Chemical Characterization of “Alcaparras” Stoned Table Olives from Northeast Portugal
Abstract
:1. Introduction
2. Results and Discussion
2.1. Proximate Chemical Composition
| Samples | Moisture | Total fat | Crude protein | Ash | Total sugars * |
|---|---|---|---|---|---|
| 2004 | |||||
| 1 | 74.8 ± 0.5 | 13.4 ± 0.0 | - | - | |
| 2 | 74.5 ± 0.2 | 14.2 ± 0.0 | - | - | |
| 3 | 78.3 ± 0.5 | 14.3 ± 0.0 | - | - | |
| 4 | 71.6 ± 1.6 | 17.6 ± 0.0 | - | - | |
| 5 | 75.1 ± 0.7 | 13.8 ± 0.0 | - | - | |
| 6 | 66.8 ± 3.3 | 15.7 ± 0.0 | - | - | |
| 7 | 74.1 ± 1.6 | 13.1 ± 0.0 | - | - | |
| 8 | 70.1 ± 1.5 | 22.0 ± 0.0 | - | - | |
| 9 | 72.6 ± 1.4 | 9.1 ± 0.0 | - | - | |
| 10 | 73.7 ± 1.8 | 12.6 ± 0.0 | - | - | |
| Average | 73.2 ± 3.2 a | 14.6 ± 0.0 a,b | - | - | |
| 2005 | |||||
| 11 | 72.2 ± 0.2 | 13.3 ± 1.6 | 0.9 ± 0.0 | 5.6 ± 0.0 | 5.3 |
| 12 | 69.0 ± 2.8 | 21.4 ± 0.2 | 1.4 ± 0.0 | 1.8 ± 0.1 | 3.6 |
| 13 | 73.3 ± 0.9 | 12.9 ± 3.6 | 0.9 ± 0.0 | 3.6 ± 0.0 | 6.5 |
| 14 | 73.2 ± 1.3 | 10.8 ± 0.8 | 0.8 ± 0.0 | 3.1 ± 0.3 | 9.4 |
| 15 | 71.8 ± 0.6 | 10.4 ± 4.6 | 0.8 ± 0.0 | 4.7 ± 1.1 | 9.6 |
| 16 | 70.2 ± 0.9 | 18.5 ± 1.0 | 0.9 ± 0.0 | 3.6 ± 0.4 | 4.1 |
| 17 | 59.5 ± 0.1 | 28.4 ± 4.3 | 1.7 ± 0.0 | 1.8 ± 0.8 | 5.3 |
| 18 | 58.9 ± 0.5 | 29.3 ± 2.1 | 1.5 ± 0.0 | 5.1 ± 0.5 | 2.3 |
| 19 | 60.3 ± 1.6 | 22.8 ± 0.1 | 1.7 ± 0.0 | 2.9 ± 0.1 | 9.6 |
