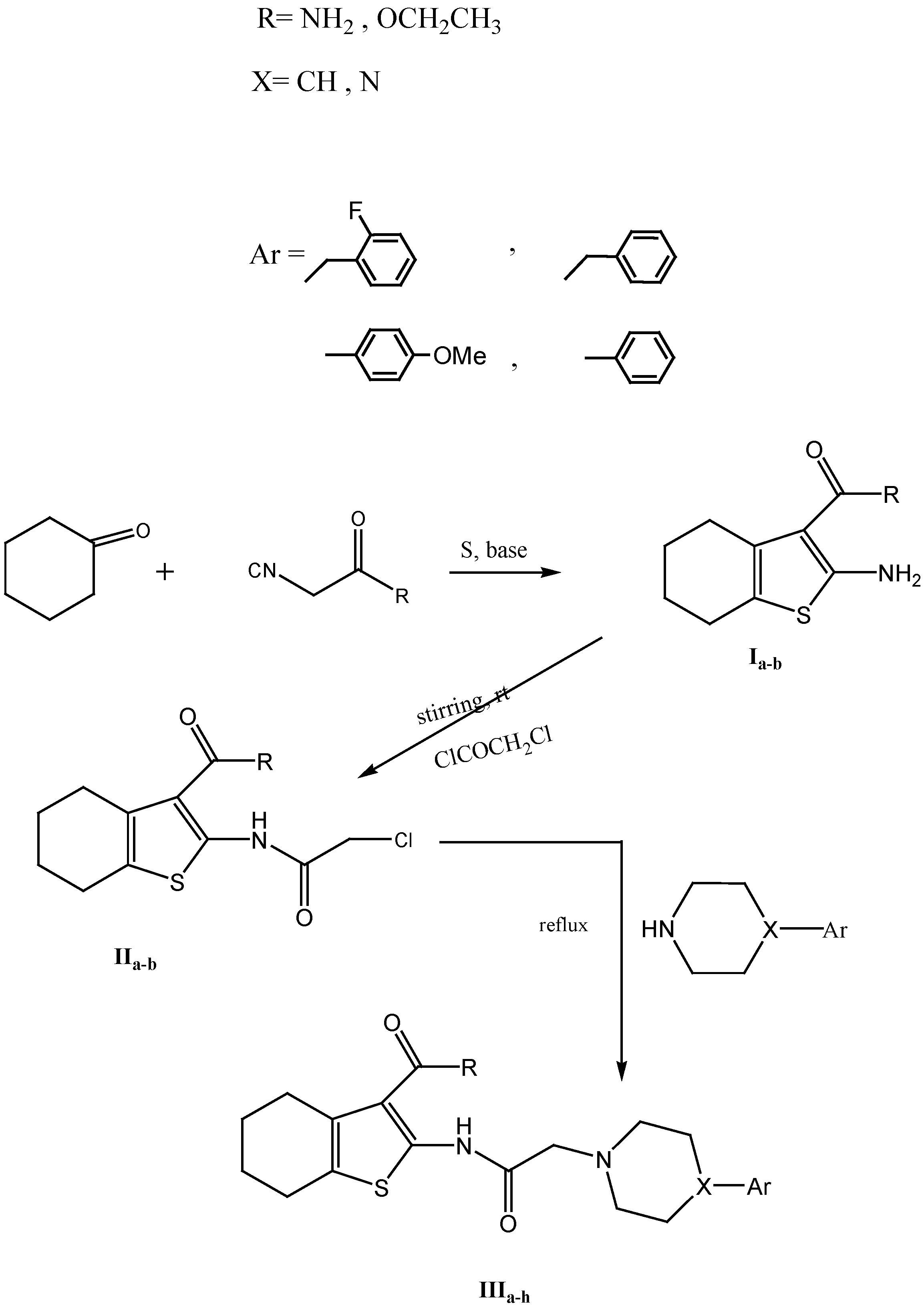
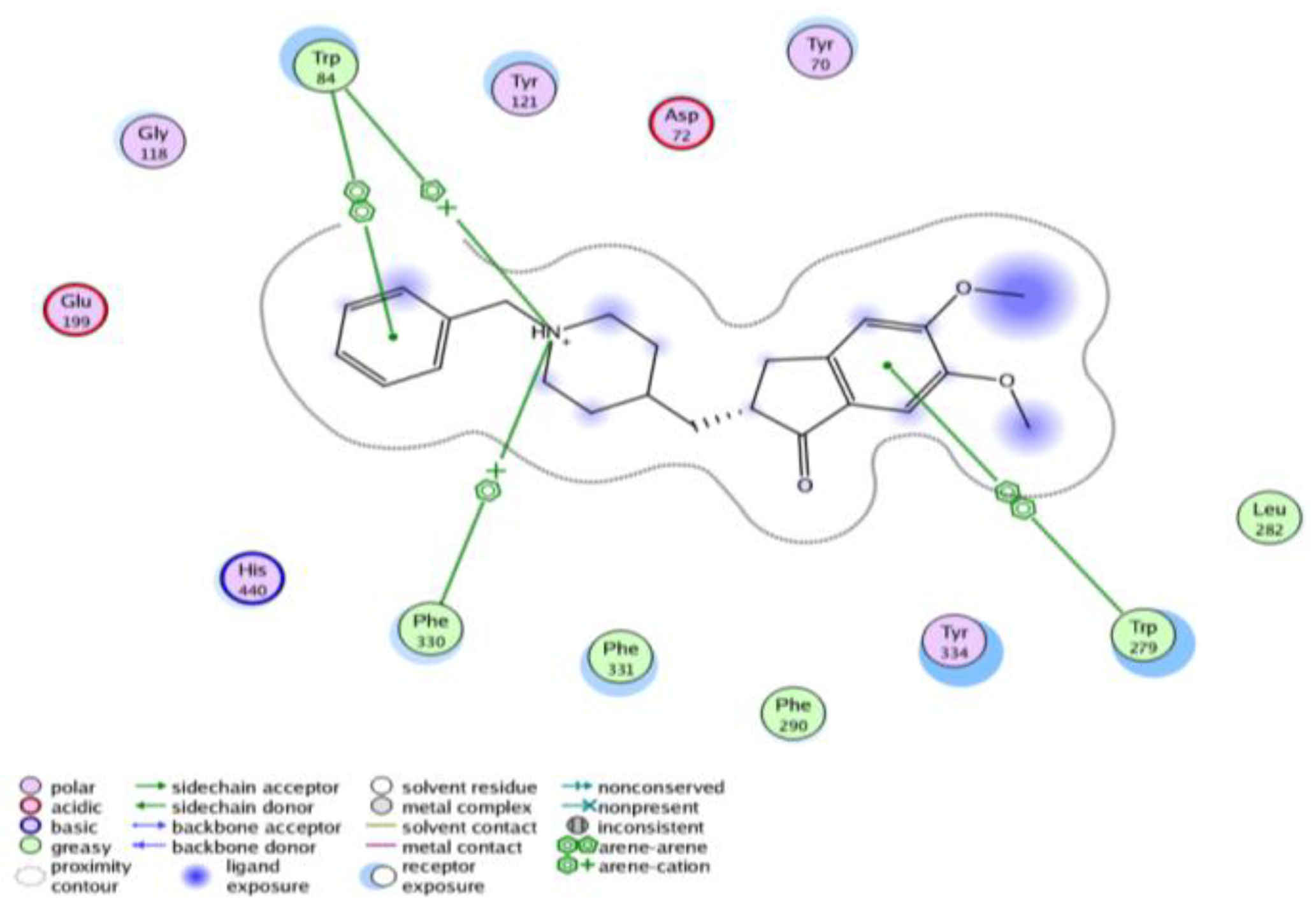
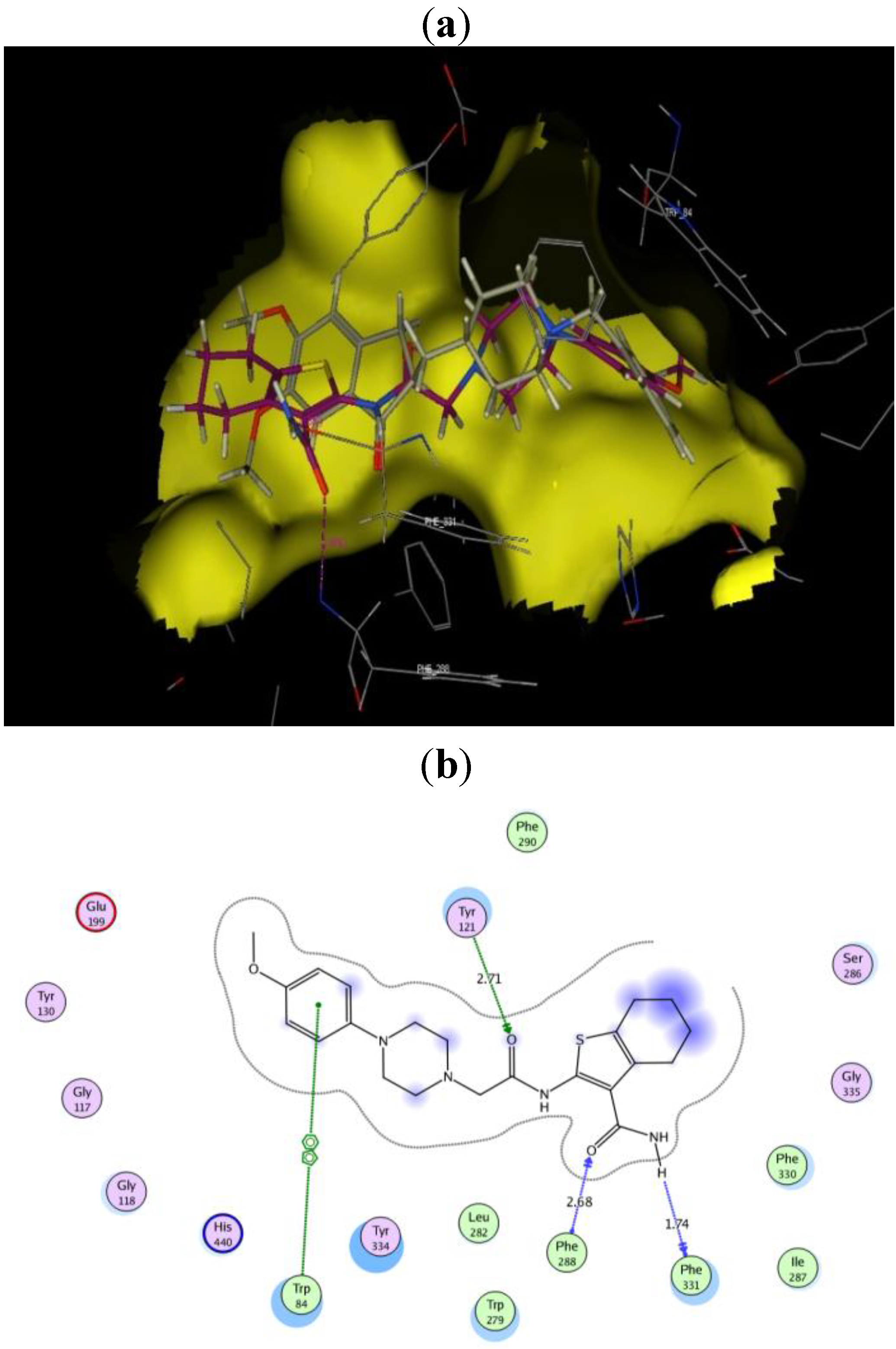
3.1.1. General Procedure for the Synthesis of IIIa–h
To a solution of each of IIa,b (0.01 mol) in an appropriate solvent (20 mL), an appropriate amine (0.02 mol) was added and the mixture was heated under reflux for 4–12 h. The product was cooled, the separated solid was filtered, dried and crystallized from ethanol.
2-(2-(4-Benzylpiperidin-1-yl)acetamido)-4,5,6,7-tetrahydrobenzo[b]thiophene-3-carboxamide (IIIa). Solvent: ethanol; time of reflux: 12 h; m.p.: 192–194 °C; yield: 80%; IR: 3406, 3290 (NH2, NH), 3050 (CH aromatic), 