High Functionalization of 5-Nitro-1H-imidazole Derivatives: The TDAE Approach
Abstract
:1. Introduction
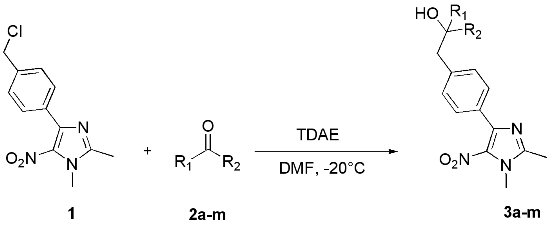
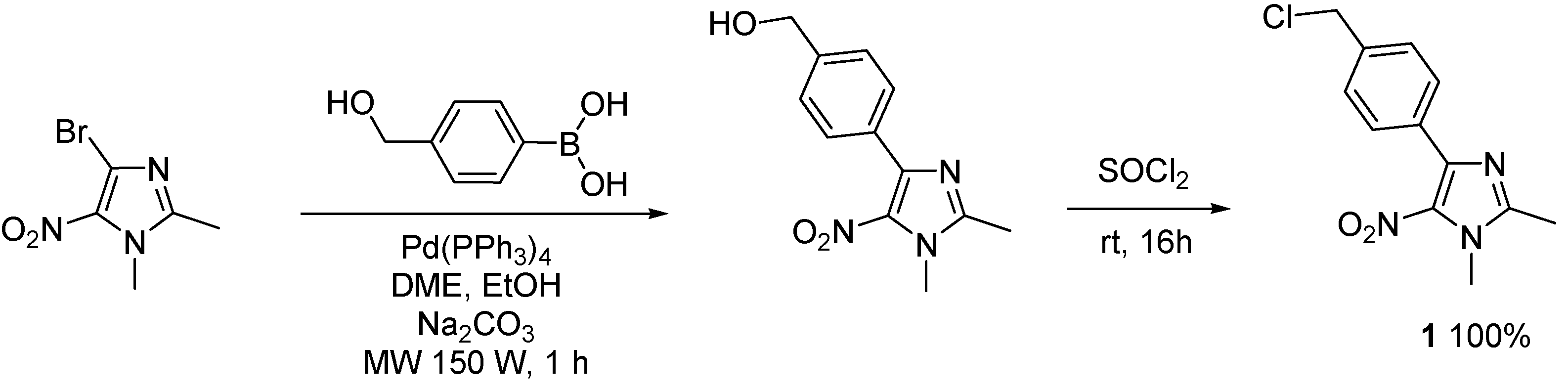
2. Results and Discussion
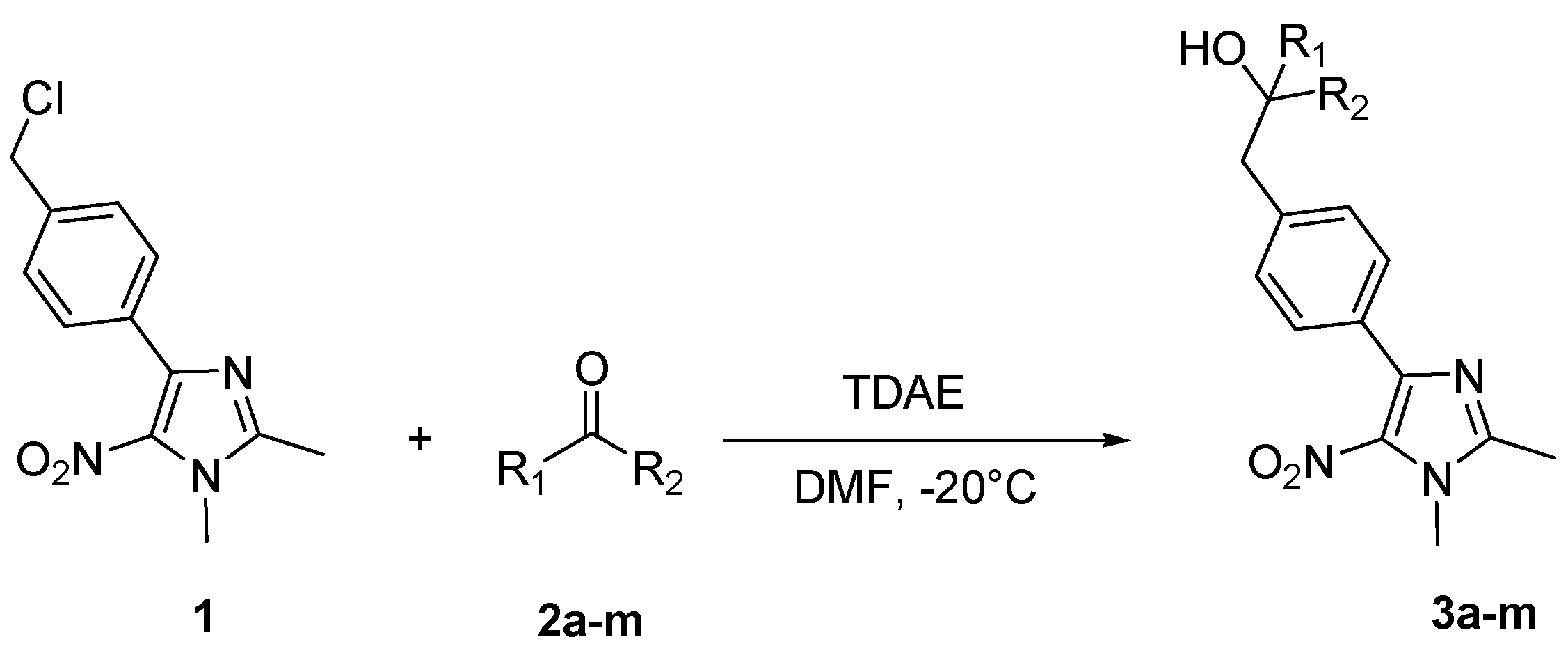
| Carbonyl compound | Product a | Product number | Yield (%) b | |
|---|---|---|---|---|
| 2a | 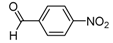 | 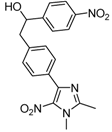 | 3a | 69 |
| 2b | 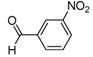 | 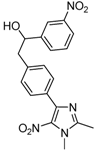 | 3b | 46 |
| 2c | 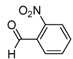 |  | 3c | 37 |
| 2d | 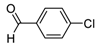 | 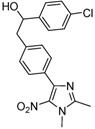 | 3d | 24 |
| 2e | 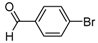 | 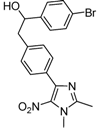 | 3e | 60 |
| 2f | 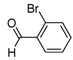 |  | 3f | 68 |
| 2g | 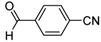 | 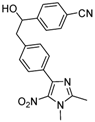 | 3g | 78 |
| 2h | 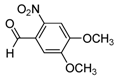 |  | 3h | 30 |
| 2i | 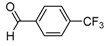 | 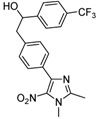 | 3i | 45 |
| 2j | 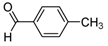 | 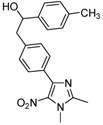 | 3j | 25 |
| 2k | 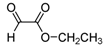 | 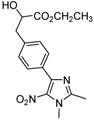 | 3k | 42 |
| 2l | 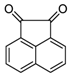 | 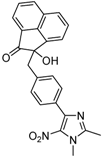 | 3l | 45 |
| 2mc | 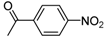 | 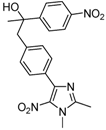 | 3m | 64 |
3. Experimental