| 20 | 65.7 ± 0.7 | 18.7 ± 0.8 | 1.5 ± 0.0 | 1.7 ± 0.0 | 9.7 |
| Average | 67.5 ± 5.9 b | 18.3 ± 6.1 a | 1.2 ± 0.4 a | 3.5 ± 1.4 a | 6.5 ± 2.8 |
| 2006 | |||||
| 21 | 77.3 ± 0.5 | 10.4 ± 1.6 | 1.3 ± 0.0 | 2.8 ± 0.1 | 5.5 |
| 22 | 67.0 ± 0.1 | 17.8 ± 0.4 | 1.4 ± 0.0 | 2.3 ± 0.1 | 8.7 |
| 23 | 74.7 ± 0.2 | 15.8 ± 0.2 | 1.0 ± 0.0 | 1.0 ± 0.0 | 4.8 |
| 24 | 75.2 ± 0.9 | 14.2 ± 0.4 | 1.0 ± 0.0 | 1.5 ± 0.1 | 5.3 |
| 25 | 77.4 ± 0.8 | 9.5 ± 0.7 | 0.8 ± 0.0 | 8.3 ± 0.0 | 1.2 |
| 26 | 80.0 ± 0.4 | 7.6 ± 0.2 | 1.0 ± 0.0 | 3.3 ± 0.1 | 5.1 |
| 27 | 78.7 ± 0.3 | 9.4 ± 2.8 | 1.1 ± 0.0 | 4.6 ± 0.1 | 3.5 |
| 28 | 73.3 ± 0.7 | 17.6 ± 0.9 | 1.2 ± 0.0 | 1.2 ± 0.0 | 4.0 |
| 29 | 78.7 ± 0.9 | 9.1 ± 0.2 | 1.0 ± 0.0 | 4.6 ± 0.1 | 3.8 |
| 30 | 76.6 ± 0.1 | 14.0 ± 0.2 | 0.9 ± 0.0 | 2.1 ± 0.0 | 3.6 |
| Average | 76.0 ± 3.5 a | 12.6 ± 3.8 b | 1.1 ± 0.2 a | 3.2 ± 2.2 a | 4.6 ± 1.9 |
2.2. Fatty Acids Composition
| Fatty acid | 2004 | 2005 | 2006 | Average |
|---|---|---|---|---|
| Palmitic acid (C16:0) | 12.49 ± 0.94 b | 12.88 ± 0.95 a,b | 13.66 ± 0.76 a | 13.3 ± 0.9 |
| Palmitoleic acid (C16:1) | 0.84 ± 0.15 b | 0.95 ± 0.21 a,b | 1.05 ± 0.08 a | 1.0 ± 0.2 |
| Heptadecanoic acid (C17:0) | 0.11 ± 0.07 a | 0.12 ± 0.08 a | 0.08 ± 0.06 a | 0.1 ± 0.1 |
| Heptadecenoic acid (C17:1) | 0.14 ± 0.10 a | 0.18 ± 0.13 a | 0.15 ± 0.09 a | 0.2 ± 0.1 |