2927, 2912 (CH aliphatic), 1681, 1662 (C=O); 1H-NMR (CDCl3) δ: 1.62–1.69 (m, 5H, 2CH2 + CH piperidine), 1.87 (m, 4H, 2CH2), 2.6 (m, 4H, 2CH2), 2.74 (m, 4H, 2 CH2 piperidine), 3.01 (s, 2H, CH2 benzyl), 5.8 (s, 2H, CO-CH2-N), 7.22 (s, 2H, NH2, D2O exchangeable), 7.14–7.31 (m, 5H, aromatic H), 12.63 (s, 1H, NH, D2O exchangeable) ppm; MS (m/z, % abundance): 411 (M+, 8.42%). Anal. calcd. for C23H29N3O2S: C 67.12, H 7.10, N 10.21; found C 66.90, H 6.88, N 9.99.
2-(2-(4-Benzylpiperazin-1-yl)acetamido)-4,5,6,7-tetrahydrobenzo[b]thiophene-3-carboxamide (IIIb). Solvent: ethanol; time of reflux: 12 h; m.p.: 200–202 °C; yield: 80%; IR: 3394, 3209 (NH2, NH), 3030 (CH aromatic), 2927, 2850 (CH aliphatic), 1685, 1650 (C=O); 1H-NMR (DMSO-d6) δ: 1.72 (s, 4H, 2CH2), 2.49 (m, 4H, 2CH2), 2.6 (m, 4H, 2CH2 piperazine), 2.9 (m, 4H, 2CH2 piperazine), 3.18(s, 2H, CO-CH2-N), 3.47 (s, 2H, CH2 benzyl), 7.27 (s, 2H, NH2, D2O exchangeable), 7.26–7.32 (m, 5H, aromatic protons), 12.27 (s, 1H, NH, D2O exchangeable) ppm; 13C-NMR (DMSO) δ: 22.41–25.17 (4CH2), 52.47 (2CH2 piperazine), 52.96 (2CH2 piperazine), 60.49 (CO-CH2-N), 62.01 (CH2 benzyl), 116.23–141.68 (aromatic C), 166.98 (C=O), 167.31 (C=O) ppm; MS (m/z, % abundance): 412 (M+, 12.31%); Anal. calcd. for C22H28N4O2S: C 64.05, H 6.84, N 13.58; found: C 64.19, H 6.92, N 13.54.