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Jorgensen, M.A.; Manos, J.; Mendz, G.L.; Hazell, S.L. The mode of action of metronidazole in Helicobacter pylori: Futile cycling or reduction? J. Antimicrob. Chemother. 1998, 41, 67–75. [Google Scholar]
- Upcroft, J.A.; Campbell, R.W.; Benakli, K.; Upcroft, P.; Vanelle, P. Efficacy of new 5-nitroimidazoles against metronidazole-susceptible and -resistant Giardia, Trichomonas, and Entamœba spp. Antimicrob. Agents Chemother. 1999, 43, 73–76. [Google Scholar]
- Upcroft, J.A.; Dunn, L.A.; Wright, J.M.; Benakli, K.; Upcroft, P.; Vanelle, P. 5-Nitroimidazole drugs effective against metronidazole-resistant Trichomonas vaginalis and Giardia duodenalis. Antimicrob. Agents Chemother. 2006, 50, 344–347. [Google Scholar] [CrossRef]
- Leitsch, D.; Kolarich, D.; Wilson, I.B.H.; Altmann, F.; Duchêne, M. Nitroimidazole action in Entamœba histolytica: A central role for thioredoxin reductase. PLoS Biol. 2007, 5, 1820–1834. [Google Scholar]
- Crozet, M.D.; Botta, C.; Gasquet, M.; Curti, C.; Remusat, V.; Hutter, S.; Chapelle, O.; Azas, N.; de Méo, M.; Vanelle, P. Lowering of 5-nitroimidazole’s mutagenicity: Towards optimal antiparasitic pharmacophore. Eur. J. Med. Chem. 2009, 44, 653–659. [Google Scholar] [CrossRef]
- Kim, P.; Zhang, L.; Manjunatha, U.H.; Singh, R.; Patel, S.; Jiricek, J.; Keller, T.H.; Boshoff, H.I.; Barry, C.E., 3rd; Dowd, C.S. Structure-activity relationships of antitubercular nitroimidazoles. 1. Structural features associated with aerobic and anaerobic activities of 4- and 5-nitroimidazoles. J. Med. Chem. 2009, 52, 1317–1328. [Google Scholar] [CrossRef]
- Çelik, A.; Ares Ateş, N. The frequency of sister chromatid exchanges in cultured human peripheral blood lymphocyte treated with metronidazole in vitro. Drug Chem. Toxicol. 2006, 29, 85–94. [Google Scholar] [CrossRef]
