Abstract
Reactivation of Epstein-Barr virus (EBV) from latency to the lytic cycle is required for the production of viral particles. Here, we examine the capacity of resveratrol to inhibit the EBV lytic cycle. Our results show that resveratrol inhibits the transcription of EBV immediate early genes, the expression of EBV lytic proteins, including Rta, Zta, and EA-D and reduces viron production, suggesting that this compound may be useful for preventing the proliferation of the virus.
1. Introduction
Epstein-Barr virus (EBV) is a gamma-herpesvirus, which infects human lymphoid cells and epithelial cells [1]. Infection by this virus is associated with a number of human cancers, including Burkitt’s lymphoma [2], nasopharyngeal carcinoma (NPC) [3] and Hodgkin’s disease [4]. The reactivation of EBV from latency to the lytic cycle is necessary for the virus to produce virions and establish infections [5,6]. At the onset of the lytic cycle, EBV expresses two transcription factors, Rta and Zta, which are transcribed from BRLF1 and BZLF1, respectively [7]. These two proteins trigger an ordered cascade of the expression of viral lytic genes, including that of BMRF1 and BALF5, which encode diffused early antigen (EA-D) and DNA polymerase [8]. Rta and Zta can also activate IL-6 transcription in lytically-infected B cells, which leads to immortalized B cells [9]. Therefore, to reduce the disease risk and to improve the clinical outcome an effective strategy to block the viral lytic cycle is of value. Earlier studies have established that lytic EBV replication is inhibited by acyclovir and ganciclovir, which specifically inhibit the function of viral-encoded DNA polymerase [10]. An earlier study showed that epigallocatechin gallate (EGCG) inhibits the expression of the EBV immediate-early genes transcription at concentration 50 μM [11]. Ethanolic extract from Andrographis paniculata and andrograpolide also inhibit the expression of Rta, Zta and EA-D at 25 μg/mL and 5 μg/mL, respectively [12].
Resveratrol is a non-flavonoid polyphenol compound present in many plants and fruits. This compound has a high bioactivity and its cytoprotective action has been demonstrated. Previous studies showed that resveratrol inhibits the replication of human cytomegalovirus (HCMV) [13], herpes simplex virus type 1 (HSV-1) [14] and Varicella-zoster virus (VZV) [15]. Furthermore, resveratrol also has been reported to inhibit the expression of EBV early antigen induced by 12-O- tetradecanoylphorbol 13-acetate (TPA) in Raji cells [16]. However, no preexisting study has been reported on resveratrol mediated the inhibition of EBV lytic cycle. Thus, the purpose of our study was to evaluate whether resveratrol inhibits the transcription of EBV immediate early genes, the expression of EBV lytic proteins, including Rta, Zta, and EA-D and viron production.
2. Results and Discussion
2.1. Determining the toxicity of resveratrol to P3HR1 cells
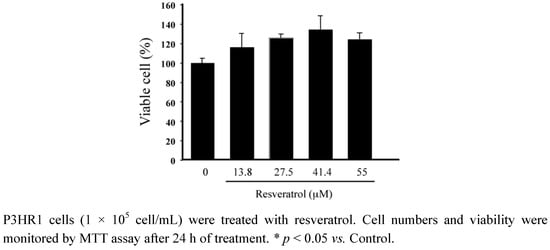
Resveratrol was added into 1 × 105 cells/mL P3HR1 cells. After 24 h of treatment, cell viability was determined by the MTT assay. Results showed that resveratrol exhibited no cytotoxicity toward P3HR1 cells at concentrations below 55 μM (Figure 1).