2-(2-(4-(2-Fluorobenzyl)piperazin-1-yl)acetamido)-4,5,6,7-tetrahydrobenzo[b]thiophene-3-carboxamide (IIIc). Solvent: dioxane; time of reflux: 4 h; m.p.: 196–198 °C; yield: 76%; IR: 3495, 3309 (NH2, NH), 3100 (CH aromatic), 2939, 2873 (CH aliphatic), 1675, 1651 (C=O); 1H-NMR (DMSO-d6) δ: 1.62 (m, 4H, 2CH2), 1.87 (m, 4H, 2CH2), 2.65–2.75 (m, 8H, 4CH2 piperazine), 3.21 (s, 2H, CO-CH2-N), 3.63 (s, 2H, CH2 benzyl), 7.22 (s, 2H,NH2, D2O exchangeable), 6.99–7.42 (m, 4H, aromatic H), 12.63 (s, 1H, NH, D2O exchangeable) ppm; MS (m/z, % abundance): 430 (M+, 15.45%); Anal. calcd. for C22H27FN4O2S: C 61.37, H 6.32, N 13.01; found: C 61.29, H 6.36, N 13.22.
2-(2-(4-(4-Methoxyphenyl)piperazin-1-yl)acetamido)-4,5,6,7-tetrahydrobenzo[b]thiophene-3-carboxamide (IIId). Solvent: ethanol; time of reflux: 8 h; m.p.: 234–236 °C; yield: 70%; IR: 3500, 3310 (NH2, NH), 3050 (CH aromatic), 2921 (CH aliphatic), 1681, 1651 (C=O); 1H-NMR (DMSO-d6) δ: 1.72 (m, 4H, 2CH2), 2.49–2.5 (m, 4H, 2CH2), 2.63–2.65 (m, 4H, CH2 piperazine), 3.18 (m, 4H, 2CH2 piperazine), 3.3 (s, 2H, CO-CH2-N), 3.69 (s, 3H, OCH3), 6.88 (s, 2H, NH2, D2O exchangeable), 6.81–6.91 (dd, 4H, aromatic H), 12.35 (s, 1H, NH ,D2O exchangeable) ppm; MS (m/z, % abundance): 428 (M+, 31.44%); Anal. calcd. for C22H28N4O3 S: C 61.66, H 6.59, N 13.07; found C 62.21, H 6.36, N 13.12.
Ethyl 2-(2-(4-benzylpiperidin-1-yl)acetamido)-4,5,6,7-tetrahydrobenzo[b]thiophene-3-carboxylate (IIIe). Solvent: ethanol; time of reflux: 8 h; m.p.: 80–82 °C; yield: 65%; IR: 3190 (NH), 3028 (CH aromatic), 2931, 2850 (CH aliphatic), 1670 (2C=O); 1H-NMR (CDCl3) δ: 1.35 (t, 3H, OCH2CH3), 1.54–1.58 (m, 5H, 2CH2 + CH piperidine), 1.71 (m, 4H, 2CH2), 2.11 (m, 4H, 2CH2), 2.76 (m, 4H, 2CH2 piperidine), 3.17 (s, 2H, CH2 benzyl), 3.36 (s, 2H, CO-CH2-N), 4.3(q, 2H, OCH2CH3), 7.15–7.30 (m, 5H, aromatic H), 12.63 (s, 1H, NH, D2O exchangeable) ppm; MS (m/z, % abundance): 440 (M+, 23.98%); Anal. calcd. for C25H32N2O3: C 68.15, H 7.32, N 6.36; found C 67.55, H6.80, N 6.22.
Ethyl 2-(2-(4-benzylpiperazin-1-yl)acetamido)-4,5,6,7-tetrahydrobenzo[b]thiophene-3-carboxylate (IIIf). Solvent: Acetonitrile; time of reflux: 12 h; m.p.: 96–98 °C; yield: 60%; IR: 3178 (NH), 3028 (CH aromatic), 2935, 2858 (CH aliphatic), 1670 (2C=O); 1H-NMR (CDCl3) δ: 1.38–1.42 (t, 3H, OCH2CH3), 1.79 (m, 4H, 2CH2), 2.65 (m, 4H, 2CH2), 2.68 (m, 4H, 2CH2 piperazine), 2.8 (m, 4H, 2CH2 piperazine), 3.3 (s, 2H, CO-CH2-N), 3.66 (s, 2H, CH2 benzyl), 4.37 (q, 2H, OCH2CH3), 7.27–7.39 (m, 5H, aromatic H), 12.18 (s,1H, NH, D2O exchangeable) ppm; MS (m/z, % abundance): 441 (M+, 9.77%); Anal. calcd. for C24H31N3O3S: C 65.28, H 7.08, N 9.52; found: C 65.32, H7.12, N9.58.
Ethyl 2-(2-(4-(2-fluorobenzyl)piperazin-1-yl)acetamido)-4,5,6,7-tetrahydrobenzo[b]thiophene-3 carboxylate (IIIg). Solvent: ethanol; time of reflux: 8 h; m.p.: 134–136 °C; yield: 62%; IR: 3450 (NH), 3105 (CH aromatic), 2920 (CH aliphatic), 1674 (2C=O); 1H-NMR (DMSO-d6) δ: 1.29 (t, 3H, OCH2CH3), 1.71 (m, 4H, 2CH2), 2.5 (m, 4H, 2CH2), 2.62 (m, 4H, 2CH2 piperazine), 3.06 (m, 4H, 2CH2 piprazine), 3.3 (s, 2H, CO-CH2-N), 3.62 (s, 2H, CH2 benzyl), 4.19 (q, 2H, OCH2CH3), 7.16–7.45 (m, 4H, aromatic H), 12.02 (s, 1H, NH, D2O exchangeable) ppm; MS (m/z, % abundance): 459 (M+, 0.02%); Anal. calcd. for C24H30FN3O3S: C 62.72, H 6.58, N 9.14; found: C 62.79, H6.53, N9.16.