- Walsh, J.S.; Wang, R.; Bagan, E.; Wang, C.C.; Wislocki, P.; Miwa, G.T. Structural alterations that differentially affect the mutagenic and antitrichomonal activities of 5-nitroimidazoles. J. Med. Chem. 1987, 30, 150–156. [Google Scholar]
- Cudmore, S.L.; Delgaty, K.L.; Hayward-McClelland, S.F.; Petrin, D.P.; Garber, G.E. Treatment of infections caused by metronidazole-resistant Trichomonas vaginalis. Clin. Microbiol. Rev. 2004, 17, 783–793. [Google Scholar] [CrossRef]
- Schwebke, J.R.; Burgess, D. Trichomoniasis. Clin. Microbiol. Rev. 2004, 17, 794–803. [Google Scholar] [CrossRef]
- Crowell, A.L.; Sanders-Lewis, K.A.; Evan Secor, W. In vitro metronidazole and tinidazole activities against metronidazole-resistant strains of Trichomonas vaginalis. Antimicrob. Agents Chemother. 2003, 47, 1407–1409. [Google Scholar] [CrossRef]
- Gellis, A.; Vanelle, P.; Kaafarani, M.; Benakli, K.; Crozet, M.P. Synthesis and SRN1 reactions of nitrothiazoles. Tetrahedron 1997, 53, 5471–5484. [Google Scholar] [CrossRef]
- Crozet, M.P.; Gellis, A.; Pasquier, C.; Vanelle, P.; Aune, J.-P. Electron transfer reactivity in 5-nitrouracil series. Tetrahedron Lett. 1995, 36, 525–528. [Google Scholar] [CrossRef]
- Roubaud, C.; Vanelle, P.; Maldonado, J.; Crozet, M.P. Synthesis and SRN1 reactions of 3-nitroimidazo[1,2-a]pyrimidines. Tetrahedron 1995, 51, 9643–9656. [Google Scholar] [CrossRef]
- Zink, L.; Crozet, M.D.; Remusat, V.; Terme, T.; Vanelle, P. Strong thermal effect upon competition between SN2 and SRN1 reactions of nitronate anions in 4- and 5-nitroimidazole series. (unpublished results)..
