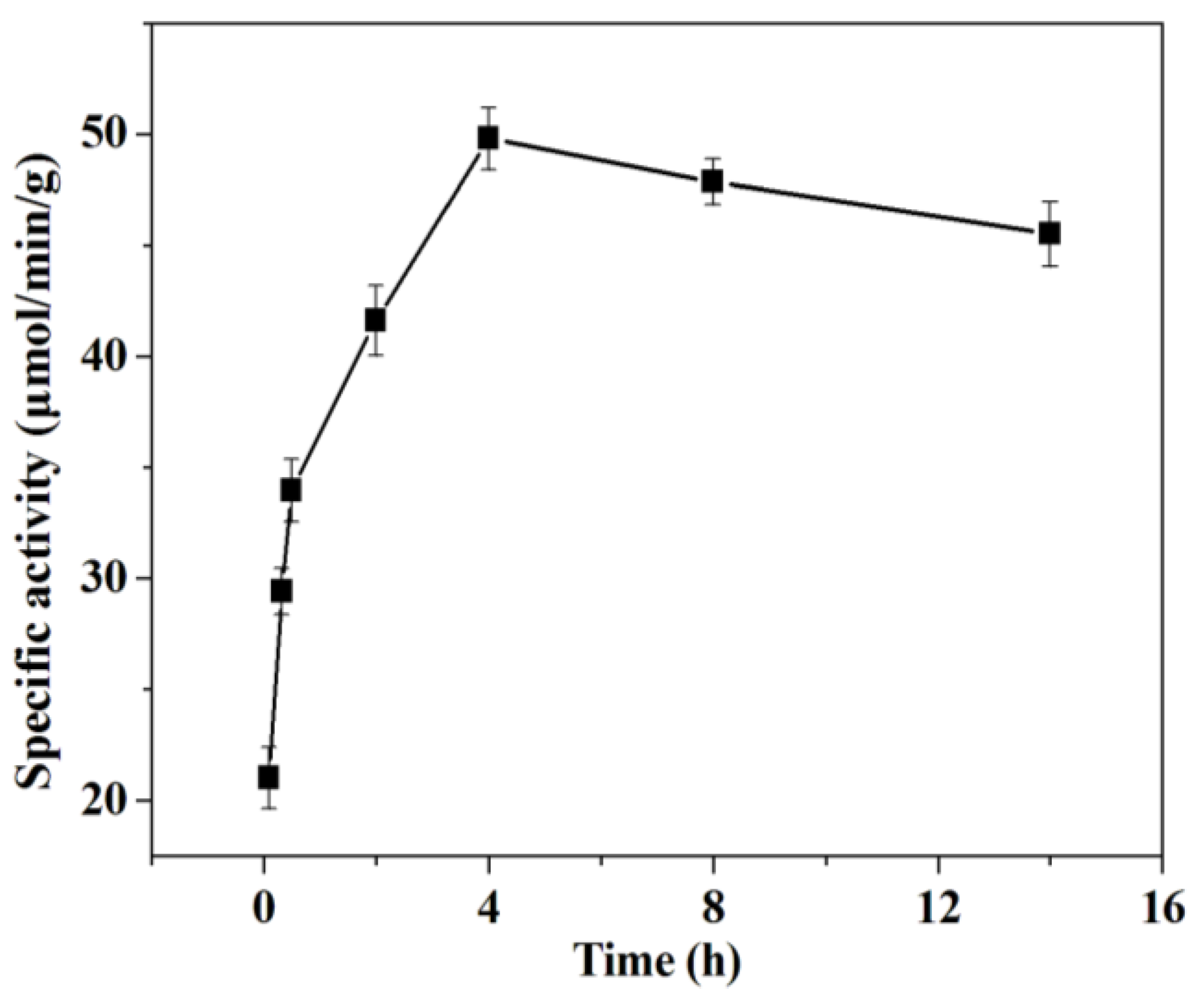
2.2.3. Effect of Immobilization Time
2.3. Enzyme Performance of the Immobilized Trypsin
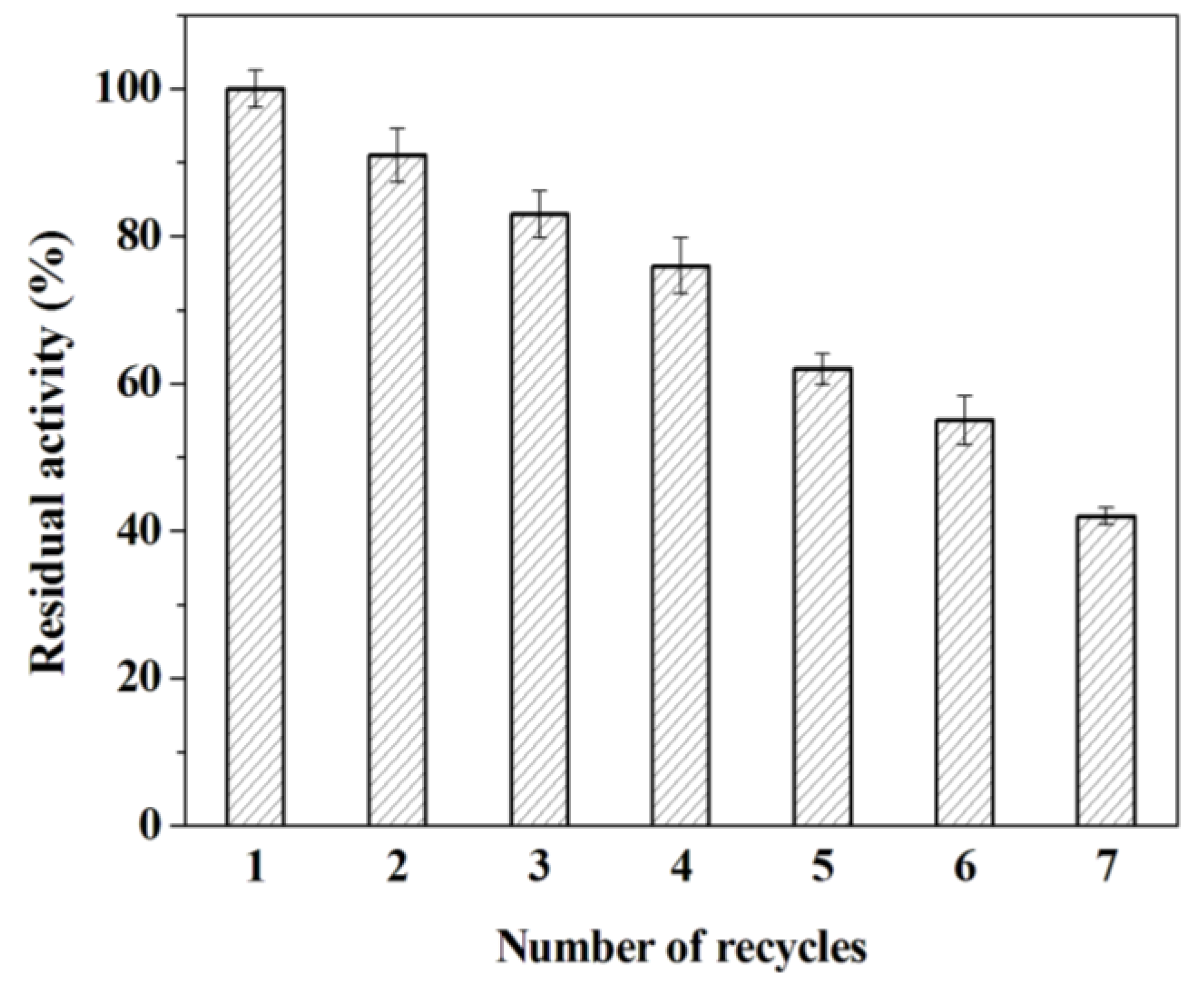
2.3.1. Reusability
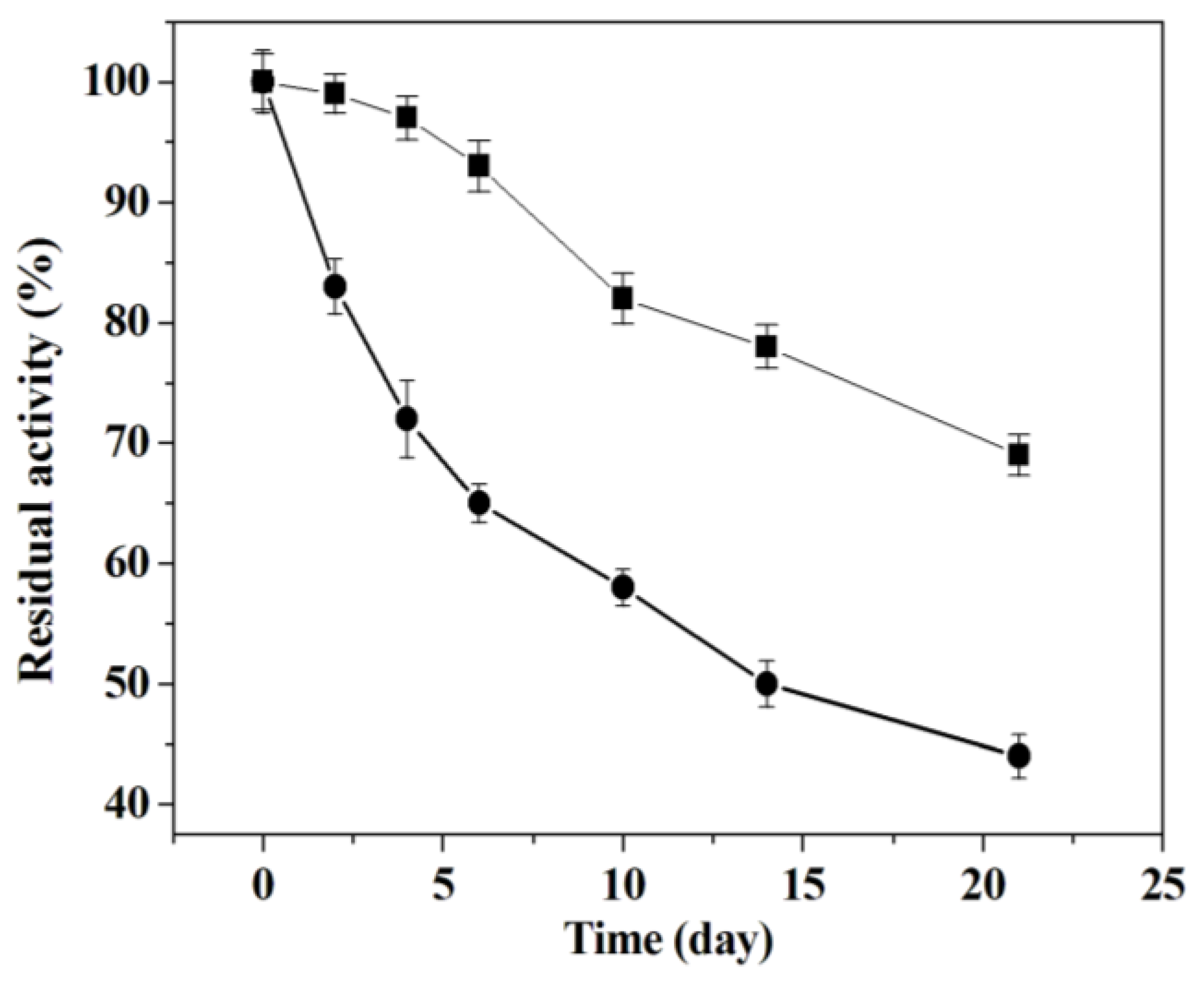
2.3.2. Storage Stability
3. Experimental
3.1. Materials
3.2. The Synthesis of Mesoporous Molecular Sieve
3.3. Preparation the Immobilization Solution
3.4. The Preparation of Adsorbed Trypsin
3.5. Optimization of the Immobilization Process
3.5.1. Optimization of Immobilization pH
3.5.2. Optimization of the Ratio of Trypsin to β-Cyclodextrin
3.5.3. Optimization of Immobilization Time
3.6. Enzyme Activity Assay
3.7. Reusability
3.8. Assessment of Storage Stability
3.9. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Cao, L.; Langen, L.V.; Sheldon, R.A. Immobilised enzymes: Carrier-bound or carrier-free? Curr. Opin. Chem. Biol. 2003, 14, 387–394. [Google Scholar] [CrossRef]
- Fernández-Lafuente, R.; Rodrı́guez, V.; Mateo, C.; Penzol, G.; Hernández-Justiz, O.; Irazoqui, G.; Villarino, A.; Ovsejevi, K.; Batista, F.; Guisán, J.M. Stabilization of multimeric enzymes via immobilization and post-immobilization techniques. J. Mol. Catal. B Enzym. 1999, 7, 181–189. [Google Scholar] [CrossRef]
- Bernal, C.; Sierra, L.; Mesa, M. Improvement of thermal stability of β-galactosidase from Bacillus circulans by multipoint covalent immobilization in hierarchical macro-mesoporous silica. J. Mol. Catal. B Enzym. 2012, 84, 166–172. [Google Scholar] [CrossRef]
- Wu, Z.; Dong, M.; Lu, M.; Li, Z. Encapsulation of β-galactosidase from Aspergillus oryzae based on “fish-in-net” approach with molecular imprinting technique. J. Mol. Catal. B Enzym. 2010, 63, 75–80. [Google Scholar] [CrossRef]
