Tipping the Balance: A Criticality Perspective
Abstract
1. Introduction
2. Model of Tumour Heterogeneity
2.1. Similarity with Population Genetics Model
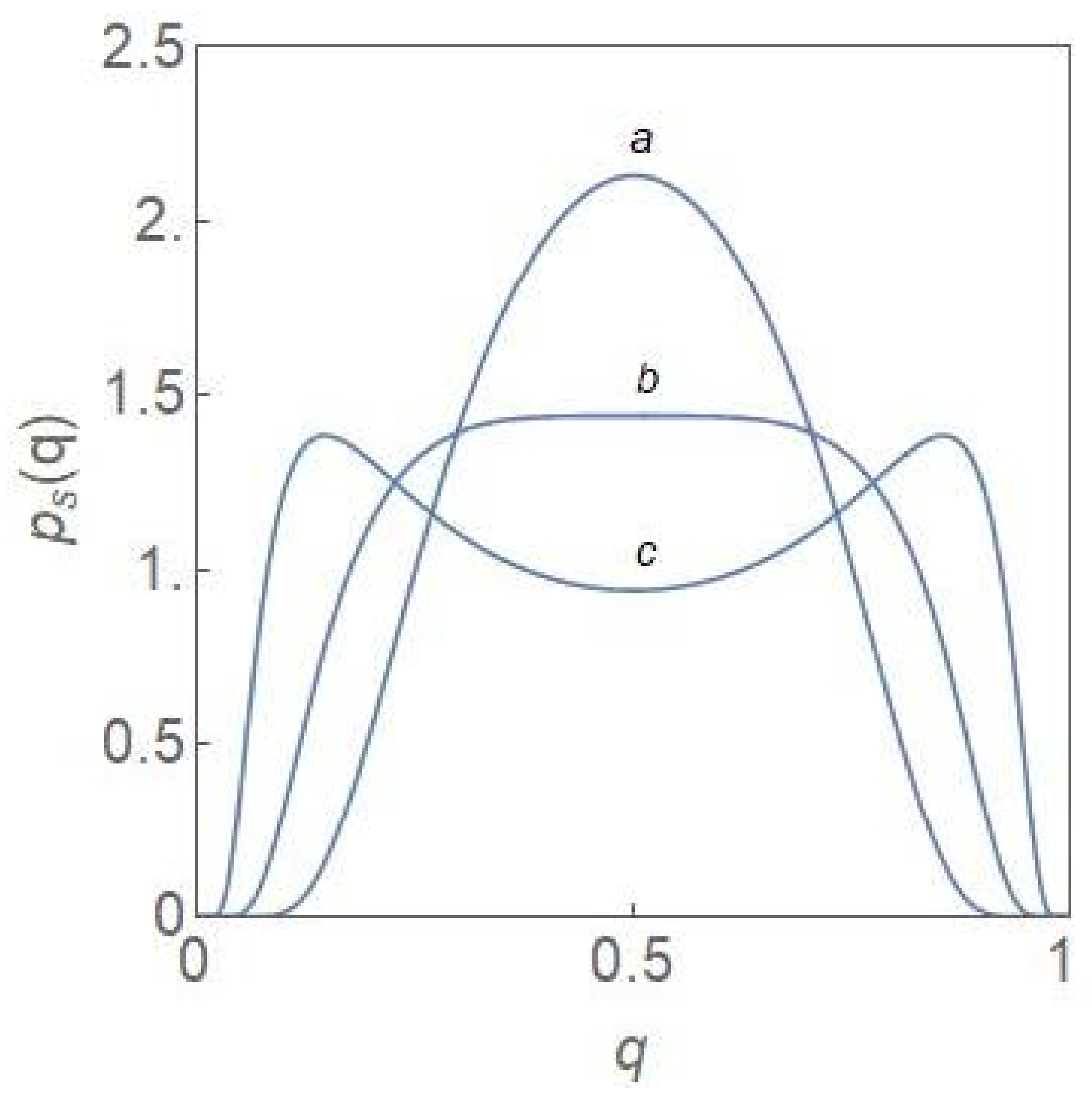
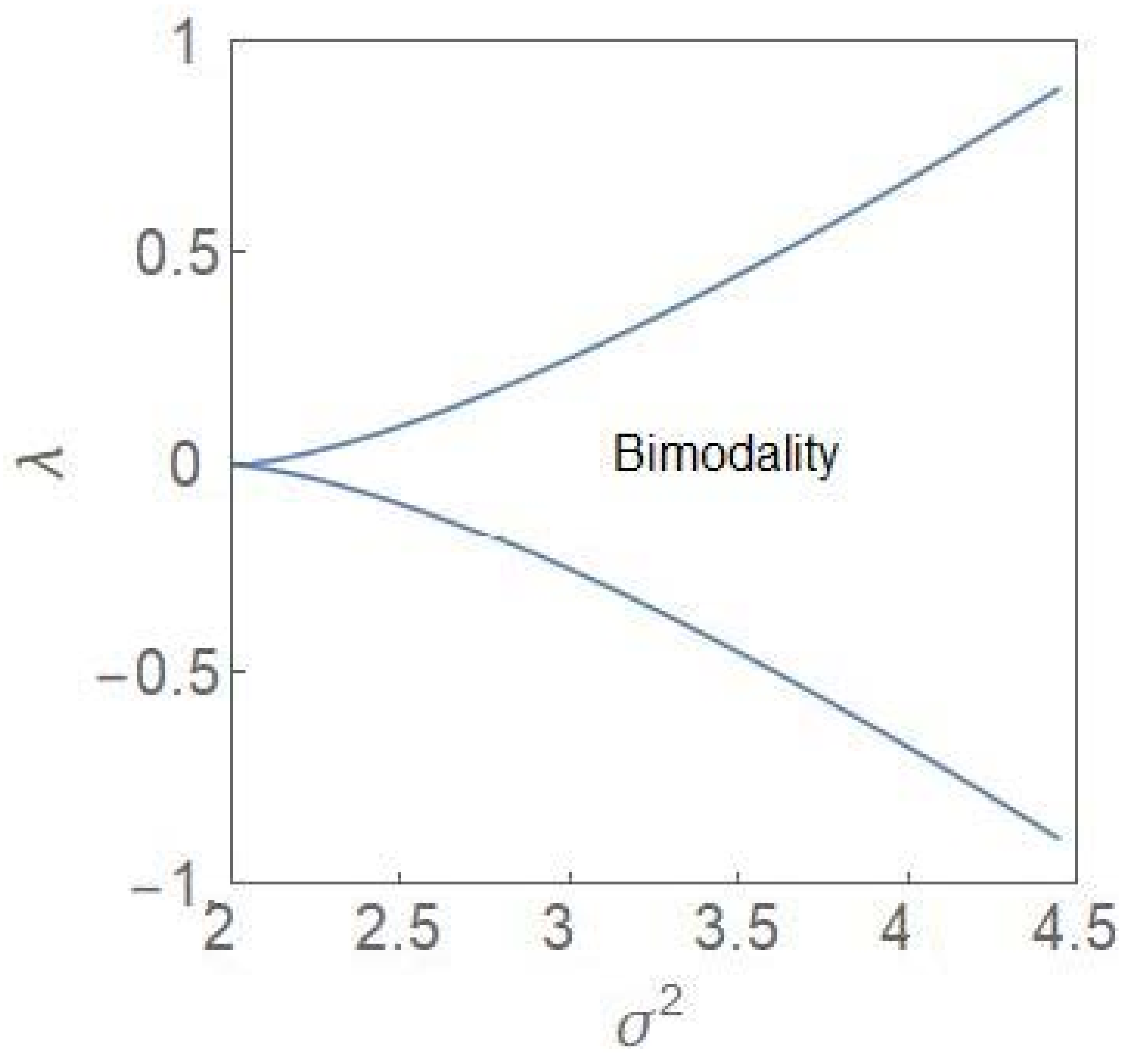
2.2. Critical-Point Transition to Bimodality
2.3. Critical Exponents
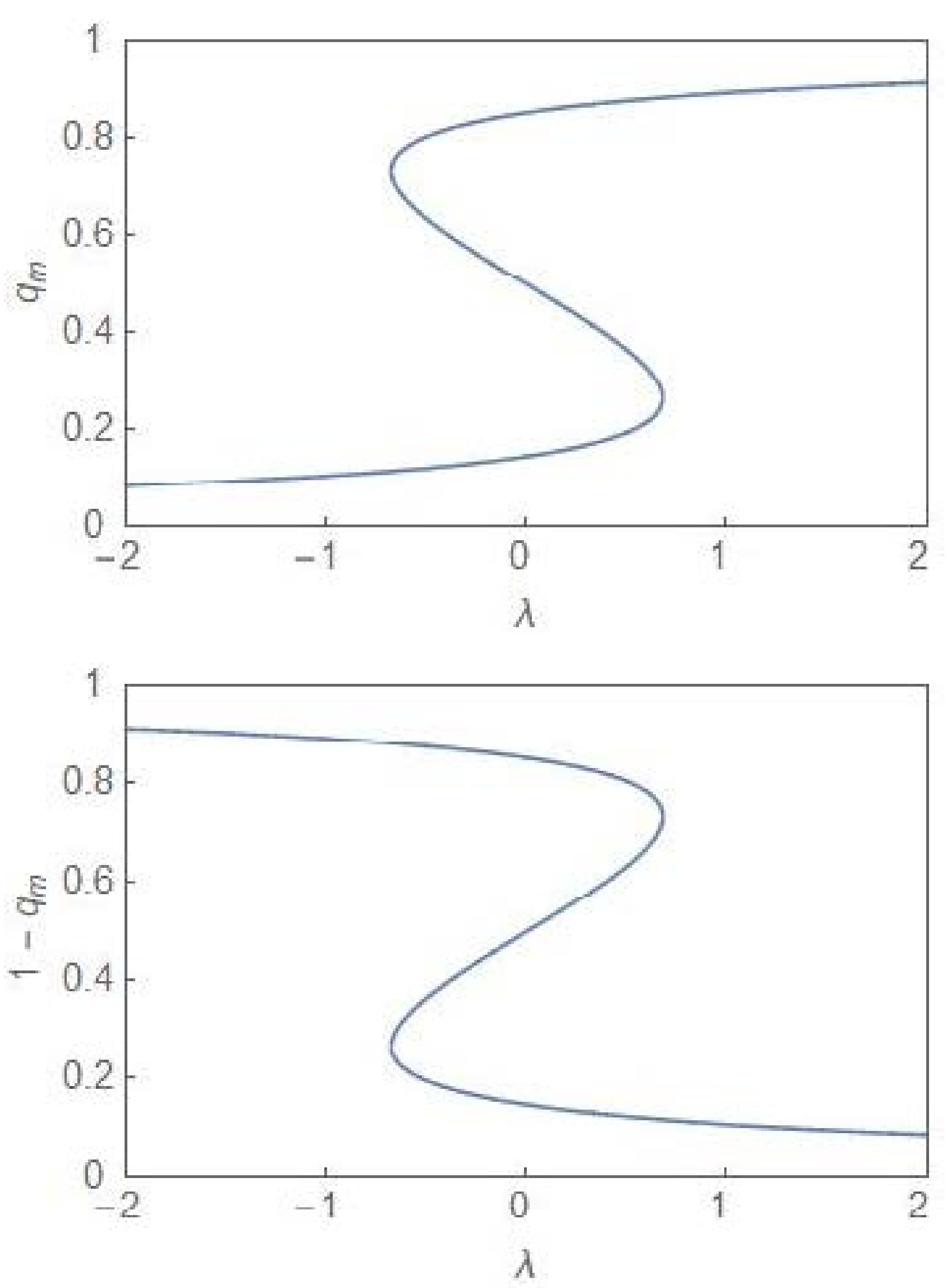
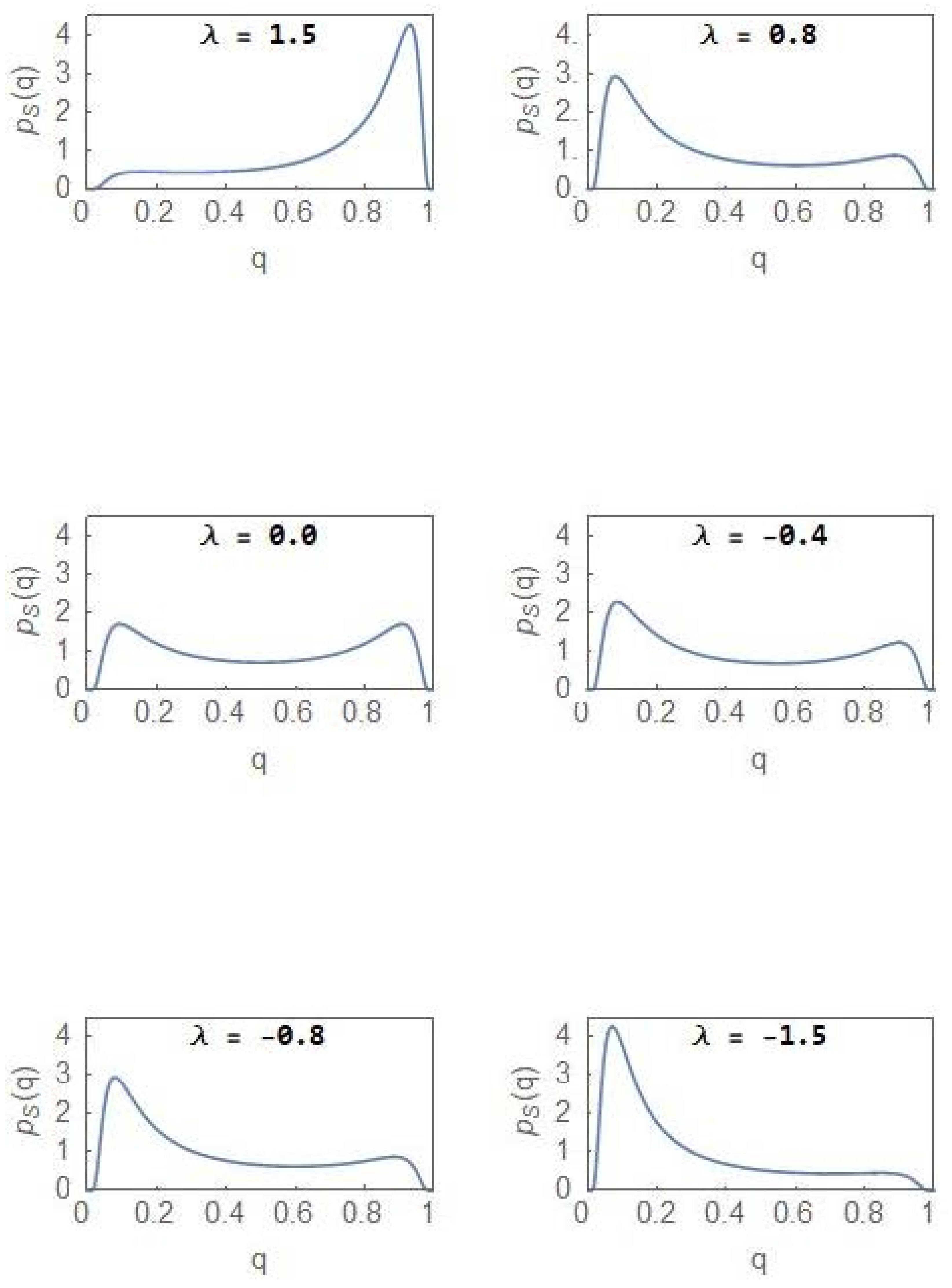
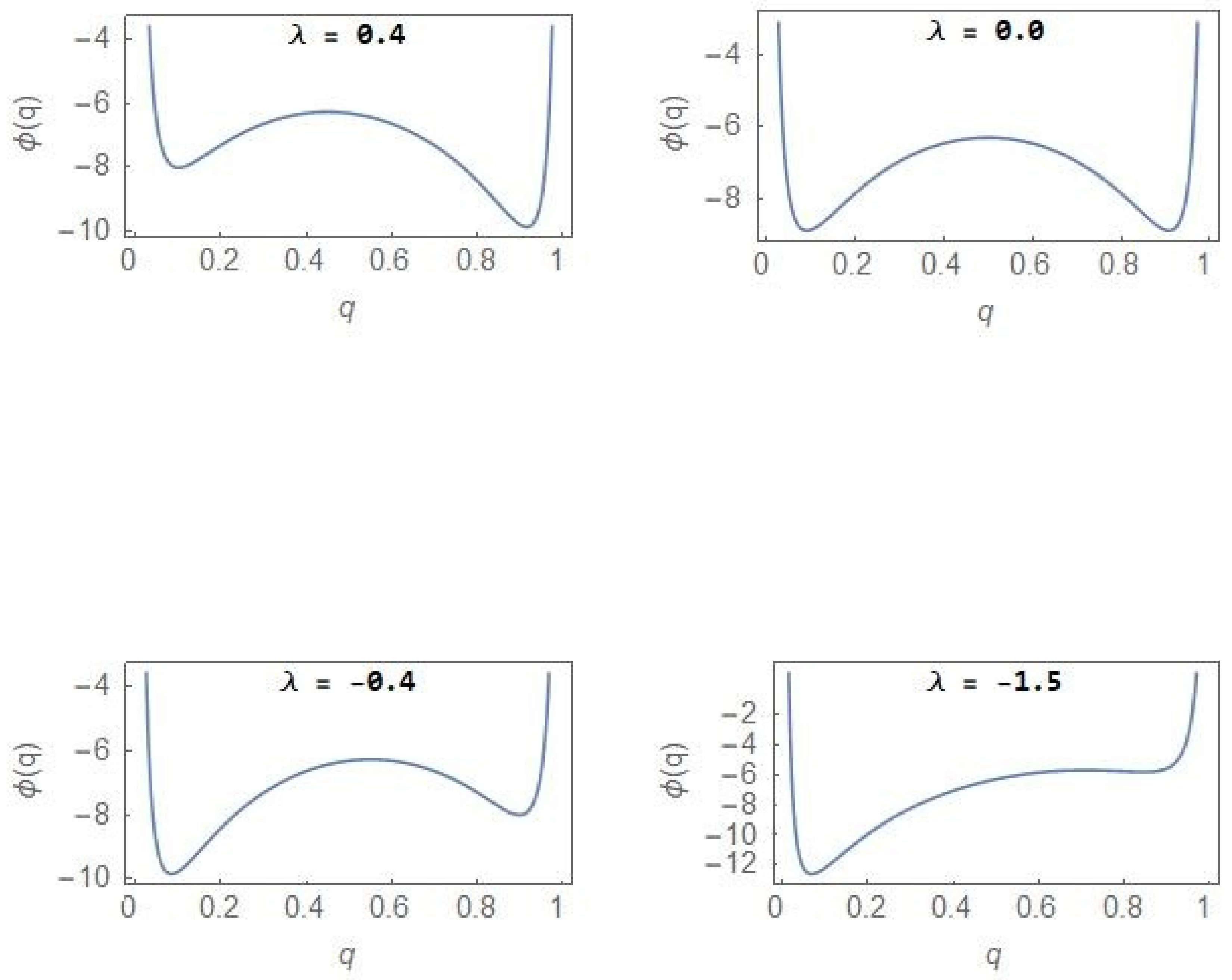
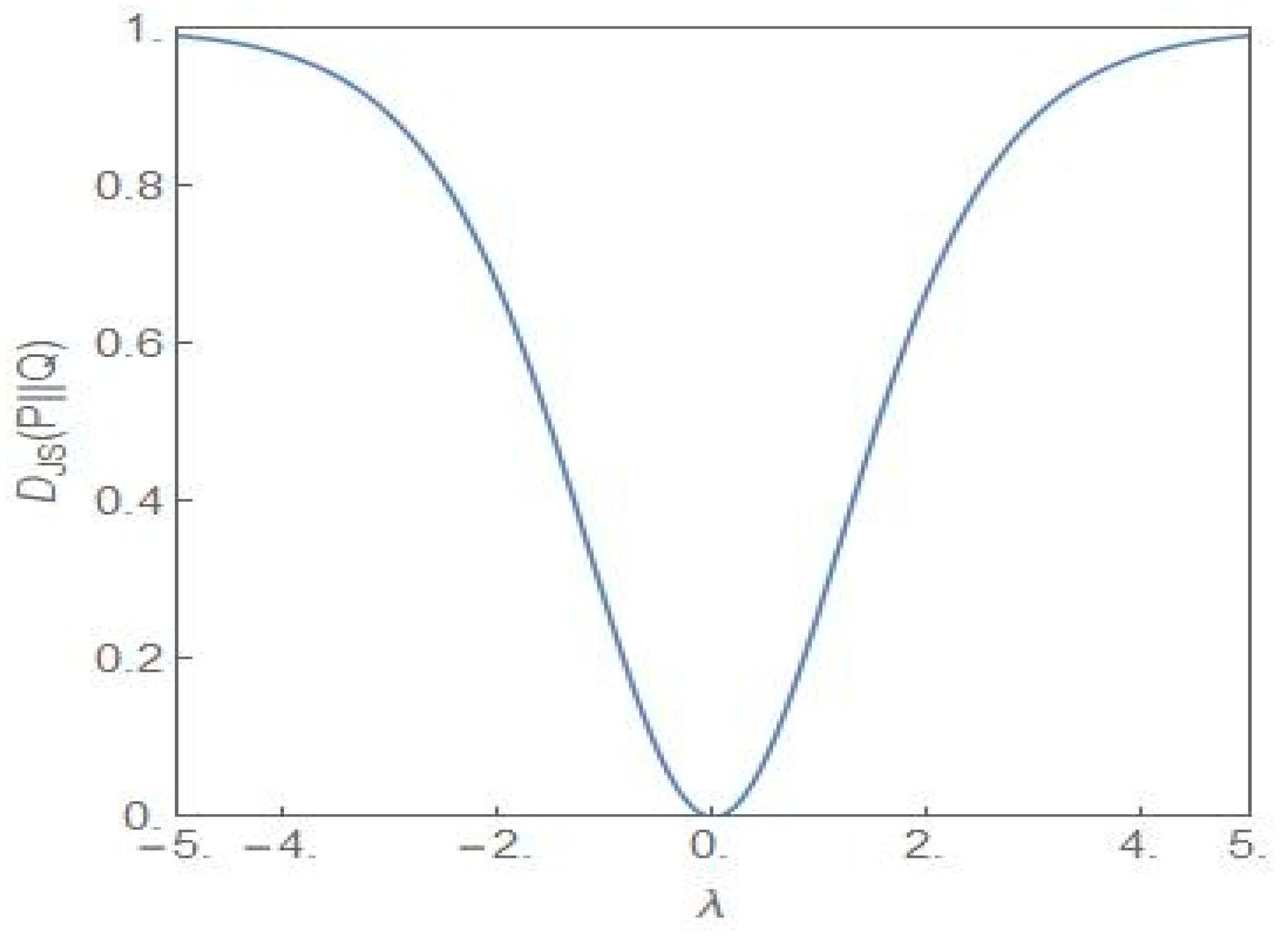
3. Nonequilibrium Model with λ ≠ 0
Cancer as a Phase Transition
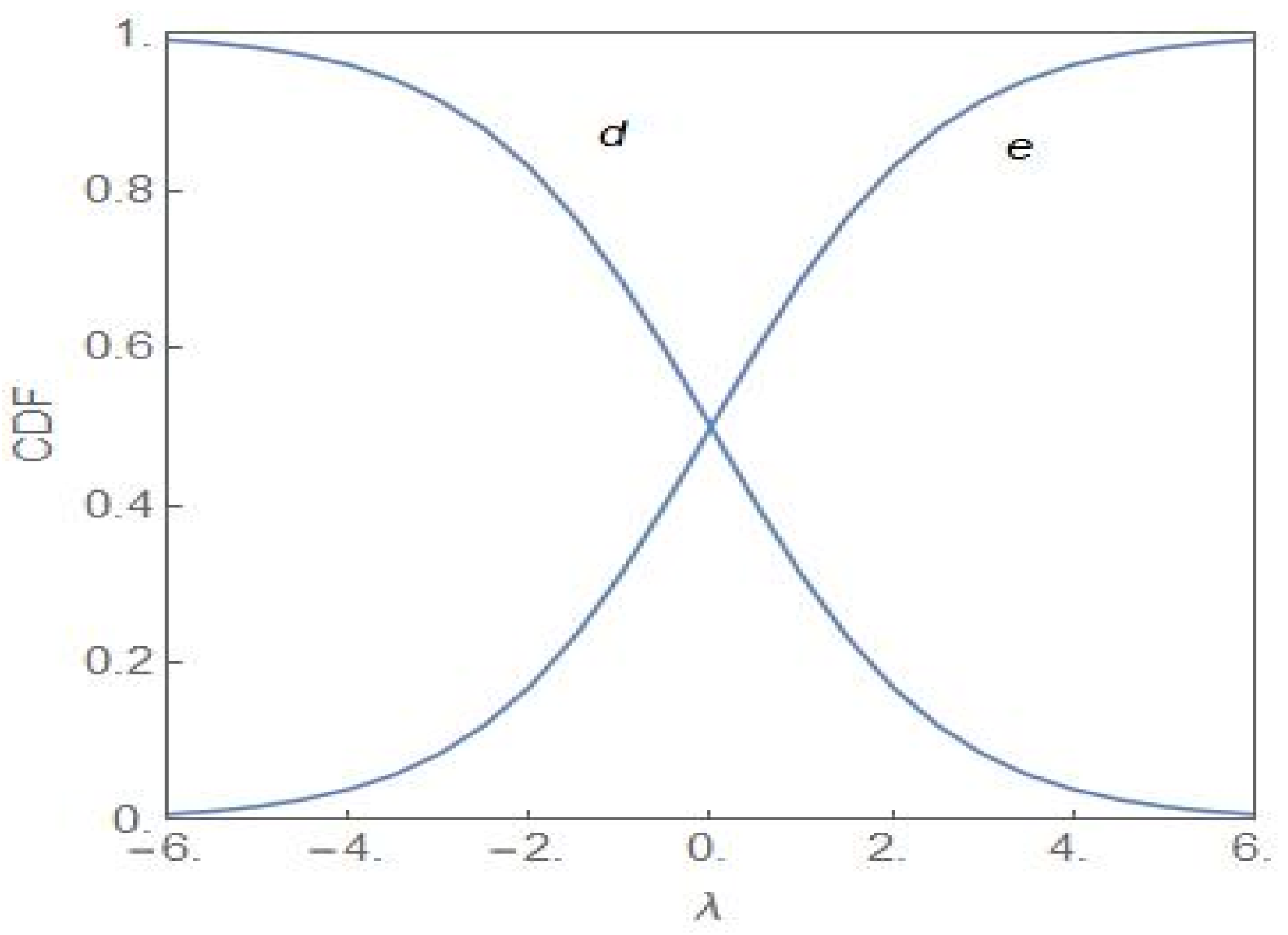
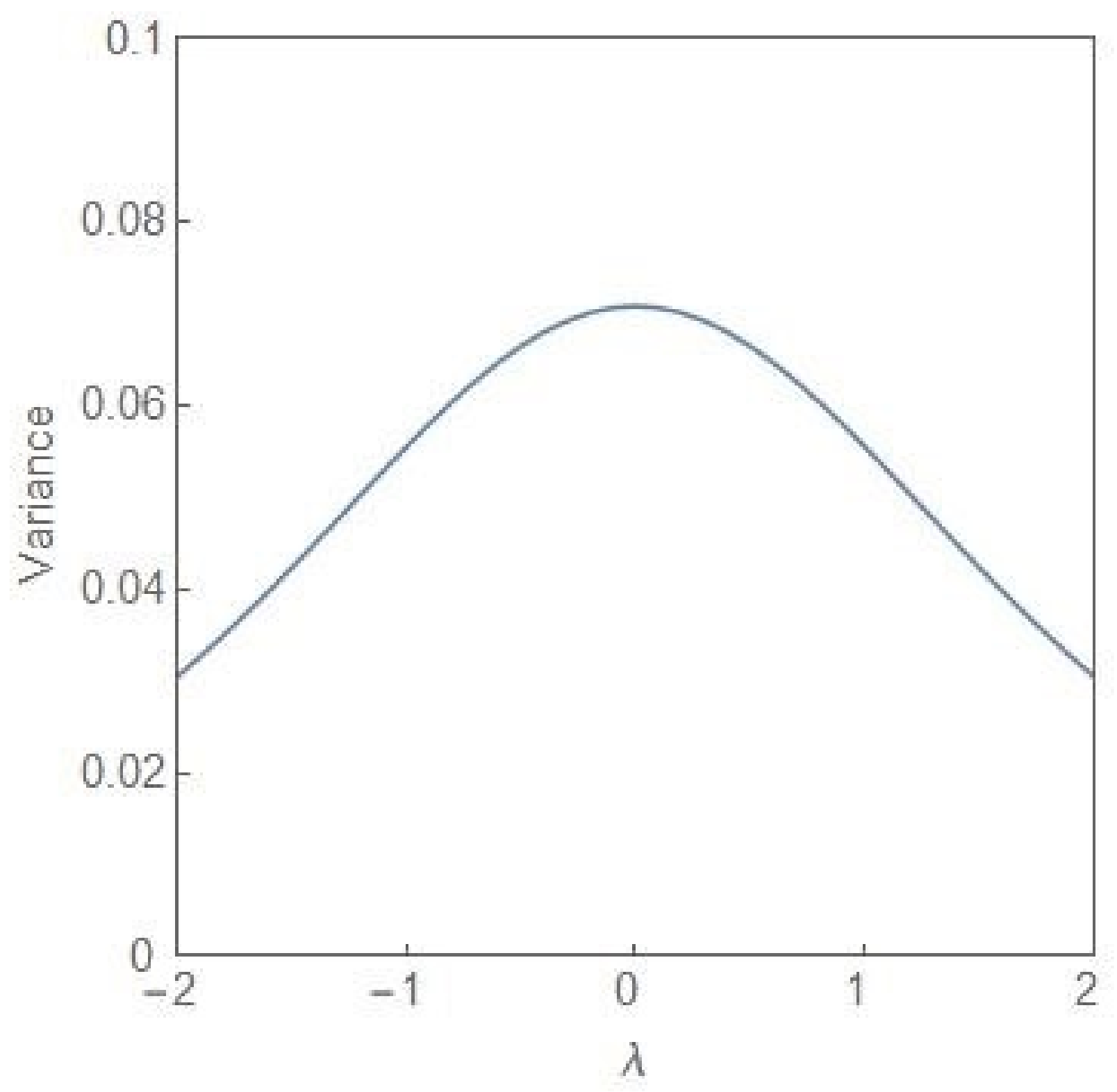
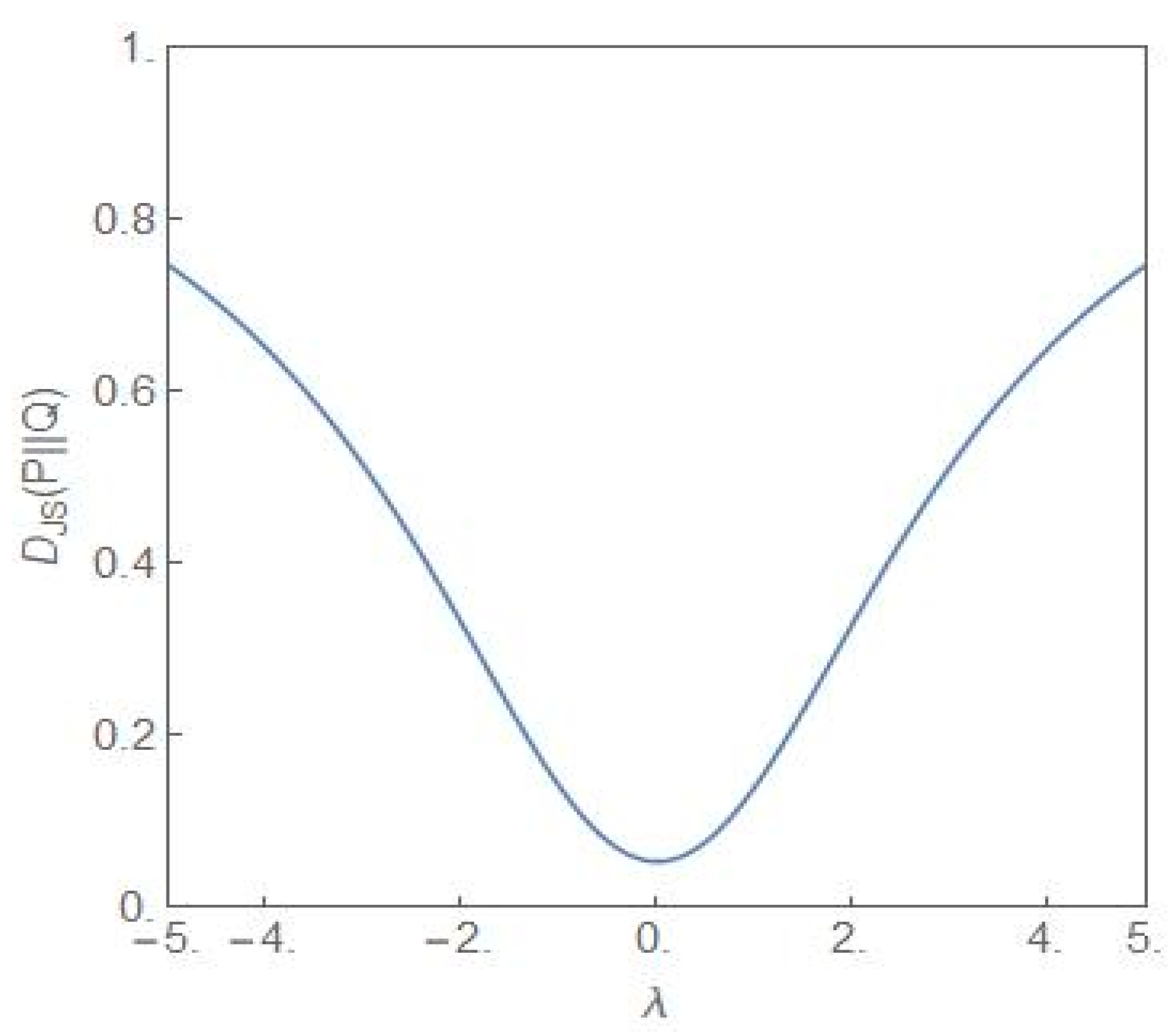
4. Quantitative Signatures of the Onset of Dominance
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
References
- Kærn, M.; Elston, T.C.; Blake, W.J.; Collins, J.J. Stochasticity in gene expression: From theories to phenotypes. Nat. Rev. Genet. 2005, 6, 451–464. [Google Scholar] [CrossRef] [PubMed]
- Raj, A.; van Oudenaarden, A. Nature, nurture or chance: Stochastic gene expression and its consequences. Cell 2018, 135, 216–226. [Google Scholar] [CrossRef] [PubMed]
