Synthesis of N-Substituted 5-Iodouracils as Antimicrobial and Anticancer Agents
Abstract
:Introduction

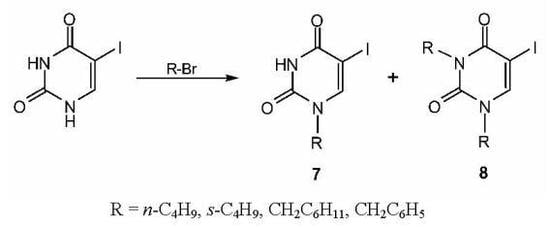
Results and Discussion
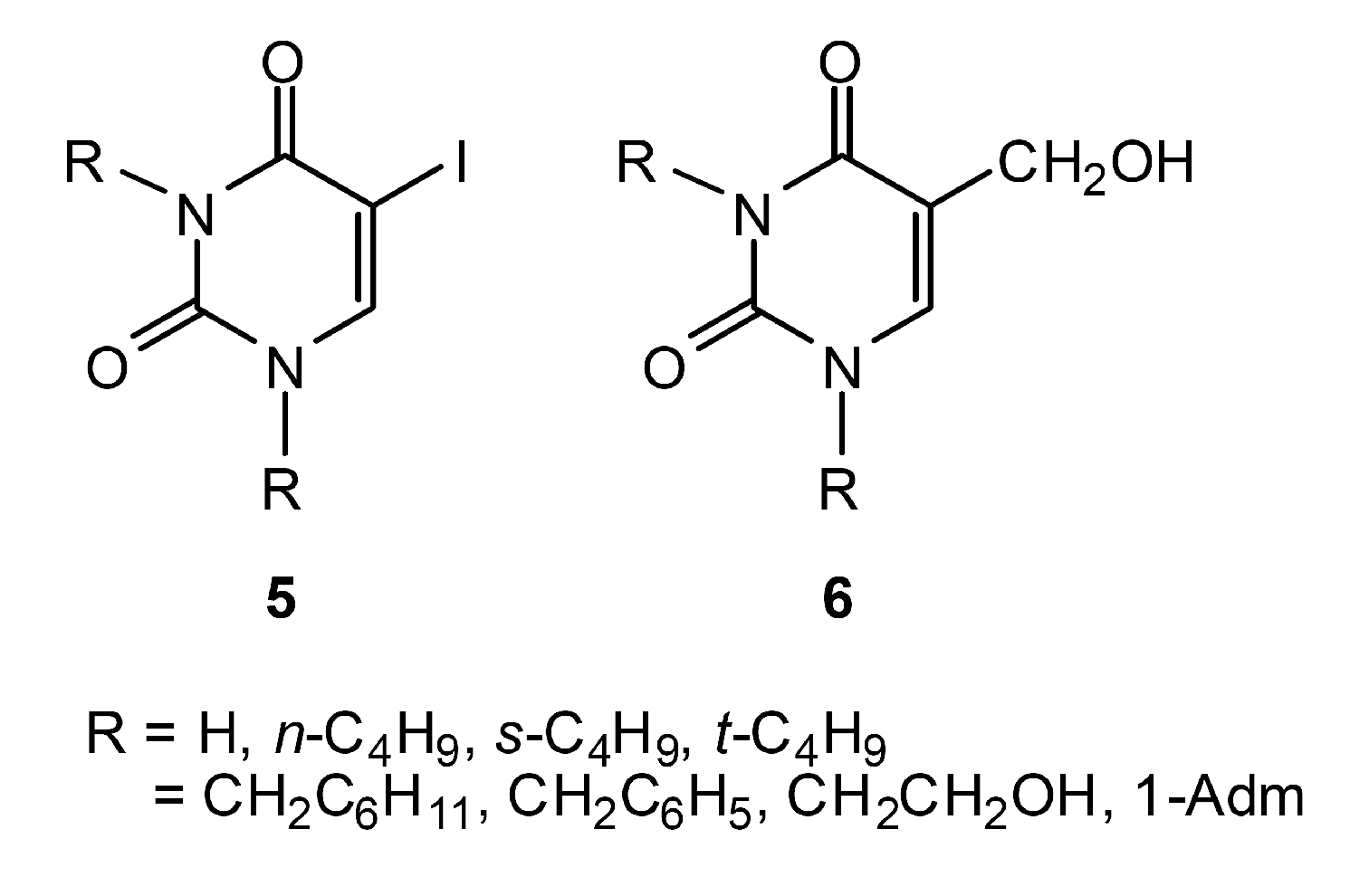
Chemistry
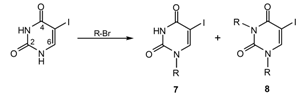 | |||
| Entry | R | Substitution Products (%) | |
| N1- | N1, N3- | ||
| 1 |  | 7a (28.0) | 8a (6.7) |
| 2 |  | 7b (6.1) | ― |
| 3 |  | 7c (28.1) | 8b (7.5) |
| 4 | 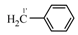 | 7d (11.4) | ― |
| 5 | t-C4H9 | ― | ― |
| 6 | 1-Adm | ― | ― |
| 7 | CH2CH2−OH | ― | ― |
| Compound | δ (ppm) | υmax (cm-1) | Mass spectra (m/z) | |||||
|---|---|---|---|---|---|---|---|---|
| H-6 | C-6 | C=O | NH | Molecular ion | Base peak | |||
| 7a | 7.59 | 148.87 | 1715,1667 | 3022 | 294 | 294 | ||
| 7b | 7.53 | 145.21 | 1716, 1700 | 3159 | 294 | 237 | ||
| 7c | 7.54 | 149.36 | 1701, 1660 | 3159 | 334 | 238 | ||
| 7d | 8.17 | 149.48 | 1714, 1669 | 3112 | 328 | 91 | ||
| 8a | 7.60 | 146.74 | 1698, 1651 | ° | 350 | 333 | ||
| 8b | 7.53 | 147.29 | 1701, 1653 | ° | 430 | 238 | ||
Antibacterial activity
| Compound** | Activity | Inhibition (%) | |
|---|---|---|---|
| 7a | Active | 50a | 25b,c |
| 7c | Active | 50a | 25b,c |
| 7d | Active | 50a | 25b,c |
| 8b | Inactive | 0 | 0 |
Antimalarial activity
Anticancer activity
| Cell line | IC50 (μg/mL)a,b | ||||||
|---|---|---|---|---|---|---|---|
| 7a | 7b | 7c | 7d | 8a | 8b | Etoposide(Doxorubicin) | |
| HepG2 | >50 | >50 | 36.00 | >50 | >50 | 16.50 | 12.00 |
| HuCCA-1 | >50 | >50 | >50 | >50 | >50 | 49.00 | (0.50) |
| A549 | >50 | >50 | >50 | >50 | >50 | 33.00 | 0.60 (0.45) |
| MOLT-3 | NA | >50 | NA | NA | 37.53 | >50 | 0.019 |
| KB | >50 | NA | 35.00 | >50 | NA | NA | 0.25 |
| HCC-S102 | >50 | NA | >50 | >50 | NA | NA | 6.00 |
| HL60 | >50 | NA | >50 | >50 | NA | NA | 0.85 |
| P388 | >50 | NA | 41.47 | >50 | NA | NA | 0.12 |
| HeLa | >50 | NA | 46.00 | >50 | NA | NA | 0.38 |
