Trypanocidal Activity of Oxoaporphine and Pyrimidine-β-Carboline Alkaloids from the Branches of Annona foetida Mart. (Annonaceae)
Abstract
:1. Introduction
2. Results and Discussion
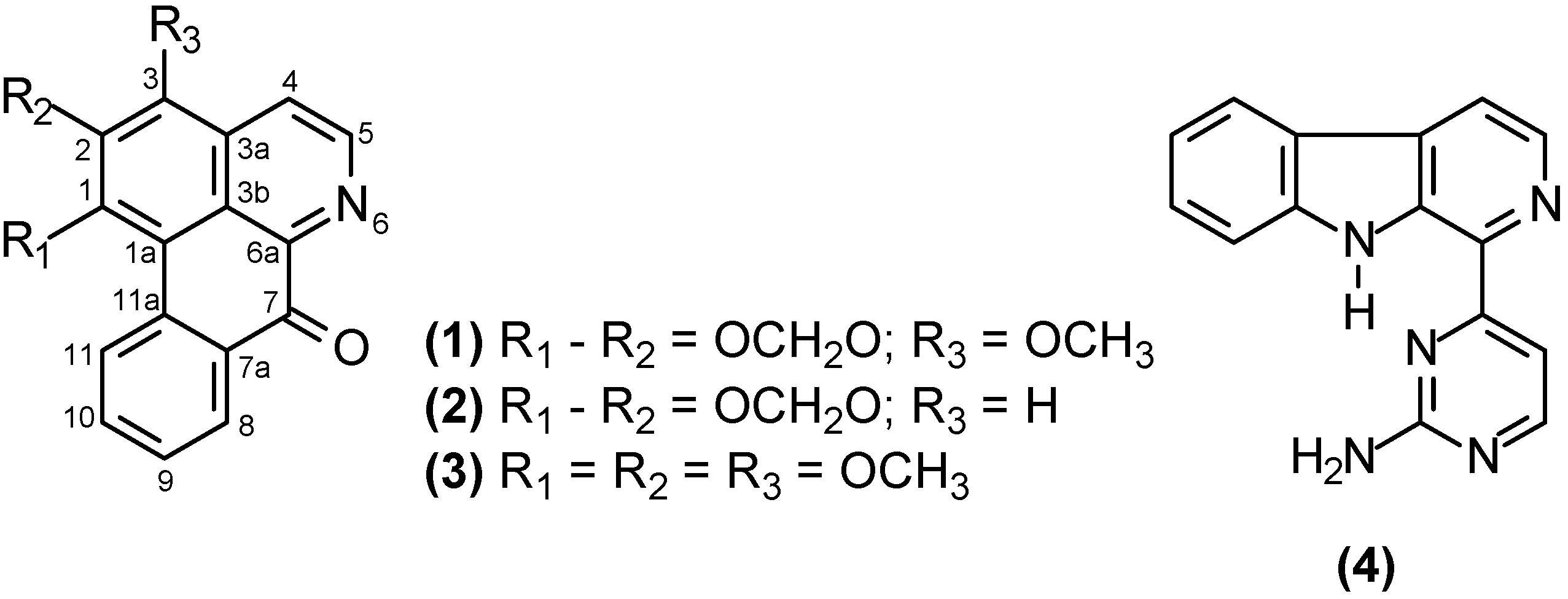
| Compounds | IC50 (µg/mL) | EC50(µg/mL) |
|---|---|---|
| Epimastigote forms | Trypomastigote forms | |
| Liriodenine (2) | 177.0 ± 10.6 | 4.0 ± 0.2 |
| O-methylmoschatoline (3) | 92.0 ± 18.4 | 3.8 ± 1.8 |
| Annomontine (4) | 198.0 ± 4.2 | 4.2 ± 1.9 |
| Benznidazole a | 2.0 ± 0.9 | |
| Crystal violet b | 12.8± 0.9 |
3. Experimental
3.1. General
3.2. Plant Material
3.3. Extraction and Isolation Procedures
3.4. In Vitro Trypanocidal Assay
4. Conclusions
Acknowledgments
Conflict of Interest
References and Notes
- Chatrou, L.W.; Escribano, M.P.; Viruel, M.A.; Maas, J.W.; Richardson, J.E.; Hormaza, J.I. Flanking regions of monomorphic microsatellite loci provide a new source of data for plant species-level phylogenetics. Mol. Phylogenet. Evol. 2009, 53, 726–733. [Google Scholar] [CrossRef]
- Corrêa, M.P. Dicionário das plantas úteis do Brasil e das exóticas cultivadas; IBDF: Rio de Janeiro, RJ, Brazil, 1984. [Google Scholar]
- Alali, F.Q.; Liu, X.-X.; McLaughlin, J.L. Annonaceous acetogenins: Recent progress. J. Nat. Prod. 1999, 62, 504–540. [Google Scholar] [CrossRef]
- Wu, Y.-C.; Chang, G.-Y.; Duh, C.-Y.; Wang, S.-K. Cytotoxic alkaloids of Annona montana. Phytochemistry 1993, 33, 497–500. [Google Scholar]
- Fatope, M.O.; Audu, O.T.; Takeda, Y.; Zeng, L.; Shi, G.; Shimada, H.; McLaughlin, J.L. Bioactive ent-kaurene diterpenoids from Annona senegalensis. J. Nat. Prod. 1996, 59, 301–303. [Google Scholar] [CrossRef]
- Costa, E.V.; Dutra, L.M.; De Jesus, H.C.R.; Nogueira, P.C.L.; Moraes, V.R.S.; Salvador, M.J.; Cavalcanti, S.C.H.; Dos Santos, R.L.C.; Prata, A.P.N. Chemical composition and antioxidant, antimicrobial, and larvicidal activities of the essential oils of Annona salzmannii and A. pickelii (Annonaceae). Nat. Prod. Commun. 2011, 6, 907–912. [Google Scholar]
- Costa, E.V.; Pinheiro, M.L.B.; Silva, J.R.A.; Maia, B.H.L.N.S.; Duarte, M.C.T.; Amaral, A.C.F.; Machado, G.M.C.; Leon, L.L. Antimicrobial and antileishmanial activity of essential oil from the leaves of Annona foetida (Annonaceae). Quim. Nova 2009, 32, 78–81. [Google Scholar] [CrossRef]
- Yang, Y.-L.; Chang, F.-R.; Wu, C.-C.; Wang, W.-Y.; Wu, Y.-C. New ent-kaurane diterpenoids with anti-platelet aggregation activity from Annona squamosa. J. Nat. Prod. 2002, 65, 1462–1467. [Google Scholar] [CrossRef]