- Balázsi, G.; van Oudenaarden, A.; Collins, J.J. Cellular decision making and biological noise: From microbes to mammals. Cell 2011, 144, 910–925. [Google Scholar] [CrossRef] [PubMed]
- Veening, J.-W.; Smits, W.K.; Kuipers, O.P. Bistability, epigenetics and bet-hedging in bacteria. Annu. Rev. Microbiol. 2008, 62, 193–210. [Google Scholar] [CrossRef] [PubMed]
- To, T.L.; Maheshri, N. Noise can induce bimodality in positive transcriptional feedback loops without bistability. Science 2010, 327, 1142–1145. [Google Scholar] [CrossRef] [PubMed]
- Arnold, L.; Horsthemke, W.; Lefever, R. White and coloured external noise and transition phenomena in nonlinear systems. Z. Physik B 1978, 29, 367–373. [Google Scholar] [CrossRef]
- Horsthemke, W.; Lefever, R. Noise-Induced Transitions, 1st ed.; Springer: Berlin/Heidelberg, Germany, 1984; pp. 108–200. [Google Scholar]
- Erez, A.; Byrd, T.A.; Vogel, R.M.; Altan-Bonnet, G.; Mugler, A. Universality of biochemical feedback and its application to immune cells. Phys. Rev. E 2019, 99, 022422. [Google Scholar] [CrossRef] [PubMed]
- Sornette, D. Critical Phenomena in Natural Sciences, 2nd ed.; Springer: Berlin/Heidelberg, Germany, 2006; pp. 241–265. [Google Scholar]
- Bose, I.; Ghosh, S. Bifurcation and criticality. J. Stat. Mech. Theory Exp. 2019, 4, 3403. [Google Scholar] [CrossRef]
