NMR Spectra of Sparteine N1-oxide and α-Isosparteine N-oxide
Abstract
:Introduction
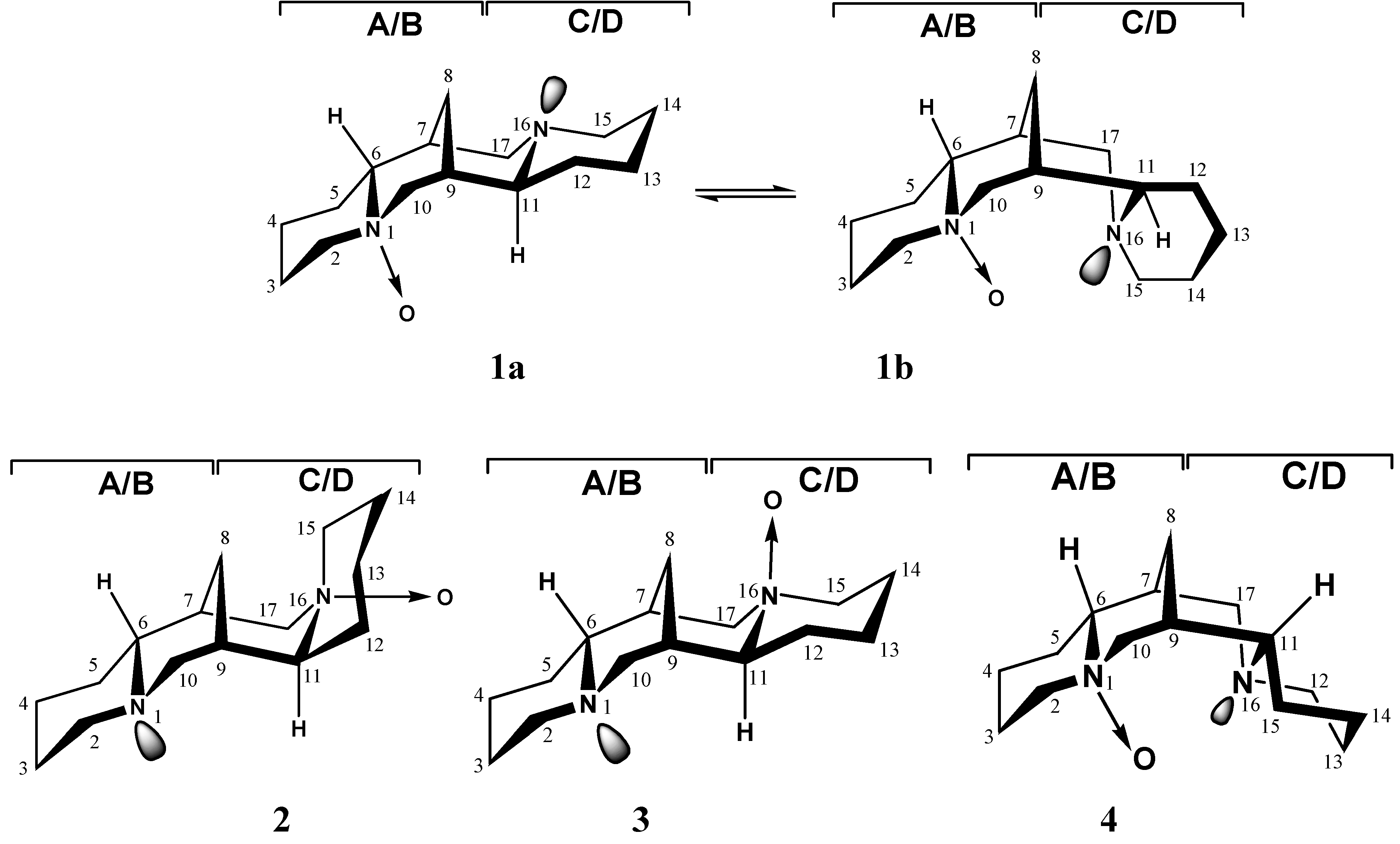
Results and Discussion
| C | Sparteine [see ref. 22] CDCl3 | Sparteine N1-oxide (1a) DMSO-d6 | N-oxide effects | Sparteine N1-oxide hydrochloride (1-HCl) CDCl3 | N-oxide and protonation effects | |||
|---|---|---|---|---|---|---|---|---|
| δC | δH | δC | δH | Δ δC | δC | δH | Δ δC | |
| 2 | 56.0 | 1.79 2.53 | 69.4 | 4.80 3.10 | +13.4 | 66.9 | 5.00 3.41 | +10.9 |
| 3 | 25.6 | 1.38 1.38 | 19.9 | 1.35 2.35 | –5.7 | 20.3 | 1.65 2.18 | –5.3 |
| 4 | 24.5 | 1.05 1.55 | 23.1 | 1.30 1.60 | –1.4 | 21.8 | 1.85 1.85 | –2.7 |
| 5 | 29.1 | 1.24 1.12 | 26.0 | 1.35 2.25 | –3.1 | 24.3 | 1.45 2.20 | –4.8 |
| 6 | 66.3 | 1.58 | 70.6 | 3.20 | +4.3 | 72.9 | 4.70 | +6.6 |
| 7 | 32.9 | 1.69 | 32.1 | 1.95 | –0.8 | 33.8 | 2.10 | +0.9 |
| 8 | 27.4 | 0.90 1.91 | 26.1 | 1.25 2.15 | –1.3 | 24.0 | 1.45 2.15 | –3.4 |
| 9 | 35.9 | 1.32 | 35.5 | 1.60 | –0.4 | 34.2 | 2.10 | –1.7 |
| 10 | 61.8 | 1.84 2.38 | 70.5 | 4.15 3.10 | +8.7 | 70.9 | 4.50 3.30 | +9.1 |
| 11 | 64.2 | 1.83 | 57.8 | 3.70 | –6.4 | 58.4 | 3.05 | –5.8 |
| 12 | 34.5 | 1.35 1.21 | 35.2 | 1.10 1.40 | +0.7 | 24.5 | 1.45 1.90 | –10.0 |
| 13 | 24.6 | 1.15 1.55 | 24.3 | 2.30 1.15 | –0.3 | 23.2 | 2.50 1.45 | –1.4 |
| 14 | 25.8 | 1.43 1.43 | 24.8 | 1.50 1.60 | –1.0 | 18.4 | 1.50 2.18 | –7.4 |
| 15 | 55.2 | 1.86 2.63 | 53.9 | 2.60 2.00 | –1.3 | 51.8 | 2.90 2.00 | –3.4 |
| 17 | 53.4 | 2.20 2.54 | 48.4 | 2.40 3.80 | –5.0 | 43.9 | 2.60 3.20 | –9.5 |
| C | α-isosparteine DMSO-d6 | α-isosparteine N-oxide (4) DMSO-d6 | N-oxide effects | ||
|---|---|---|---|---|---|
| δC | δH | δC | δH | Δ δC | |
| 2 | 55.5 | 1.76 2.74 | 66.5 | 3.42 3.50 | +11.0 |
| 3 | 23.8 | 1.75 1.40 | 19.9 | 1.60 2.02, | –3.9 |
| 4 | 22.9 | 1.24 1.70 | 21.8 | 1.44 1.76 | –1.1 |
| 5 | 28.0 | 1.69 1.19 | 23.8 | 1.70 1.40 | –4.2 |
| 6 | 65.7 | 1.86 | 72.9 | 3.74 | +7.2 |
| 7 | 32.1 | 1.45 | 32.4 | 2.07 | +0.3 |
| 8 | 32.8 | 1.31 1.52 | 31.7 | 1.80 2.00 | –1.1 |
| 9 | 32.1 | 1.31 | 33.0 | 2.18 | +0.9 |