| Stearic acid (C18:0) | 2.34 ± 0.36 a,b | 2.75 ± 0.51 a | 2.15 ± 0.34 b | 2.4 ± 0.5 |
| Oleic acid (C18:1) | 78.43 ± 2.80 a | 77.11 ± 1.37 a | 77.79 ± 1.30 a | 77.4 ± 1.3 |
| Linoleic acid (C18:2) | 4.11 ± 2.39 a | 4.26 ± 1.17 a | 3.06 ± 0.55 a | 3.7 ± 1.1 |
| Linolenic acid (C18:3) | 0.66 ± 0.06 b | 0.73 ± 0.07 b | 0.88 ± 0.09 a | 0.8 ± 0.1 |
| Arachidic acid (C20:0) | 0.23 ± 0.06 b | 0.44 ± 0.06 a | 0.47 ± 0.04 a | 0.5 ± 0.0 |
| Eicosenopic acid (C20:1) | 0.34 ± 0.03 b | 0.30 ± 0.03 c | 0.39 ± 0.02 a | 0.4 ± 0.1 |
| Behenic acid (C22:0) | 0.14 ± 0.02 a,b | 0.13 ± 0.02 b | 0.16 ± 0.03 a | 0.2 ± 0.0 |
| Lignoceric acid (C24:0) | 0.08 ± 0.01 b | 0.03 ± 0.01 c | 0.14 ± 0.06 a | 0.1 ± 0.1 |
| SFA | 15.43 ± 0.77 b | 16.41 ± 0.78 a,b | 16.46 ± 1.25 a | 16.5 ± 0.9 |
| MUFA | 79.75 ± 2.86 a | 78.56 ± 1.46 a | 79.89 ± 1.27 a | 79.2 ± 1.5 |
| PUFA | 4.77 ± 2.41 a | 4.99 ± 1.21 a | 3.44 ± 0.55 a | 4.2 ± 1.2 |
| trans isomers | 0.05 ± 0.01 a | 0.04 ± 0.01 a | 0.05 ± 0.05 a | 0.04 ± 0.03 |
2.3. Tocopherols Composition
| Samples | α-tocopherol | β-tocopherol | γ-tocopherol | δ-tocopherol | Total |
|---|---|---|---|---|---|
| 2005 | |||||
| 11 | 67.4 ± 1.0 | 1.7 ± 0.2 | 1.2 ± 0.1 | 0.3 ± 0.0 | 70.7 ± 1.2 |
| 12 | 68.7 ± 0.2 | 0.7 ± 0.0 | 9.7 ± 0.3 | 0.4 ± 0.0 | 79.7 ± 0.2 |
