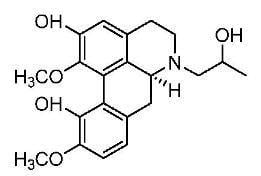
(+)-N-(2-Hydroxypropyl)lindcarpine: A New Cytotoxic Aporphine Isolated from Actinodaphne pruinosa Nees
Abstract
:Introduction
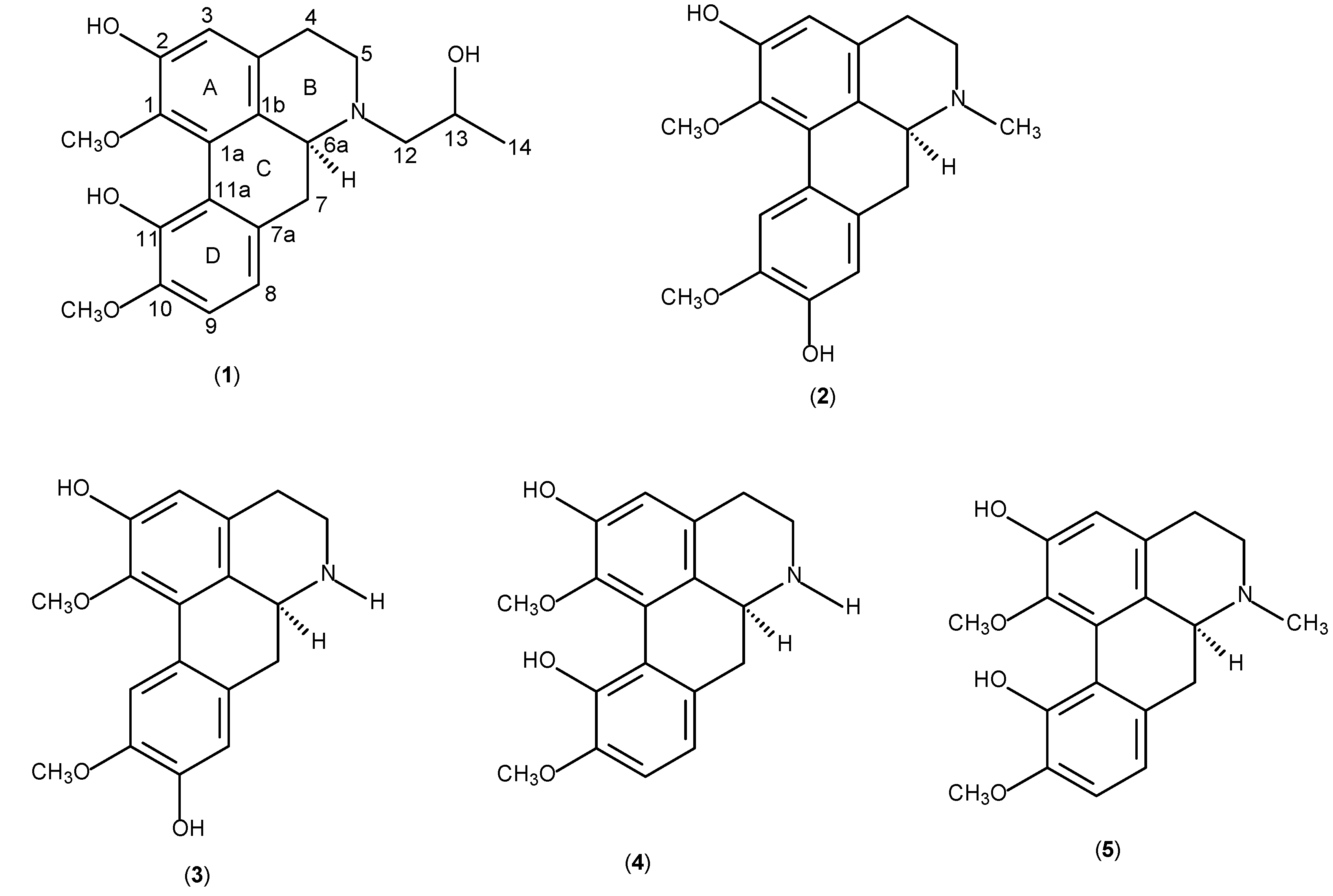
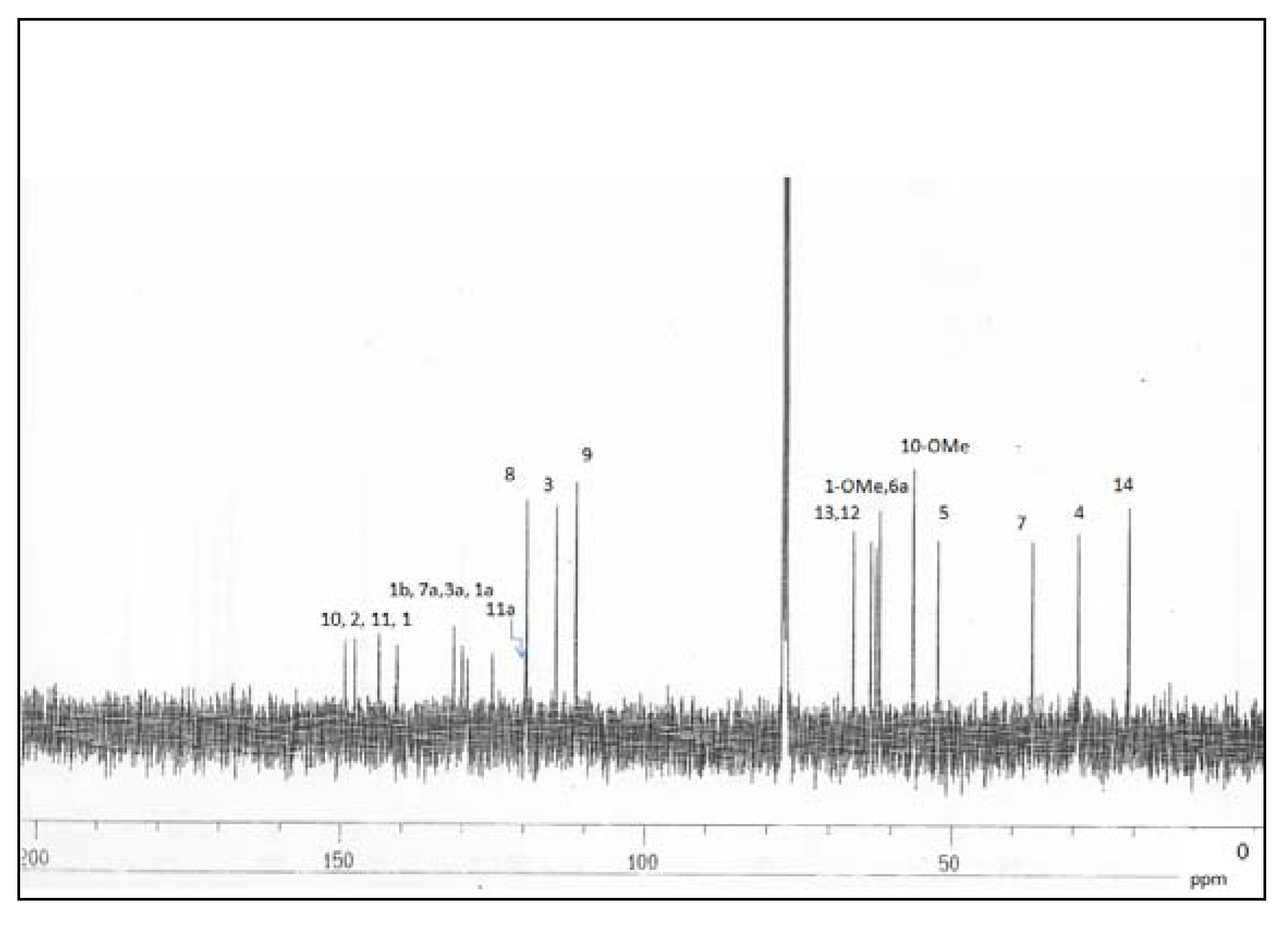
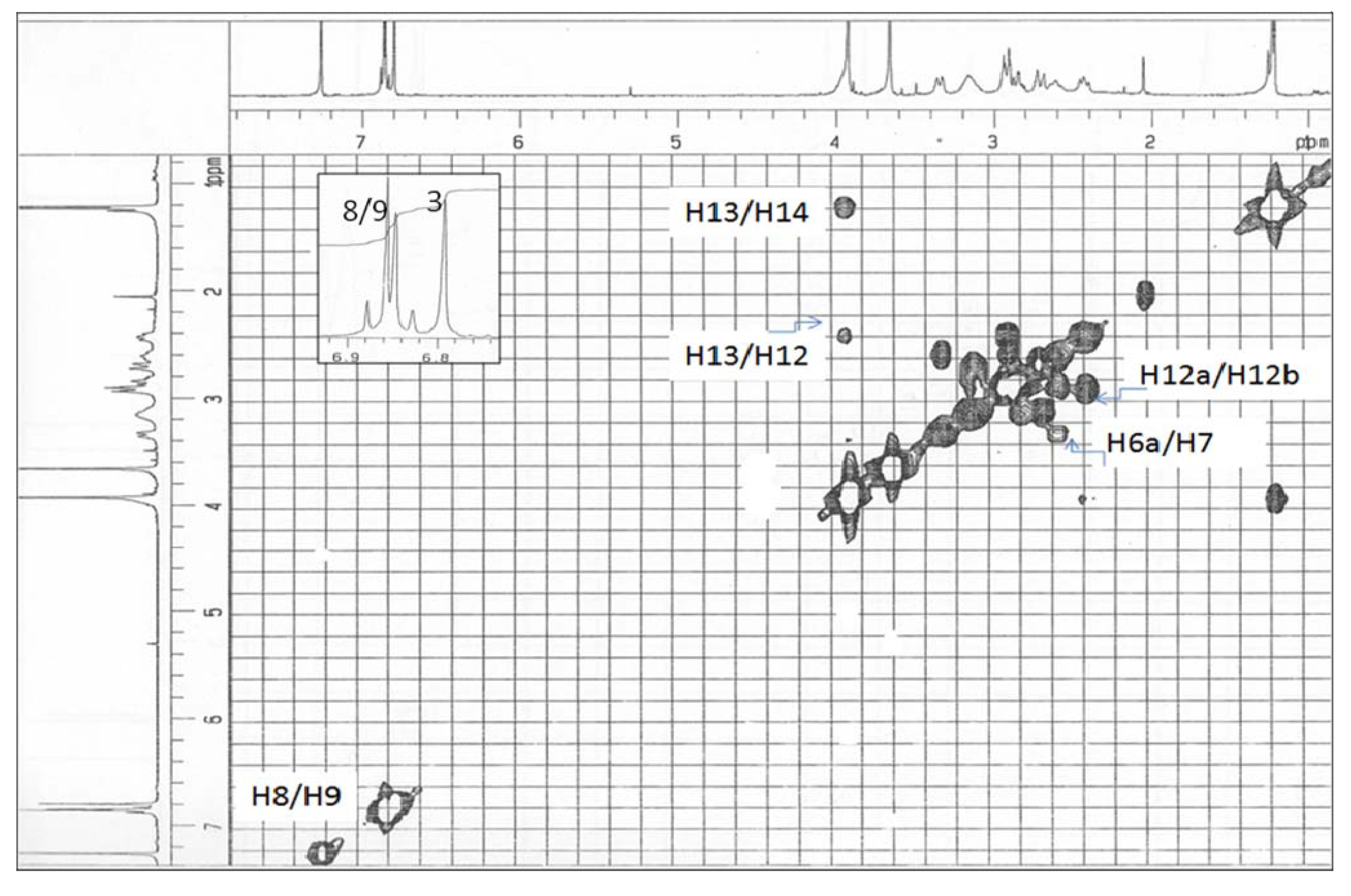
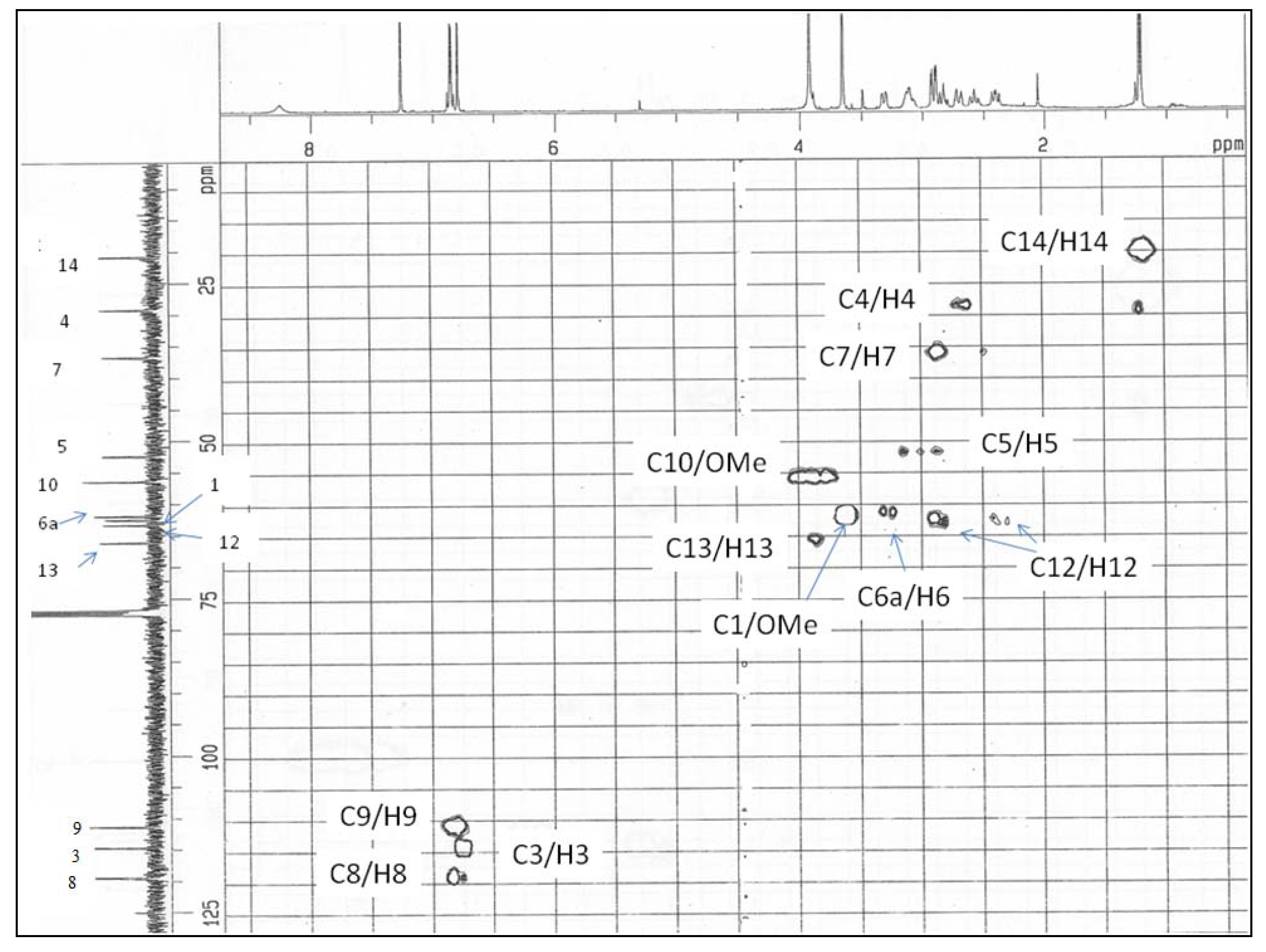
Results and Discussion
| Position | δ 1H (Hz) | δ 13C | HMBC (2J, 3J) |
|---|---|---|---|
| 1 | 140.6 | ||
| 1a | 125.0 | ||
| 1b | 131.2 | ||
| 2 | 147.6 | ||
| 3 | 6.79 s | 114.0 | 1, 2, 3a, 4 |
| 3a | 129.2 | ||
| 4 | 2.67 m | 29.1 | |
| 2.71 m | 1b | ||
| 5 | 3.08 m | 52.3 | 12, 6a |
| 6a | 3.30 dd (13.1, 3.2) | 61.9 | 12 |
| 7 | 2.90 dd (13.1, 3.2) | 36.7 | 6a |
| 2.56 t (13.1) | |||
| 7a | 129.9 | ||
| 8 | 6.84 d (8.0) | 119.5 | 7, 11a, 10 |
| 9 | 6.86 d (8.0 | 111.4 | 7a, 11 |
| 10 | 149.2 | ||
| 11 | 143.2 | ||
