Effects of Different Maturity Stages on Antioxidant Content of Ivorian Gnagnan (Solanum indicum L.) Berries
Abstract
:1. Introduction
2. Results and Discussion
2.1. Colour analysis

| Colourimetera | |||
|---|---|---|---|
| L* | a* | b* | |
| Green | 44.1 ± 5.5b | -5.1 ± 3.4c | 26.4 ± 8.4b |
| Yellow | 48.8 ± 3.8a | 28.5 ± 3.9b | 40.9 ± 6.4a |
| Red | 42.9 ± 2.2b | 33.7 ± 3.5a | 30.7 ± 30.6b |
| Image analysisa | |||
| Green | 51.0 ± 6.3a | -20.1 ± 2.5b | 51.8 ± 7.9ab |
| Yellow | 49.5 ± 2.2ab | 31.8 ± 8.9a | 57.6 ± 2.1a |
| Red | 40.1 ± 3.7b | 41.0 ± 8.7a | 44.7 ± 3.9b |
2.2. Ascorbic acid
| Ascorbic acid | α-carotene* | β-carotene | lycopene | |
|---|---|---|---|---|
| Green | 8.46 ± 0.81a | N.Q. | 0.02 ± 0.01a | N.D. |
| Yellow | 8.54 ± 0.28a | N.Q. | 0.06 ± 0.06a | N.D. |
| Red | 6.67 ± 0.32b | 0.15 ± 0.06 | 1.16 ± 0.34b | 1.84 ± 0.52 |
2.3. Carotenoids
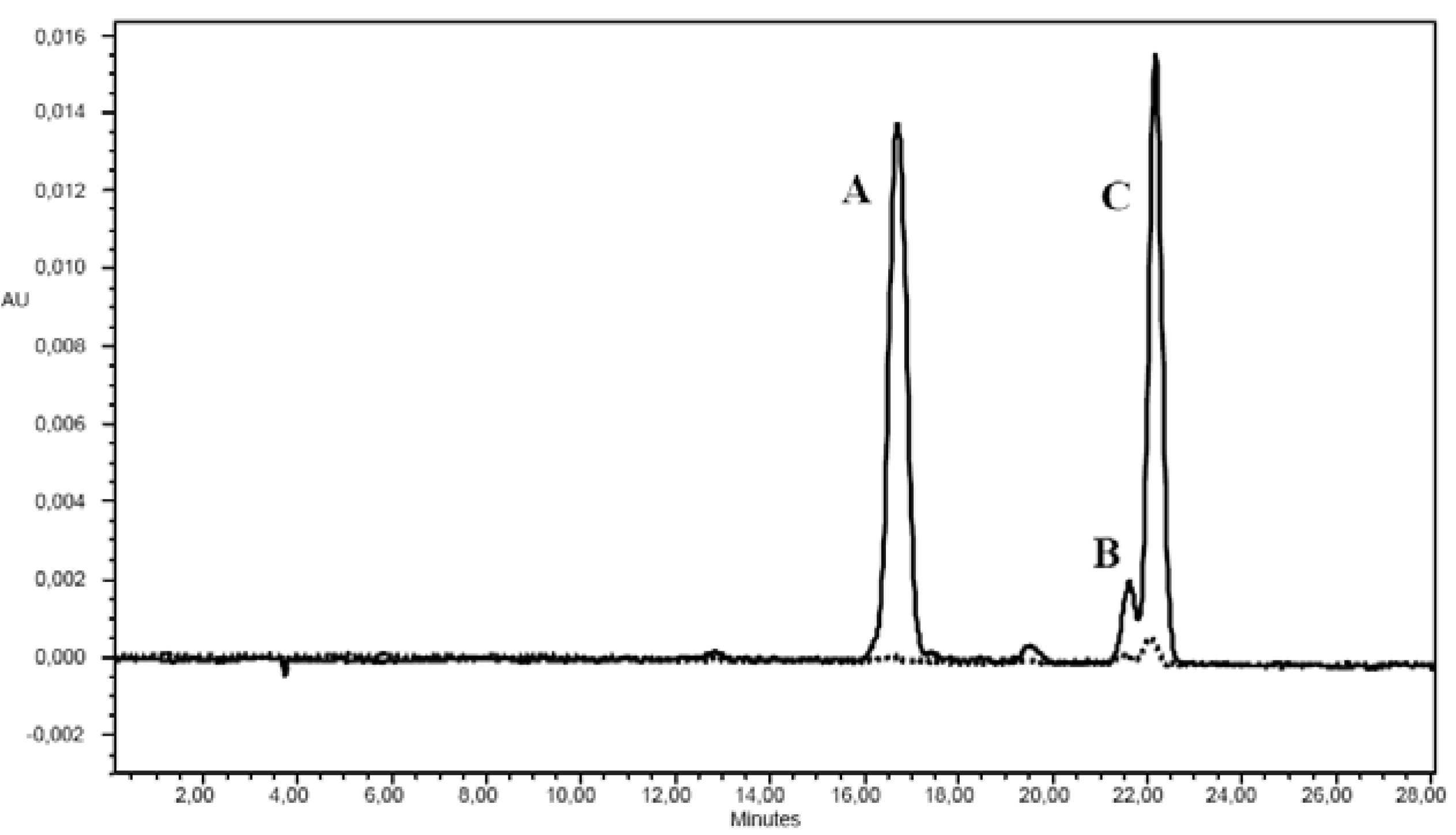
2.4. TAC and Total Polyphenols
| FRAP * | Total polyphenols ** | |
|---|---|---|
| Green | 0.58 ± 0.06a | 102.09 ± 22.64a |
| Yellow | 0.63 ± 0.05a | 135.15 ± 47.04a |
| Red | 0.88 ± 0.11b | 123.48 ± 34.38a |
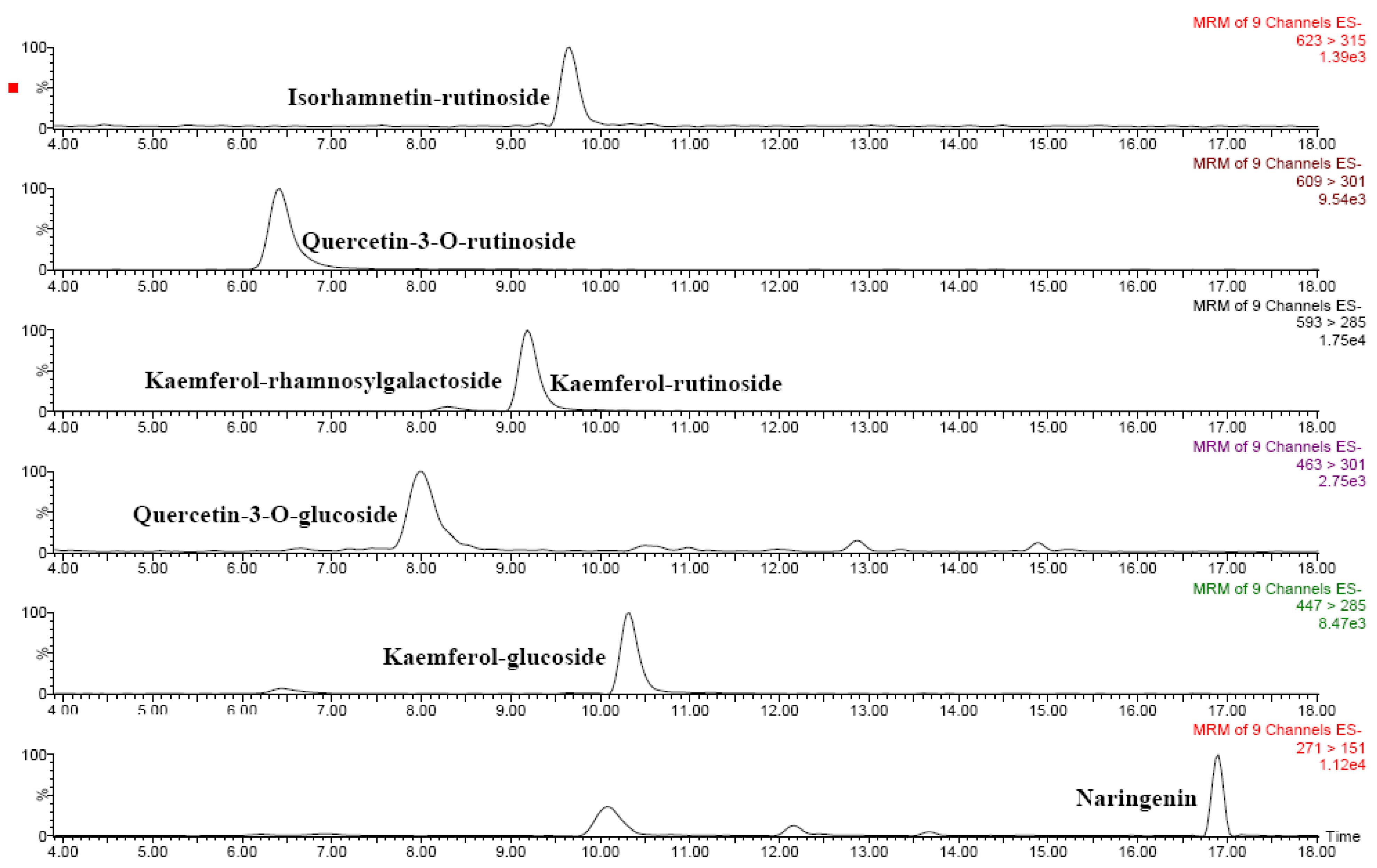
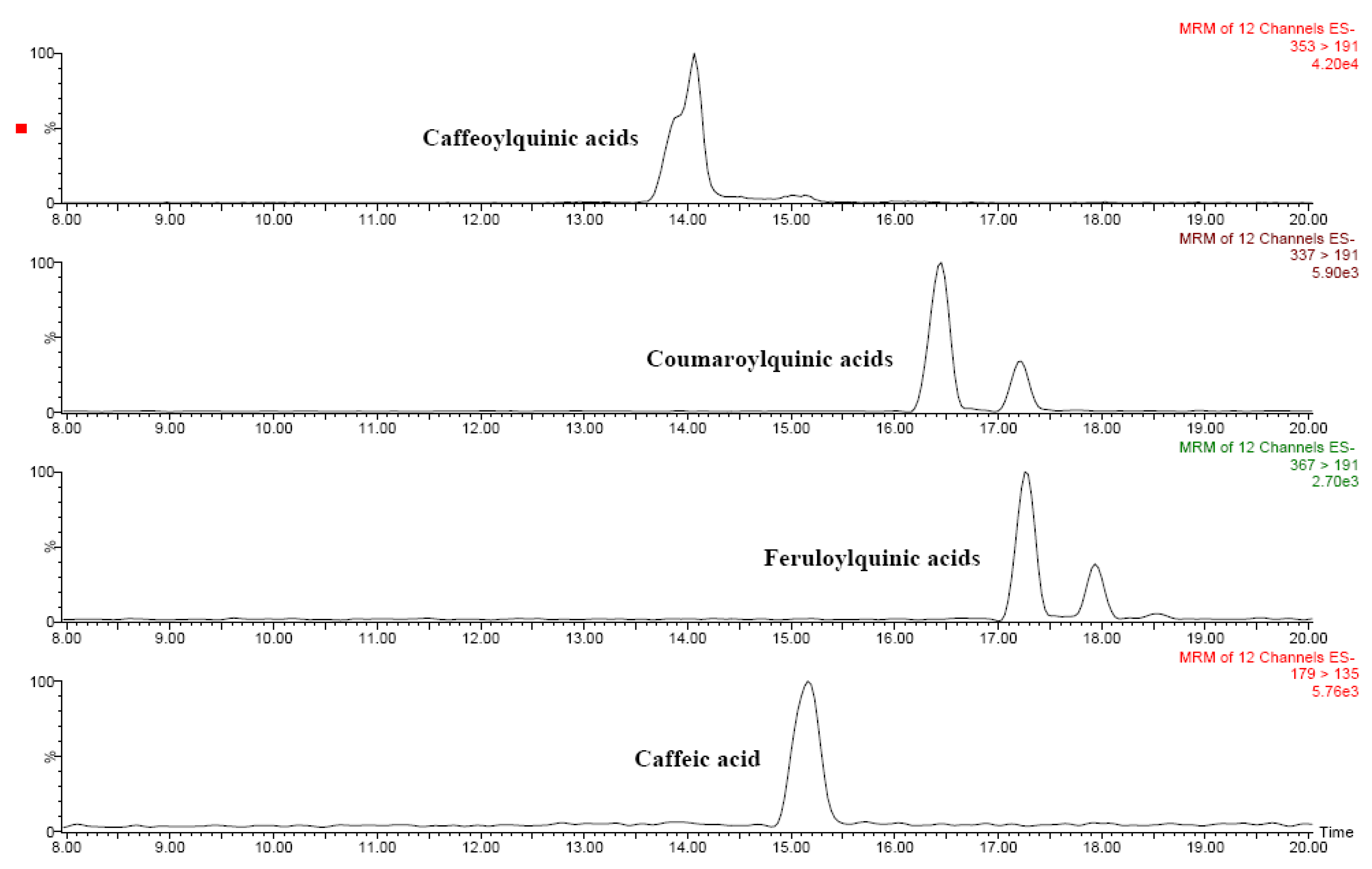
2.5. Phenolic compounds
| Phenolic acids | ||
|---|---|---|
| Compound | [M-H]- (m/z) | MS2 ions (m/z) |
| p-Coumaric acid | 163 | 119 |
| Caffeic acid | 179 | 135 |
| Coumaroylquinic acids | 337 | 191, 173, 163 |