| 13 | 77.1 ± 0.3 | 1.2 ± 0.0 | 1.0 ± 0.1 | 0.1 ± 0.0 | 79.3 ± 0.4 |
| 14 | 78.5 ± 1.5 | 1.1 ± 0.2 | 3.4 ± 0.2 | 0.1 ± 0.0 | 83.1 ± 1.8 |
| 15 | 65.8 ± 0.7 | 1.0 ± 0.1 | 2.2 ± 0.2 | 0.1 ± 0.0 | 69.1 ± 0.9 |
| 16 | 52.5 ± 1.7 | 1.0 ± 0.1 | 2.3 ± 0.4 | 0.1 ± 0.0 | 55.8 ± 2.1 |
| 17 | 47.8 ± 0.3 | 1.0 ± 0.1 | 7.3 ± 0.2 | 0.3 ± 0.0 | 56.4 ± 0.5 |
| 18 | 62.5 ± 2.9 | 0.8 ± 0.1 | 0.7 ± 0.2 | 0.1 ± 0.0 | 64.2 ± 3.2 |
| 19 | 81.5 ± 3.9 | 1.4 ± 0.2 | 2.7 ± 0.4 | 0.1 ± 0.0 | 85.7 ± 4.3 |
| 20 | 57.4 ± 5.1 | 1.0 ± 0.1 | 2.5 ± 0.2 | 0.2 ± 0.0 | 60.4 ± 6.1 |
| Average | 65.9 ± 11.0 a | 1.0 ± 0.3 a | 3.3 ± 2.9 a | 0.2 ± 0.1 a | 70.4 ± 11.0 a |
| 2006 | |||||
| 21 | 60.8 ± 3.8 | 7.0 ± 0.5 | 3.0 ± 0.1 | 0.3 ± 0.0 | 71.2 ± 4.2 |
| 22 | 45.0 ± 2.0 | 1.2 ± 0.1 | 1.0 ± 0.1 | 0.4 ± 0.0 | 47.6 ± 2.1 |
| 23 | 66.3 ± 2.7 | 0.6 ± 0.0 | 3.0 ± 0.2 | 0.1 ± 0.0 | 70.1 ± 2.8 |
| 24 | 58.1 ± 3.5 | 1.0 ± 0.1 | 3.2 ± 0.4 | 0.1 ± 0.0 | 62.5 ± 3.9 |
| 25 | 83.3 ± 4.1 | 0.7 ± 0.0 | 4.4 ± 0.2 | 0.3 ± 0.0 | 88.8 ± 4.3 |
| 26 | 87.7 ± 4.6 | 1.4 ± 0.1 | 2.3 ± 0.2 | 0.1 ± 0.0 | 91.6 ± 4.9 |
| 27 | 53.9 ± 0.3 | 1.4 ± 0.0 | 0.9 ± 0.1 | 0.3 ± 0.2 | 56.4 ± 0.1 |
| 28 | 30.7 ± 0.6 | 1.2 ± 0.0 | 1.5 ± 0.0 | 0.2 ± 0.1 | 33.7 ± 0.5 |
| 29 | 55.7 ± 4.4 | 0.7 ± 0.0 | 3.9 ± 0.3 | 0.5 ± 0.0 | 60.8 ± 4.6 |
| 30 | 106.3 ± 4.2 | 1.6 ± 0.2 | 1.1 ± 0.1 | 0.6 ± 0.0 | 109.5 ± 4.4 |
