Invasive Plant Species Establishment and Range Dynamics in Sri Lanka under Climate Change
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Modeling Method
2.3. Species Occurrence Data
2.4. Environmental Predictors
2.5. MaxEnt Settings
2.6. Evaluating Model Performance
2.7. Development of Climatic Suitability Maps
3. Results
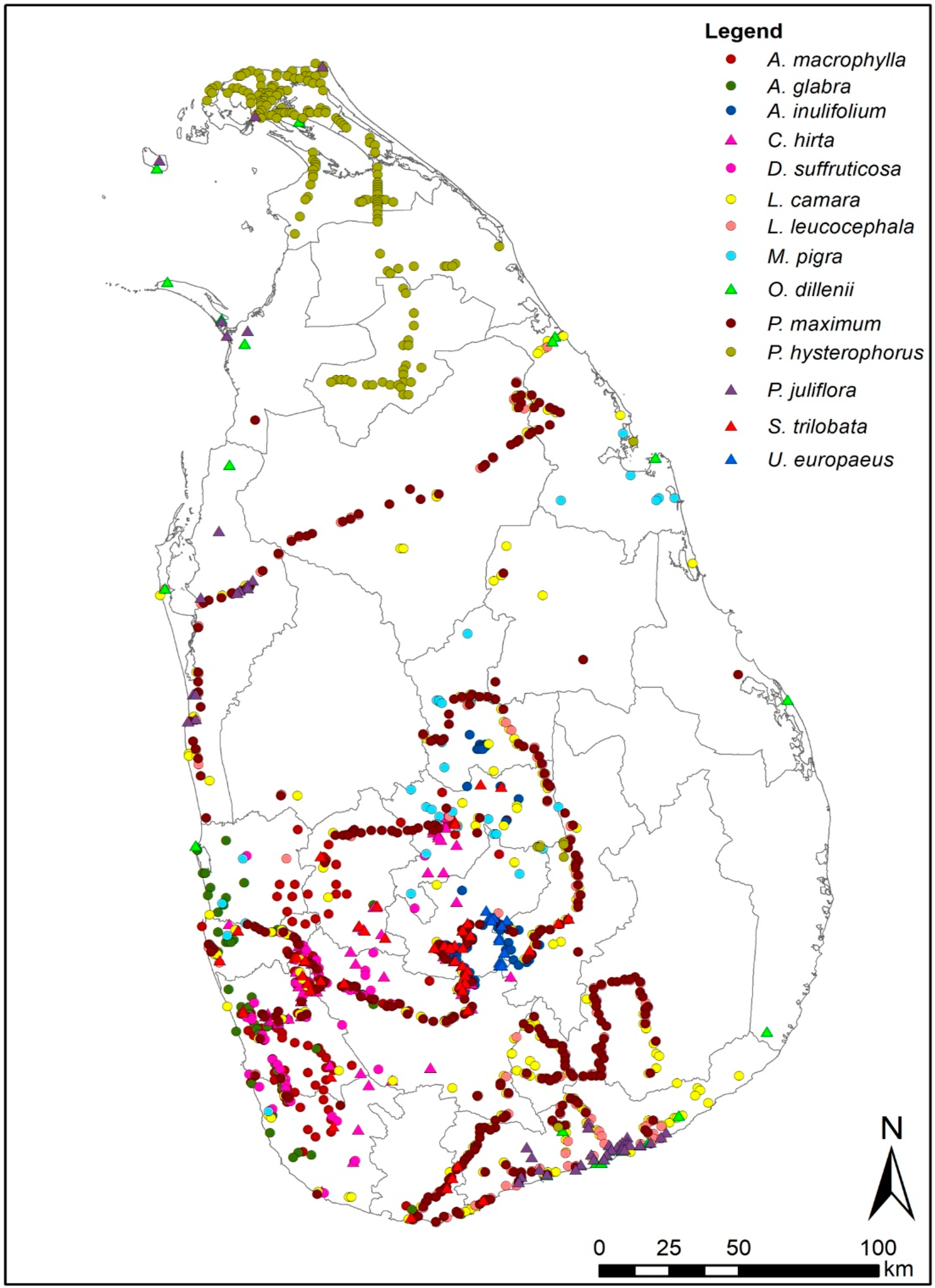
3.1. Species Distribution Models of 14 IAPS
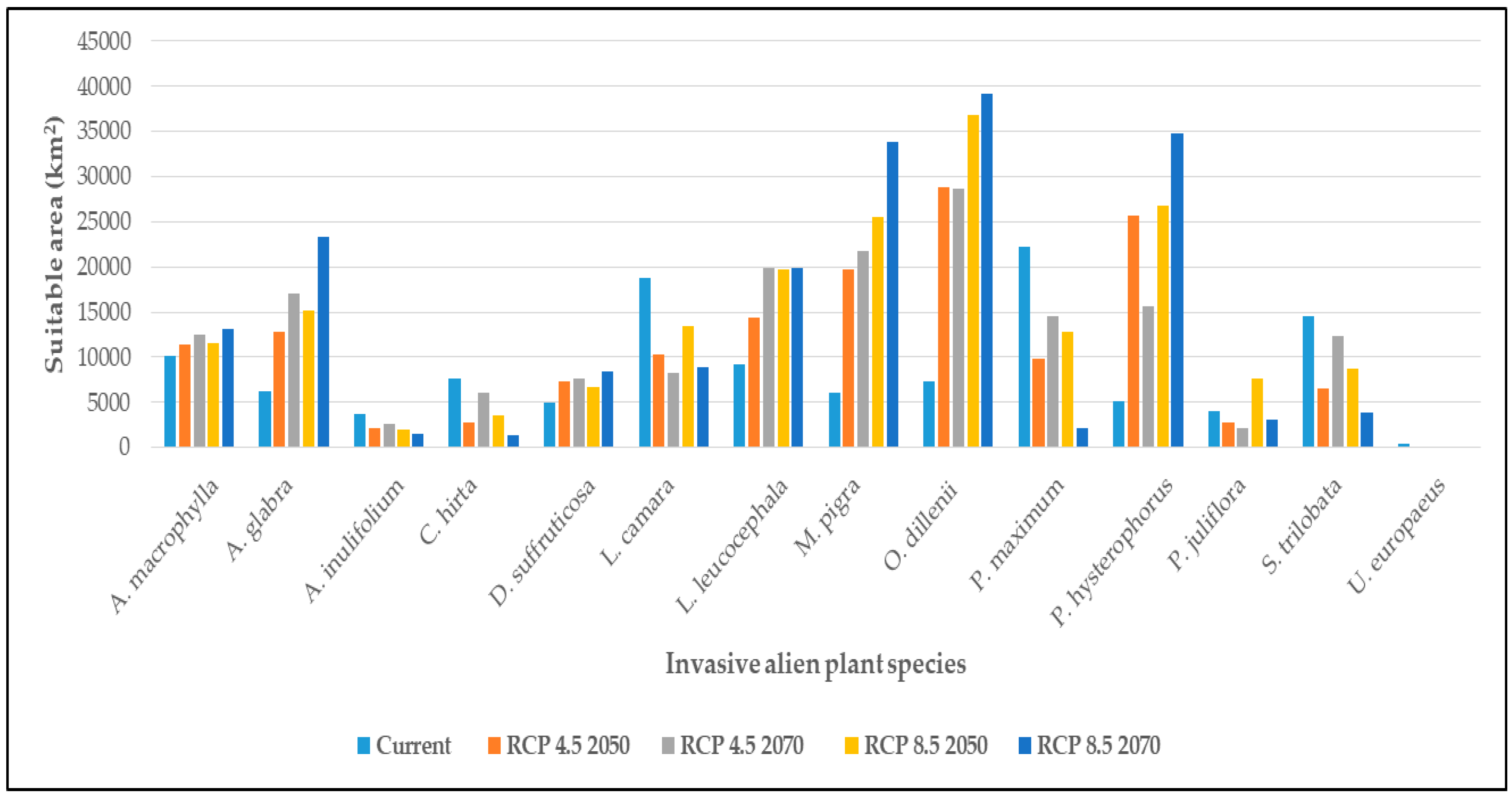
3.2. Climatic Suitability of Individual IAPS
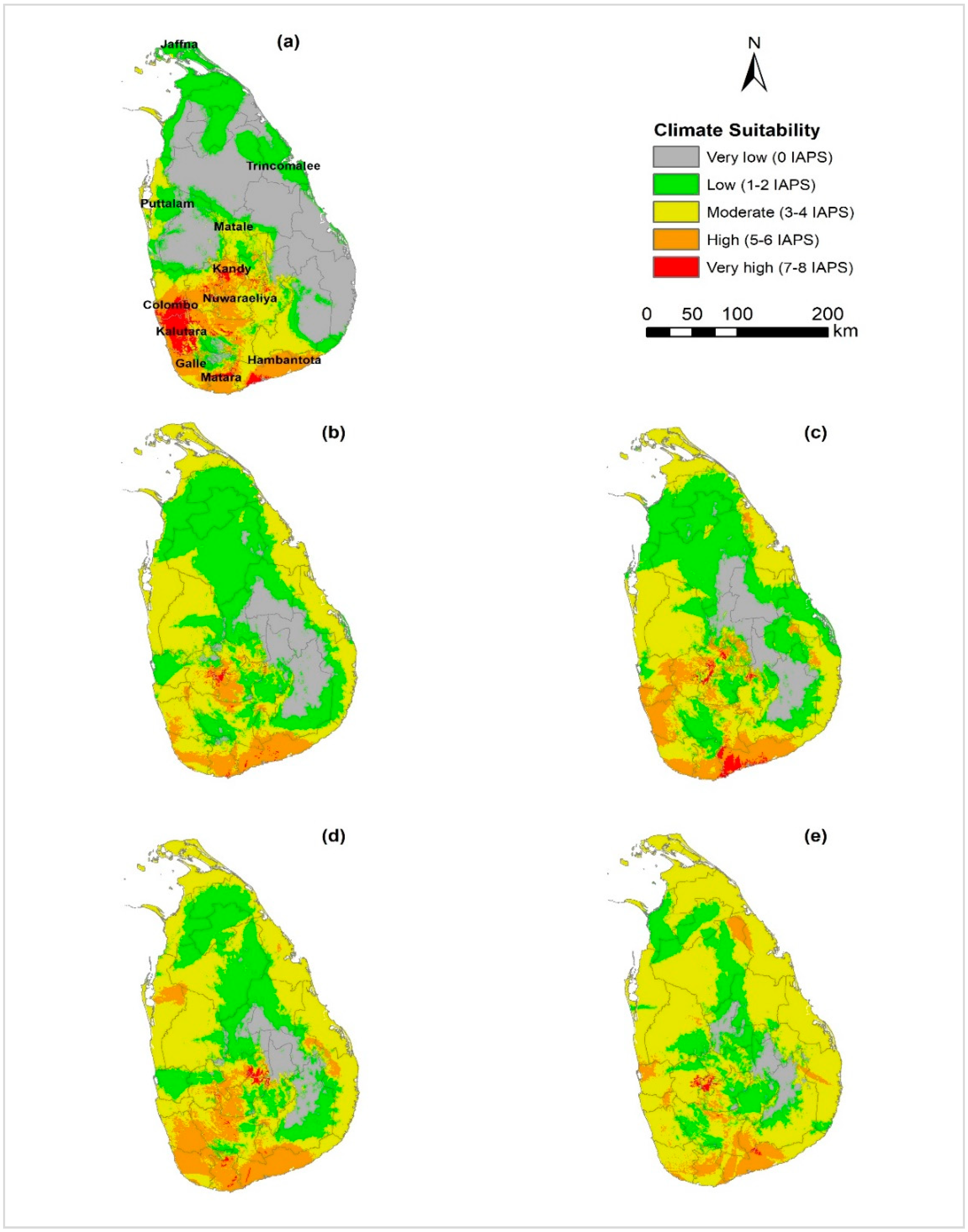
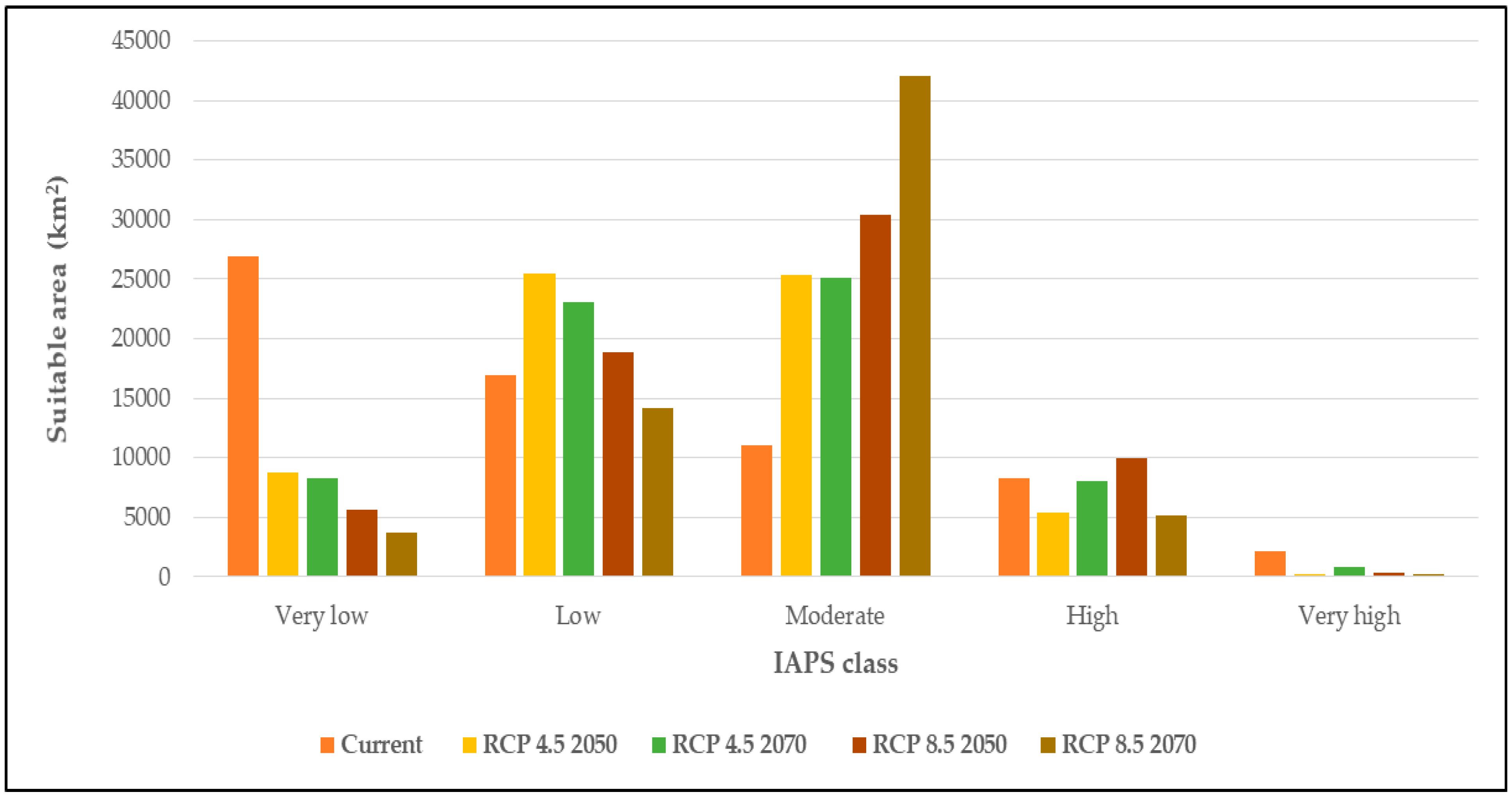
3.3. Climatic Suitability for Multiple Species Establishment
4. Discussion
4.1. Species Distribution Models of 14 IAPS
4.2. Climatic Suitability of Individual IAPS
4.3. Climate Suitability for Multiple Species Establishment
4.4. Study Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dukes, J.S.; Mooney, H.A. Does global change increase the success of biological invaders? Trends Ecol. Evol. 1999, 14, 135–139. [Google Scholar] [CrossRef]
- Lowe, S.; Browne, M.; Boudjelas, S.; De Poorter, M. 100 of the World’s Worst Invasive Alien Species: A Selection from the Global Invasive Species Database; Invasive Species Specialist Group (ISSG) a specialist group of the Species Survival Commission (SSC) of the World Conservation union (IUCN): Grand, Switzerland, 2000; Volume 12. [Google Scholar]