- Chang, F.-R.; Wei, J.-L.; Teng, C.-M.; Wu, Y.-C. Two new 7-dehydroaporphine alkaloids and antiplatelet action aporphines from the leaves of Annona purpurea. Phytochemistry 1998, 49, 2015–2018. [Google Scholar] [CrossRef]
- Siqueira, C.A.T.; Oliani, J.; Sartoratto, A.; Queiroga, C.L.; Moreno, P.R.H.; Reimão, J.Q.; Tempone, A.G.; Fischer, D.C.H. Chemical constituents of the volatile oil from leaves of Annona coriacea Mart., Annonaceae, and in vitro antiprotozoal activity. Rev. Bras. Farmacogn. 2011, 21, 33–40. [Google Scholar]
- Costa, E.V.; Pinheiro, M.L.B.; Xavier, C.M.; Silva, J.R.A.; Amaral, A.C.F.; Souza, A.D.L.; Barison, A.; Campos, F.R.; Ferreira, A.G.; Machado, G.M.C.; Leon, L.L.P. A pyrimidine-β-carboline and other alkaloids from Annona foetida with antileishmanial activity. J. Nat. Prod. 2006, 69, 292–294. [Google Scholar] [CrossRef]
- Queiroz, E.F.; Roblot, F.; Cavé, A.; Paulo, M.Q.; Fournet, A. Pessoine and spinosine, two catecholic berbines from Annona spinescens. J. Nat. Prod. 1996, 59, 438–440. [Google Scholar] [CrossRef]
- Pinheiro, M.L.B.; Xavier, C.M.; de Souza, A.D.L.; Rabelo, D.M.; Batista, C.L.; Batista, R.L.; Costa, E.V.; Campos, F.R.; Barison, A.; Valdez, R.H.; et al. Acanthoic acid and other constituents from the stem of Annona amazonica (Annonaceae). J. Braz. Chem. Soc. 2009, 20, 1095–1102. [Google Scholar]
- Maas, P.J.M.; Maas, H.; Miralha, J.M.S.; Junikka, L. Flora da Reserva Ducke, Amazonas, Brasil: Annonaceae. Rodriguésia 2007, 58, 617–662. [Google Scholar]
- Chen, C.-Y.; Chang, F.-R.; Wu, Y.-C. The constituents from the stems of Annona cherimola. J. Chin. Chem. Soc. 1997, 44, 313–319. [Google Scholar]
- Costa, E.V.; Marques, F.A.; Pinheiro, M.L.B.; Vaz, N.P.; Duarte, M.C.T.; Delarmelina, C. 7,7-Dimethylaporphine alkaloids from the stem of Guatteriopsis friesiana. J. Nat. Prod. 2009, 72, 1516–1519. [Google Scholar] [CrossRef]
- Costa, E.V.; Pinheiro, M.L.B.; Souza, A.D.L.; Santos, A.G.; Campos, F.R.; Ferreira, A.G.; Barison, A. Full NMR analysis of annomontine, methoxyannomontine and N-hydroxyannomontine pyrimidine-β-carboline alkaloids. Magn. Reson. Chem. 2008, 46, 69–74. [Google Scholar] [CrossRef]
- Leboeuf, M.; Cavé, A.; Forgacs, P.; Provost, J.; Chiaroni, A.; Riche, C. Alkaloids of the Annonaceae. Part 33. Annomontine and methoxyannomontine, two new pyrimidine-β-carboline-type alkaloids from Annona montana. J. Chem. Soc. Perkin Trans. 1 1982, 1205–1208. [Google Scholar]
- Yang, T.H.; Cheng, M.Y. The alkaloids of Annona reticulata L. II. Taiwan Yaoxue Zazhi 1987, 39, 195–201. [Google Scholar]