| MDA-MB231 | >50 | NA | >50 | >50 | NA | NA | 0.24 |
| T47D | >50 | NA | 20.00 | 43.00 | NA | NA | 0.05 |
| H69AR | >50 | NA | 50.00 | >50 | NA | NA | 30.00 |
Conclusions
Experimental
General
Synthesis of N-substituted 5-iodouracil analogs 7a-d and 8a-b
Chloroquine resistant Plasmodium falciparum (T9.94)
Cancer cells
Antimicrobial assay
| Microorganisms | |
|---|---|
| Gram-negative bacteria | |
| Escherichia coli ATCC 25922 | Morganella morganii |
| Klebsiella pneumoniae ATCC 700603 | Vibrio cholera |
| Salmonella typhimurium ATCC 13311 | Vibrio mimicus |
| Salmonella choleraesuis ATCC 10708 | Aeromonas hydrophila |
| Pseudomonas aeruginosa ATCC 15442 | Plesiomonas shigelloides |
| Edwardsiella tarda | Xanthomonas maltophilia |
| Shigella dysenteriae | Neisseria mucosa |
| Citrobacter freundii | Branhamella catarrhalis |
| Gram-positive bacteria | |
| Stapphylococcus aureus ATCC 25923 | Bacillus subtilis ATCC 6633 |
| Stapphylococcus epidermidis ATCC 12228 | Streptococcus pyogenes |
| Enterococcus faecalis ATCC 29212 | Listeria monocytogenes |
| Micrococcus lutens ATCC 10240 | Bacillus cereus |
| Corynebacterium diphtheriae NCTC10356 | Micrococcusflavas |
| Diploid fungus (Yeast) | |
| Candida albicans | |
Antimalarial assay
Cytotoxicity assays
| Cell lines | |
|---|---|
| Human hepatocellular liver carcinoma cell line (HepG2) | Human promyelocytic leukemia cell line (HL-60) |
| Human cholangiocarcinoma cancer cells (HuCCA-1) | Murine leukemia cell line (P388) |
| Human lung carcinoma cell line (A549) | Cervical adenocarcinoma cell line (HeLa) |
| T-lymphoblast (MOLT-3, acute lymphoblastic leukemia) | Hormone-independent breast cancer cell line (MDA-MB231) |
| Human epidermoid carcinoma of the mouth (KB) | Hormone-dependent breast cancer cell line (T47D) |
| Hepatocellular carcinoma cell line (HCC-S102) | Multidrug-resistance small cell lung cancer cell line (H69AR) |
Acknowledgements
- Samples Availability: Contact the authors.