- Muñoz, M.A. Criticality and dynamical scaling in living systems. Rev. Mod. Phys. 2018, 90, 031001. [Google Scholar] [CrossRef]
- Marusyk, A.; Almendro, V.; Polyak, K. Intra-tumour heterogeneity: A looking glass for cancer? Nat. Rev. Cancer 2012, 12, 323–334. [Google Scholar] [CrossRef]
- Marusyk, A.; Janiszewska, M.; Polyak, K. Intratumour heterogeneity: The Rosetta Stone of therapy resistance. Cancer Cell 2020, 37, 471–484. [Google Scholar] [CrossRef] [PubMed]
- Jordan, N.V.; Bardia, A.; Wittner, B.S.; Benes, C.; Ligorio, M.; Zheng, Y.; Yu, M.; Sundaresan, T.K.; Licausi, J.A.; Desai, R.; et al. HER2 expression identifies dynamical functional states within circulating breast cancer cells. Nature 2016, 537, 102–106. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Thirumalai, D. A mathematical model for phenotypic heterogeneity in breast cancer with implications for therapeutic strategies. J. R. Soc. Interface 2022, 18, 20210803. [Google Scholar] [CrossRef]
- Liu, R.; Wang, X.; Aihara, K.; Chen, L. Early diagnosis of complex diseases by molecular biomarkers, network biomarkers, and dynamical network biomarkers. Med. Res. Rev. 2014, 34, 455–478. [Google Scholar] [CrossRef] [PubMed]