| 10 | 55.7 | 2.04 2.96 | 65.5 | 3.74 3.74 | +9.8 |
| 11 | 65.7 | 1.86 | 60.6 | 2.68 | –5.1 |
| 12 | 28.0 | 1.69 1.19 | 27.4 | 1.60 1.74 | –0.6 |
| 13 | 22.9 | 1.24 1.70 | 22.6 | 1.44 1.76 | –0.3 |
| 14 | 23.8 | 1.75 1.40 | 23.3 | 2.16 1.40 | –0.5 |
| 15 | 55.5 | 1.76 2.74 | 52.4 | 2.48 3.08 | –3.1 |
| 17 | 55.7 | 2.04 2.96 | 50.7 | 2.58 3.00 | –5.0 |
| C | δ exper. (1a) | δ theor. (1a) | ΔδC (1a) | δ exper. (1-HCl) | δ theor. (1-HCl) | ΔδC (1-HCl) | δ exper. (2) | δ theor. (2) | ΔδC (2) |
|---|---|---|---|---|---|---|---|---|---|
| C2 | 69.4 | 72.2 | 2.8 | 66.9 | 66.3 | –0.6 | 52.4 | 48.9 | –3.8 |
| C3 | 19.9 | 21.3 | 1.4 | 20.3 | 17.9 | –2.4 | 23.3 | 23.4 | 0.1 |
| C4 | 23.1 | 23.0 | –0.1 | 21.8 | 21.4 | –0.4 | 22.6 | 23.4 | 0.8 |
| C5 | 26.0 | 27.1 | 1.1 | 24.3 | 26.2 | 1.9 | 27.4 | 28.9 | 1.5 |
| C6 | 70.6 | 70.3 | –0.3 | 72.9 | 70.9 | –2.0 | 60.6 | 57.5 | –3.1 |
| C7 | 32.1 | 33.3 | 1.2 | 33.8 | 31.2 | –2.6 | 33.0 | 34.0 | 1.0 |
| C8 | 26.1 | 23.4 | –2.7 | 24.0 | 22.3 | –1.7 | 31.7 | 33.3 | 1.6 |
| C9 | 35.5 | 35.0 | –0.5 | 34.2 | 32.5 | –1.7 | 32.4 | 33.4 | 1.0 |
| C10 | 70.5 | 71.8 | 1.3 | 70.9 | 67.3 | –3.6 | 50.7 | 45.5 | –5.2 |
| C11 | 57.8 | 60.8 | 3.0 | 58.4 | 62.9 | 4.5 | 72.9 | 71.8 | –1.1 |
| C12 | 35.2 | 34.4 | –0.8 | 24.5 | 25.4 | 0.9 | 23.8 | 23.5 | –0.3 |
| C13 | 24.3 | 26.6 | 2.3 | 23.2 | 22.9 | –0.3 | 21.8 | 23.5 | 1.7 |
| C14 | 24.8 | 21.5 | –3.3 | 18.4 | 17.6 | –0.8 | 19.9 | 19.6 | –0.3 |
| C15 | 53.9 | 53.2 | –0.7 | 51.8 | 53.1 | 1.3 | 66.5 | 69.4 | 2.9 |
| C17 | 48.4 | 52.2 | 3.8 | 43.9 | 45.9 | 2.0 | 65.5 | 65.9 | 0.4 |
Conclusions
Experimental
General
Syntheses
DFT calculations
References
- Wiewiórowski, M.; Edwards, O.E.; Bratek-Wiewiórowska, M.D. Conformation of the C15 lupine alkaloids. Can. J. Chem. 1967, 45, 1447–1457. [Google Scholar] [CrossRef]
- Wysocka, W.; Brukwicki, T. Conformational equilibria in quinolizidine alkaloids. J. Mol. Struct. 1996, 385, 23–33. [Google Scholar] [CrossRef]
