Anti-Adhesive Activities of Flavan-3-ols and Proanthocyanidins in the Interaction of Group A-Streptococci and Human Epithelial Cells
Abstract
:1. Introduction
2. Results and Discussion
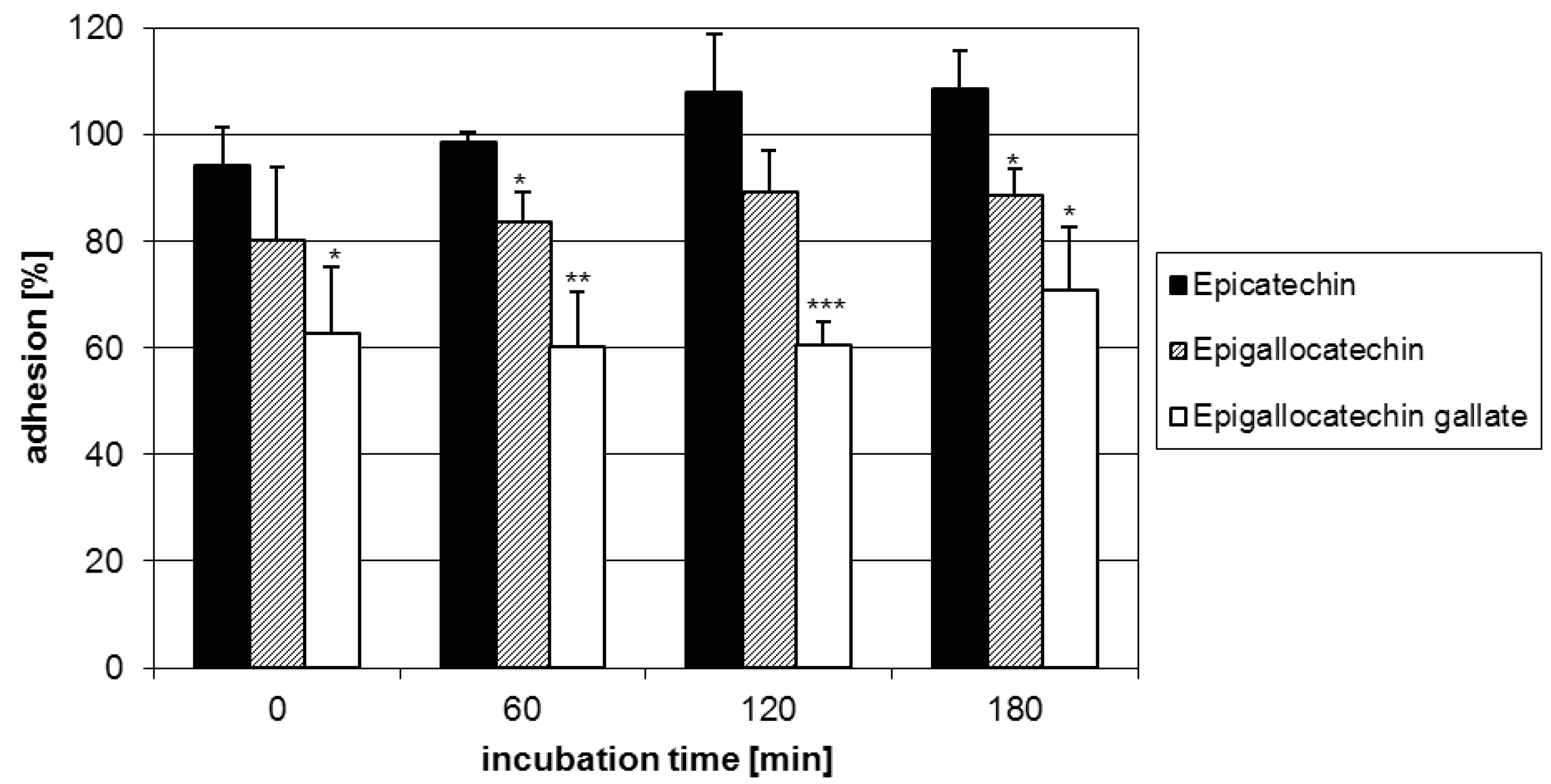
2.1. Anti-adhesive potency of flavan-3-ols

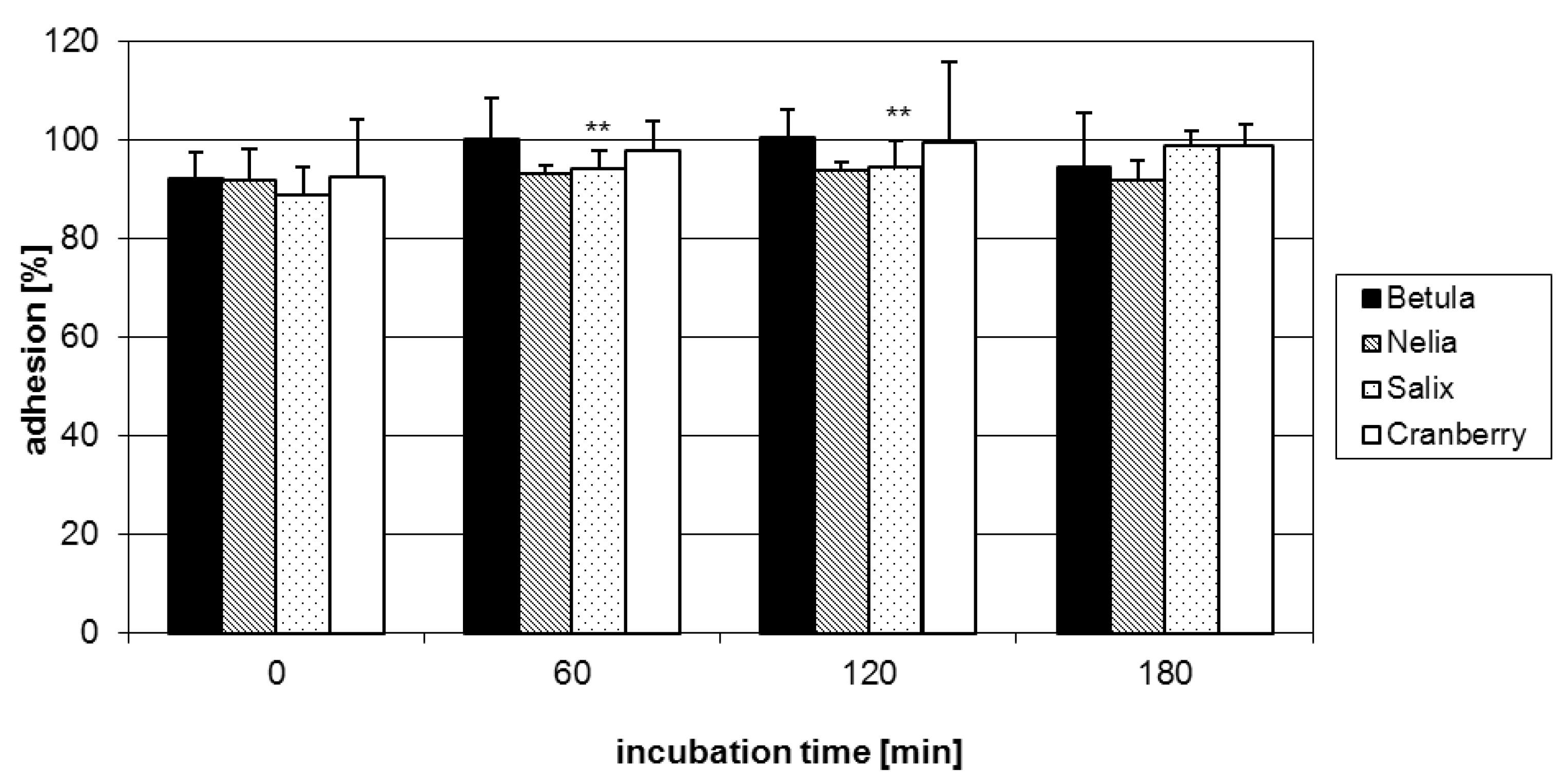
2.2. Anti-adhesive potency of proanthocyanidins: structure-activity relationships
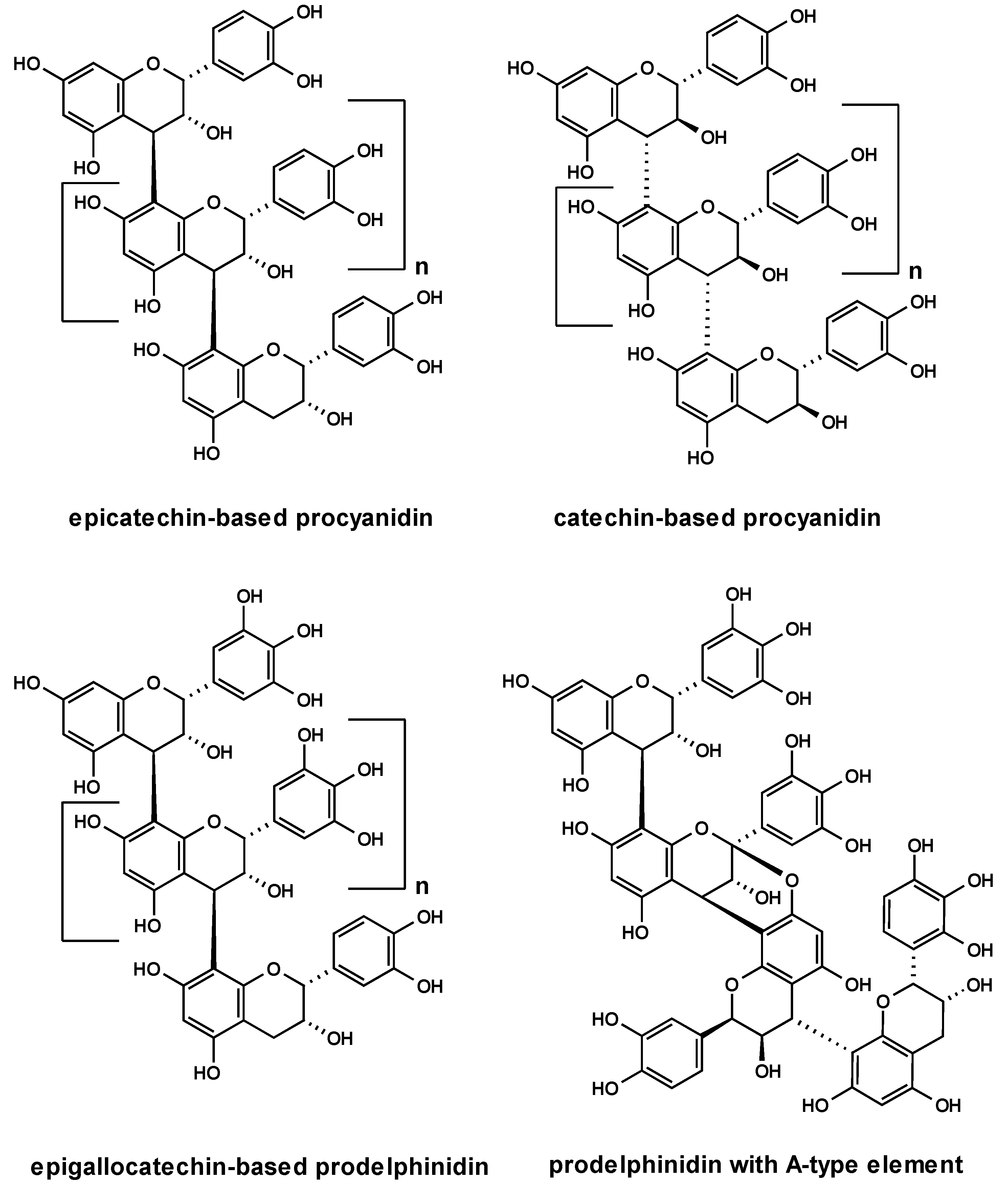

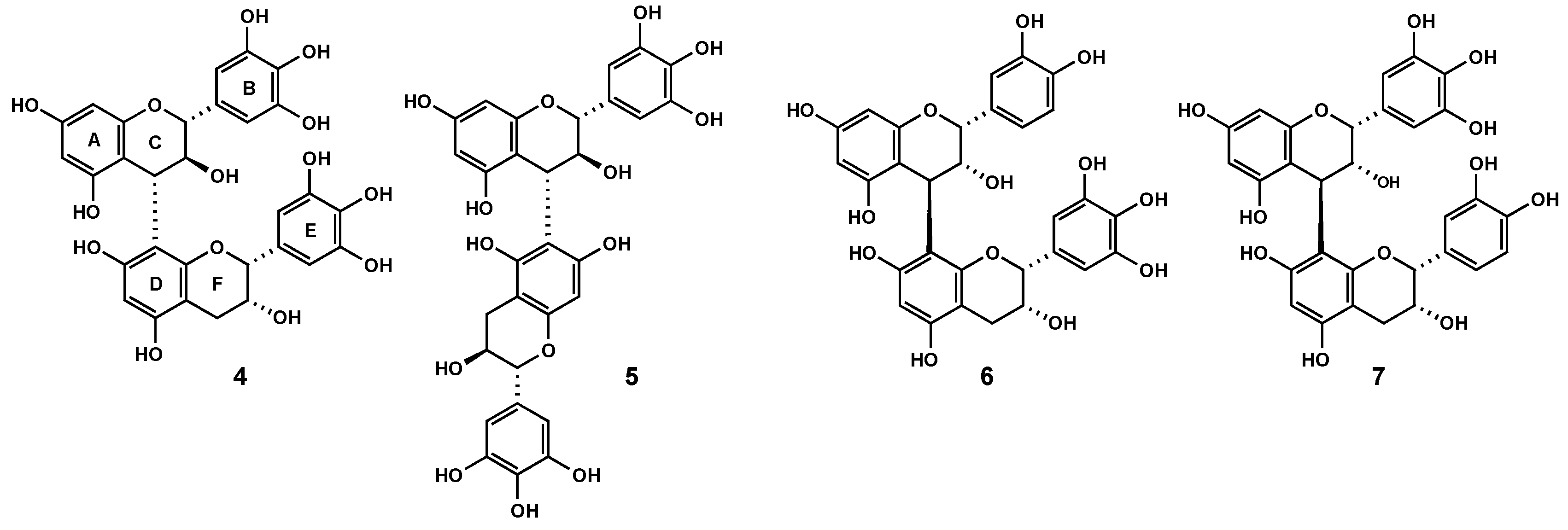
2.3. Characterization of proanthocyanidins from Pelargonium sidoides
3. Experimental Section
3.1. Test substances and plant extracts
3.2. Bacterial strain and culture conditions
3.3. Human laryngeal epithelial cells (HEp-2 cells)
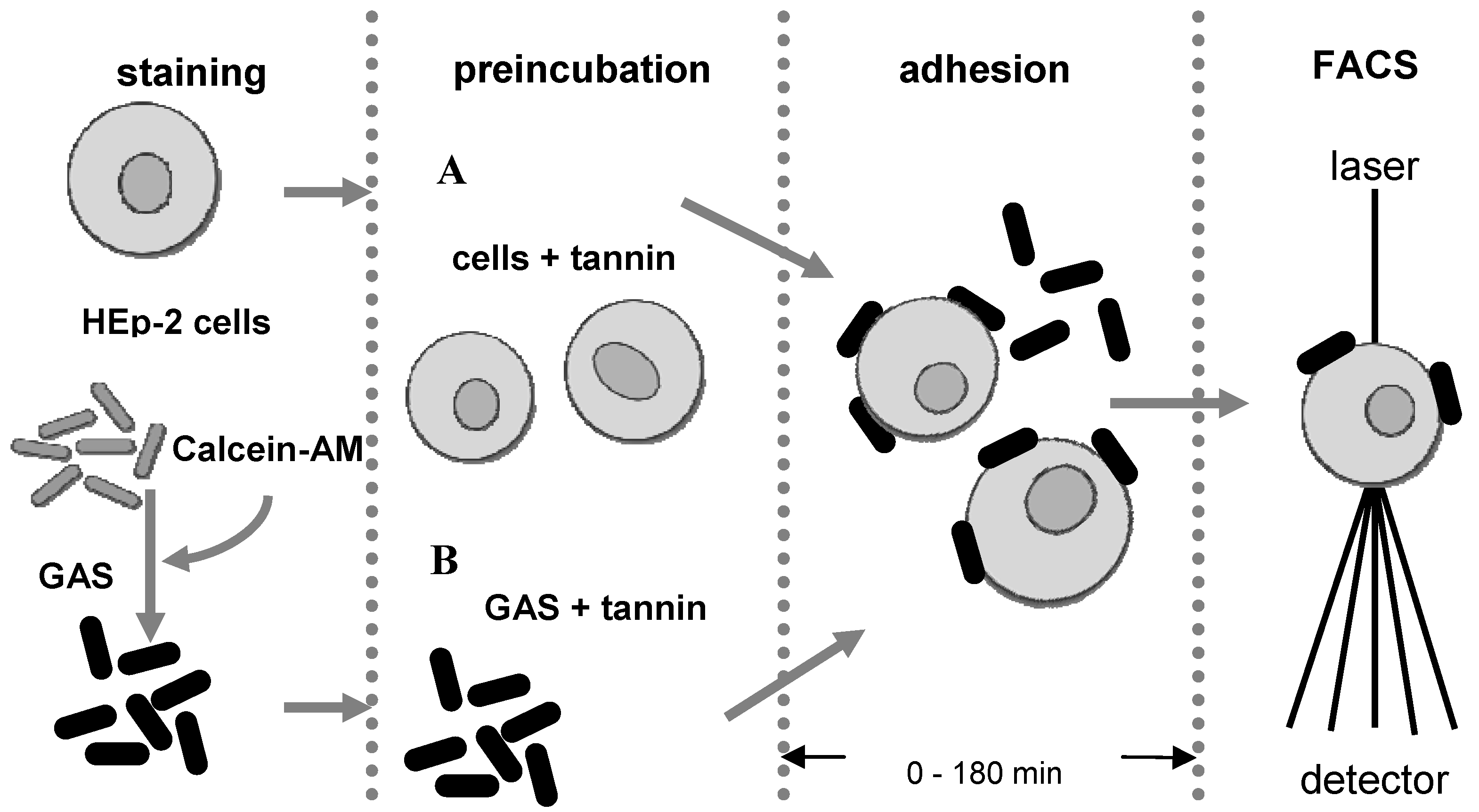
3.4. Anti-adhesion assay
3.5. Flow cytometry
3.6. Statistical analysis
3.7. Isolation of proanthocyanidins from P. sidoides
4. Conclusions
Acknowledgements
References and Notes
- Feeny, P. Plant apparency and chemical defense. Rec. Adv. Phytochem. 1976, 10, 1–40. [Google Scholar]
- Zucker, W.V. Tannins: does structure determine function? An ecological perspective. Am. Nat. 1981, 121, 331–365. [Google Scholar]
- Scalbert, A. Antimicrobial properties of tannins. Phytochemistry 1991, 30, 3875–3883. [Google Scholar] [CrossRef]
- Kolodziej, H.; Kayser, O.; Lattè, K.P.; Ferreira, D. Evaluation of the antimicrobial potency of tannins and related compounds using the microdilution broth method. Planta Med. 1999, 65, 444–446. [Google Scholar] [CrossRef]