| Caffeoylquinic acids | 353 | 191, 173, 179 |
| Feruloylquinic acids | 367 | 191, 173, 193 |
| Flavonoids | ||
| Compound | [M-H]- (m/z) | MS2 ions (m/z) |
| Naringenin | 271 | 151 |
| Quercetin* | 301 | 151 |
| Quercetin-3- O-glucoside | 463 | 301 |
| Kaemferol-glucoside | 447 | 285 |
| Quercetin-3- O-rutinoside | 609 | 301 |
| Kaemferol-rutinoside | 593 | 285 |
| Kaemferolrhamnosyl-galactoside* | 593 | 285 |
| Isorhamnetin-rutinoside | 623 | 315 |
| Phenolic acids | ||||||
|---|---|---|---|---|---|---|
| p-CoA | CA | CoQA | CQA | FQA | ||
| Green | 18.8 ± 0.7 | 2.0 ± 1.0 | 356.0 ± 26.4 | 932.3 ± 184.8 | 277.9 ± 11.2 | |
| Yellow | 19.2 ± 0.9 | 3.4 ± 0.3 | 348.4 ± 27.7 | 887.3 ± 42.4 | 246.8 ± 7.7 | |
| Red | 15.8 ± 1.1 | 18.9 ± 0.8 | 517.4 ± 34.4 | 5303.3 ± 523.6 | 250.2 ± 20.4 | |
| Flavonoids | ||||||
| NAR | Q-3-Glu | K-Glu | Q-3-Rut | K-Rut | IR-Rut | |
| Green | 95.5 ± 16.3 | 38.6 ± 19.3 | 154.7 ± 8.5 | 422.4 ± 31.2 | 378.8 ± 4.5 | NQ |
| Yellow | 128.2 ± 18.5 | 36.0 ± 18.0 | 167.4 ± 13.3 | 311.3 ± 28.4 | 363.2 ± 22.4 | NQ |
| Red | 461.7 ± 71.7 | 386.8 ± 30.1 | 800.6 ± 32.3 | 3820.5 ± 215.1 | 5869.5 ± 321.2 | 440.9 ± 15.0 |
| FRAP | total phenols | α-carotene | β-carotene | lycopene | ascorbic acid | L* | a* | b* | |
|---|---|---|---|---|---|---|---|---|---|
| FRAP | 1.000 | ||||||||
| polyphenols | 0.068 | 1.000 | |||||||
| α-carotene | 0.812** | 0.136 | 1.000 | ||||||