- Masuda, Y.; Kugimiya, S.-I.; Murai, K.; Hayashi, A.; Kato, K. Enhancement of activity and stability of the formaldehyde dehydrogenase by immobilizing onto phenyl-functionalized mesoporous silica. Colloid. Surfaces B 2013, 101, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Huo, Q.; Feng, J.; Chmelka, B.F.; Stucky, G.D. Nonionic Triblock and Star Diblock Copolymer and Oligomeric Surfactant Syntheses of Highly Ordered, Hydrothermally Stable, Mesoporous Silica Structures. J. Am. Chem. Soc. 1998, 120, 6024–6036. [Google Scholar] [CrossRef]
- Jang, S.; Kim, D.; Choi, J.; Row, K.; Ahn, W. Trypsin immobilization on mesoporous silica with or without thiol functionalization. J. Porous Mater. 2006, 13, 385–391. [Google Scholar] [CrossRef]
- Yang, X.Y.; Li, Z.Q.; Liu, B.; Klein-Hofmann, A.; Tian, G.; Feng, Y.F.; Ding, Y.; Su, D.S.; Xiao, F.S. “Fish-in-Net” Encapsulation of Enzymes in Macroporous Cages for Stable, Reusable, and Active Heterogeneous Biocatalysts. Adv. Mater. 2006, 18, 410–414. [Google Scholar] [CrossRef]
- Liu, J.; Guan, J.; Lu, M.; Kan, Q.; Li, Z. Hemoglobin immobilized with modified “fish-in-net” approach for the catalytic removal of aniline. J. Hazard. Mater. 2012, 217–218, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Chouyyok, W.; Panpranot, J.; Thanachayanant, C.; Prichanont, S. Effects of pH and pore characters of mesoporous silicas on horseradish peroxidase immobilization. J. Mol. Catal. B Enzym. 2009, 56, 246–252. [Google Scholar] [CrossRef]
- Díaz, J.F.; Balkus, K.J., Jr. Enzyme immobilization in MCM-41 molecular sieve. J. Mol. Catal. B Enzym. 1996, 2, 115–126. [Google Scholar] [CrossRef]