- Kolodziej, H.; Kayser, O.; Kierlen, A.F.; Ito, H.; Hatano, T.; Yoshida, T.; Foo, L.Y. Antileishmanial activity of hydrolysable tannins and their modulatory effects on nitrice oxide and tumour necrosis factor a release in macrophages in vitro. Planta Med. 2001, 67, 825–832. [Google Scholar]
- Kolodziej, H.; Kayser, O.; Kierlen, A.F.; Ito, H.; Hatano, T.; Yoshida, T.; Foo, L.Y. Proanthocyanidins and related compounds: antileishmanial activity and modulatory effects on nitric oxide and tumour necrosis factor a release in the murine macrophage-like cell line RAW 264.7. Biol. Pharm. Bull. 2001, 24, 1016–1021. [Google Scholar] [CrossRef]
- Howell, A.B.; Vora, N.; Der Marderosian, A.; Foo, L.Y. Inhibition of the adherence of p-frimbrinated Escherichia coli to uroepithelial cell surfaces by proanthocyanidin extracts from cranberries. N. Engl. J. Med. 1998, 339, 1085–1086. [Google Scholar] [CrossRef]
- Schmidt, D.R.; Sobota, A.E. An examination of the anti-adherence activity of cranberry juice on urinary and non-urinary bacterial isolates. Microbios 1988, 55, 173–181. [Google Scholar]
- Busscher, H.J; Norde, W., vander Wei. Specific molecular recognition and nonspecific contributions to bacterial interaction forces. Appl. Environ. Microbiol. 2008, 74, 2559–2564. [Google Scholar] [CrossRef]
