Physicochemical Properties, in Vitro Antioxidant Activities and Inhibitory Potential against α-Glucosidase of Polysaccharides from Ampelopsis grossedentata Leaves and Stems
Abstract
:1. Introduction
2. Results
2.1. Physicochemical Properties of ALPS and ASPS
| Sample | ALPS | ASPS |
|---|---|---|
| Yield (%) | 1.1 ± 0.1 a | 1.0 ± 0.1 a |
| Protein (%) | 3.1 ± 0.3 a | 8.8 ± 0.4 b |
| Neutral sugar (%) | 34.7 ± 2.4 b | 24.2 ± 1.7 b |
| Uronic acid (%) | 27.5 ± 3.1 a | 40.0 ± 2.4 b |
| Neutral sugar composition (mol ratio) | ||
| L-rhamnose | 0.6 | 1.3 |
| D-fucose | 0.2 | 0.2 |
| L-arabinose | 2.5 | 2.2 |
| D-xylose | 0.1 | 0.3 |
| D-mannose | 0.8 | 3.7 |
| D-glucose | 1.6 | 0.8 |
| D-galactose | 4.2 | 1.6 |
2.2. Antioxidant Activities of ALPS and ASPS
2.2.1. DPPH Assay
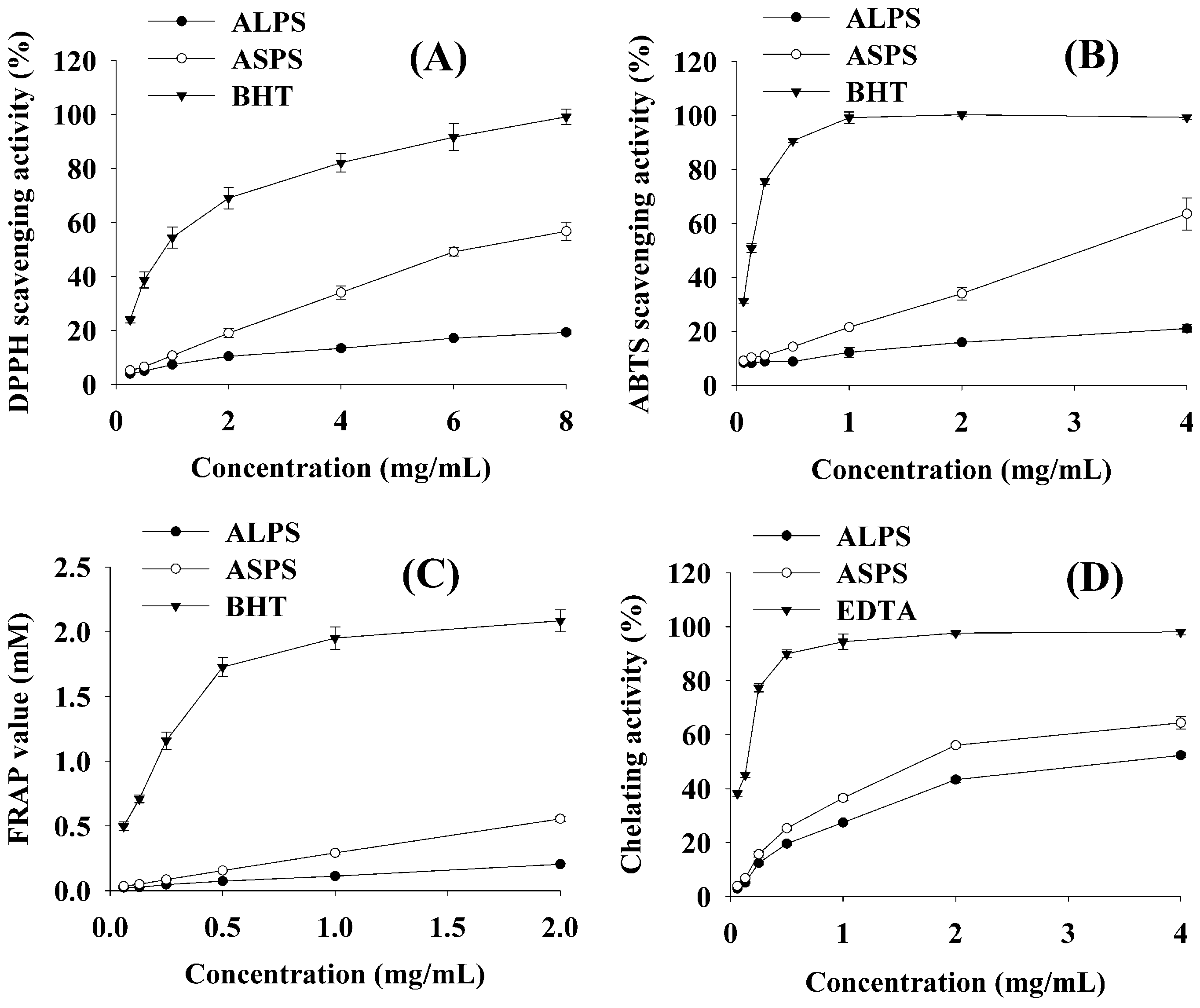
2.2.2. ABTS Assay
2.2.3. FRAP Assay
2.2.4. FIC Assay
2.3. Inhibitory Activity on α-Glucosidase of ALPS and ASPS
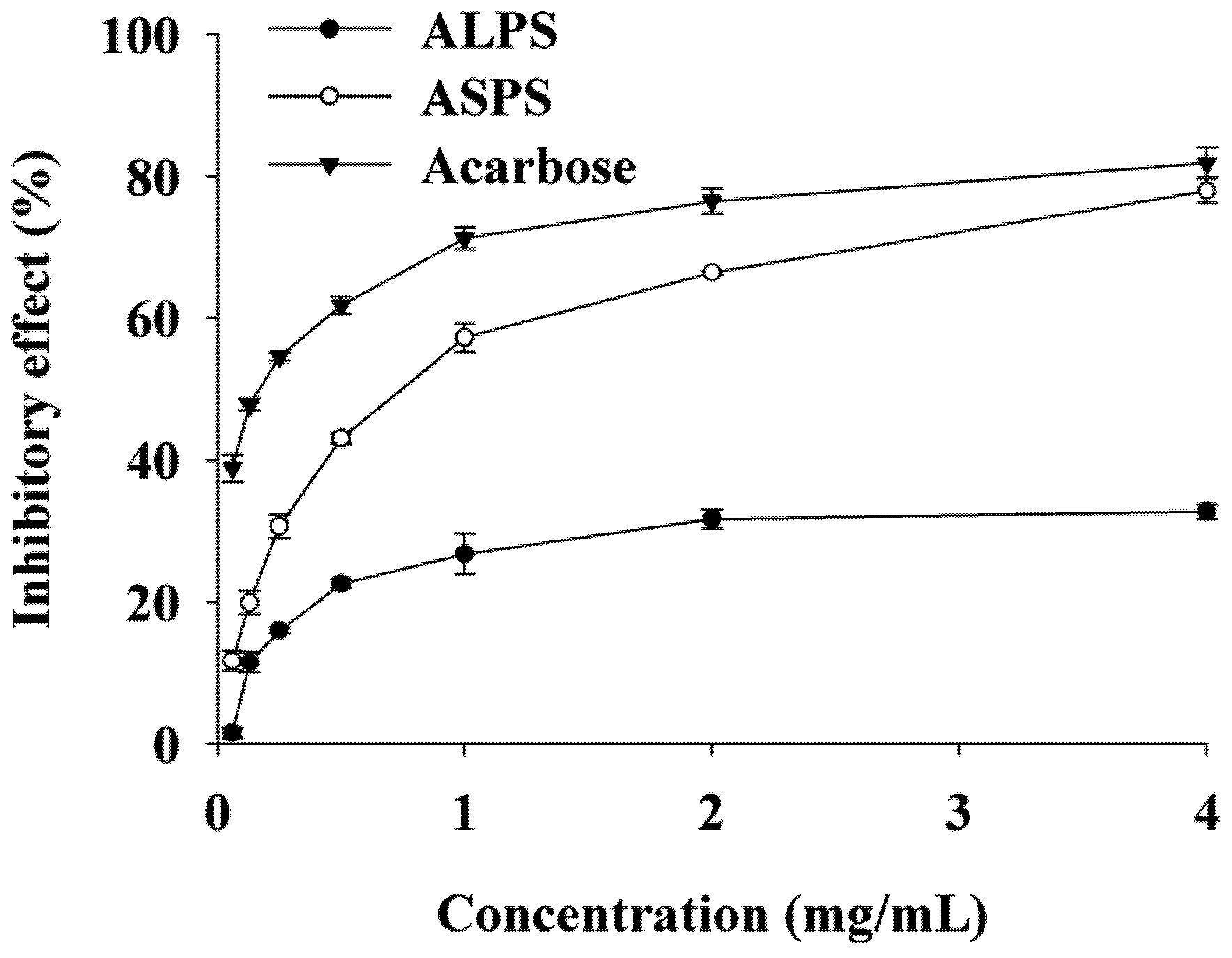
3. Discussion
4. Experimental
4.1. Materials and Chemicals
4.2. Extraction and Isolation of ALPS and ASPS
4.3. Chemical Component Analysis
4.4. Monosaccharide Composition Analysis
4.5. Molecular Weight (MW) Determination
4.6. Infrared Spectral Analysis
4.7. Antioxidant Activities Determination
4.7.1. DPPH Assay
4.7.2. ABTS Assay
4.7.3. Ferric-Reducing Antioxidant Power (FRAP) Assay
4.7.4. Ferrous Ion Chelating (FIC) Assay
4.8. α-Glucosidase Inhibition Assay
4.9. Statistical Analysis
5. Conclusions
Conflict of Interest
References
- Apostolidis, E.; Lee, C. In vitro potential of Ascophyllum nodosum phenolic antioxidant-mediated α-glucosidase and α-amylase inhibition. J. Food Sci. 2010, 75, H97–H102. [Google Scholar] [CrossRef]
- Haffner, S.M. The importance of hyperglycemia in the nonfasting state to the development of cardiovascular disease. Endocr. Rev. 1998, 19, 583–592. [Google Scholar] [CrossRef]
- Yao, Y.; Sang, W.; Zhou, M.; Ren, G. Antioxidant and α-glucosidase inhibitory activity of colored grains in china. J. Agric. Food Chem. 2009, 58, 770–774. [Google Scholar]