- Pysek, P.; Richardson, D.M. Invasive Species, Environmental Change and Management, and Health. Annu. Rev. Environ. Resour. 2010, 35, 25–55. [Google Scholar] [CrossRef]
- Early, R.; Bradley, B.A.; Dukes, J.S.; Lawler, J.J.; Olden, J.D.; Blumenthal, D.M.; Gonzalez, P.; Grosholz, E.D.; Ibañez, I.; Miller, L.P. Global threats from invasive alien species in the twenty-first century and national response capacities. Nat. Commun. 2016, 7, 12485. [Google Scholar] [CrossRef]
- Millennium Ecosystem Assessment. Ecosystems and Human Well-Being: Synthesis; Island Press: Washington, DC, USA, 2005. [Google Scholar]
- Vitousek, P.M.; D’antonio, C.M.; Loope, L.L.; Rejmanek, M.; Westbrooks, R. Introduced species: A significant component of human-caused global change. N. Z. J. Ecol. 1997, 21, 1–16. [Google Scholar]
- McNeely, J.A. Strangers in our midst: The problem of invasive alien species. Environment 2004, 46, 16. [Google Scholar] [CrossRef]
- Kumar, L.; Tehrany, M.S. Climate change impacts on the threatened terrestrial vertebrates of the Pacific Islands. Sci. Rep. 2017, 7, 5030. [Google Scholar] [CrossRef] [PubMed]