- Lu, S.; An, Z.; Li, J.; He, J. pH-Triggered Adsorption-Desorption of Enzyme in Mesoporous Host to Act on Macrosubstrate. J. Phys. Chem. B 2011, 115, 13695–13700. [Google Scholar] [CrossRef] [PubMed]
- Yasutaka, K.; Takato, Y.; Takashi, K.; Kohsuke, M.; Hiromi, Y. Enhancement in Adsorption and Catalytic Activity of Enzymes Immobilized on Phosphorus- and Calcium-Modified MCM-41. J. Phys. Chem. B 2011, 115, 10335–10345. [Google Scholar] [CrossRef] [PubMed]
- Shah, P.; Sridevi, N.; Prabhune, A.; Ramaswamy, V. Structural features of Penicillin acylase adsorption on APTES functionalized SBA-15. Micropor. Mexopor. Mat. 2008, 116, 157–165. [Google Scholar] [CrossRef]
- Ding, Y.; Yin, G.; Liao, X.; Huang, Z.; Chen, X.; Yao, Y.; Li, J. A convenient route to synthesize SBA-15 rods with tunable pore length for lysozyme adsorption. Micropor. Mesopor. Mat. 2013, 170, 45–51. [Google Scholar] [CrossRef]
- Salis, A.; Meloni, D.; Ligas, S.; Casula, M.F.; Monduzzi, M.; Solinas, V.; Dumitriu, E. Physical and Chemical Adsorption of Mucor javanicus Lipase on SBA-15 Mesoporous Silica. Synthesis, Structural Characterization, and Activity Performance. Langmuir 2005, 21, 5511–5516. [Google Scholar] [CrossRef] [PubMed]
- Steri, D.; Monduzzi, M.; Salis, A. Ionic Strength Affects Lysozyme Adsorption and Release from SBA-15 Mesoporous Silica. Micropor. Mesopor. Mat. 2013, 170, 164–172. [Google Scholar] [CrossRef]
- Vinoba, M.; Kim, D.H.; Lim, K.S.; Jeong, S.K.; Lee, S.W.; Alagar, M. Biomimetic Sequestration of CO2 and Reformation to CaCO3 Using Bovine Carbonic Anhydrase Immobilized on SBA-15. Energ. Fuel. 2010, 25, 438–445. [Google Scholar] [CrossRef]