| Average | 64.8 ± 21.6 a | 1.7 ± 1.8 a | 2.4 ± 1.2 a | 0.3 ± 0.2 a | 69.2 ± 21.9 a |
2.4. Statistical Analysis
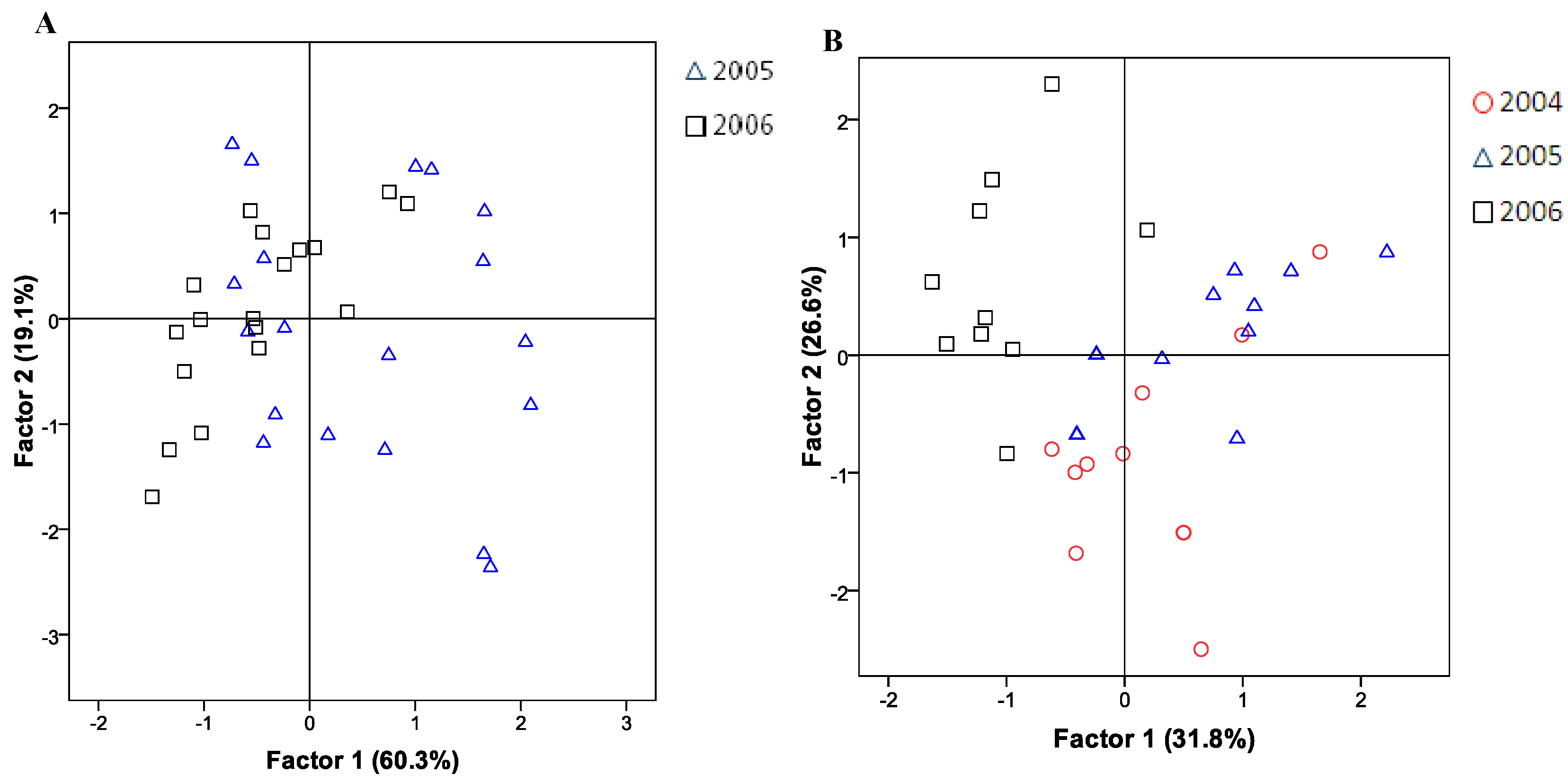
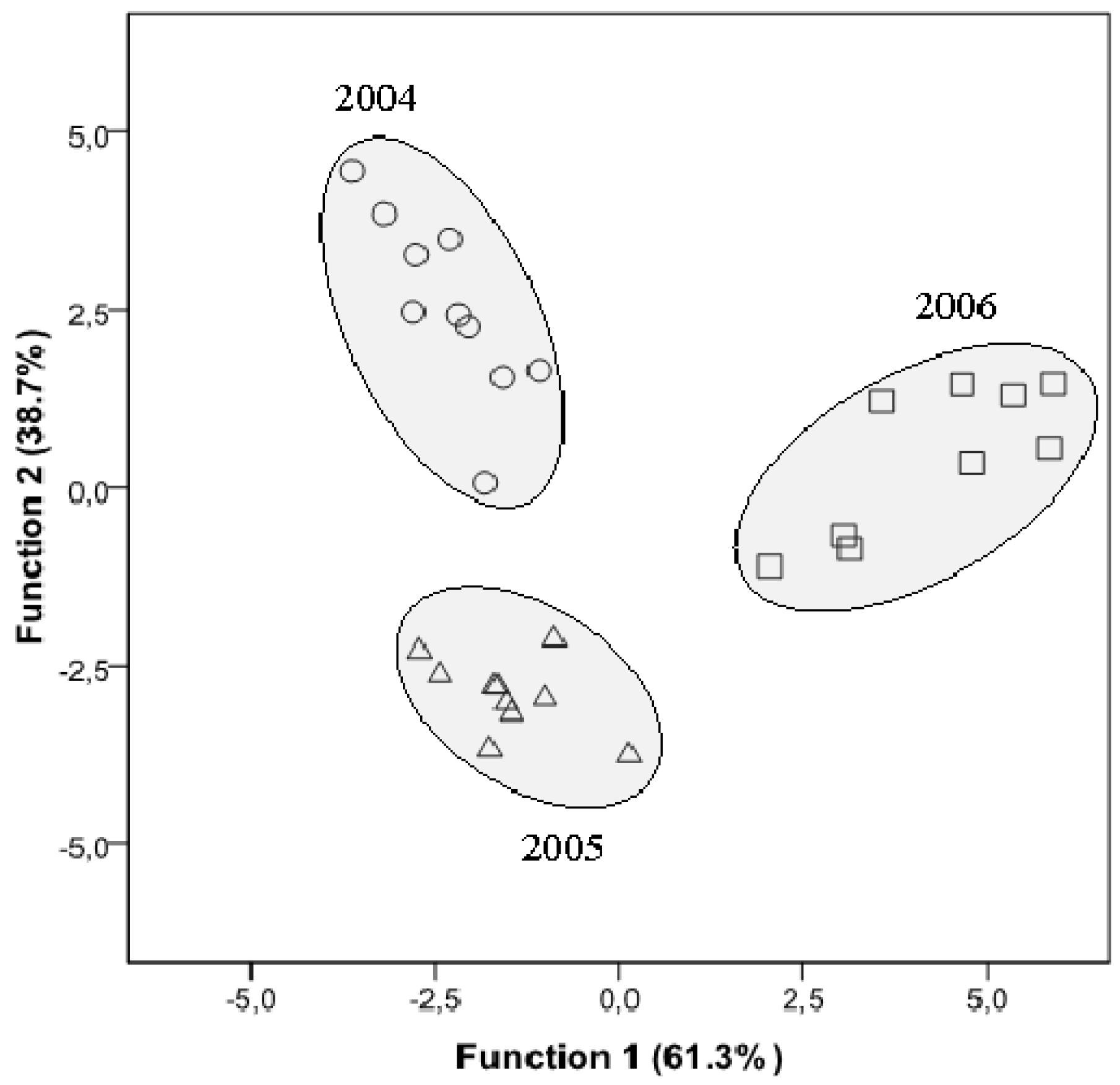
3. Experimental
3.1. Samples
3.2. Samples Preparation
3.3. Chemical Analysis
3.3.1. Pulp Analysis
3.4. Oil Analysis
3.4.1. Fatty Acid Composition
3.4.2. Tocopherol Composition
3.5. Statistical Analysis
4. Conclusions
| 100 g | Serving size a | |
|---|---|---|
| Energy (kcal) | 156 | 31 |
| Energy (kJ) | 652 | 130 |
| Edible part | 100% | |
| Moisture (g) | 72.5 | 14.5 |
| Proteins (g) | 1.1 | 0.2 |
| Minerals (g) | 3.4 | 0.7 |
| Carbohydrates (g) b | 4.6 | 0.9 |
| Fibre (g) | 2.7 c | 0.5 |
| Fat (g) | 14.6 | 2.5 |
| Saturated fatty acids | 2.1 | 0.4 |
| Monounsaturated fatty acids | 10.0 | 2.5 |
| Polyunsaturated fatty acids | 0.5 | 0.1 |
| Trans fatty acids | tr | tr |
| PUFA:SFA | 0.3 | |
| (MUFA + PUFA):SFA | 5.0 | |
| Vitamin E (mg) | 1.2 | 0.3 |
| Polyphenols (g) d | 1.3 | 0.3 |
Acknowledgments
Conflict of Interest
References
- Trichopoulou, A.; Lagiou, P. Healthy traditional Mediterranean diet: An expression of culture, history and lifestyle. Nutr. Rev. 1997, 55, 383–389. [Google Scholar] [CrossRef]
- COI/OT/NC n°1. Trade standard applying to table olives. International Olive Oil Council. 2004; p. 17.