2.3. Flow cytometric analysis of the expression of EBV lytic proteins
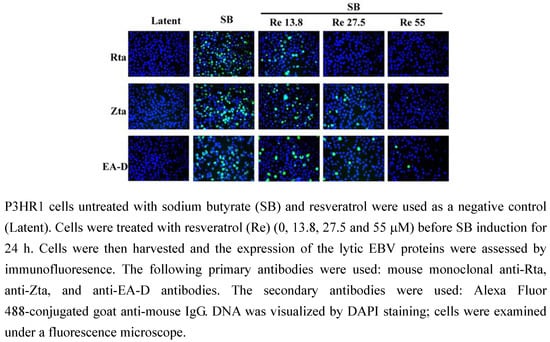
The presence of EBV lytic proteins in P3HR1 cells was further analyzed by flow cytometric analysis. The P3HR1 populations not treated with SB that expressed Rta, Zta and EA-D wwere 4.5%, 2.1% and 4.9%, respectively (Figure 3). The populations that expressed the three proteins after SB treatment increased to 51.2%, 61.4% and 46.8%, respectively. After treating the cells with 13.8 μM of resveratrol, the populations that expressed Rta, Zta and EA-D decreased to 29.6%, 37.0% and 29.6%, respectively (Figure 3A The populations that expressed Rta, Zta, and EA-D further decreased when the concentration of resveratrol was increased to 27.5 μM and 55 μM. At 27.5 μM, the populations that expressed Rta, Zta, and EA-D decreased to 15.4%, 20.2%, and 17.9%, respectively (Figure 3). At 55 μM, the population that expressed Rta was reduced from 51.2% to 4.7% (Figure 3); the population that expressed Zta, from 61.4% to 5.6%; and the population that expressed EA-D from 46.8% to 4.5%, respectively (Figure 3). These results show that resveratrol inhibited the expression of Rta, Zta and EA-D in a dose-dependent manner (Figure 3B).
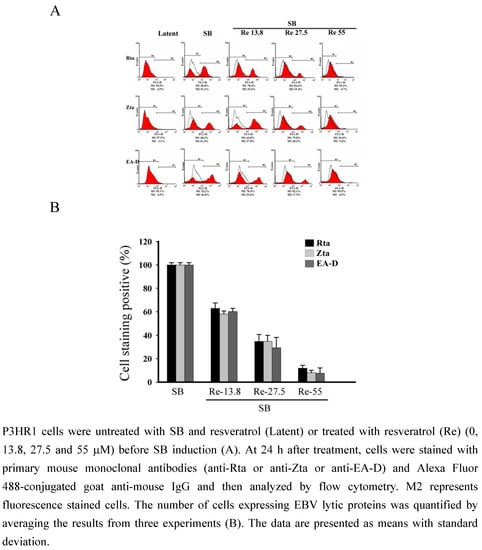
Figure 3.
Inhibition of the expression of EBV lytic proteins by resveratrol.
Figure 3.
Inhibition of the expression of EBV lytic proteins by resveratrol.
2.4. Inhibiting the transcription of EBV immediate-early genes
The activity of the BRLF1 and BZLF1 promoters was analyzed by transient transfection assay in P3HR1 cells using the reporter plasmids pRLUC and pZLUC, respectively. Results showed that resveratrol at 6.9 μM inhibited the activity of BRLF1 and BZLF1 promoters by 56% and 83%. Resveratrol at 13.8 μM decreased the BRLF1 and BZLF1 promoters’ activity to background (Figure 4).
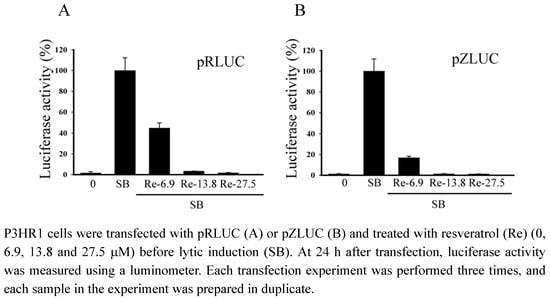
Figure 4.
Inhibition of the transcription of the BRLF1 and BZLF1 promoters by resveratrol.
Figure 4.
Inhibition of the transcription of the BRLF1 and BZLF1 promoters by resveratrol.
2.5. Inhibition of the production of EBV particles
P3HR1 cells were then treated with 13.8, 27.5 and 55 μM of resveratrol after lytic induction. After culturing for five days, EBV particles released into medium were isolated. Real-time qPCR showed that resveratrol at a concentration of 27.5 μM decreased virus production by 42% (Figure 5). Resveratrol at aconcentration of 55 μM reduced the production of EBV particles by 74% (Figure 5).
