Effects of Sorghum [Sorghum bicolor (L.) Moench] Crude Extracts on Starch Digestibility, Estimated Glycemic Index (EGI), and Resistant Starch (RS) Contents of Porridges
Abstract
:1. Introduction
2. Results and Discussion
| Sorghum Brans | Total phenols (mg/g gallic acid eq.) | Tannins (mg catechin eq./g) | Anthocyanins (mg/g) |
|---|---|---|---|
| White corn | 2.8 ± 0.1 f,1 | 0.0 ± 0.0 d | 0.0 ± 0.0 c |
| White corn residue | 0.1 ± 0.0 g | 0.0 ± 0.0 d | 0.0 ± 0.0 c |
| White bran | 4.3 ± 0.7 e | 0.0 ± 0.0 d | 0.0 ± 0.0 c |
| White bran residue | 0.0 ± 0.0 g | 0.0 ± 0.0 d | 0.0 ± 0.0 c |
| Black bran | 15.0 ± 1.2 c | 2.0 ± 0.4 c | 4.2 ± 1.2 a |
| Black bran residue | 4.6 ± 0.8 e | 0.0 ± 0.0 d | 0.0 ± 0.0 c |
| Black with tannin bran | 24.0 ± 2.4 b | 24.0 ± 4.3 b | 2.0 ± 0.1 b |
| Black with tannin bran residue | 4.6 ± 0.4 e | 1.8 ± 0.3 c | 0.0 ± 0.0 c |
| Tannin bran | 40.3 ± 3.2 a | 75.0 ± 4.8 a | 0.0 ± 0.0 c |
| Tannin bran residue | 7.2 ± 1.0 d | 2.6 ± 0.7 c | 0.0 ± 0.0 c |
2.1. The Effects of 70% Aqueous Acetone Extracts from Sorghum Bran on Starch Digestibility of Corn Starch Porridges
| Porridges | Peak Viscosity (RVU) | Trough (RVU) | Breakdown (RVU) | Final Viscosity (RVU) | Setback (RVU) | Peak time (min) | Pasting Temp. (°C) | Peak Temp. (°C) |
|---|---|---|---|---|---|---|---|---|