- Hagerman, A.E.; Butler, L.G. The specificity of proanthocyanidin-protein interactions. J. Biol. Chem. 1981, 256, 4494–4497. [Google Scholar]
- Haslam, E. Plant polyphenols – Vegetable tannins revisited; Cambridge University Press: Cambridge, Great Britain, 1989. [Google Scholar]
- Dangles, O.; Dufour, C. Flavonoid-protein interactions. In Flavonoids: Chemistry, Biochemistry and Application; Andersen, M., Markham, K.R., Eds.; Taylor and Francis: Boca Raton, FL, USA, 2006; pp. 443–469. [Google Scholar]
- Sethman, C.R.; Doyle, R.J.; Cowan, M.M. Flow cytometric evaluation of adhesion of Streptococcus pyogenes to epithelial cells. J. Microbiol. Meth. 2002, 51, 35–42. [Google Scholar] [CrossRef]
- LaPenta, D.; Rubens, C.; Chi, E.; Clearly, P. Group A streptococci efficiently invade human respiratory epithelial cells. Proc. Natl. Acad. Sci. USA 1994, 91, 12115–12119. [Google Scholar] [CrossRef]
- Kolodziej, H. Occurrence of procyanidins in Nelia meyeri. Phytochemistry 1984, 23, 1745–1752. [Google Scholar] [CrossRef]
- Kolodziej, H. Oligomeric flavan-3-ols from medicinal willow bark. Phytochemistry 1990, 29, 955–960. [Google Scholar] [CrossRef]
- Kolodziej, H. Procyanidins from medicinal birch: bonding patterns and sequence of units in triflavanoids of mixed stereochemistry. Phytochemistry 1989, 28, 3487–3492. [Google Scholar] [CrossRef]