References
- Ogilvie, K.K.; Hamilton, R.G.; Gillen, M.F.; Radatus, B.K.; Smith, K.O.; Galloway, K.S. Uracil analogues of the acyclonucleoside 9-[[2-hydroxy-1-(hydroxymethyl)ethoxy]- methyl]guanine (BIOLF-62). Can. J. Chem. 1984, 62, 16–21. [Google Scholar] [CrossRef]
- Zenker, N. Thyroid function and thyroid drugs. In Principles of Medicinal Chemistry; Foye, W.O., Ed.; Lea & Febiger: Philadelphia, London, UK, 1990; pp. 603–621. [Google Scholar]
- del Carmen Núñeza, M.; Entrena, A.; Rodríguez-Serrano, F.; Marchal, J.A.; Aránega, A.; Gallo, M.A.; Espinosa, A.; Campos, J.M. Synthesis of novel 1-(2,3-dihydro-5H-4,1-benzoxathiepin-3-yl)-uracil and -thymine, and their corresponding S-oxidized derivatives. Tetrahedron 2005, 61, 10363–10369. [Google Scholar] [CrossRef]
- De Clercq, E. Antiviral drugs in current clinical use. J. Clin. Virol. 2004, 30, 115–133. [Google Scholar] [CrossRef]
- Tanaka, H.; Takashima, H.; Ubasawa, M.; Sekiya, K.; Nitta, I.; Baba, M.; Shigeta, S.; Walker, R.T.; De Clercq, E.; Miyasaka, T. Synthesis and antiviral activity of deoxy analogs of 1-[(2-hydroxyethoxy)methyl]-6-(phenylthio)thymine (HEPT) as potent and selective anti-HIV-1 agents. J. Med. Chem. 1992, 35, 4713–4719. [Google Scholar] [CrossRef]
- Semenov, V.E.; Voloshina, A.D.; Toroptzova, E.M.; Kulik, N.V.; Zobov, V.V.; Giniyatullin, R.K.; Mikhailov, A.S.; Nikolaev, A.E.; Akamsin, V.D.; Reznik, V.S. Antibacterial and antifungal activity of acyclic and macrocyclic uracil derivatives with quaternized nitrogen atoms in spacers. Eur. J. Med. Chem. 2006, 41, 1093–1101. [Google Scholar] [CrossRef]
- Kamalakannan, P.; Venkappayya, D.; Balasubramanian, T. A new antimetabolite, 5-morpholinomethyl-2-thiouracil—spectral properties, thermal profiles, antibacterial, antifungal and antitumour studies of some of its metal chelates. J. Chem. Soc., Dalton Trans. 2002, 3381–3391. [Google Scholar]
- Motawia, M.S.; Abdel-Megied, A.E.S.; Pedersen, E.B.; Nielsen, C.M.; Ebbesen, P. Synthesis of 5-alkoxymethyl derivatives of 3'-amino-2',3'-dideoxyuridine and evaluation of their activity against HIV and cancer. Acta Chem. Scand. 1992, 46, 77–81. [Google Scholar] [CrossRef]
- Fabrissin, S.; de Nardo, M.; Nisi, C.; Morasca, L.; Dolfini, E.; Franchi, G. Synthesis and anticancer activity of 5-diethylaminomethyl derivatives and nitrogen mustards of uracil and 2-thiouracils. J. Med. Chem. 1976, 19, 639–642. [Google Scholar] [CrossRef]
- Gondela, A.; Walczak, K. N-Functionalization of uracil derivatives: synthesis of chiral 2-(3-methyl-5-nitro-2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)alkanoic acids and their methyl esters. Tetrahedron: Asymmetry 2005, 16, 2107–2112. [Google Scholar] [CrossRef]
- Malik, V.; Singh, P.; Kumar, S. Regioselective synthesis of 1-allyl- and 1-arylmethyl uracil and thymine derivatives. Tetrahedron 2005, 61, 4009–4014. [Google Scholar]