- Teschendorff, A.E.; Feinberg, A.P. Statistical mechanics meets single-cell biology. Nat. Rev. Genet. 2021, 22, 459–476. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Liu, R.; Liu, Z.P.; Li, M.; Aihara, K. Detecting early-warning signals for sudden deterioration of complex diseases by dynamical network biomarkers. Sci. Rep. 2012, 2, 342–349. [Google Scholar] [CrossRef]
- Zhong, J.; Liu, R.; Chen, P. Identifying critical state of complex diseases by single-sample Kullback-Leibler divergence. BMC Genom. 2020, 21, 87. [Google Scholar] [CrossRef]
- Scheffer, M.; Bascompte, J.; Brock, W.A.; Brovkin, V.; Carpenter, S.R.; Dakos, V.; Held, H.; van Nes, E.H.; Rietkerk, M.; Sugihara, G. Early-warning signals for critical transitions. Nature 2009, 461, 53–59. [Google Scholar] [CrossRef]
- Thattai, M.; van Oudenaarden, A. Stochastic gene expression in fluctuating environments. Genetics 2004, 167, 523–530. [Google Scholar] [CrossRef] [PubMed]
- Nevozhay, D.; Adams, R.M.; Van Itallie, E.; Bennett, M.R.; Balázsi, G. Mapping the environmental fitness landscape of a synthetic gene circuit. PLoS Comput. Biol. 2012, 8, e1002480. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.X.; Pisco, A.O.; Qian, H.; Huang, S. Nonequilibrium population dynamics of phenotype conversion of cancer cells. PLoS ONE 2014, 9, e110714. [Google Scholar] [CrossRef] [PubMed]