- Singh, B.; Kaur, P.; Gopichand; Singh, R.D.; Ahuja, P.S. Biology and chemistry of Ginkgo biloba. Fitoterapia 2008, 79, 401–419. [Google Scholar] [CrossRef]
- Schötz, K.; Nöldner, M. Mass spectroscopic characterization of oligomeric proanthocyanidins derived from an extract of Pelargonium sidoides (EPs® 7630) and pharmacological screening in CNS models. Phytomedicine 2007, 14, 32–39. [Google Scholar]
- Foo, L.Y.; Lu, Y.; Howell, A.B.; Vorsa, N. A-type proanthocyanidin trimers from cranberry that inhibit adherence of uropathogenic p-fimbriated E. coli. J. Nat. Prod. 2000, 63, 1225–1228. [Google Scholar] [CrossRef]
- Foo, L.Y.; Lu, Y.; Howell, A.B.; Vorsa, N. The structure of cranberry proanthocyanidins which inhibit adherence of uropathogenic P-fimbriated Escherichia coli in vitro. Phytochemistry 2000, 54, 173–181. [Google Scholar]
- Johnson, J.R.; Kaster, N.; Kuskowski, M.A.; Ling, G.V. Identification of urovirulence traits in Escherichia coli by comparison of urinary and rectal E. coli isolates from dogs with urinary tract infection. J. Clin. Microbiol. 2003, 41, 337–345. [Google Scholar] [CrossRef]
- Salminen, A.; Loimaranta, V.; Joosten, J.A.F.; Salam Khan, A.; Hacker, J.; Pieters, R.J.; Finne, J. Inhibition of P-fimbriated Escherichia coli adhesion by multivalent galabiose derivatives studied by a live-bacteria application of suface plasmon resonance. J. Antimicrob. Chemother. 2007, 60, 495–501. [Google Scholar] [CrossRef]