- Frieden, M.; Giraud, M.; Reese, C.B.; Song, Q. Synthesis of 1-[cis-3-(hydroxymethyl)-cyclobutyl]-uracil, -thymine and -cytosine. J. Chem. Soc., Perkin Trans. 1998, 1, 2827–2832. [Google Scholar]
- Kundu, N.G.; Das, P.; Balzarini, J.; De Clercq, E. Palladium-catalyzed synthesis of [E]-6-(2-acylvinyl)uracils and [E]-6-(2-acylvinyl)-1-[(2-hydroxyethoxy)methyl]uracils-their antiviral and cytotoxic activities. Bioorg. Med. Chem. 1997, 5, 2011–2018. [Google Scholar] [CrossRef]
- Dolakova, P.; Dracinsky, M.; Masojidkova, M.; Solinova, V.; Kasicka, V.; Holy, A. Acyclic nucleoside bisphosphonates: synthesis and properties of chiral 2-amino-4,6-bis[(phosphono-methoxy)alkoxy]pyrimidines. Eur. J. Med. Chem. 2009, 44, 2408–2424. [Google Scholar]
- Prachayasittikul, S.; Worachartcheewan, A.; Lawang, R.; Ruchirawat, S.; Prachayasittikul, V. Activities of thiotetrahydropyridines as antioxidant and antimicrobial agents. EXCLI J. 2009, 8, 107–114. [Google Scholar]
- Trager, W.; Jensen, J.B. Human malaria parasites in continuous culture. Science 1976, 193, 673–675. [Google Scholar]
- Tengchaisri, T.; Chawengkirttikul, R.; Rachaphaew, N.; Reutrakul, V.; Sangsuwan, R.; Sirisinha, S. Antitumor activity of triptolide against cholangiocarcinoma growth in vitro and in hamsters. Cancer Lett. 1998, 133, 169–175. [Google Scholar] [CrossRef]
- Weaver, D.F.; Guillain, B.M.; Carran, J.R.; Jones, K. Preparation of pyrimidine derivatives as anti-ictogenic and or/ anti-epileptogenic agents. WO Patent 2002083651, 2002. [Chem. Abstr. 2002, 137, 325-426]. [Google Scholar]
- Lambros, C.; Vanderberg, J.P. Synchronization of Plasmodium falciparum erythrocytic stages in culture. J. Parasitol. 1979, 65, 418–420. [Google Scholar] [CrossRef]
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Prachayasittikul, S.; Sornsongkhram, N.; Pingaew, R.; Worachartcheewan, A.; Ruchirawat, S.; Prachayasittikul, V. Synthesis of N-Substituted 5-Iodouracils as Antimicrobial and Anticancer Agents. Molecules 2009, 14, 2768-2779. https://doi.org/10.3390/molecules14082768
Prachayasittikul S, Sornsongkhram N, Pingaew R, Worachartcheewan A, Ruchirawat S, Prachayasittikul V. Synthesis of N-Substituted 5-Iodouracils as Antimicrobial and Anticancer Agents. Molecules. 2009; 14(8):2768-2779. https://doi.org/10.3390/molecules14082768
Chicago/Turabian StylePrachayasittikul, Supaluk, Nirun Sornsongkhram, Ratchanok Pingaew, Apilak Worachartcheewan, Somsak Ruchirawat, and Virapong Prachayasittikul. 2009. "Synthesis of N-Substituted 5-Iodouracils as Antimicrobial and Anticancer Agents" Molecules 14, no. 8: 2768-2779. https://doi.org/10.3390/molecules14082768
APA StylePrachayasittikul, S., Sornsongkhram, N., Pingaew, R., Worachartcheewan, A., Ruchirawat, S., & Prachayasittikul, V. (2009). Synthesis of N-Substituted 5-Iodouracils as Antimicrobial and Anticancer Agents. Molecules, 14(8), 2768-2779. https://doi.org/10.3390/molecules14082768