- Goldenfeld, N. Lectures on Phase Transitions and the Renormalizationgroup; Frontiers in Physics 85; CRC Press: New York, NY, USA, 2018; pp. 23–163. [Google Scholar]
- Strogatz, S.H. Nonlinear Dynamics and Chaos, 1st ed.; Addision-Wesley: New York, NY, USA, 1994; pp. 15–92. [Google Scholar]
- Davies, P.C.W.; Demetrius, L.; Tuszynski, T.A. Cancer as a dynamical phase transition. Theor. Biol. Med. Model. 2011, 8, 1–16. [Google Scholar] [CrossRef]
- Tsuchiya, M.; Giuliani, A.; Hashimoto, M.; Erenpreisa, J.; Yoshikawa, K. Emergent self-organised criticality in gene expression dynamics: Temporal development of global phase transition revealed in a cancer cell line. PLoS ONE 2015, 10, e0128565. [Google Scholar] [CrossRef]
- Tsuchiya, M.; Hashimoto, M.; Takenaka, Y.; Motoike, I.N.; Yoshikawa, K. Global genetic response in a cancer cell: Self-organised coherent expression dynamics. PLoS ONE 2014, 9, e97411. [Google Scholar] [CrossRef] [PubMed]
- Pal, M.; Ghosh, S.; Bose, I. Non-genetic heterogeneity, criticality and cell differentiation. Phys. Biol. 2015, 12, 016001. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mojtahedi, M.; Skupin, A.; Zhou, J.; Castaño, I.G.; Leong-Quong, R.Y.; Chang, H.; Trachana, K.; Giuliani, A.; Huang, S. Cell fate decision as high-dimensional critical state transition. PLoS Biol. 2016, 14, e2000640. [Google Scholar] [CrossRef] [PubMed]
- Cover, T.M.; Thomas, J.A. Elements of Information Theory, 2nd ed.; Wiley-Interscience: Hoboken, NJ, USA, 2006; pp. 13–55. [Google Scholar]
- Karolak, A.; Branciamore, S.; McCune, J.S.; Lee, P.P.; Rodin, S.S.; Rockne, R.C. Concepts and applications of information theory to immuno-oncology. Trends Cancer 2021, 7, 335–346. [Google Scholar] [CrossRef] [PubMed]
- Lin, J. Divergence measures based on the Shannon entropy. IEEE Trans. Inf. Theory 1991, 37, 145–151. [Google Scholar] [CrossRef]
- Jinling, Y.; Peiluan, L.; Gao, R.; Ying, L.; Luonan, C. Identifying critical states of complex diseases by single-sample Jensen-Shannon divergence. Front. Oncol. 2021, 11, 684781. [Google Scholar]
- Klein, I.; Doll, M. (Generalized) maximum cumulative direct, residual, and paired φ entropy approach. Entropy 2020, 22, 91. [Google Scholar] [CrossRef]