- Mack, R.N.; Simberloff, D.; Mark Lonsdale, W.; Evans, H.; Clout, M.; Bazzaz, F.A. Biotic invasions: Causes, epidemiology, global consequences, and control. Ecol. Appl. 2000, 10, 689–710. [Google Scholar] [CrossRef]
- McNeely, J.A.; Mooney, H.A.; Neville, L.E.; Schei, P.J.; Waage, J.K. A Global Strategy on Invasive Alien Species; IUCN: Gland, Switzerland; Cambridge, UK, 2001. [Google Scholar]
- Taylor, S.; Kumar, L. Global climate change impacts on pacific islands terrestrial biodiversity: A review. Trop. Conserv. Sci. 2016, 9, 203–223. [Google Scholar] [CrossRef]
- Hulme, P.E. Trade, transport and trouble: Managing invasive species pathways in an era of globalization. J. Appl. Ecol. 2009, 46, 10–18. [Google Scholar] [CrossRef]
- Kearney, M.; Phillips, B.L.; Tracy, C.R.; Christian, K.A.; Betts, G.; Porter, W.P. Modelling species distributions without using species distributions: The cane toad in Australia under current and future climates. Ecography 2008, 31, 423–434. [Google Scholar] [CrossRef]
- Ward, D.F. Modelling the potential geographic distribution of invasive ant species in New Zealand. Biol. Invasions 2007, 9, 723–735. [Google Scholar] [CrossRef]