- Chen, S.-Y.; Chen, Y.-T.; Lee, J.-J.; Cheng, S. Tuning pore diameter of platelet SBA-15 materials with short mesochannels for enzyme adsorption. J. Mater. Chem. 2011, 21, 5693–5703. [Google Scholar] [CrossRef]
- Bai, W.; Yang, Y.-J.; Tao, X.; Chen, J.-F.; Tan, T.-W. Immobilization of lipase on aminopropyl-grafted mesoporous silica nanotubes for the resolution of (R, S)-1-phenylethanol. J. Mol. Catal. B Enzym. 2012, 76, 82–88. [Google Scholar] [CrossRef]
- Manyar, H.G.; Gianotti, E.; Sakamoto, Y.; Terasaki, O.; Coluccia, S.; Tumbiolo, S. Active Biocatalysts Based on Pepsin Immobilized in Mesoporous SBA-15. J. Phys. Chem. C. 2008, 112, 18110–18116. [Google Scholar] [CrossRef]
- Yiu, H.H.P.; Wright, P.A.; Botting, N.P. Enzyme immobilisation using SBA-15 mesoporous molecular sieves with functionalised surfaces. J. Mol. Catal. B Enzym. 2001, 15, 81–92. [Google Scholar] [CrossRef]
- Fernández, M.; Villalonga, M.L.; Caballero, J.; Fragoso, A.; Cao, R.; Villalonga, R. Effects of β-cyclodextrin-dextran polymer on stability properties of trypsin. Biotechnol. Bioeng. 2003, 83, 743–747. [Google Scholar] [CrossRef] [PubMed]
- Fernández, M.; Fragoso, A.; Cao, R.; Baños, M.; Villalonga, R. Chemical conjugation of trypsin with monoamine derivatives of cyclodextrins: Catalytic and stability properties. Enzyme Microb. Technol. 2002, 31, 543–548. [Google Scholar] [CrossRef]
- Fernández, M.; Fragoso, A.; Cao, R.; Baños, M.; Ansorge-Schumacher, M.; Hartmeier, W.; Villalonga, R. Functional properties and application in peptide synthesis of trypsin modified with cyclodextrin-containing dicarboxylic acids. J. Mol. Catal. B Enzym. 2004, 31, 47–52. [Google Scholar] [CrossRef]