| 11a | 119.6 | ||
| 12 | 2.80 m | 63.3 | 5, 6a |
| 2.38 dd (13.7, 9.0) | 5, 13 | ||
| 13 | 3.89 m | 66.1 | |
| 14 (Me) | 1.22 d (6.1) | 20.8 | 13 |
| 1-OMe | 3.65 s | 62.4 | 1 |
| 10-OMe | 3.92 s | 56.4 | 10 |
Conclusions
Experimental
General
Plant material
Extraction and isolation of the alkaloids
Cytotoxic assays
Acknowledgements
References
- Burkill, I.H. A Dictionary of the Economic Products of the Malay Peninsula; Ministry of Agriculture Malaysia: Kuala Lumpur, Malaysia, 1935; pp. 42–43. [Google Scholar]
- Ridley, H.N. The Flora of the Malay Peninsula; Reeve & Co. Ltd.: London, UK, 1924; pp. 107–112. [Google Scholar]
- Kim, M.R.; Jung, H.J.; Min, B.S.; Oh, S.R.; Kim, C.H.; Ahn, K.S.; Kang, W.S.; Lee, H.K. Constituents from the stems of Actinodaphne lancifolia. Phytochemistry 2002, 59, 861–865. [Google Scholar] [CrossRef]
- Uprety, H.; Bhakuni, D.S.; Dhar, M.M. Aporphine Alkaloids of Litsea Sebifera, L. Wightiana and Actinodaphne Obovata. Phytochemistry 1972, 11, 3057–3059. [Google Scholar] [CrossRef]
- Leboeuf, M.; Cavé, A.; Bhaumik, P.K.; Mukherjee, B.; Mukherjee, R. The Phytochemistry of the Annonaceae. Phytochemistry 1980, 21, 2783–2813. [Google Scholar] [CrossRef]
- Kittisak, L.; Angerhofer, C.K.; Chai, H.; Pezzuto, J.M.; Cordell, G.A.; Ruangrungsi, N. Cytotoxic and Antimalarial Alkaloids from the Tubers of Stephania pierrei. J. Nat. Prod. 1993, 56, 1468–1478. [Google Scholar] [CrossRef]
- Guinaudeau, H.; Leboeuf, M.; Cavé, A. Aporphine alkaloids. Lloydia 1975, 38, 275–288. [Google Scholar]
- Shao, W.S.; Shoei, S.L.; Li, Y.C.; Chien, K.C. Separation Of Aporphine Alkaloids By Micellar Electrokinetic Chromatography. J. Chromatogr. A 1997, 767, 277–284. [Google Scholar] [CrossRef]