| β-carotene | 0.853** | 0.074 | 0.937** | 1.000 | |||||
| lycopene | 0.846** | 0.073 | 0.938** | 0.999** | 1.000 | ||||
| ascorbic acid | -0.833** | -0.032 | -0.908** | -0.966** | -0.966 | 1.000 | |||
| L* | -0.239 | 0.057 | -0.400* | -0.360 | -0.381* | 0.267 | 1.000 | ||
| a* | 0.658** | 0.355 | 0.538** | 0.605** | 0.585** | -0.537** | 0.048 | 1.000 | |
| b* | -0.017 | 0.259 | -0.196 | -0.159 | -0.186 | 0.143 | 0.852** | 0.396* | 1.000 |
2.6. Factor Analysis
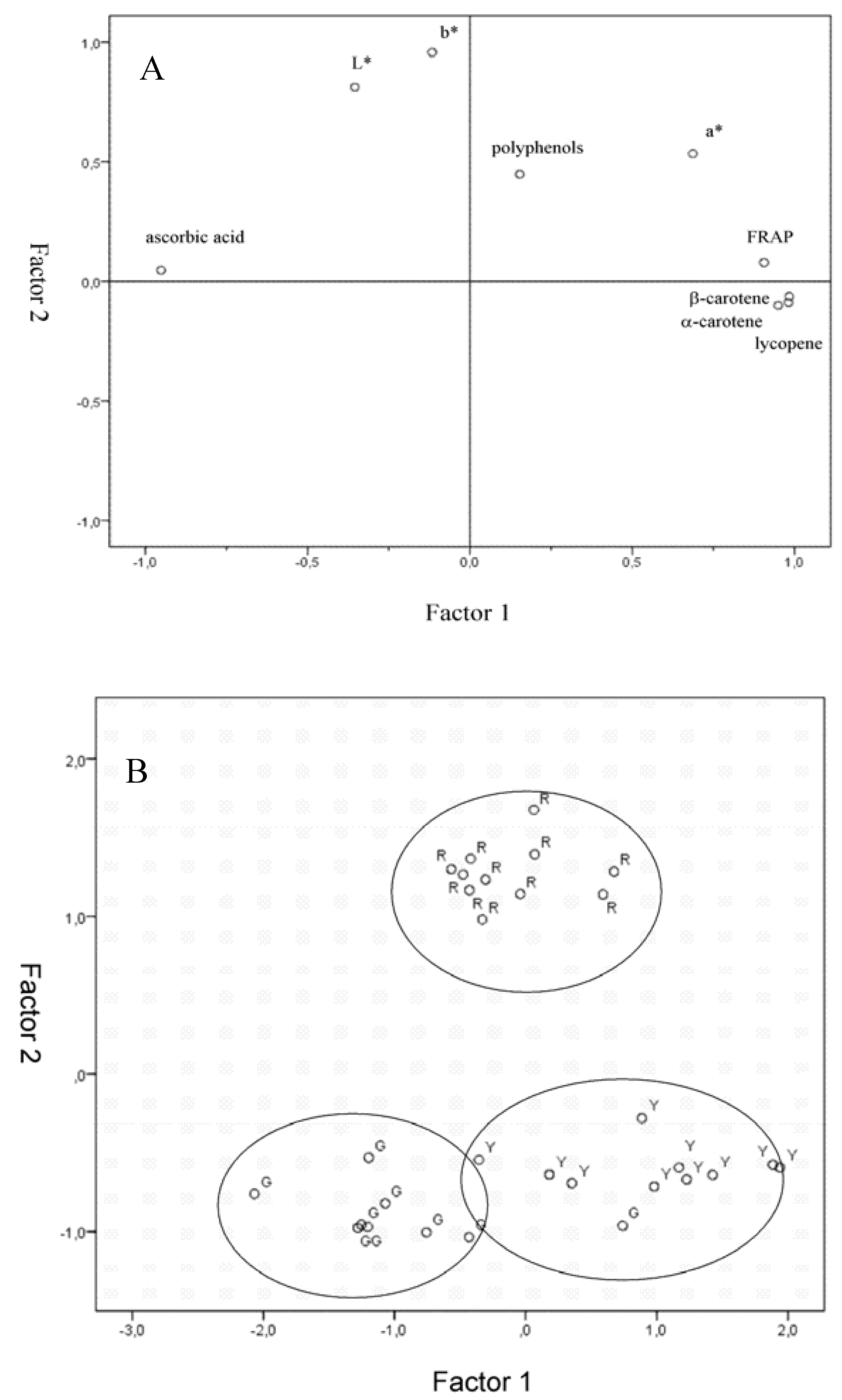
3. Experimental
3.1. Chemical
3.2. Solanum indicum L. fruits
3.3. Analysis
3.4. HPLC-DAD analysis
3.5. HPLC-ESI-MS /MS analysis of phenolic compounds
3.6. Colour determination
3.7. Statistical Analysis
4. Conclusions
- Sample Availability: Contact the authors.
References
- N'dri, M.T.K.; Gnahoua, M.G.; Konan, E.K.; Traoré, D. Plantes alimentaires spontanées de la région du Fromager (Centre Ouest de la Côte d'Ivoire) flore, habitats et organes consommés. Sci. & Nat. 2008, 5, 61–70. [Google Scholar]
- Bahgat, A.; Abdel-Aziz, H.; Raafat, M.; Mahdy, A.; El-Khatib, A.S.; Ismail, A.; Khayyal, M.T. Solanum indicum ssp. distichum extract is effective against L-NAME-induced hypertension in rats. Fundam. Clin. Pharmacol. 2008, 22, 693–699. [Google Scholar] [CrossRef]
- Aberoumand, A.; Deokule, S.S. Comparison of phenolic compounds of some edible plants of Iran and India. Pak. J. Nutr. 2008, 7, 582–585. [Google Scholar] [CrossRef]