- Zhao, D.; Feng, J.; Huo, Q.; Melosh, N.; Fredrickson, G.H.; Chmelka, B.F.; Stucky, G.D. Triblock copolymer syntheses of mesoporous silica with periodic 50 to 300 angstrom pores. Science 1998, 279, 548–552. [Google Scholar] [CrossRef] [PubMed]
- Tompsett, G.; Krogh, L.; Griffin, D.; Conner, W. Hysteresis and scanning behavior of mesoporous molecular sieves. Langmuir 2005, 21, 8214–8225. [Google Scholar] [CrossRef] [PubMed]
- Goradia, D.; Cooney, J.; Hodnett, B.K.; Magner, E. The adsorption characteristics, activity and stability of trypsin onto mesoporous silicates. J. Mol. Catal. B Enzym. 2005, 32, 231–239. [Google Scholar] [CrossRef]
- Kadoya, S.; Fujii, K.; Izutsu, K.-I.; Yonemochi, E.; Terada, K.; Yomota, C.; Kawanishi, T. Freeze-drying of proteins with glass-forming oligosaccharide-derived sugar alcohols. Int. J. Pharm. 2010, 389, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Santagapita, P.R.; Brizuela, L.G.; Mazzobre, M.F.; Ramirez, H.L.; Corti, H.R.; Santana, R.V.; Buera, M.P. Structure/Function Relationships of Several Biopolymers as Related to Invertase Stability in Dehydrated Systems. Biomacromolecules 2008, 9, 741–747. [Google Scholar] [CrossRef] [PubMed]
- Santagapita, P.R.; Brizuela, L.G.; Mazzobre, M.F.; Ramírez, H.L.; Corti, H.R.; Santana, R.V.; Buera, M.P. β-Cyclodextrin modifications as related to enzyme stability in dehydrated systems: Supramolecular transitions and molecular interactions. Carbohyd. Polym. 2011, 83, 203–209. [Google Scholar] [CrossRef]
- Li, Y.; Zhou, G.; Li, C.; Qin, D.; Qiao, W.; Chu, B. Adsorption and catalytic activity of Porcine pancreatic lipase on rod-like SBA-15 mesoporous material. Colloids Surf. A Physicochem. Eng. Asp. 2009, 341, 79–85. [Google Scholar] [CrossRef]