Previous studies showed that lytic EBV proteins actually induce the expression of B-cell growth factor, IL-6, cellular IL-10 and viral IL-10, allowing B cells to grow more efficiently [17,18]. Lytically-infected cells also produce VEGF and thus contribute to angiogenesis in both B-cell and epithelial-cell malignancies [18]. Therefore, new treatment strategies aimed at completely suppressing the expression of all lytic viral proteins is usefully in controlling early EBV-associated malignancies. This study finds that resveratrol significantly reduces the expression of EBV immediate-early proteins, Rta, Zta and EA-D in a dose-dependent manner (Figure 2 and Figure 3). In other words, resveratrol interferes with an early step of EBV replication cycle. The concentration of resveratrol needed to inhibit EBV immediate-early proteins expression by 50% (EC50) is approximately 24 μM. The cytotoxicity of resveratrol was determined by the MTT assay and resveratrol under the concentration of 55 μM did not affect the P3HR1 cells viability. Earlier study showed that EGCG inhibits the expression of EBV lytic proteins at concentration 50 μM [11]. The concentration of glycyrrhizic acid required to inhibit EBV EA/VCA expression by 50% is 42 μM [19]. These result suggested that resveratrol is more effective than EGCG and glycyrrhizic acid in inhibiting the expression of EBV immediate-early genes.
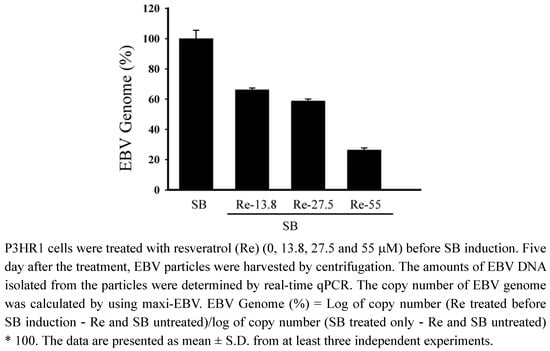
Figure 5.
Resveratrol treatment and production of EBV particles.
Figure 5.
Resveratrol treatment and production of EBV particles.
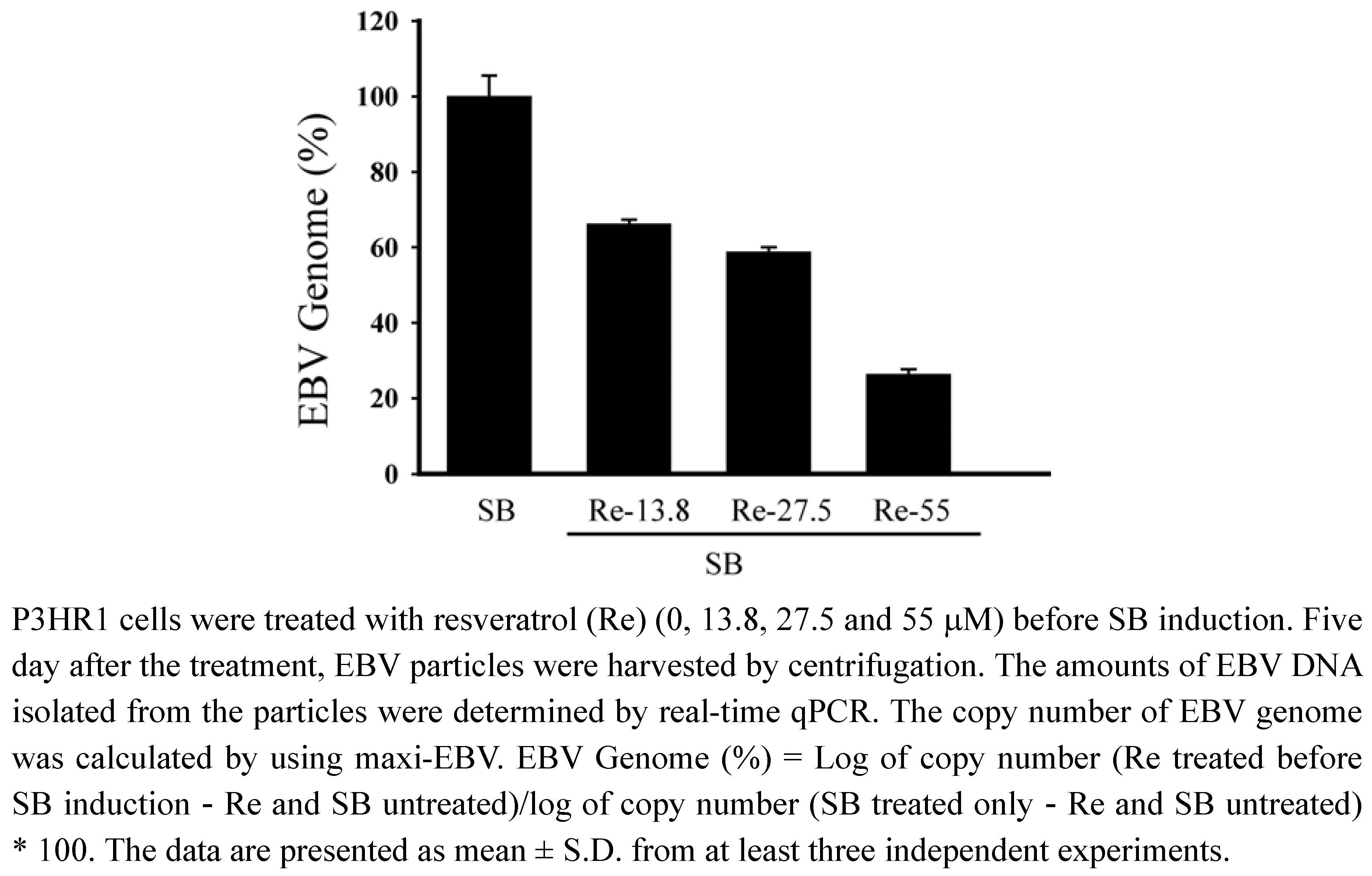
Our study also demonstrates that resveratrol inhibits the transcriptional activity of BRLF1 and BZLF1 promoters, in turn, affect viral lytic proteins expression. Moreover, the inhibition actually decreases the production of mature viral particles. Real-time qPCR indicates that the effective concentration of resveratrol required to inhibit EBV genome copy numbers by 50% (EC50) is 52.2 μM (Figure 5). The EC50 values of resveratrol obtained by real-time qPCR is approximately 52.2 μM, in contrast to the 24 μM value determined by flow cytometry. However, the inhibitory profile remains unchanged, indicating that flow cytometry is as reliable as the real-time qPCR method for initial drug screening.