- Aberoumand, A.; Deokule, S.S. Studies of nutritional values of some wild edible plants from Iran and India. Pak. J. Nutr. 2009, 8, 26–31. [Google Scholar] [CrossRef]
- Aberoumand, A.; Deokule, S.S. Total phenolic contents of some plant foods as antioxidant compound. J. Food Technol. 2010, 8, 131–133. [Google Scholar] [CrossRef]
- Lopez Camelo, A.F.; Gomez, P.A. Comparison of color indexes for tomato ripening. Hortic. Bras. 2004, 22, 534–537. [Google Scholar] [CrossRef]
- Abushita, A.A.; Daood, H.G.; Biacs, P.A. Change in carotenoids and antioxidant vitamins in tomato as a function of varietal and technological factors. J. Agric. Food Chem. 2000, 48, 2075–2081. [Google Scholar] [CrossRef]
- Raffo, A.; La Malfa, G.; Fogliano, V.; Maiani, G.; Quaglia, G. Seasonal variations in antioxidant components of cherry tomatoes (Lycopersicon esculentum cv. Naomi F1). J. Food Compos. Anal. 2006, 19, 11–19. [Google Scholar] [CrossRef]
- Abushita, A.A.; Hebshi, E.A.; Daood, H.G.; Biacs, P.A. Determination of antioxidants vitamins in tomatoes. Food Chem. 1997, 60, 207–212. [Google Scholar] [CrossRef]
- Raffo, A.; Leonardi, C.; Fogliano, V.; Ambrosino, P.; Salucci, M.; Gennaro, L.; Bugianesi, R.; Giuffrida, F.; Quaglia, G. Nutritional value of cherry tomatoes (Lycopersicon esculentum Cv. Naomi F1) harvested at different ripening stages. J. Agric. Food Chem. 2002, 50, 6550–6556. [Google Scholar]