- Lei, C.; Soares, T.A.; Shin, Y.; Liu, J.; Ackerman, E.J. Enzyme specific activity in functionalized nanoporous supports. Nanotechnology 2008, 19, 125102–125111. [Google Scholar] [CrossRef] [PubMed]
- Schlossbauer, A.; Schaffert, D.; Kecht, J.; Wagner, E.; Bein, T. Click Chemistry for High-Density Biofunctionalization of Mesoporous Silica. J. Am. Chem. Soc. 2008, 130, 12558–12559. [Google Scholar] [CrossRef] [PubMed]
- Kannan, K.; Jasra, R.V. Immobilization of alkaline serine endopeptidase from Bacillus licheniformis on SBA-15 and MCF by surface covalent binding. J. Mol. Catal. B Enzym. 2009, 56, 34–40. [Google Scholar] [CrossRef]
Sample Availability: Samples of the immobilized trypsin are available from the authors. |
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, S.; Wu, Z.; Lu, M.; Wang, Z.; Li, Z. Improvement of the Enzyme Performance of Trypsin via Adsorption in Mesoporous Silica SBA-15: Hydrolysis of BAPNA. Molecules 2013, 18, 1138-1149. https://doi.org/10.3390/molecules18011138
Li S, Wu Z, Lu M, Wang Z, Li Z. Improvement of the Enzyme Performance of Trypsin via Adsorption in Mesoporous Silica SBA-15: Hydrolysis of BAPNA. Molecules. 2013; 18(1):1138-1149. https://doi.org/10.3390/molecules18011138
Chicago/Turabian StyleLi, Shanshan, Zhuofu Wu, Ming Lu, Zhi Wang, and Zhengqiang Li. 2013. "Improvement of the Enzyme Performance of Trypsin via Adsorption in Mesoporous Silica SBA-15: Hydrolysis of BAPNA" Molecules 18, no. 1: 1138-1149. https://doi.org/10.3390/molecules18011138
APA StyleLi, S., Wu, Z., Lu, M., Wang, Z., & Li, Z. (2013). Improvement of the Enzyme Performance of Trypsin via Adsorption in Mesoporous Silica SBA-15: Hydrolysis of BAPNA. Molecules, 18(1), 1138-1149. https://doi.org/10.3390/molecules18011138