- Chen, H.; Qu, Z.; Fu, L.; Dong, P.; Zhang, X. Physicochemical properties and antioxidant capacity of 3 polysaccharides from green tea, oolong tea, and black tea. J. Food Sci. 2009, 74, C469–C474. [Google Scholar] [CrossRef]
- Matsui, T.; Ogunwande, I.; Abesundara, K.; Matsumoto, K. Anti-hyperglycemic potential of natural products. Mini-Rev. Med. Chem. 2006, 6, 349–356. [Google Scholar] [CrossRef]
- Kwon, Y.I.; Apostolidis, E.; Shetty, K. In vitro studies of eggplant (Solanum melongena) phenolics as inhibitors of key enzymes relevant for type 2 diabetes and hypertension. Bioresour. Technol. 2008, 99, 2981–2988. [Google Scholar] [CrossRef]
- Chen, H.; Lu, X.; Qu, Z.; Wang, Z.; Zhang, L. Glycosidase inhibitory activity and antioxidant properties of a polysaccharide from the mushroom Inonotus obliquus. J. Food Biochem. 2010, 34, 178–191. [Google Scholar] [CrossRef]
- Cheng, Y. Research Development of Ampelopsis grossedentata. Econ. Forest Res. 2004, 3, 51–56. [Google Scholar]
- Gao, J.; Liu, B.; Ning, Z.; Zhao, R.; Zhang, A.; Wu, Q. Characterization and antioxidant activity of flavonoid-rich extracts from leaves of Ampelopsis grossedentata. J. Food Biochem. 2009, 33, 808–820. [Google Scholar] [CrossRef]
- Zhang, Y.; Ning, Z.; Yang, S.; Wu, H. Antioxidation properties and mechanism of action of dihydromyricetin from Ampelopsis grossedentata. Acta Pharm. Sinica. 2003, 38, 241–244. [Google Scholar]
- Zhang, Y.; Zhang, Q.; Li, L.; Wang, B.; Zhao, Y.; Guo, D. Simultaneous determination and pharmacokinetic studies of dihydromyricetin and myricetin in rat plasma by HPLC-DAD after oral administration of Ampelopsis grossedentata decoction. J. Chromatogr. B 2007, 860, 4–9. [Google Scholar] [CrossRef]
- Soare, J.R.; Dinis, T.C.P.; Cunha, A.P.; Almeida, L. Antioxidant activities of some extracts of Thymus zygis. Free Radical Res. 1997, 26, 469–478. [Google Scholar] [CrossRef]