Ethyl 2-(2-(4-(4-methoxyphenyl)piperazin-1-yl)acetamido)-4,5,6,7-tetrahydrobenzo[b]thiophene-3-carboxylate (IIIh). Solvent: ethanol; time of reflux: 8 h; m.p.: 145–147 °C; yield: 73%; IR: 3240 (NH), 3050 (CH aromatic), 2947, 2904 (CH aliphatic), 1675, 1666 (C=O); 1H-NMR (DMSO-d6) δ: 1.25–1.3 (t, 3H, OCH2CH3), 1.72 (m, 4H, 2CH2), 2.49–2.6 (m, 4H, 2CH2), 2.67 (m, 4H, 2CH2, piperazine), 3.13 (m, 4H, 2CH2 piperazine), 3.3 (s, 2H, CO-CH2-N), 3.7 (s, 3H,OCH3), 4.26 (q, 2H, OCH2CH3), 6.81–6.93 (dd, 4H, aromatic H), 12.04 (s,1H, NH, D2O exchangeable) ppm; 13C-NMR (DMSO): 13.98 (OCH2CH3), 22.19–25.71 (4CH2), 49.31 (2CH2 piperazine), 52.80 (2CH2 piperazine), 55.12 (OCH3), 59.96 (CO-CH2-N), 60.26 (OCH2CH3), 114.25–152.84 (aromatic C), 164.52 (C=O), 167.68 (C=O) ppm; MS (m/z, % abundance): 457 (M+, 28.79%); Anal. calcd. for C24H31N3O4S; calcd. C 63.00, H 6.83, N 9.18; found: C 63.13, H6.89, N9.21.
2-(2-Chloroacetamido)-5,6,7,8-tetrahydro-4H-cyclohepta[b]thiophene-3-carboxamide (Va). Chloroacetyl chloride (3.69 g, 0.033 mol) was added to a solution of each of IVa (6.3g, 0.03 mol) in acetic acid (20 mL). The reaction mixture was stirred at room temperature for 1 hour. The product was filtered, dried and crystallized from ethanol. M.p.: 220–222 °C; yield 55%; IR: 3390–3209 (NH2, NH), 2920 (CH aliphatic), 1670, 1627 (C=O); 1H-NMR (DMSO-d6) δ: 1.55–1.59 (m, 4H, 2CH2), 1.73–1.75 (m, 2H, CH2), 2.67–2.71 (m, 4H, 2CH2), 4.43 (s, 2H, CO-CH2-Cl), 7.5 (s, 2H, NH2, D2O exchangeable), 11.11 (s, 1H, NH, D2O exchangeable) ppm; MS (m/z, % abundance): 286 (M+, 40.43%), 288 (M + 2, 19.42%); Anal. calcd. for C12H15ClN2O2S: C 50.26, H 5.27, N 9.77; found: C 50.29, H5.28, N9.73.
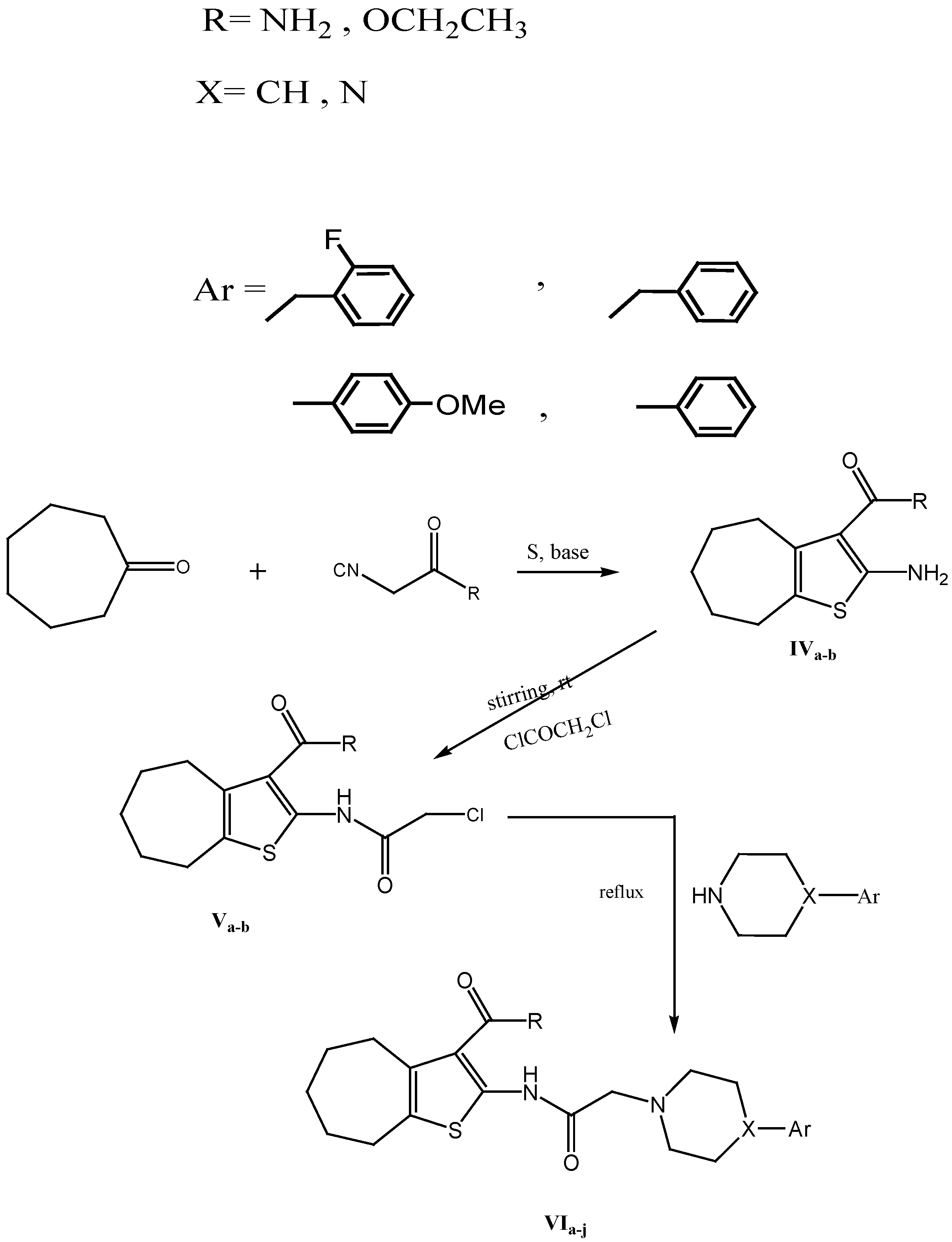
3.1.2. General Procedure of the Preparation of Compounds VIa–j