- Mahesh, M.; Murphy, J.A.; LeStrat, F.; Wessel, H.P. Reduction of arenediazonium salts by tetrakis(dimethylamino)ethylene (TDAE): Efficient formation of products derived from aryl radicals. Beilstein J. Org. Chem. 2009, 5. [Google Scholar] [CrossRef]
- Burkholder, C.; Dolbier, W.R., Jr.; Médebielle, M. Tetrakis(dimethylamino)ethylene as a useful reductant of some bromodifluoromethyl heterocycles. Application to the synthesis of new gem-difluorinated heteroarylated compounds. J. Org. Chem. 1998, 63, 5385–5394. [Google Scholar] [CrossRef]
- Médebielle, M.; Kato, K.; Dolbier, W.R., Jr. Difluoromethylation reactions of ethyl pyruvate with TDAE – A mild approach to the synthesis of ethyl 3,3-difluoro-2-hydroxy-2-methyl-4-oxobutyrates. Synlett 2002, 1541–1543. [Google Scholar]
- Giuglio-Tonolo, G.; Terme, T.; Médebielle, M.; Vanelle, P. Original reaction of p-nitrobenzyl chloride with aldehydes using tetrakis(dimethylamino)ethylene (TDAE). Tetrahedron Lett. 2003, 44, 6433–6435. [Google Scholar]
- Amiri-Attou, O.; Terme, T.; Vanelle, P. Functionalization of 6-nitrobenzo[1,3]dioxole with carbonyl compounds via TDAE methodology. Molecules 2005, 10, 545–551. [Google Scholar] [CrossRef]
- Montana, M.; Terme, T.; Vanelle, P. Original synthesis of oxiranes via TDAE methodology: Reaction of 2,2-dibromomethylquinoxaline with aromatic aldehydes. Tetrahedron Lett. 2005, 46, 8373–8376. [Google Scholar] [CrossRef]
- Khoumeri, O.; Montana, M.; Terme, T.; Vanelle, P. First TDAE approach in quinonic series: Synthesis of new 2-substituted 1,4-dimethoxy-9,10-anthraquinones. Tetrahedron 2008, 64, 11237–11242. [Google Scholar] [CrossRef]
- Giuglio-Tonolo, G.; Terme, T.; Médebielle, M.; Vanelle, P. Nitrobenzylation of α-carbonyl ester derivatives using TDAE approach. Tetrahedron Lett. 2004, 45, 5121–5124. [Google Scholar] [CrossRef]
- Montana, M.; Crozet, M.D.; Castera-Ducros, C.; Terme, T.; Vanelle, P. Rapid synthesis of new azaheterocyclic hydroxy-malonate derivatives using TDAE approach. Heterocycles 2008, 75, 925–932. [Google Scholar] [CrossRef]
- Juspin, T.; Laget, M.; Terme, T.; Azas, N.; Vanelle, P. TDAE-assisted synthesis of new imidazo[2,1-b]thiazole derivatives as anti-infectious agents. Eur. J. Med. Chem. 2010, 45, 840–845. [Google Scholar]
- Amiri-Attou, O.; Terme, T.; Vanelle, P. Original and rapid access to new alkaloid analogues of neocryptolepine: Synthesis of substituted 6-methyl-6H-indolo[2,3-b]quinolines via TDAE strategy. Synlett 2005, 3047–3050. [Google Scholar]
- Juspin, T.; Terme, P.; Vanelle, P. TDAE strategy using α-diketones: Rapid access to 2,3-diphenylquinoline and acenaphtho[1,2-b]quinoline derivatives. Synlett 2009, 1485–1489. [Google Scholar]
- Montana, M.; Terme, T.; Vanelle, P. TDAE-initiated synthesis of oxiranes in heterocyclic series: Reaction of 2-(dibromomethyl)quinoxaline with α-dicarbonyl derivatives. Lett. Org. Chem. 2010, 7, 453–456. [Google Scholar] [CrossRef]
- Crozet, M.D.; Zink, L.; Remusat, V.; Curti, C.; Vanelle, P. Efficient microwave-assisted palladium-catalyzed Suzuki-Miyaura cross-coupling reactions in 5-nitroimidazole series. Synthesis 2009, 3150–3156. [Google Scholar]
- Sample Availability: Samples of the compounds 3a-m are available from the authors.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Juspin, T.; Zink, L.; Crozet, M.D.; Terme, T.; Vanelle, P. High Functionalization of 5-Nitro-1H-imidazole Derivatives: The TDAE Approach. Molecules 2011, 16, 6883-6893. https://doi.org/10.3390/molecules16086883
Juspin T, Zink L, Crozet MD, Terme T, Vanelle P. High Functionalization of 5-Nitro-1H-imidazole Derivatives: The TDAE Approach. Molecules. 2011; 16(8):6883-6893. https://doi.org/10.3390/molecules16086883
Chicago/Turabian StyleJuspin, Thierry, Laura Zink, Maxime D. Crozet, Thierry Terme, and Patrice Vanelle. 2011. "High Functionalization of 5-Nitro-1H-imidazole Derivatives: The TDAE Approach" Molecules 16, no. 8: 6883-6893. https://doi.org/10.3390/molecules16086883
APA StyleJuspin, T., Zink, L., Crozet, M. D., Terme, T., & Vanelle, P. (2011). High Functionalization of 5-Nitro-1H-imidazole Derivatives: The TDAE Approach. Molecules, 16(8), 6883-6893. https://doi.org/10.3390/molecules16086883