- Juroszek, P.; Lumpkin, H.M.; Yang, R.Y.; Ledesma, D.R.; Ma, C.H. Fruit quality and bioactive compounds with antioxidant activity of tomatoes grown on-farm: comparison of organic and conventional management systems. J. Agric. Food Chem. 2010, 57, 1188–1194. [Google Scholar]
- Frusciante, L.; Carli, P.; Ercolano, M. R.; Pernice, R.; Di Matteo, A.; Fogliano, V.; Pellegrini, N. Antioxidant nutritional quality of tomato. Mol. Nutr. Food Res. 2007, 51, 609–617. [Google Scholar] [CrossRef]
- Tang, G. Bioconversion of dietary provitamin A carotenoids to vitamin A in humans. Am. J. Clin. Nutr. 2010, 91, 1468–1473. [Google Scholar] [CrossRef]
- Trumbo, P.; Yates, A.A.; Schlicker, S.; Poos, M. Dietary Reference Intakes for vitamin A, vitamin K, boron, chromium, copper, iodine, iron, manganese, molybdenum, nickel, silicon, vanadium and zinc. J. Am. Diet. Assoc. 2001, 101, 294–301. [Google Scholar] [CrossRef]
- World Health Organization, Global Prevalence of Vitamin A Deficiency in Populations at Risk 1995–2005: WHO Global Database on Vitamin A Deficiency. WHO: Geneva, Switzerland, 2009.
- Livny, O.; Reifen, R.; Levy, I.; Madar, Z.; Faulks, R.; Southon, S.; Schwartz, B. Beta-carotene bioavailability from differently processed carrot meals in human ileostomy volunteers. Eur. J. Nutr. 2003, 42, 338–345. [Google Scholar] [CrossRef]