To a solution of each of IVa,b (0.01 mol) in an appropriate solvent (20 mL), an appropriate amine (0.02 mol) was added and the mixture was heated under reflux for 4–12 h. The product was poured into ice-cold water (25 mL), and then extracted with chloroform. The chloroform layer was dried over anhydrous sodium sulphate. The reaction was filtered and the solvent was evaporated under reduced pressure to give an oily product. The oily product was treated with ether (25 mL) and the solid obtained was filtered, dried and crystallized from ethanol.
2-(2-(4-Benzylpiperidin-1-yl)acetamido)-5,6,7,8-tetrahydro-4H-cyclohepta[b]thiophene-3-carboxamide (VIa). Solvent: Acetonitrile; time of reflux: 12 h; m.p.: 83–85 °C; yield: 65%; IR: 3325 (NH2, NH), 3059, 3024 (CH aromatic), 2920 (CH aliphatic), 1654 (2C=O); 1H-NMR (DMSO-d6) δ: 1.55–1.58 (m, 4H, 2CH2,), 1.78 (m, 2H, CH2), 2.49 (m, 4H, 2CH2), 2.67–2.74 (m, 5H, 2CH2+ CH piperidine), 3.3 (m, 4H, 2CH2 piperidine), 3.8 (s, 2H, CH2 benzyl), 5.1(s, 2H, CO-CH2-N), 7.17–7.29 (m, 5H, aromatic H), 7.45 (s, 2H, NH2, D2O exchangeable), 10.8 (s, 1H, NH, D2O exchangeable) ppm; MS (m/z, % abundance): 425.558 (M+); Anal. calcd. for C24H31N3O2S: C 67.73, H 7.34, N 9.87; found: C 67.48, H7.35, N9.81.
2-(2-(4-Benzylpiperazin-1-yl)acetamido)-5,6,7,8-tetrahydro-4H-cyclohepta[b]thiophene-3-carboxamide (VIb). Solvent: Acetonitrile; time of reflux: 12; m.p.:108–110 °C; yield: 60%; IR: 3441, 3340 (NH2, NH), 3062, 3028 (CH aromatic), 2916 (CH aliphatic), 1651(2C=O); 1H-NMR (DMSO-d6) δ: 1.52–1.56 (m, 4H, 2CH2), 1.78 (m, 2H, CH2), 2.49 (m, 4H, 2CH2), 2.7 (m, 4H, 2CH2 piperazine), 3.13 (m, 4H, 2CH2 piperazine), 3.55 (s, 2H, CO-CH2-N), 4.09 (s, 2H,CH2 benzyl), 7.28–7.32 (m, 5H, aromatic H), 7.45 (s, 2H, NH2, D2O exchangeable), 11.17 (s, 1H, NH, D2O exchangeable) ppm; MS (m/z, % abundance): 426.560 (M+); Anal. calcd. for C23H30N4O2S: C 64.76, H 7.09, N 13.13; found: C 64.82, H 7.11, N13.18.