- Johns, S.R.; Lamberton, J.A.; Sioumis, A.A. Alkaloids of Actinodaphne nitida (Lauraceae). Aust. J. Chem. 1969, 22, 2257–2257. [Google Scholar] [CrossRef]
- Lu, S.T.; Wang, S.J.; Lin, F.S. Studies on the Alkaloids of Formosan Lauraceous Plants. XIV.: Alkaloids of Actinodaphne acutivena (Hayata) Nakai and Litsea hayatae. J. Pharm. Soc. Jap. 1969, 89, 1313–1317. [Google Scholar]
- Alley, M.C.; Scudiero, D.A.; Monks, A.; Hursey, M.L.; Czerwinski, M.J.; Fine, D.L.; Abbott, B. J.; Mayo, J.G.; Shoemaker, R.H.; Boyd, M.R. Feasibility of Drug Screening with Panels of Human Tumor Cell Lines Using a Microculture Tetrazolium Assay. Cancer Res. 1988, 48, 589–610. [Google Scholar]
- Mukhtar, M.R.; Martin, M.T.; Domansky, M.; Pais, M.; Hadi, A.H.A.; Awang, K. Phoebegrandines A and B, Proaporphine-Tryptamine Dimers, from Phoebe grandis. Phytochemistry 1997, 45, 1543–1546. [Google Scholar] [CrossRef]