- Omoni, A.O.; Aluko, R.E. The anti-carcinogenic and anti-atherogenic effects of lycopene: a review. Trends Food Sci. Tech. 2005, 16, 344–350. [Google Scholar]
- Lenucci, M.S.; Cadinu, D.; Taurino, M.; Piro, G.; Dalessandro, G. Antioxidant composition in cherry and high-pigment tomato cultivars. J. Agric. Food Chem. 2006, 54, 2606–2613. [Google Scholar]
- Carlsen, M.H.; Halvorsen, B.L.; Holte, K.; Bøhn, S.K.; Dragland, S.; Sampson, L.; Willey, C.; Senoo, H.; Umezono, Y.; Sanada, C.; Barikmo, I.; Berhe, N.; Willett, W.C.; Phillips, K.M.; Jacobs, D.R. Jr.; Blomhoff, R. The total antioxidant content of more than 3100 foods, beverages, spices, herbs and supplements used worldwide. Nutr. J. 2010, 9, 3. [Google Scholar] [CrossRef]
- Pellegrini, N.; Serafini, M.; Colombi, B.; Del Rio, D.; Salvatore, S.; Bianchi, M.; Brighenti, F. Total antioxidant capacity of plant foods, beverages and oils consumed in Italy assessed by three different in vitro assays. J. Nutr. 2003, 133, 2812–2819. [Google Scholar]
- Scalfi, L.; Fogliano, V.; Pentangelo, A.; Graziani, G.; Giordano, I.; Ritieni, A. Antioxidant activity and general fruit characteristics in different ecotypes of Corbarini small tomatoes. J. Agric. Food Chem. 2000, 48, 1363–1366. [Google Scholar]
- Gokmen, V; Serpen, A; Fogliano, V. Direct measurement of the total antioxidant capacity of foods: a Quencher approach. Trends Food Sci. Tech. 2009, 20, 278–288. [Google Scholar] [CrossRef]