2-(2-(4-(2-Fluorobenzyl)piperazin-1-yl)acetamido)-5,6,7,8-tetrahydro-4H-cyclohepta[b]thiophene-3-carboxamide (VIc). Solvent: Dioxane; time of reflux: 4 h; m.p.:170–172 °C; yield: 60%; IR: 3394, 3321 (NH2, NH), 3070 (CH aromatic), 2920 (CH aliphatic), 1660–1631 (C=O); 1H-NMR(CDCl3) δ: 1.52–1.56 (m, 4H, 2CH2), 1.78 (m, 2H, CH2), 2.49 (m, 4H, 2CH2), 2.59 (m, 4H, 2CH2 piperazine), 3.05 (m, 4H, 2CH2 piperazine), 3.29 (s, 2H, CO-CH2-N), 3.6 (s, 2H, CH2 benzyl), 7.15–7.44 (m, 4H, aromatic H), 7.22 (s, 2H, NH2, D2O exchangeable), 8.84 (s, 1H, NH, D2O exchangeable) ppm; MS (m/z, % abundance): 444.917 (M+); Anal. calcd. for C23H29FN4O2S: C 62.14, H 6.58, N 12.60; found: C 62.23, H6.56, N12.65.
2-(2-(4-(4-Methoxyphenyl)piperazin-1-yl)acetamido)-5,6,7,8-tetrahydro-4H-cyclohepta[b]thiophene-3-carboxamide (VId). Solvent: acetonitrile; time of reflux: 12 h; m.p.: 190–192 °C; yield: 60%; IR: 3345, 3440 (NH2, NH), 3089 (CH aromatic), 2954 (CH aliphatic), 1778, 1724 (C=O); 1H-NMR (DMSO-d6) δ: 1.56 (m, 4H, 2CH2), 1.8 (m, 2H, CH2), 2.49 (m, 4H, 2CH2), 2.7 (m, 4H, 2CH2 piperazine), 3.16 (m, 4H, 2CH2 piperazine), 3.8 (s, 2H, CO-CH2-N), 4.23 (s, 3H,OCH3), 6.83–7.00 (dd, 4H, aromatic H), 7.48 (s, 2H,NH2, D2O exchangeable), 11.17 (s, 1H, NH, D2O exchangeable) ppm; MS (m/z, % abundance): 442.657 (M+); Anal. calcd. for C23H30N4O3S: C 62.42, H 6.83, N 12.66; found C 62.47, H 6.88, N 12.60.
2-(2-(4-Phenylpiperazin-1-yl)acetamido)-5,6,7,8-tetrahydro-4H-cyclohepta[b]thiophene-3-carboxamide (VIe). Solvent: acetonitrile; time of reflux: 12 h; m.p.: 158–160 °C; yield: 60%; IR: 3394 (NH2, NH), 3050 (CH aromatic), 2920 (CH aliphatic), 1651 (2C=O); 1H-NMR (DMSO-d6) δ: 1.56 (m, 4H, 2CH2), 1.79 (m, 2H, CH2), 2.49 (m, 4H, 2CH2), 2.7 (m, 4H, 2CH2 piperazine), 3.2 (m, 4H, 2CH2 piperazine), 3.8 (s, 2H, CO-CH2-N), 6.80–7.27 (m, 5H, aromatic H), 7.48 (s, 2H, NH2, D2O exchangeable), 11.17 (s, 1H, NH, D2O exchangeable) ppm; MS (m/z, % abundance): 412 (M+, 0.75%); Anal. calcd. for C22H28N4O2S: C 64.05, H 6.84, N 13.58; found C 63.94, H6.84, N 13.52.
Ethyl 2-(2-(4-benzylpiperidin-1-yl)acetamido)-5,6,7,8-tetrahydro-4H-cyclohepta[b]thiophene-3-carboxylate (VIf). Solvent: ethanol; time of reflux: 8 h; m.p.: 155–157 °C; yield: 56%; IR: 3332 (NH), 3000 (CH aromatic), 2916 (CH aliphatic), 1716, 1658 (C=O); 1H-NMR(CDCl3) δ: 1.37–1.42 (t, 3H, OCH2CH3), 1.59–1.67 (m, 6H, 3CH2), 1.83 (m, 4H, 2CH2), 2.7 (m, 5H, 2CH2 + CH piperidine), 3.03 (m, 4H, 2CH2 piperidine), 3.56 (s, 2H, CH2 benzyl), 4.33 (s, 2H, CO-CH2-N), 4.39 (q, 2H, OCH2CH3), 7.26–7.27 (m, 5H, aromatic H), 11.78( s,1H, NH, D2O exchangeable) ppm; MS (m/z, % abundance): 453 (M+−H, 1.05%); Anal. calcd. for C26H34N2O3S: C 68.69, H 7.54, N 6.16; found C 68.73, H 7.58, N 6.18.