- Gormley, A.M.; Forsyth, D.M.; Griffioen, P.; Lindeman, M.; Ramsey, D.S.L.; Scroggie, M.P.; Woodford, L. Using presence-only and presence-absence data to estimate the current and potential distributions of established invasive species. J. Appl. Ecol. 2011, 48, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Franklin, J. Mapping Species Distributions: Spatial Inference and Prediction; Cambridge University Press: Cambridge, UK, 2009; p. 320. [Google Scholar]
- Wiens, J.A.; Stralberg, D.; Jongsomjit, D.; Howell, C.A.; Snyder, M.A. Niches, models, and climate change: Assessing the assumptions and uncertainties. Proc. Natl. Acad. Sci. USA 2009, 106, 19729–19736. [Google Scholar] [CrossRef] [PubMed]
- Guisan, A.; Zimmermann, N.E. Predictive habitat distribution models in ecology. Ecol. Model. 2000, 135, 147–186. [Google Scholar] [CrossRef]
- Guisan, A.; Thuiller, W. Predicting species distribution: Offering more than simple habitat models. Ecol. Lett. 2005, 8, 993–1009. [Google Scholar] [CrossRef]
- Jiménez-Valverde, A.; Peterson, A.T.; Soberón, J.; Overton, J.; Aragón, P.; Lobo, J.M. Use of niche models in invasive species risk assessments. Biol. Invasions 2011, 13, 2785–2797. [Google Scholar] [CrossRef]
- Phillips, S.; Anderson, R.P.; Schapire, R.E. Maximum entropy modelling of species geographic distributions. Ecol. Model. 2006, 190, 231–259. [Google Scholar] [CrossRef]