- Dürüst, N.; Sümengen, D.; Dürüst, Y. Ascorbic acid and element contents of foods of Trabzon (Turkey). J. Agric. Food Chem. 1997, 45, 2085–2087. [Google Scholar] [CrossRef]
- Gökmen, V.; Kahraman, N.; Demir, N.; Acar, J. Enzymatically validated liquid chromatographic method for the determination of ascorbic and dehydroascorbic acids in fruit and vegetables. J. Chromatogr. A 2000, 881, 309–316. [Google Scholar] [CrossRef]
- Leonardi, C.; Ambrosino, P.; Esposito, F.; Fogliano, V. Antioxidative activity and carotenoid and tomatine contents in different typologies of fresh consumption tomatoes. J. Agric. Food Chem. 2000, 48, 4723–4727. [Google Scholar]
- Crozier, A.; Lean, M. E.; McDonald, M.S.; Black, C. Quantitative analysis of the flavonoid content of commercial tomatoes, onions, lettuce and celery. J. Agric. Food Chem. 1997, 43, 590–595. [Google Scholar]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic–phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Benzie, I.F.; Strain, J.J. Ferric reducing/antioxidant power assay: direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration. Methods Enzymol. 1999, 299, 15–27. [Google Scholar]
- Romani, S., Rocculi. Image characterization of potato chip appearance during frying. J. Food Eng. 2009, 93, 487–494. [Google Scholar] [CrossRef]