- Hassapidou, M.N.; Balatsouras, D.D.; Manoukas, A.G. Effect of processing upon the tocopherol and tocotrienol composition of table olives. Food Chem. 1994, 50, 111–114. [Google Scholar] [CrossRef]
- Marsilio, V.; Campestre, C.; Lanza, B. Phenolic compounds change during California-style ripe olive processing. Food Chem. 2001, 74, 55–60. [Google Scholar] [CrossRef]
- Montaño, A.; Casado, F.J.; Castro, A.; Sánchez, A.H.; Rejano, L. Influence of processing, storage time, and pasteurization upon the tocopherol and amino acid contents of treated green table olives. Eur. Food Res. Technol. 2005, 220, 255–260. [Google Scholar] [CrossRef]
- Georget, D.M.R.; Smith, A.C.; Waldron, K.W. Effect of ripening on the mechanical properties of Portuguese and Spanish varieties of olive (Olea europaea L). J. Sci. Food Agric. 2001, 81, 448–454. [Google Scholar] [CrossRef]
- Sakouhi, F.; Harrabi, S.; Absalon, C.; Sbei, K.; Boukhchina, S.; Kallel, H. α-Tocopherol and fatty acids contents of some Tunisian table olives (Olea europea L.): Changes in their composition during ripening and processing. Food Chem. 2008, 108, 833–839. [Google Scholar] [CrossRef]
- Romero, C.; Brenes, M.; Yousfi, K.; García, P.; García, A.; Garrido, A. Effect of cultivar and processing method on the contents of polyphenols in table olives. J. Agric. Food Chem. 2004, 52, 479–484. [Google Scholar] [CrossRef]
- Marsilio, V.; Campestre, C.; Lanza, B.; de Angelis, M. Sugar and polyol compositions of some European olive fruit varieties (Oleae europaea L.) suitable for table purposes. Food Chem. 2001, 72, 485–490. [Google Scholar] [CrossRef]
- Montaño, A.; Sánchez, A.H.; Casado, F.J.; de Castro, A.; Rejano, L. Chemical profile of industrially fermented green olives of different varieties. Food Chem. 2003, 82, 297–302. [Google Scholar]
- Marsilio, V.; d’Andria, R.; Lanza, B.; Russi, F.; Iannucci, E.; Lavini, A.; Morelli, G. Effect of irrigation and lactic acid bacteria inoculants on the phenolic fraction, fermentation and sensory characteristics of olive (Olea europaea L. cv. Ascolana tenera) fruits. J. Sci. Food Agric. 2006, 86, 1005–1013. [Google Scholar] [CrossRef]
- Bianchi, G. Lipids and phenols in table olives. Eur. J. Lipid Sci. Technol. 2003, 105, 229–242. [Google Scholar] [CrossRef]
- Simopoulos, A.P. The Mediterranean diets: What is so special about the diet of Greece? The scientific evidence. J. Nutr. 2001, 131, 3065–3073. [Google Scholar]
- Blekas, G.; Vassilakis, C.; Harizanis, C.; Tsimidou, M.; Boskou, D.G. Biophenols in table olives. J. Agric. Food Chem. 2002, 50, 3688–3692. [Google Scholar] [CrossRef]