The molecular mechanism underlying the inhibition of EBV early gene expression by resveratrol is unclear. Previous studies showed that resveratrol inhibits the activation of p38 MAPK, ERK and JNK signaling and affects the activation of the promoters that are activated by AP-1 or ATF2 [20]. Because both the BRLF1 and BZLF1 promoters are strongly activated by AP-1 and ATF2 [21,22,23]. The inhibition of the activation of signaling pathways may explain how resveratrol inhibits the EBV lytic cycle. Resveratrol also has been reported to activate the deacetylase activity of sirtuin protein [24]. Histone acetylation at the BRLF1 promoter allows the virus to express Rta and to activate the viral lytic cycle [25]. Moreover, Zta-directed nucleosome acetylation activates the transcriptional activity of viral immediate-early gene [26]. Thus, the inhibition of resveratrol on deacetylase activity of sirtuin protein may involve in the transcriptional inhibition of BRLF1 and BZLF1 promoters.
3. Experimental
3.1. Compound
Resveratrol (Sigma, St. Louis, MO, USA) was dissolved in dimethylsulfoxide (DMSO) before use.
3.2. Cell culture and lytic induction of EBV
P3HR1, a Burkitt’s lymphoma cell line which is latently infected by EBV, was cultured in RPMI 1640 medium containing 10% fetal calf serum (Biological Industries, Beit Haemek, Israel). Cells were treated with 3 mM of sodium butyrate (SB) to induce the EBV lytic cycle [27].
3.3. Cell viability assay
A 1 mg/mL solution of [3-(4,5-dimethyldiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) in RPMI 1640 medium was added to 1 × 105 P3HR1 cells. The dehydrogenase activity of the viable cells was measured using the method of Carmichael et al. [28].