- Mukhtar, M.R.; Hadi, A.H.A.; Sevenet, T.; Martin, MT.; Awang, K. Phoebegrandine C A Novel Proaporphine-Tryptamine Dimer from Phoebe grandis (Nees) Merr. Nat. Prod. Res. 2004, 18, 163–167. [Google Scholar] [CrossRef]
- Sample Availability: Samples of compounds 2-4 are available from the authors.
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Rachmatiah, T.; Mukhtar, M.R.; Nafiah, M.A.; Hanafi, M.; Kosela, S.; Morita, H.; Litaudon, M.; Awang, K.; Omar, H.; Hadi, A.H.A. (+)-N-(2-Hydroxypropyl)lindcarpine: A New Cytotoxic Aporphine Isolated from Actinodaphne pruinosa Nees. Molecules 2009, 14, 2850-2856. https://doi.org/10.3390/molecules14082850
Rachmatiah T, Mukhtar MR, Nafiah MA, Hanafi M, Kosela S, Morita H, Litaudon M, Awang K, Omar H, Hadi AHA. (+)-N-(2-Hydroxypropyl)lindcarpine: A New Cytotoxic Aporphine Isolated from Actinodaphne pruinosa Nees. Molecules. 2009; 14(8):2850-2856. https://doi.org/10.3390/molecules14082850
Chicago/Turabian StyleRachmatiah, Tiah, Mat Ropi Mukhtar, Mohd Azlan Nafiah, Muhammad Hanafi, Soleh Kosela, Hiroshi Morita, Marc Litaudon, Khalijah Awang, Hanita Omar, and A. Hamid A. Hadi. 2009. "(+)-N-(2-Hydroxypropyl)lindcarpine: A New Cytotoxic Aporphine Isolated from Actinodaphne pruinosa Nees" Molecules 14, no. 8: 2850-2856. https://doi.org/10.3390/molecules14082850
APA StyleRachmatiah, T., Mukhtar, M. R., Nafiah, M. A., Hanafi, M., Kosela, S., Morita, H., Litaudon, M., Awang, K., Omar, H., & Hadi, A. H. A. (2009). (+)-N-(2-Hydroxypropyl)lindcarpine: A New Cytotoxic Aporphine Isolated from Actinodaphne pruinosa Nees. Molecules, 14(8), 2850-2856. https://doi.org/10.3390/molecules14082850