Ethyl 2-(2-(4-benzylpiperazin-1-yl)acetamido)-5,6,7,8-tetrahydro-4H-cyclohepta[b]thiophene-3-carboxylate (VIg). Solvent: Acetonitrile; time of reflux: 12; m.p.: 153–155 °C; yield: 56%; IR: 3345 (NH), 3020 (CH aromatic), 2920 (CH aliphatic), 1716, 1658 (C=O); 1H-NMR (CDCl3) δ: 1.37–1.42 (t, 3H, OCH2CH3), 1.59–1.69 (m, 6H, 3CH2), 1.83 (m, 4H, 2CH2), 2.7 (m, 4H, 2CH2 piperazine), 3.03 (m, 4H, 2CH2 piperazine), 3.56 (s, 2H, CO-CH2-N), 4.33 (s, 2H, CH2 benzyl), 4.39 (q, 2H, OCH2CH3), 7.26–7.37 (m, 5H, aromatic H), 11.78 (s, 1H, NH, D2O exchangeable) ppm; MS: (m/z, % abundance) 455 (M+, 12.34%); Anal. calcd. for C25H33N3O3S: C 65.90, H 7.30, N 9.22; found C 65.94, H 7.31, N 9.29.
Ethyl 2-(2-(4-(2-fluorobenzyl)piperazin-1-yl)acetamido)-5,6,7,8-tetrahydro-4H-cyclohepta [b]thiophene-3-carboxylate (VIh). Solvent: Ethanol; time of reflux: 8 h; m.p.: 163–165 °C; yield: 55%; IR: 3345 (NH), 3020 (CH aromatic), 2920 (CH aliphatic), 1716, 1658 (C=O); 1H-NMR (CDCl3) δ: 1.37–1.42 (t, 3H, OCH2CH3), 1.57–1.67 (m, 6H, 3CH2), 1.85 (m, 4H, 2CH2), 2.71 (m, 4H, 2CH2 piperazine), 3.03 (m, 4H, 2CH2 piperazine), 3.56 (s, 2H, CO-CH2-N), 4.33 (s, 2H, CH2 benzyl), 4.39 (q, 2H, OCH2CH3), 7.16–7.45 (m, 4H, aromatic H), 11.78 (s, 1H, NH, D2O exchangeable) ppm; MS (m/z, % abundance): 473 (M+, 100%); Anal. calcd. for C25H32FN3O3S: C 63.40, H 6.81, N 8.87; found: C 63.49, H 6.84, N 8.83.
Ethyl 2-(2-(4-(4-methoxyphenyl)piperazin-1-yl)acetamido)-5,6,7,8-tetrahydro-4H-cyclohepta[b]thiophene-3-carboxylate (VIi). Solvent: Ethanol; time of reflux: 8; m.p.: 128–130 °C; yield: 60%; IR: 3259 (NH), 3020 (CH aromatic), 2924, 2850 (CH aliphatic), 1683 (C=O); 1H-NMR (DMSO-d6) δ :1.26–1.33 (t, 3H, OCH2CH3), 1.54 (m, 4H, 2CH2), 1.8 (m, 2H, CH2), 2.5 (m,4H, 2CH2), 2.66 (m, 4H, 2CH2 piperazine), 3.0 (m, 4H, 2CH2 piperazine), 3.3 (s, 2H, CO-CH2-N), 3.68 (s, 3H, OCH3), 4.27 (q, 2H, OCH2CH3), 6.81–6.92 (dd, 4H, aromatic H), 11.8(s,1H, NH, D2O exchangeable) ppm; 13C-NMR (DMSO-d6) δ: 14.13 (OCH2CH3), 26.54–31.54 (5CH2), 49.42 (2CH2 piperazine), 52.89 (2CH2 piperazine), 55.12 (OCH3), 60.36 (CO-CH2-N + OCH2CH3), 113.05–152.89 (aromatic C), 164.58 (C=O), 167.73 (C=O) ppm; MS (m/z, % abundance): 471 (M+, 28.99%); Anal. calcd. for C25H33N3O4S: C 63.67, H 7.05, N 8.91; found C 63.6, H 7.05, N 8.90.
Ethyl 2-(2-(4-phenylpiperazin-1-yl)acetamido)-5,6,7,8-tetrahydro-4H-cyclohepta[b]thiophene-3-carbox-ylate (VIj). Solvent: Ethanol; time of reflux: 8 h; m.p.: 138–140 °C; yield: 65%;IR: 3332 (NH), 3100 (CH aromatic), 2920 (CH aliphatic), 1716, 1658 (C=O); 1H-NMR (DMSO-d6) δ: 1.29–1.30 (t, 3H, OCH2CH3), 1.32 (m, 2H, CH2), 1.54 (m, 4H, 2CH2), 1.8 (m, 2H, CH2), 2.71 (m, 4H, 2CH2 piperazine), 2.98 (m, 2H, CH2), 3.32 (m, 4H, CH2 piperazine), 3.3 (s, 2H, CO-CH2-N), 4.26 (q, 2H, OCH2CH3), 6.93–7.24 (m, 5H, aromatic H), 11.83 (s, 1H, NH, D2O exchangeable) ppm. MS (m/z, % abundance): 441 (M+, 17.79%); Anal. calcd. for C24H31N3O3S: C 65.28, H 7.08, N 9.52; found C 65.26, H 7.09, N 9.47.