- Podbielski, A.; Kreikemeyer, B. The background of tissue specificity of Streptococcus pyogenes. Bioforum 2006, 29, 32–34. [Google Scholar]
- Kolodziej, H. Fascinating metabolic pools of Pelargonium sidoides and Pelargonium reniforme, traditional and phytomedicinal sources of the herbal medicine Umckaloabo®. Phytomedicine 2007, 14, 9–17. [Google Scholar] [CrossRef]
- DeBruyne, T.; Pieters, L.A.; Dommisse, R.A.; Kolodziej, H.; Wray, V.; Domke, T.; Vlietinck, A. Unambiguous assignments for free dimeric proanthocyanidin phenols from 2D NMR. Phytochemistry 1996, 43, 265–272. [Google Scholar]
- Fletcher, A.C.; Porter, L.J.; Haslam, E. Conformational and configurational studies of natural procyanidins. J. Chem. Soc. Perkin Trans. I 1977, 1628–1637. [Google Scholar]
- Foo, L.Y.; Porter, L.J. Prodelphinidin polymers: Definition of structural units. J. Chem. Soc. Perkin Trans. I 1978, 1186–1190. [Google Scholar]
- Tits, M.; Angenot, L.; Poukens, P.; Warin, R.; Diercxsens, Y. Prodelphinidins from Ribes nigrum. Phytochemistry 1992, 31, 971–973. [Google Scholar]
- Hashimoto, F.; Nonaka, G.; Nishioka, I. Tannins and related compounds. XC. 8-C-Ascorbyl (-)-epigalloatechin 3-O-gallate and novel dimeric flavan-3-ols, Oolonghomobisflavans A and B, from Oolong tea. Chem. Pharm. Bull. 1989, 37, 3255–3263. [Google Scholar] [CrossRef]
- Foo, L.Y.; Lu, Y.; Molan, A.L.; Woodfield, D.R.; McNabb, W.C. The phenols and prodelphinidins of white clover flowers. Phytochemistry 2000, 54, 539–548. [Google Scholar] [CrossRef]