- Pereira, J.A.; Pereira, A.P.; Ferreira, I.C.F.R.; Valentão, P.; Andrade, P.; Seabra, R.; Estevinho, L.; Bento, A. Table olives from Portugal: Phenolic compounds, antioxidant potential, and antimicrobial activity. J. Agric. Food Chem. 2006, 54, 8425–8431. [Google Scholar]
- Romero, C.; García, P.; Brenes, M.; García, A.; Garrido, A. Phenolic compounds in natural black Spanish olive varieties. Eur. Food Res. Technol. 2002, 215, 489–496. [Google Scholar] [CrossRef]
- Lanza, B.; di Serio, M.; Iannucci, E.; Russi, F.; Marfisi, P. Nutritional, textural and sensorial characterisation of Italian table olives (Olea europeae L. cv “Intosso d’Abruzzo”). Int. J. Food Sci. Technol. 2010, 45, 67–74. [Google Scholar]
- Bogani, P.; Galli, C.; Villa, M.; Visioli, F. Postprandial anti-inflammatory and antioxidant effects of extra virgin olive oil. Atherosclerosis 2007, 190, 181–186. [Google Scholar] [CrossRef]
- Cheeseeman, K.H.; Slater, T.F. An introduction to free radical biochemistry. Br. Med. Bull. 1993, 49, 481–493. [Google Scholar]
- Sousa, A.; Ferreira, I.C.F.R.; Barros, L.; Bento, A.; Pereira, J.A. Effect of solvent and extraction temperatures on the antioxidant potential of traditional stoned table olives alcaparras. LWT-Food Sci. Technol. 2008, 41, 739–745. [Google Scholar] [CrossRef]
- Sousa, A.; Ferreira, I.C.F.R.; Calhelha, R.; Andrade, P.B.; Valentão, P.; Seabra, R.; Estevinho, L.; Bento, A.; Pereira, J.A. Phenolics and antimicrobial activity of traditional stoned table olives “alcaparra”. Bioorg. Med. Chem. 2006, 14, 8533–8538. [Google Scholar]
- Malheiro, R.; Sousa, A.; Casal, S.; Bento, A.; Pereira, J.A. Cultivar effect on the phenolic composition and antioxidant potential of stoned table olives. Food Chem. Toxicol. 2011, 49, 449–456. [Google Scholar]
- nal, K.; Nergiz, C. The effect of table olives preparing methods and storage on the composition and nutritive value of olives. Grasas y Aceites 2003, 54, 71–76. [Google Scholar]
- AOAC, Official Methods of Analysis, 16th ed; Association of Official Analytical Chemists: Arlington, VA, USA, 1995.
- AOAC, Official Methods of Analysis, 17th; Horwitz, W. (Ed.) Association of Official Analytical Chemists: Arlington, VA, USA, 2000; Volume II, pp. 1–3.
- Commission Directive 2008/100/EC, of 28 October 2008, amending Council Directive 90/496/EEC on nutrition labeling for foodstuffs as regards recommended daily allowances, energy conversion factors and definitions. In Off. J. Eur. Union; 2008; L285/9.
- Commission Regulation (EEC) No 2568/91 of 11 July 1991, on the characteristics of olive oil and olive-residue oil and on the relevant methods of analysis, 1991R2568, 01.01.2008, 021.001. pp. 1–112.
- ISO 9936. Animal and vegetable fats and oils-Determination of tocopherol and tocotrienol contents by high-performance liquid chromatography. 2006.