- Kauffman, S. Homeostasis and differentiation in random genetic control networks. Nature 1969, 22, 177–178. [Google Scholar] [CrossRef]
- Huang, S.; Eichler, G.; Bar-Yam, Y.; Ingber, D.E. Cell fates as high-dimensional attractor states of a complex gene regulatory network. Phys. Rev. Lett. 2005, 94, 128701. [Google Scholar] [CrossRef]
- Li, Q.; Wennborg, A.; Aurell, E.; Dekel, E.; Zou, J.Z.; Xu, Y.; Huang, S.; Ernberg, I. Dynamics inside the cancer cell attractor reveal cell heterogeneity, limits of stability, and escape. Proc. Natl. Acad. Sci. USA 2016, 113, 2672–2677. [Google Scholar] [CrossRef]
- Kauffman, S. Differentiation of malignant to benign cells. J. Theor. Biol. 1971, 3, 429–451. [Google Scholar] [CrossRef]
- Rockne, R.C.; Branciamore, S.; Qi, J.; Frankhouser, D.E.; O’Meally, D.; Hua, W.-K.; Cook, G.; Carnahan, E.; Zhang, L.; Marom, A.; et al. State-transition analysis of time-sequential gene expression identifies critical points that predict development of acute myeloid leukemia. Cancer Res. 2020, 80, 3157–3169. [Google Scholar] [CrossRef]
- Liu, R.; Yu, X.; Liu, X.; Xu, D.; Aihara, K.; Chen, L. Identifying critical transitions of complex diseases based on a single sample. Bioinformatics 2014, 30, 1579–1586. [Google Scholar] [CrossRef]