- Giovanelli, G.; Buratti, S. Comparison of polyphenolic composition and antioxidant activity of wild Italian blueberries and some cultivated varieties. Food Chem. 2009, 112, 903–908. [Google Scholar] [CrossRef]
- Kuda, T.; Ikemori, T. Minerals, polysaccharides and antioxidant properties of aqueous solutions obtained from macroalgal beach-casts in the Noto Peninsula, Ishikawa, Japan. Food Chem. 2009, 112, 575–581. [Google Scholar] [CrossRef]
- Kaviarasan, S.; Naik, G.; Gangabhagirathi, R.; Anuradha, C.; Priyadarsini, K. In vitro studies on antiradical and antioxidant activities of fenugreek (Trigonella foenum graecum) seeds. Food Chem. 2007, 103, 31–37. [Google Scholar]
- Shi, J.; Gong, J.; Liu, J.; Wu, X.; Zhang, Y. Antioxidant capacity of extract from edible flowers of Prunus mume in China and its active components. LWT-Food Sci. Technol. 2009, 42, 477–482. [Google Scholar] [CrossRef]
- Moure, A.; Dominguez, H.; Parajo, J.C. Antioxidant properties of ultrafiltration-recovered soy protein fractions from industrial effluents and their hydrolysates. Process Biochem. 2006, 41, 447–456. [Google Scholar] [CrossRef]
- Finkel, T.; Holbrook, N.J. Oxidants, oxidative stress and the biology of ageing. Nature 2000, 239–247. [Google Scholar]
- Rains, J.L.; Jain, S.K. Oxidative stress, insulin signaling and diabetes. Free Radical Biol. Med. 2011, 50, 567–575. [Google Scholar] [CrossRef]
- Chen, H.; Zhang, M.; Qu, Z.; Xie, B. Antioxidant activities of different fractions of polysaccharide conjugates from green tea (Camellia Sinensis). Food Chem. 2008, 106, 559–563. [Google Scholar] [CrossRef]
- Tian, L.; Zhao, Y.; Guo, C.; Yang, X. A comparative study on the antioxidant activities of an acidic polysaccharide and various solvent extracts derived from herbal Houttuynia cordata. Carbohydr. Polym. 2010, 83, 537–544. [Google Scholar]
- Luo, A.X.; He, X.J.; Zhou, S.D.; Fan, Y.J.; Luo, A.S.; Chun, Z. Purification, composition analysis and antioxidant activity of the polysaccharides from Dendrobium nobile Lindl. Carbohydr. Polym. 2010, 79, 1014–1019. [Google Scholar] [CrossRef]