- Clèment, A.; Dorais, M.; Vernon, M. Multivariate approach to the measurement of tomato maturity and gustatory attributes and their rapid assessment by vis-nir spectroscopy. J. Agric. Food Chem. 2008, 56, 1538–1544. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
N’Dri, D.; Calani, L.; Mazzeo, T.; Scazzina, F.; Rinaldi, M.; Del Rio, D.; Pellegrini, N.; Brighenti, F. Effects of Different Maturity Stages on Antioxidant Content of Ivorian Gnagnan (Solanum indicum L.) Berries. Molecules 2010, 15, 7125-7138. https://doi.org/10.3390/molecules15107125
N’Dri D, Calani L, Mazzeo T, Scazzina F, Rinaldi M, Del Rio D, Pellegrini N, Brighenti F. Effects of Different Maturity Stages on Antioxidant Content of Ivorian Gnagnan (Solanum indicum L.) Berries. Molecules. 2010; 15(10):7125-7138. https://doi.org/10.3390/molecules15107125
Chicago/Turabian StyleN’Dri, Denis, Luca Calani, Teresa Mazzeo, Francesca Scazzina, Massimiliano Rinaldi, Daniele Del Rio, Nicoletta Pellegrini, and Furio Brighenti. 2010. "Effects of Different Maturity Stages on Antioxidant Content of Ivorian Gnagnan (Solanum indicum L.) Berries" Molecules 15, no. 10: 7125-7138. https://doi.org/10.3390/molecules15107125
APA StyleN’Dri, D., Calani, L., Mazzeo, T., Scazzina, F., Rinaldi, M., Del Rio, D., Pellegrini, N., & Brighenti, F. (2010). Effects of Different Maturity Stages on Antioxidant Content of Ivorian Gnagnan (Solanum indicum L.) Berries. Molecules, 15(10), 7125-7138. https://doi.org/10.3390/molecules15107125