| Corn starch | 451 ± 30 a,1 | 154 ± 20 c | 297 ± 14 a | 468 ± 29 a | 314 ± 25 a | 4.6 ± 2.1 a | 73.9 ± 3.5 a | 94.8 ± 5.6 a |
| Corn starch + white bran extract | 426 ± 39 a | 136 ± 15 d | 290 ± 15 a | 405 ± 24 b | 269 ± 48 b | 4.7 ± 0.9 a | 75.2 ± 4.3 a | 94.5 ± 4.4 a |
| Corn starch + tannin bran extract | 401 ± 36 a | 210 ± 13 a | 191 ± 12 c | 353 ± 17 c | 142 ± 18 d | 4.9 ± 1.9 a | 74.2 ± 4.0 a | 93.3 ± 4.3 a |
| Corn starch + black with tannin bran extract | 412 ± 25 a | 175 ± 15 b | 237 ± 18 b | 365 ± 40 c | 159 ± 25 cd | 5.0 ± 2.0 a | 75.4 ± 1.9 a | 93.6 ± 7.2 a |
| Corn starch + black bran extract | 416 ± 26 a | 167 ± 14 b | 249 ± 33 b,l | 334 ± 21 c | 198 ± 48 c | 4.8 ± 1.2 a | 74.1 ± 2.1 a | 94.9 ± 4.6 a |
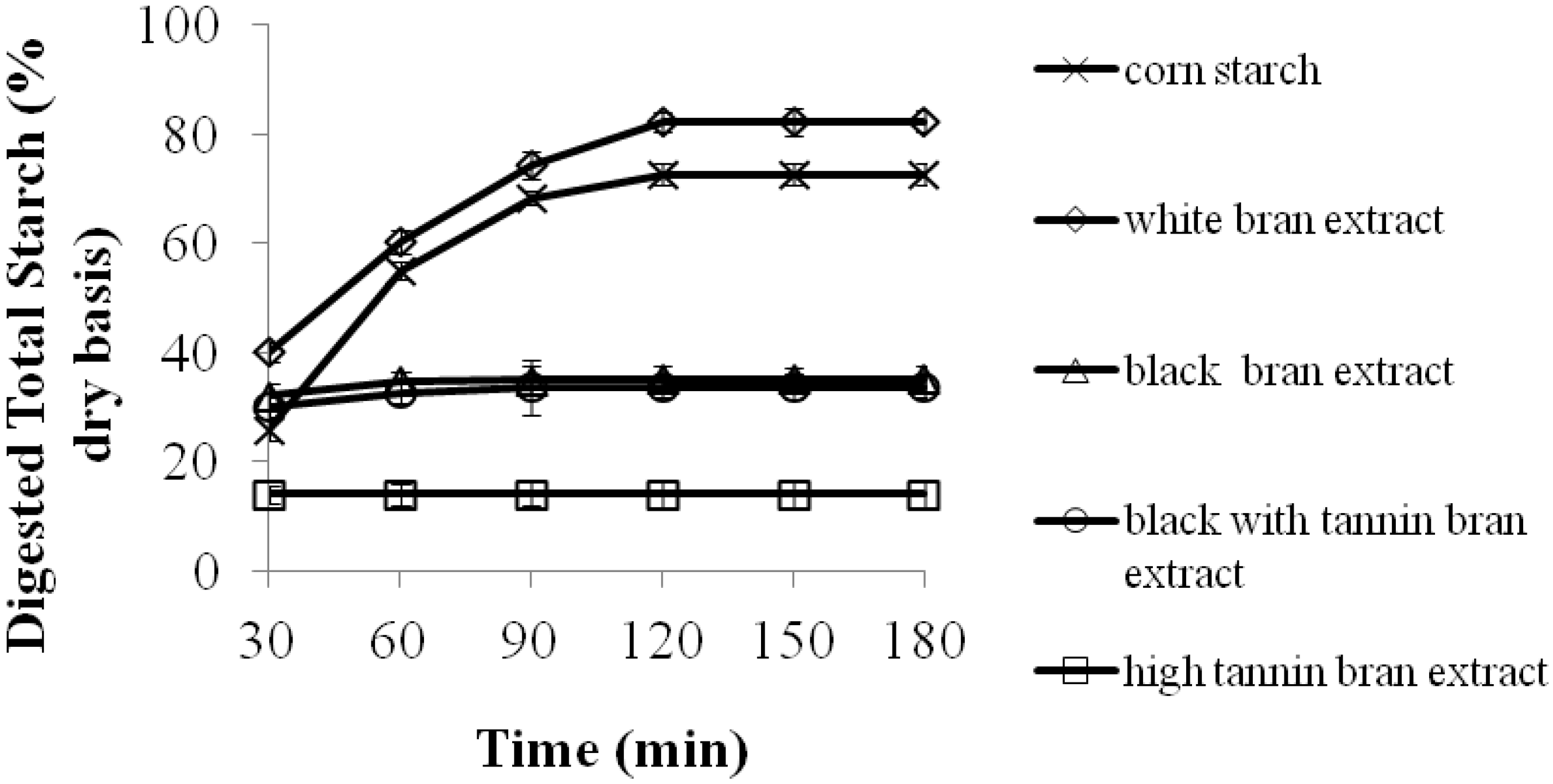
| Porridges | Estimated Glycemic Index (EGI) | Resistant Starch (% dry weight basis) |
|---|---|---|
| Corn starch | 90.0 ± 5.6 b,1 | 0.0 ± 0.0 c |
| Corn starch + white bran extract | 98.0 ± 4.2 a | 0.2 ± 0.0 c |
| Corn starch + high tannin bran extract | 49.0 ± 2.4 d | 14.9 ± 0.9 a |
| Corn starch + black bran extract | 67.0 ± 3.6 c | 9.1 ± 0.6 b |
| Corn starch + black with tannin bran extract | 65.0 ± 6.3 c | 10.6 ± 1.0 b |
2.2. The Effects of 70% Aqueous Acetone Extracts from Sorghum Brans on Digestibility of High Amylose Corn Starch Porridges