- Ananthi, S.; Raghavendran, H.R.; Sunil, A.G.; Gayathri, V.; Ramakrishnan, G.; Vasanthi, H.R. In vitro antioxidant and in vivo anti-inflammatory potential of crude polysaccharide from Turbinaria ornata (Marine Brown Alga). Food Chem. Toxicol. 2010, 48, 187–192. [Google Scholar] [CrossRef]
- Lin, C.L.; Wang, C.C.; Chang, S.C.; Inbaraj, B.S.; Chen, B.H. Antioxidative activity of polysaccharide fractions isolated from Lycium barbarum Linnaeus. Int. J. Biol. Macromol. 2009, 45, 146–151. [Google Scholar] [CrossRef]
- Chen, H.; Zhang, M.; Xie, B. Quantification of uronic acids in tea polysaccharide conjugates and their antioxidant properties. J. Agric. Food Chem. 2004, 52, 3333–3336. [Google Scholar] [CrossRef]
- Lebovitz, H. Postprandial hyperglycaemic state: importance and consequences. Diabetes Res. Clin. Pract. 1998, 40, S27–S28. [Google Scholar] [CrossRef]
- Zhang, J.; Tiller, C.; Shen, J.; Wang, C.; Girouard, G.S.; Dennis, D.; Barrow, C.J.; Miao, M.; Ewart, H.S. Antidiabetic properties of polysaccharide-and polyphenolic-enriched fractions from the brown seaweed Ascophyllum nodosum. Can. J. Physiol. Pharmacol. 2007, 85, 1116–1123. [Google Scholar] [CrossRef]
- Maritim, A.; Sanders, R.; Watkins, J.B. Effects of α-lipoic acid on biomarkers of oxidative stress in streptozotocin-induced diabetic rats. J. Nutr. Biochem. 2003, 14, 288–294. [Google Scholar] [CrossRef]
- Morris, D.L. Quantitative determination of carbohydrates with Dreywood's anthrone reagent. Science 1948, 107, 254–255. [Google Scholar]
- Bitter, T.; Muir, H.M. A modified uronic acid carbazole reaction. Anal. Biochem. 1962, 4, 330–334. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar]
- Mohsen, S.M.; Ammar, A.S.M. Total phenolic contents and antioxidant activity of corn tassel extracts. Food Chem. 2009, 112, 595–598. [Google Scholar] [CrossRef]