- Galasso, V.; Asaro, F.; Berti, F.; Kovač, B.; Habuš, I.; Sacchetti, A. On the structure and spectroscopic properties of sparteine and its diastereoisomers. Chem. Phys. 2003, 294, 155–169. [Google Scholar]
- Haasnoot, C.A.G. Conformational analysis of six-membered rings in solution: Ring puckering coordinates derived from vicinal NMR proton-proton coupling constants. J. Am. Chem. Soc. 1993, 115, 1460–1468. [Google Scholar] [CrossRef]
- Boschmann, E.; Weinstock, L.M.; Carmack, M. Metal Complexes of the Three Sparteine Diastereoisomers. Properties and Reactivities of the Copper(II) Derivatives. Inorg. Chem. 1974, 13, 1297–1300. [Google Scholar] [CrossRef]
- Jasiewicz, B.; Boczoń, W.; Warżajtis, B.; Rychlewska, U.; Rafałowicz, T. Synthesis, spactral and structural characterization of zinc(II) methacrylate complexes with sparteine and α-isosparteine; The role of hydrogen bonds and dipolar interactions in stabilizing the molecular structure. J. Mol. Struct. 2005, 753, 45–52. [Google Scholar] [CrossRef]
- Jasiewicz, B.; Boczoń, W.; Kowalczyk, A. Synthesis and spectral characterization of sparteine and α-isosparteine complexes with copper(II) sulfate. J. Coord. Chem. 2007, 60, 2441–2448. [Google Scholar] [CrossRef]
- Przybylska, M.; Barnes, W.H. The crystal and molecular structure of α-isosparteine monohydrate. Acta Cryst. 1953, 6, 377–384. [Google Scholar] [CrossRef]
- Jasiewicz, B.; Boczoń, W.; Mumot, A.; Warżajtis, B.; Rychlewska, U. Synthesis, spectroscopy and crystal structure of α-isosparteine complexes with ZnX2 (X = Br, CN, Cl). J. Mol. Struct. 2005, 737, 239–244. [Google Scholar] [CrossRef]
- Jasiewicz, B.; Boczoń, W.; Borowiak, T.; Wolska, I. Synthesis and structural characterization of zinc(II) acetate complexes with α-isosparteine. J. Mol. Struct. 2007. [Google Scholar] [CrossRef]
- Jasiewicz, B. NMR and DFT studies of bis-quinolizidine complexes with LiClO4 and LiCl. J. Mol. Struct. 2007. [Google Scholar] [CrossRef]
- Jasiewicz, B. N16-oxides of sparteine, 2-methylsparteine and 2-phenylsparteine as ligands; spectroscopic and DFT studies of complexes with ZnX2 (X=Cl, Br). J. Coord. Chem. Accepted manuscript..