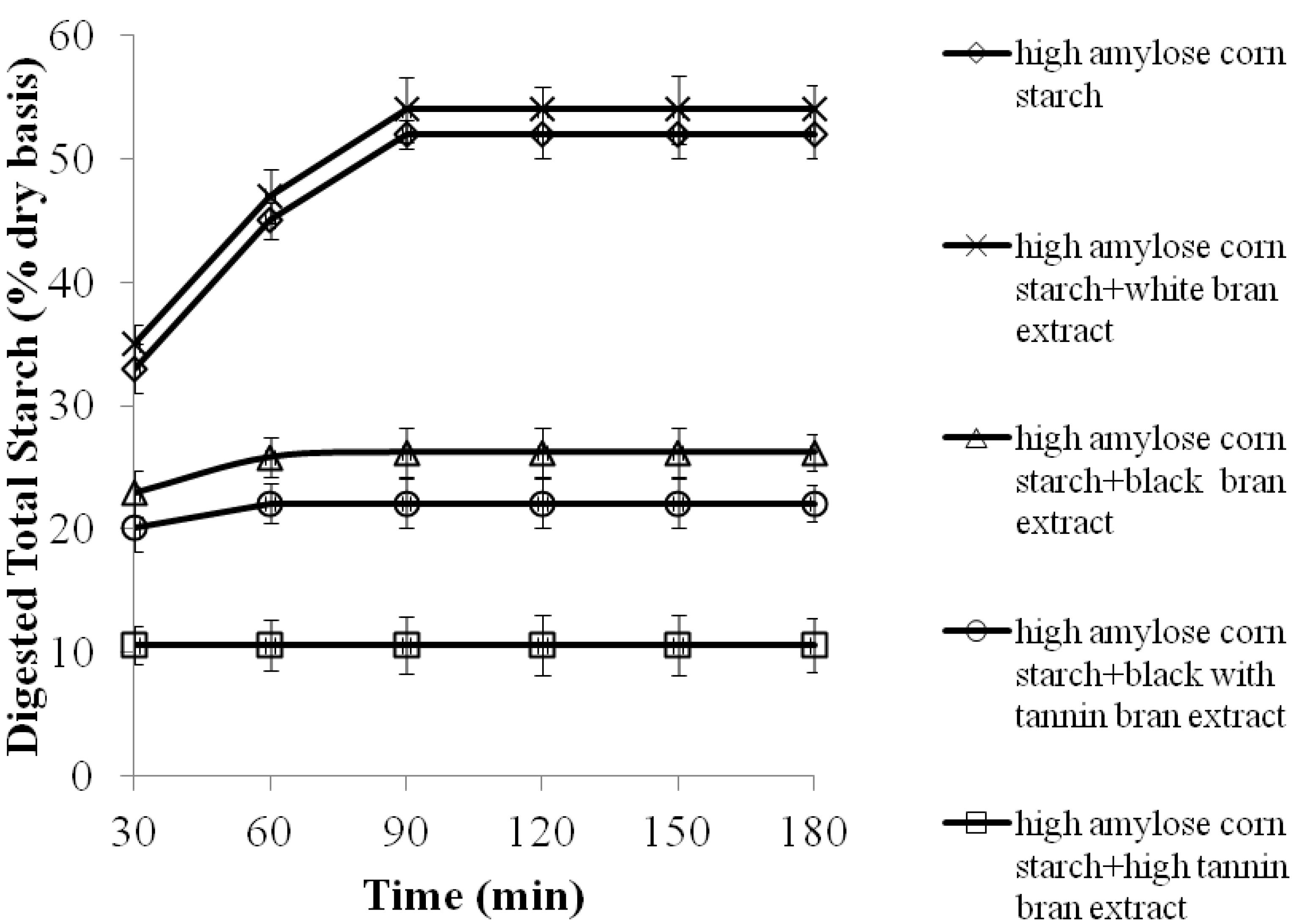
| Porridges | Estimated Glycemic Index (EGI) | Resistant Starch (% dry weight basis) |
|---|---|---|
| High amylose corn starch | 70.0± 3.0 a,1 | 47.0± 2.0 c |
| High amylose corn starch + white bran extract | 70.0± 3.0 a | 46.0± 1.0 c |
| High amylose corn starch + high tannin bran extract | 48.0± 5.0 c | 58.0± 2.0 a |
| High amylose corn starch + black bran extract | 60.0± 3.0 b | 51.0± 4.0 b |
| High amylose corn starch + black with tannin bran extract | 60.0± 2.0 b | 53.0± 4.0 b |
2.3. The Effects of 70% Aqueous Acetone Extracts from Sorghum Brans on Starch Digestibility of Whole Sorghum Flour Porridges
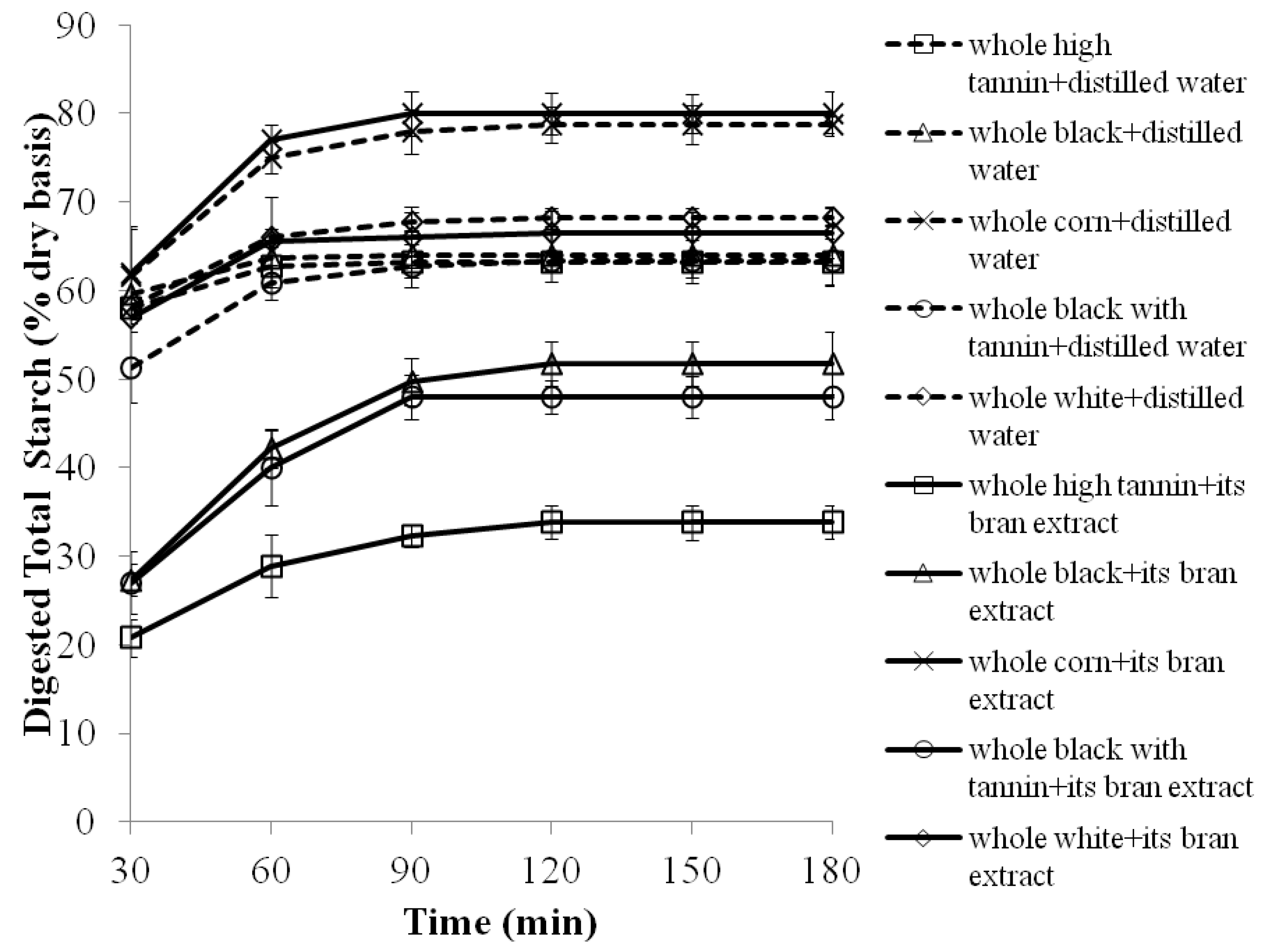
| Porridges | EGI | RS (% dry weight basis) |
|---|---|---|
| Whole white corn cooked with water | 100.0 ± 3.3 a,1 | 0.1 ± 0.0 e |
| Whole white corn cooked with its bran extract | 98.0 ± 2.1 a | 0.0 ± 0.0 e |
| Whole white sorghum cooked with water | 92.0 ± 4.3 b | 0.2 ± 0.0 e |
| Whole white sorghum cooked with its bran extract | 91.0 ± 3.0 b | 0.3 ± 0.0 e |
| Whole high tannin sorghum cooked with water | 85.0 ± 4.5 c | 2.2 ± 1.2 c |
| Whole high tannin sorghum cooked with its bran extract | 62.0 ± 4.1 e | 6.8 ± 0.3 a |
| Whole black sorghum cooked with water | 88.0 ± 5.0 c | 0.3 ± 0.0 e |