- Petereit, F.; Kolodziej, H.; Nahrstedt, A. Flavan-3-ols and proanthocyanidins from Cistus incanus. Phytochemistry 1991, 30, 981–985. [Google Scholar] [CrossRef]
- Malik, A.; Kuliev, Z.A.; Akhmedov, Y.A.; Vdovin, A.D.; Abduliaev, N.D. Proanthocyanidins of Ziziphuy jujuba. Chem. Nat. Comp. 1997, 33, 165–173. [Google Scholar] [CrossRef]
- Lengsfeld, C.; Deters, A.; Faller, G.; Hensel, A. High molecular weight polysaccharides from black currant seeds inhibit adhesion of Helicobacter pylori to human gastric mucosa. Planta Med. 2004, 70, 620–626. [Google Scholar] [CrossRef]
- Sharon, N. Carbohydrates as future anti-adhesion drugs for infection diseases. Biochim. Biophys. Acta 2006, 1760, 527–537. [Google Scholar] [CrossRef]
- Wittschier, N.; Lengsfeld, C.; Vorthems, S.; Stratmann, U.; Ernst, J.F.; Verspohl, E.J.; Hensel, A. Large molecules as anti-adhesive compounds against pathogens. J. Pharm. Pharmacol. 2007, 59, 777–786. [Google Scholar]
- Wittschier, N.; Faller, G.; Hensel, A. Aqueous extracts and polysaccharides from liquorice roots (Glycyrrhiza glabra L.) inhibit adhesion of Helicobacter pylori to human gastric mucosa. J. Ethnopharmacol. 2009, 125, 218–223. [Google Scholar] [CrossRef]
- Nowak, R.; Schmitt, W. Cranberry juice for prophylaxis of urinary tract infections – Conclusions from clinical experience and research. Phytomedicine 2008, 15, 653–667. [Google Scholar] [CrossRef]