- Boczoń, Wł.; Kozioł, B. Synthesis and Structure of 17β-Isopropyllupanine N-Oxide and Its Perchlorate Salt. Pol. J. Chem. 2000, 74, 1249–1258. [Google Scholar]
- Boczoń, Wł.; Jasiewicz, B. Synthesis and conformational analysis of di-substituted sparteine derivatives. Collect. Czech. Chem. Commun. 2003, 68, 696–710. [Google Scholar] [CrossRef]
- Jasiewicz, B.; Boczoń, Wł.; Warżajtis, B.; Rychlewska, U. rac-2-Cyano-2-(p-tolyl)sparteine and its perchlorate salt. J. Mol. Struct. 2004, 688, 111–119. [Google Scholar] [CrossRef]
- Jasiewicz, B.; Boczoń, Wł.; Kurek, J. Synthesis and conformational analysis of new 17-alkyl derivatives of lupanine and their perchlorate salts. Coll. Czech. Chem. Commun. 2004, 69, 2068–2080. [Google Scholar] [CrossRef]
- Jasiewicz, B.; Boczoń, Wł. Oxidation Reaction of bis-quinolizidine System: Synthesis of new 2-Methyl-17-oxosparteine and 2-Methyl-α-isosparteine. Lett. Org. Chem. 2007, 4, 27–31. [Google Scholar] [CrossRef]
- Brukwicki, T.; Wysocka, W. NMR spectra and geometry of epi N-oxides of sparteine and some of its derivatives in solution. J. Mol. Struct. 2003, 647, 275–286. [Google Scholar] [CrossRef]
- Wiewiórowski, M.; Baranowski, P. Amine Oxides of Lupin Alkaloids. I. The Structure of the N-oxides of N1 and N16-Sparteine, α-Isosparteine and Lupanine. Bull. Acad. Pol.: Chim. 1962, 10, 537–542. [Google Scholar]
- Bratek-Wiewiórowska, M.D.; Skolik, J.; Łangowska, K.; Wiewiórowski, M. Further Studies on the Chemistry and Structure of N-oxides of Sparteine and its derivatives. IV. Synthesis and Structure of a New Sparteine-N(16)-oxide. Conformation of Sparteine-N(1)-oxide. Bull. Acad. Pol.: Chim. 1974, 12, 1025–1036. [Google Scholar]
- Thiel, J.; Boczoń, Wł.; Fiedorow, P.; Jasiewicz, B.; Knychała, M. Conformation of some sparteine N-16 oxides revisited. J. Mol. Struct. 2002, 642, 15–23. [Google Scholar] [CrossRef]
- Jasiewicz, B.; Boczoń, Wł. A comparative study of sparteine, α-isosparteine and 2-methylsparteine monoperchlorate salts and zinc(II) complexes by NMR chemical shifts. J. Mol. Struct. 2005, 752, 115–123. [Google Scholar]
- Boczoń, Wł.; Skolik, J.; Kozioł, B. Further studies on the stereochemistry of sparteine, its isomers and derivatives: Part XXII. 13C NMR analysis of sparteine derivatives substituted in external rings – monosalts. J. Mol. Struct. 1994, 328, 1–10. [Google Scholar] [CrossRef]
- Sample Availability: Contact the author.
© 2008 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Jasiewicz, B. NMR Spectra of Sparteine N1-oxide and α-Isosparteine N-oxide. Molecules 2008, 13, 3-10. https://doi.org/10.3390/molecules13010003
Jasiewicz B. NMR Spectra of Sparteine N1-oxide and α-Isosparteine N-oxide. Molecules. 2008; 13(1):3-10. https://doi.org/10.3390/molecules13010003
Chicago/Turabian StyleJasiewicz, Beata. 2008. "NMR Spectra of Sparteine N1-oxide and α-Isosparteine N-oxide" Molecules 13, no. 1: 3-10. https://doi.org/10.3390/molecules13010003
APA StyleJasiewicz, B. (2008). NMR Spectra of Sparteine N1-oxide and α-Isosparteine N-oxide. Molecules, 13(1), 3-10. https://doi.org/10.3390/molecules13010003