| Whole black sorghum cooked with its bran extract | 73.0 ± 4.0 d | 2.4 ± 0.5 c |
| Whole black with tannin sorghum cooked with water | 87.0 ± 5.0 c | 1.0 ±0.2 d |
| Whole black with tannin sorghum cooked with its bran extract | 72.0 ± 4.3 d | 5.3 ± 0.4 b |
2.4. Discussion
3. Experimental
3.1. Sorghum Varieties
3.2. Control Samples and High Amylose Corn Starch
3.3. Preparations of Whole Sorghum Flours and Brans
3.4. Preparation of Extracts from Sorghum Brans

3.5. Tannin and Phenol Determination
3.6. Preparation of Porridges
3.7. Total Starch, in Vitro Starch Digestibility, and Resistant Starch
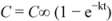
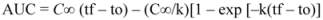
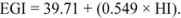
3.8. Statistical Analyses
4. Conclusions
Acknowledgments
References
- Flegal, K.M.; Carroll, M.D.; Ogden, C.L.; Curtin, L.R. Prevalence trends in obesity among US adults, 1999-2008. JAMA 2010, 303, 235–241. [Google Scholar] [CrossRef]
- O’dea, K.; Holm, J. Rate of starch hydrolysis in vitro as a predictor for metabolic responses to complex carbohydrate in vivo. Am. J. Clin. Nutr. 1985, 34, 1991–1993. [Google Scholar]
- Lund, E.K.; Johnson, I.J. Fermentable carbohydrate reaching the colon after ingestion of oats in humans. J. Nutr. 1991, 121, 311–317. [Google Scholar]