- Amaral, J.S.; Casal, S.; Oliveira, M.B.; Seabra, R.M. Development and evaluation of a Normal Phase Liquid Chromatographic method for the determination of tocopherols and tocotrienols in walnuts. J. Liquid Chromatogr. Relat. Technol. 2005, 28, 785–795. [Google Scholar] [CrossRef]
- López, A.; García, P.; Garrido, A. Multivariate characterization of table olives according to their mineral nutrient composition. Food Chem. 2008, 106, 369–378. [Google Scholar] [CrossRef]
- Maroco, J. Análise Estatística,com Utilização do SPSS; Edições Sílabo: Lisboa, Portugal, 2003. [Google Scholar]
- Rencher, A.C. Methods of Multivariate Analysis; John Willey: New York, NY, USA, 1995. [Google Scholar]
- Borzillo, A.; Iannotta, N.; Uccella, N. Oinotria table olives: Quality evaluation during ripening and processing by biomolecular components. Eur. Food Res. Technol. 2000, 212, 113–121. [Google Scholar] [CrossRef]
- López-López, A.; Montaño, A.; Ruíz-Méndez, M.V.; Garrido-Fernández, A. Sterols, fatty alcohols, and triterpenic alcohols in commercial table olives. J. Am. Oil Chem. Soc. 2008, 85, 253–262. [Google Scholar] [CrossRef]
- Gutiérrez, F.; Jímenez, B.; Ruíz, A.; Albi, M.A. Effect of olive ripeness on the oxidative stability of virgin olive oil extracted from the varieties Picual and Hojiblanca and on the different components involved. J. Agric. Food Chem. 1999, 47, 121–127. [Google Scholar] [CrossRef]
- López-López, A.; Jiménez-Araujo, A.; García-García, P.; Garrido-Fernández, A. Multivariate analysis for the evaluation of fiber, sugars, and organic acids in commercial presentations of table olives. J. Agric. Food Chem. 2007, 55, 10803–10811. [Google Scholar] [CrossRef]
- Kailis, S.; Harris, D. Producing Table Olives; Landlinks Press: Collingwood, Australia, 2007. [Google Scholar]
- Kanavouras, A.; Gazouli, M.; Leonidas, L.T.; Petrakis, C. Evaluation of black table olives in different brines. Grasas y Aceites 2005, 56, 106–115. [Google Scholar]
- López, A.; Montaño, A.; Garcia, P.; Garrido, A. Fatty acid profile of table olives and its multivarate characterization using unsupervised (PCA) and supervised (DA) chemometrics. J. Agric. Food Chem. 2006, 54, 6747–6753. [Google Scholar] [CrossRef]
- Pereira, J.A.; Casal, S.; Bento, A.; Oliveira, M.B.P.P. Influence of olive storage period on oil quality of three Portuguese cultivars of Olea europea, Cobrançosa, Madural and Verdeal Transmontana. J. Agric. Food Chem. 2002, 50, 6335–6340. [Google Scholar] [CrossRef]
- Pereira, J.A.; Alves, R.; Casal, S.; Oliveira, M.B.P.P. Effect of olive fruit fly infestation on the quality of olive oil from cultivars Cobrançosa, Madural and Verdeal Transmontana. Ital. J. Food Sci. 2004, 16, 355–365. [Google Scholar]
- Malheiro, R.; Sousa, A.; Casal, S.; Bento, A.; Pereira, J.A. Cultivar effect on the phenolic composition and antioxidant potential of stoned table olives. Food Chem. Toxicol. 2011, 49, 450–457. [Google Scholar] [CrossRef]
- Malheiro, R.; de Pinho, P.G.; Casal, S.; Bento, A.; Pereira, J.A. Determination of the volatile profile of stoned table olives from diferente varieties by using HS-SPME and GC/IT-MS. J. Sci. Food Agric. 2011, 91, 1693–1701. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sousa, A.; Casal, S.; Bento, A.; Malheiro, R.; Oliveira, M.B.P.P.; Pereira, J.A. Chemical Characterization of “Alcaparras” Stoned Table Olives from Northeast Portugal. Molecules 2011, 16, 9025-9040. https://doi.org/10.3390/molecules16119025
Sousa A, Casal S, Bento A, Malheiro R, Oliveira MBPP, Pereira JA. Chemical Characterization of “Alcaparras” Stoned Table Olives from Northeast Portugal. Molecules. 2011; 16(11):9025-9040. https://doi.org/10.3390/molecules16119025
Chicago/Turabian StyleSousa, Anabela, Susana Casal, Albino Bento, Ricardo Malheiro, M. Beatriz P.P. Oliveira, and José Alberto Pereira. 2011. "Chemical Characterization of “Alcaparras” Stoned Table Olives from Northeast Portugal" Molecules 16, no. 11: 9025-9040. https://doi.org/10.3390/molecules16119025
APA StyleSousa, A., Casal, S., Bento, A., Malheiro, R., Oliveira, M. B. P. P., & Pereira, J. A. (2011). Chemical Characterization of “Alcaparras” Stoned Table Olives from Northeast Portugal. Molecules, 16(11), 9025-9040. https://doi.org/10.3390/molecules16119025