3.4. Indirect immunofluorescence staining
Immunofluoresence staining was performed as described [12], using mouse monoclonal anti-Rta, anti-Zta (Argene, Varilhes, France), and anti-EA-D antibodies (Chemicon, Temecula, CA, USA). Secondary antibodies used in the study included Alexa Fluor 488-conjugated goat anti-mouse IgG from Invitrogen. DNA was visualized by staining with 4’,6’-diamido-2-phenylindole (DAPI). Cells were examined under the Axioskop 2 plus fluorescence microscope (Zeiss, Oberkochen, Germany).
3.5. Flow cytometric analysis
P3HR1 cells were treated with antibodies as described for indirect immunofluorescence analysis and fixed in 4% paraformaldehyde. Finally, cells were resuspended in 1% paraformaldehyde and analyzed using a flow cytometer (model FACScanTO, BD Biosciences, New Jersey, USA).
3.6. Transient transfection and luciferase assays
P3HR1 cells (5 × 106) were transfected with 10 μg of pRLUC and pZLUC with a Bio-Rad electroporator using the method of Chang et al. [25,29]. Cells were harvested at 24 h after transfection and luciferase activity was determined using a luminometer (Berthold, Bad Wildbad, Germany) according to a method described elsewhere [25].
3.7. Quantification of EBV particles produced by P3HR1 cells
P3HR1 cells were cultured for 5 days. EBV particles released into the culture medium were harvested by centrifugation. Viral DNA was extracted and the amount of EBV DNA was determined by real-time PCR using an iCycleriQ multicolor real-time PCR detection system (BioRad, CA, USA) with primer and a probe that were specific to the EBNA1 gene [30]. The copy number of the EBV genome was calculated using maxi-EBV DNA extracted from E. coli as a reference. The molecular weight of maxi-EBV is about 1.2 × 107 and 1 ng of maxi-EBV equals to 5.05 × 106 copies of the maxi-EBV genome.
3.8. Statistical analysis
Data were analyzed statistically by one-way analysis of variance (ANOVA) using the SAS JMP 6.0 software package. Data are presented as means ± S.D. and a p value of < 0.05 was regarded as significant.
4. Conclusions
To summarize the whole study, our results clearly demonstrate that resveratrol inhibits the transcription of lytic genes and the lytic cycle of EBV to reduce the production of viral particles. Resveratrol could be of potential use for the development of anti-EBV drugs.
Acknowledgements
This work was supported by research grants 96-2320-B-041-009 from the National Science Council, R.O.C., and CLFHR9620 from Chi Mei Medical Center, Liouying, Tainan, Taiwan.
- Sample Availability: Sample of the compound is available from the authors.
References
- Roizman, B.; Carmichael, L.E.; Deinhardt, F.; de-The, G.; Nahmias, A.J.; Plowright, W.; Rapp, F.; Sheldrick, P.; Takahashi, M.; Wolf, K. Herpesviridae. Definition, provisional nomenclature, and taxonomy. The Herpesvirus Study Group, the International Committee on Taxonomy of Viruses. Intervirology 1981, 16, 201–217. [Google Scholar] [CrossRef]
- Magrath, I.; Jain, V.; Bhatia, K. Epstein-Barr virus and Burkitt's lymphoma. Semin. Cancer Biol. 1992, 3, 285–295. [Google Scholar]
- zur Hausen, H.; Schulte-Holthausen, H.; Klein, G.; Henle, W.; Henle, G.; Clifford, P.; Santesson, L. EBV DNA in biopsies of Burkitt tumours and anaplastic carcinomas of the nasopharynx. Nature 1970, 228, 1056–1058. [Google Scholar] [CrossRef]