- Jarvi, B.E.K.; Granfeldt, Y.E.; Bjorck, I.E.; Asp, N.G.; Vessby, B.O. Improved glycemic control lipid profile normalized fibrinolytic activity on a low-glycemic index diet in type 2 diabetic patients. Diabetes Care 1999, 22, 10–18. [Google Scholar] [CrossRef]
- Jenkins, D.J.A.; Kendall, C.W.C.; McKeown-Eyssen, G.; Josse, G.R.; Silverberg, G.J.; Booth, G.L.; Vidgen, J.; Nguyen, E.A.R.; Corrigan, T.H.; Banach, M.S.; et al. Effect of a Low-Glycemic Index or a High-Cereal Fiber Diet on Type 2 Diabetes, A Randomized Trial. J. Am. Med. Assoc. 2008, 300, 2742–2753. [Google Scholar]
- Baghurst, P.A.; Baghurst, K.I.; Record, S.J. Dietary fiber, non-starch polysaccharides and resistant starch: A review. Food Aust. 1996, 48, 3–35. [Google Scholar]
- Hendrich, S. Battling obesity with resistant starch. Food Technol. 2010, 64, 22–30. [Google Scholar]
- Le Bourvellec, C.; Renard, C.M.G.C. Interactions between Polyphenols and Macromolecules: Quantification Methods and Mechanisms. Crit. Rev. Food Sci. Nutr. 2012, 52, 213–248. [Google Scholar] [CrossRef]
- Davis, A.B.; Hoseney, R.C. Grain sorghum condensed tannins. I. Isolation; estimation; selective adsorption by starch. Cereal Chem. 1979, 56, 310–314. [Google Scholar]
- Rahman, M.D.; Richards, G.N. Interactions of starch and other polysaccharides with condensed tannins in hot water extracts of ponderosa pine bark. J. Wood Chem. Technol. 1988, 8, 111–120. [Google Scholar] [CrossRef]