- Izumi, E.; Ueda-Nakamura, T.; Dias Filho, B.P.; Veiga Júnior, V.F.; Nakamura, C.V. Natural products and Chagas’ disease: A review of plant compounds studied for activity against Trypanosoma cruzi. Nat. Prod. Rep. 2011, 28, 809–823. [Google Scholar] [CrossRef]
- Rivas, P.; Cassels, B.K.; Morello, A.; Repetto, Y. Effects of some β-carboline alkaloids on intact Trypanosoma cruzi epimastigotes. Comp. Biochem. Physiol.-C: Pharmacol. Toxicol. Endocrinol. 1999, 122, 27–31. [Google Scholar] [CrossRef]
- Valdez, R.H; Düsman, T.L.T.; Ueda-Nakamura, T.; Dias Filho, B.P.; Morgado-Díaz, J.A.; Sarragiotto, M.H.; Nakamura, C.V. Biological activity of 1,2,3,4-tetrahydro-β-carboline-3-carboxamides against Trypanosoma cruzi. Acta Trop. 2009, 110, 7–14. [Google Scholar] [CrossRef]
- Brener, Z. Therapeutic activity criterion of cure on mice experimentally infected with Trypanosoma cruzi. Rev. Inst. Med. Trop. S. Paulo 1962, 4, 389–396. [Google Scholar]
- Sample Availability: Samples of the alkaloids 1–4 are available from the authors.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Costa, E.V.; Pinheiro, M.L.B.; Souza, A.D.L.d.; Barison, A.; Campos, F.R.; Valdez, R.H.; Ueda-Nakamura, T.; Filho, B.P.D.; Nakamura, C.V. Trypanocidal Activity of Oxoaporphine and Pyrimidine-β-Carboline Alkaloids from the Branches of Annona foetida Mart. (Annonaceae). Molecules 2011, 16, 9714-9720. https://doi.org/10.3390/molecules16119714
Costa EV, Pinheiro MLB, Souza ADLd, Barison A, Campos FR, Valdez RH, Ueda-Nakamura T, Filho BPD, Nakamura CV. Trypanocidal Activity of Oxoaporphine and Pyrimidine-β-Carboline Alkaloids from the Branches of Annona foetida Mart. (Annonaceae). Molecules. 2011; 16(11):9714-9720. https://doi.org/10.3390/molecules16119714
Chicago/Turabian StyleCosta, Emmanoel Vilaça, Maria Lúcia Belém Pinheiro, Afonso Duarte Leão de Souza, Andersson Barison, Francinete Ramos Campos, Rodrigo Hinojosa Valdez, Tânia Ueda-Nakamura, Benedito Prado Dias Filho, and Celso Vataru Nakamura. 2011. "Trypanocidal Activity of Oxoaporphine and Pyrimidine-β-Carboline Alkaloids from the Branches of Annona foetida Mart. (Annonaceae)" Molecules 16, no. 11: 9714-9720. https://doi.org/10.3390/molecules16119714
APA StyleCosta, E. V., Pinheiro, M. L. B., Souza, A. D. L. d., Barison, A., Campos, F. R., Valdez, R. H., Ueda-Nakamura, T., Filho, B. P. D., & Nakamura, C. V. (2011). Trypanocidal Activity of Oxoaporphine and Pyrimidine-β-Carboline Alkaloids from the Branches of Annona foetida Mart. (Annonaceae). Molecules, 16(11), 9714-9720. https://doi.org/10.3390/molecules16119714