- Weiss, L.M.; Movahed, L.A.; Warnke, R.A.; Sklar, J. Detection of Epstein-Barr viral genomes in Reed-Sternberg cells of Hodgkin's disease. N. Engl. J. Med. 1989, 320, 502–506. [Google Scholar] [CrossRef]
- Schwarzmann, F.; Jager, M.; Prang, N.; Wolf, H. The control of lytic replication of Epstein-Barr virus in B lymphocytes (Review). Int. J. Mol. Med. 1998, 1, 137–142. [Google Scholar]
- Hopwood, P.A.; Brooks, L.; Parratt, R.; Hunt, B.J.; Bokhari, M.; Thomas, J.A.; Yacoub, M.; Crawford, D.H. Persistent Epstein-Barr virus infection: unrestricted latent and lytic viral gene expression in healthy immunosuppressed transplant recipients. Transplantation 2002, 74, 194–202. [Google Scholar] [CrossRef]
- Giot, J.F.; Mikaelian, I.; Buisson, M.; Manet, E.; Joab, I.; Nicolas, J.C.; Sergeant, A. Transcriptional interference between the EBV transcription factors EB1 and R: both DNA-binding and activation domains of EB1 are required. Nucleic Acids Res. 1991, 19, 1251–1258. [Google Scholar] [CrossRef]
- Fixman, E.D.; Hayward, G.S.; Hayward, S.D. trans-acting requirements for replication of Epstein-Barr virus ori-Lyt. J. Virol. 1992, 66, 5030–5039. [Google Scholar]
- Tosato, G.; Tanner, J.; Jones, K.D.; Revel, M.; Pike, S.E. Identification of interleukin-6 as an autocrine growth factor for Epstein-Barr virus-immortalized B cells. J. Virol. 1990, 64, 3033–3041. [Google Scholar]
- Meerbach, A.; Holy, A.; Wutzler, P.; De Clercq, E.; Neyts, J. Inhibitory effects of novel nucleoside and nucleotide analogues on Epstein-Barr virus replication. Antivir. Chem. Chemother. 1998, 9, 275–282. [Google Scholar]
- Chang, L.K.; Wei, T.T.; Chiu, Y.F.; Tung, C.P.; Chuang, J.Y.; Hung, S.K.; Li, C.; Liu, S.T. Inhibition of Epstein-Barr virus lytic cycle by (-)-epigallocatechin gallate. Biochem. Biophys. Res. Commun. 2003, 301, 1062–1068. [Google Scholar] [CrossRef]
- Lin, T.P.; Chen, S.Y.; Duh, P.D.; Chang, L.K.; Liu, Y.N. Inhibition of the epstein-barr virus lytic cycle by andrographolide. Biol. Pharm. Bull. 2008, 31, 2018–2023. [Google Scholar] [CrossRef]
- Evers, D.L.; Wang, X.; Huong, S.M.; Huang, D.Y.; Huang, E.S. 3,4',5-Trihydroxy-trans-stilbene (resveratrol) inhibits human cytomegalovirus replication and virus-induced cellular signaling. Antivir. Res. 2004, 63, 85–95. [Google Scholar]
- Docherty, J.J.; Fu, M.M.; Stiffler, B.S.; Limperos, R.J.; Pokabla, C.M.; DeLucia, A.L. Resveratrol inhibition of herpes simplex virus replication. Antivir. Res. 1999, 43, 145–155. [Google Scholar]
- Docherty, J.J.; Sweet, T.J.; Bailey, E.; Faith, S.A.; Booth, T. Resveratrol inhibition of varicella-zoster virus replication in vitro. Antivir. Res. 2006, 72, 171–177. [Google Scholar] [CrossRef]
- Kapadia, G.J.; Azuine, M.A.; Tokuda, H.; Takasaki, M.; Mukainaka, T.; Konoshima, T.; Nishino, H. Chemopreventive effect of resveratrol, sesamol, sesame oil and sunflower oil in the Epstein-Barr virus early antigen activation assay and the mouse skin two-stage carcinogenesis. Pharmacol. Res. 2002, 45, 499–505. [Google Scholar] [CrossRef]
- Jones, R.J.; Seaman, W.T.; Feng, W.H.; Barlow, E.; Dickerson, S.; Delecluse, H.J.; Kenney, S.C. Roles of lytic viral infection and IL-6 in early versus late passage lymphoblastoid cell lines and EBV-associated lymphoproliferative disease. Int. J. Cancer 2007, 121, 1274–1281. [Google Scholar] [CrossRef]