- Belitz, H.D; Grosch, W. Food Chemistry, 2nd ed; Springer: Berlin, Germany, 1999. [Google Scholar]
- Dlamini, N.R. Effect of sorghum type and processing on the antioxidant properties of sorghum [Sorghum bicolor (L.) Moench] based foods. Ph.D. Thesis, Texas A&M University, College Station, TX, USA, 2007. [Google Scholar]
- Le Bourvellec, C.; Renard, C.M.G.C. Non-covalent interaction between procyanidins and cell wall material. Part II: Quantification and impact of the cell wall drying. Biochim. Biophys. Acta Gen. Subj. 2005, 1725, 1–9. [Google Scholar]
- Asquith, T.N.; Butler, L.G. Interactions of condensed tannins with selected proteins. Phytochemistry 1986, 25, 1591–1593. [Google Scholar]
- Vonk, R.J.; Hagedoorn, R.E.; de Graaff, R.; Elzinga, H.; Tabak, S.; Yang, Y.X.; Stellaard, F. Digestion of so called resistant starch sources in the human small intestine. Am. J. Clin. Nutr. 2000, 72, 432–438. [Google Scholar]
- Waniska, R.D. Structure; phenolic compounds; antifungal proteins of sorghum caryopses. In Proceedings of Technical Institutional Options for Sorghum Grain Mold Management: Proceedings of an International Consultation, ICRISAT, Patancheru, India, 18–19 May 2000; pp. 72–106.