- Huang, S.; Kauffman, S. How to escape the cancer attractor: Rationale and limitations of multi-target drugs. Semin. Cancer Biol. 2013, 23, 270–278. [Google Scholar] [CrossRef]
- Dar, R.D.; Hosmane, N.N.; Arkin, M.R.; Siliciano, R.F.; Weinberger, L.S. Screening for noise in gene expression identifies drug synergies. Science 2014, 344, 1392–1396. [Google Scholar] [CrossRef]
- Desai, R.V.; Chen, X.; Martin, B.; Chaturvedi, S.; Hwang, D.W.; Li, W.; Yu, C.; Ding, S.; Thomson, M.; Singer, R.H.; et al. A DNA repair pathway can regulate transcriptional noise to promote cell-fate transitions. Science 2021, 373, 870. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bose, I. Tipping the Balance: A Criticality Perspective. Entropy 2022, 24, 405. https://doi.org/10.3390/e24030405
Bose I. Tipping the Balance: A Criticality Perspective. Entropy. 2022; 24(3):405. https://doi.org/10.3390/e24030405
Chicago/Turabian StyleBose, Indrani. 2022. "Tipping the Balance: A Criticality Perspective" Entropy 24, no. 3: 405. https://doi.org/10.3390/e24030405
APA StyleBose, I. (2022). Tipping the Balance: A Criticality Perspective. Entropy, 24(3), 405. https://doi.org/10.3390/e24030405