- Hong, G.K.; Kumar, P.; Wang, L.; Damania, B.; Gulley, M.L.; Delecluse, H.J.; Polverini, P.J.; Kenney, S.C. Epstein-Barr virus lytic infection is required for efficient production of the angiogenesis factor vascular endothelial growth factor in lymphoblastoid cell lines. J. Virol. 2005, 79, 13984–13992. [Google Scholar] [CrossRef]
- Lin, J.C.; Cherng, J.M.; Hung, M.S.; Baltina, L.A.; Baltina, L.; Kondratenko, R. Inhibitory effects of some derivatives of glycyrrhizic acid against Epstein-Barr virus infection: structure-activity relationships. Antivir. Res. 2008, 79, 6–11. [Google Scholar]
- Kang, O.H.; Jang, H.J.; Chae, H.S.; Oh, Y.C.; Choi, J.G.; Lee, Y.S.; Kim, J.H.; Kim, Y.C.; Sohn, D.H.; Park, H.; Kwon, D.Y. Anti-inflammatory mechanisms of resveratrol in activated HMC-1 cells: pivotal roles of NF-kappaB and MAPK. Pharmacol. Res. 2009, 59, 330–337. [Google Scholar] [CrossRef]
- Matusali, G.; Arena, G.; De Leo, A.; Di Renzo, L.; Mattia, E. Inhibition of p38 MAP kinase pathway induces apoptosis and prevents Epstein Barr virus reactivation in Raji cells exposed to lytic cycle inducing compounds. Mol. Cancer 2009, 8, 18. [Google Scholar] [CrossRef]
- Rahman, M.M.; Kukita, A.; Kukita, T.; Shobuike, T.; Nakamura, T.; Kohashi, O. Two histone deacetylase inhibitors, trichostatin A and sodium butyrate, suppress differentiation into osteoclasts but not into macrophages. Blood 2003, 101, 3451–3459. [Google Scholar] [CrossRef]
- Adamson, A.L.; Darr, D.; Holley-Guthrie, E.; Johnson, R.A.; Mauser, A.; Swenson, J.; Kenney, S. Epstein-Barr virus immediate-early proteins BZLF1 and BRLF1 activate the ATF2 transcription factor by increasing the levels of phosphorylated p38 and c-Jun N-terminal kinases. J. Virol. 2000, 74, 1224–1233. [Google Scholar] [CrossRef]
- Borra, M.T.; Smith, B.C.; Denu, J.M. Mechanism of human SIRT1 activation by resveratrol. J. Biol. Chem. 2005, 280, 17187–17195. [Google Scholar] [CrossRef]
- Chang, L.K.; Liu, S.T. Activation of the BRLF1 promoter and lytic cycle of Epstein-Barr virus by histone acetylation. Nucleic Acids Res. 2000, 28, 3918–3925. [Google Scholar] [CrossRef]
- Deng, Z.; Chen, C.J.; Chamberlin, M.; Lu, F.; Blobel, G.A.; Speicher, D.; Cirillo, L.A.; Zaret, K.S.; Lieberman, P.M. The CBP bromodomain and nucleosome targeting are required for Zta-directed nucleosome acetylation and transcription activation. Mol. Cell Biol. 2003, 23, 2633–2644. [Google Scholar] [CrossRef]
- Luka, J.; Kallin, B.; Klein, G. Induction of the Epstein-Barr virus (EBV) cycle in latently infected cells by n-butyrate. Virology 1979, 94, 228–231. [Google Scholar] [CrossRef]
- Carmichael, J.; DeGraff, W.G.; Gazdar, A.F.; Minna, J.D.; Mitchell, J.B. Evaluation of a tetrazolium-based semiautomated colorimetric assay: assessment of chemosensitivity testing. Cancer Res. 1987, 47, 936–942. [Google Scholar]
- Chang, P.J.; Chang, Y.S.; Liu, S.T. Role of Rta in the translation of bicistronic BZLF1 of Epstein-Barr virus. J. Virol. 1998, 72, 5128–5136. [Google Scholar]
- Chiu, Y.F.; Tung, C.P.; Lee, Y.H.; Wang, W.H.; Li, C.; Hung, J.Y.; Wang, C.Y.; Kawaguchi, Y.; Liu, S.T. A comprehensive library of mutations of Epstein Barr virus. J. Gen. Virol. 2007, 88, 2463–2472. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).