- Cai, Y.; Luo, Q.; Sun, M.; Corke, H. Antioxidant activity and phenolic compounds of 112 traditional Chinese medicinal plants associated with anticancer. Life sci. 2004, 74, 2157–2184. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J. Ferric reducing/antioxidant power assay: Direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration. Method. Enzymol. 1999, 299, 15–27. [Google Scholar]
- Yuan, J.F.; Zhang, Z.Q.; Fan, Z.C.; Yang, J.X. Antioxidant effects and cytotoxicity of three purified polysaccharides from Ligusticum chuanxiong Hort. Carbohydr Polym. 2008, 74, 822–827. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are available from the authors.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, Y.; Bian, X.; Park, J.; Ying, L.; Qian, L.; Xu, P. Physicochemical Properties, in Vitro Antioxidant Activities and Inhibitory Potential against α-Glucosidase of Polysaccharides from Ampelopsis grossedentata Leaves and Stems. Molecules 2011, 16, 7762-7772. https://doi.org/10.3390/molecules16097762
Wang Y, Bian X, Park J, Ying L, Qian L, Xu P. Physicochemical Properties, in Vitro Antioxidant Activities and Inhibitory Potential against α-Glucosidase of Polysaccharides from Ampelopsis grossedentata Leaves and Stems. Molecules. 2011; 16(9):7762-7772. https://doi.org/10.3390/molecules16097762
Chicago/Turabian StyleWang, Yuefei, Xiaoying Bian, Jinhyouch Park, Le Ying, Lisheng Qian, and Ping Xu. 2011. "Physicochemical Properties, in Vitro Antioxidant Activities and Inhibitory Potential against α-Glucosidase of Polysaccharides from Ampelopsis grossedentata Leaves and Stems" Molecules 16, no. 9: 7762-7772. https://doi.org/10.3390/molecules16097762
APA StyleWang, Y., Bian, X., Park, J., Ying, L., Qian, L., & Xu, P. (2011). Physicochemical Properties, in Vitro Antioxidant Activities and Inhibitory Potential against α-Glucosidase of Polysaccharides from Ampelopsis grossedentata Leaves and Stems. Molecules, 16(9), 7762-7772. https://doi.org/10.3390/molecules16097762