- Dykes, L.; Rooney, L.W. Review. Sorghum millet phenols antioxidants. J. Cereal Sci. 2006, 44, 236–251. [Google Scholar] [CrossRef]
- Daiber, K.H. Enzyme inhibition by polyphenols of sorghum grain malt. J. Sci. Food Agric. 1975, 26, 1399–1411. [Google Scholar] [CrossRef]
- Beta, T.; Rooney, L.W.; Marovatsanga, L.T.; Taylor, J.R.N. Effect of chemical treatment on polyphenols malt quality in sorghum. J. Cereal Sci. 2000, 31, 295–302. [Google Scholar] [CrossRef]
- De Castro Palomino Siller, A. In Vitro Starch Digestibility Estimated Glycemic Index of Sorghum Products. M.Sc. Thesis, Texas A&M University, College Station, TX, USA, 2006. [Google Scholar]
- Jankowski, A.; Jankowski, B.; Niedworok, J. The influence of anthocyanin dye from grapes on experimental diabetes. Folica Med. Cracov. 2000, 41, 5–15. [Google Scholar]
- Tsuda, T. Regulation of adipocyte function by anthocyanins; possibility of preventing the metabolic syndrome. J. Agric. Food Chem. 2003, 56, 642–646. [Google Scholar] [CrossRef]
- Hargrove, J.L.; Greenspan, P.; Hartle, D.K.; Dowd, C. Inhibition of Aromatase and α-Amylase by flavonoids and proanthocyanidins from Sorghum bicolor bran extracts. J. Med. Food 2011, 14, 799–807. [Google Scholar] [CrossRef]
- Austin, D.L.; Turner, N.D.; McDonough, C.M.; Rooney, L.W. Effects of Brans from Specialty Sorghum Varieties on in Vitro Starch Digestibility of Soft and Hard Sorghum Endosperm Porridges. Cereal Chem. 2012, 89, 190–197. [Google Scholar] [CrossRef]
- Gularte, M.A.; Rosell, C.M. Physicochemical properties and enzymatic hydrolysis of different starches in the presence of hydrocolloids. Carbohydr. Polym. 2011, 85, 237–244. [Google Scholar] [CrossRef]
- Magaletta, R.L.; DiCataldo, S.N.; Dong, L.; Hong, L.L.; Borwankar, R.P.; Martini, M.C. In Vitro Method for Predicting Glycemic Index of Foods Using Simulated Digestion and an Artificial Neural Network. Cereal Chem. 2010, 87, 363–369. [Google Scholar] [CrossRef]
- Ferruzi, M.G.; Green, R.J. Analysis of catechins from milk-tea beverages by enzyme assisted extraction followed by high performance liquid chromatography. Food Chem. 2006, 99, 484–491. [Google Scholar] [CrossRef]
- Rhon, S.; Rawel, H.M.; Kroll, J. Inhibitory effects of plant phenols on the activity of selected enzymes. J. Agric. Food. Chem. 2002, 50, 3566–3571. [Google Scholar] [CrossRef]
- Behall, K.M.; Hallfrisch, J. Plasma glucose insulin reduction after consumption of breads varying in amylose content. Eur. J. Clin. Nutr. 2002, 56, 913–920. [Google Scholar] [CrossRef]
- Arts, M.J.; Haenen, G.R.; Wilms, L.C.; Beetstra, S.A.; Heijnen, C.G.; Voss, H.P.; Bast, A. Interactions between flavonoids proteins, effect on the total antioxidant capacity. J. Agric. Food Chem. 2002, 50, 1184–1187. [Google Scholar]
- Kim, J.S.; Kwon, C.S.; Son, K.H. Inhibition of alpha-glucosidase amylase by luteolin; a flavonoid. Biosci. Biotechnol. Biochem. 2000, 64, 2458–2461. [Google Scholar] [CrossRef]
- Etoundi, C.B.; Kuaté, D.; Ngondi, J.L.; Oben, J. Anti-amylase; anti-lipase antioxidant effects of aqueous extracts of some Cameroonian spices. J. Nat. Prod. 2010, 3, 165–171. [Google Scholar]
- Hamaker, B.R.; Bugusu, B. Overview, Sorghum Proteins Food Quality. In Workshop on the Proteins of Sorghum Millets; Enhancing Nutritional Functional Properties for Africa, AFRIPRO, EU funded project; University of Pretoria: Pretoria, South Africa, 2003. [Google Scholar]