- Ngueyemem, T.A.; Brusotti, G.; Marubini, G.; Grisoli, P.; Dacarro, C.; Vidari, G.; Vita Finzi, P.; Caccialanza, G. Validation of use of a traditional remedy from Bridelia grandis (Pierre ex Hutch) stem bark against oral Streptococci. J. Ethnopharmacol. 2008, 120, 13–16. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Janecki, A.; Kolodziej, H. Anti-Adhesive Activities of Flavan-3-ols and Proanthocyanidins in the Interaction of Group A-Streptococci and Human Epithelial Cells. Molecules 2010, 15, 7139-7152. https://doi.org/10.3390/molecules15107139
Janecki A, Kolodziej H. Anti-Adhesive Activities of Flavan-3-ols and Proanthocyanidins in the Interaction of Group A-Streptococci and Human Epithelial Cells. Molecules. 2010; 15(10):7139-7152. https://doi.org/10.3390/molecules15107139
Chicago/Turabian StyleJanecki, Aneta, and Herbert Kolodziej. 2010. "Anti-Adhesive Activities of Flavan-3-ols and Proanthocyanidins in the Interaction of Group A-Streptococci and Human Epithelial Cells" Molecules 15, no. 10: 7139-7152. https://doi.org/10.3390/molecules15107139
APA StyleJanecki, A., & Kolodziej, H. (2010). Anti-Adhesive Activities of Flavan-3-ols and Proanthocyanidins in the Interaction of Group A-Streptococci and Human Epithelial Cells. Molecules, 15(10), 7139-7152. https://doi.org/10.3390/molecules15107139