- Zhang, G.; Hamaker, B.R. Low alpha-amylase starch digestibility of cooked sorghum flours the effect of protein. Cereal Chem. 1998, 75, 710–713. [Google Scholar] [CrossRef]
- Duodu, K.G.; Taylor, J.R.N.; Belton, P.S.; Hamaker, B.R. Factors affecting sorghum protein digestibility. J. Cereal Sci. 2002, 38, 117–131. [Google Scholar]
- Porter, L.J. Structure Chemical properties of the condensed tannins. In Plant Polyphenols, Synthesis, Properties, Significance; Hemmingway, R.W., Laks, P.E., Eds.; Plenum Press: New York, NY, USA, 1992; pp. 245–257. [Google Scholar]
- Dykes, L.; Rooney, L.W.; Waniska, R.D.; Rooney, W.L. Phenolic compounds antioxidant activity of sorghum grains of varying genotypes. J. Agric. Food Chem. 2005, 53, 6813–6818. [Google Scholar] [CrossRef]
- Kaluza, W.Z.; McGrath, R.M.; Roberts, T.C.; Schroder, H.H. Separation of phenolics of Sorghum bicolor (L.) Moench grain. J. Agric. Food Chem. 1980, 28, 1191–1196. [Google Scholar]
- Fuleki, T.; Francis, F.J. Quantitative methods for anthocyanins. Determination of total anthocyanin degradation index for cranberry juice. J. Food Sci. 1968, 33, 78–83. [Google Scholar] [CrossRef]
- Awika, J.M.; Rooney, L.W. Sorghum phytochemicals potential impact on human health. Phytochemistry 2004, 65, 1199–1221. [Google Scholar]
- McCleary, B.V.; Gibson, T.S.; Mugford, D.C. Measurement of total starch in cereal products by amyloglucosidase—A-amylase method, Collaborative study. J. AOAC Int. 1997, 80, 571–579. [Google Scholar]
- Goñi, I.; Garcia-Alonso, A.; Saura-Calixto, F. A starch hydrolysis procedure to estimate glycemic index. Nutr. Res. 1997, 17, 427–437. [Google Scholar] [CrossRef]
- Goñi, I.; Garcia-Diaz, L.; Manas, E.; Saura-Calixto, F. Analysis of resistant starch; a method for foods food products. Food Chem. 1996, 56, 445–449. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the sorghum varieties are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lemlioglu-Austin, D.; Turner, N.D.; McDonough, C.M.; Rooney, L.W. Effects of Sorghum [Sorghum bicolor (L.) Moench] Crude Extracts on Starch Digestibility, Estimated Glycemic Index (EGI), and Resistant Starch (RS) Contents of Porridges. Molecules 2012, 17, 11124-11138. https://doi.org/10.3390/molecules170911124
Lemlioglu-Austin D, Turner ND, McDonough CM, Rooney LW. Effects of Sorghum [Sorghum bicolor (L.) Moench] Crude Extracts on Starch Digestibility, Estimated Glycemic Index (EGI), and Resistant Starch (RS) Contents of Porridges. Molecules. 2012; 17(9):11124-11138. https://doi.org/10.3390/molecules170911124
Chicago/Turabian StyleLemlioglu-Austin, Dilek, Nancy D. Turner, Cassandra M. McDonough, and Lloyd W. Rooney. 2012. "Effects of Sorghum [Sorghum bicolor (L.) Moench] Crude Extracts on Starch Digestibility, Estimated Glycemic Index (EGI), and Resistant Starch (RS) Contents of Porridges" Molecules 17, no. 9: 11124-11138. https://doi.org/10.3390/molecules170911124
APA StyleLemlioglu-Austin, D., Turner, N. D., McDonough, C. M., & Rooney, L. W. (2012). Effects of Sorghum [Sorghum bicolor (L.) Moench] Crude Extracts on Starch Digestibility, Estimated Glycemic Index (EGI), and Resistant Starch (RS) Contents of Porridges. Molecules, 17(9), 11124-11138. https://doi.org/10.3390/molecules170911124