- Tsoar, A.; Allouche, O.; Steinitz, O.; Rotem, D.; Kadmon, R. A comparative evaluation of presence-only methods for modelling species distribution. Divers. Distrib. 2007, 13, 397–405. [Google Scholar] [CrossRef]
- IPCC. Climate Change 2014: Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Core Writing Team, Pachauri, R.K., Meyer, L.A., Eds.; IPCC: Geneva, Switzerland, 2014. [Google Scholar]
- Zhang, B.; Cui, L.; Shi, J.; Wei, P. Vegetation dynamics and their response to climatic variability in China. Adv. Meteorol. 2017. [Google Scholar] [CrossRef]
- Sun, J.; Cheng, G.; Li, W. Meta-analysis of relationships between environmental factors and aboveground biomass in the alpine grassland on the Tibetan Plateau. Biogeosciences 2013, 10, 1707–1715. [Google Scholar] [CrossRef]
- Woodward, F.I. Climate and Plant Distribution; Cambridge University Press: New York, NY, USA, 1987. [Google Scholar]
- Pearson, R.G.; Dawson, T.P. Predicting the impacts of climate change on the distribution of species: Are bioclimate envelope models useful? Glob. Ecol. Biogeogr. 2003, 12, 361–371. [Google Scholar] [CrossRef]
- Beaumont, L.J.; Hughes, L.; Poulsen, M. Predicting species distributions: Use of climatic parameters in BIOCLIM and its impact on predictions of species’ current and future distributions. Ecol. Model. 2005, 186, 251–270. [Google Scholar] [CrossRef]
- Elith, J. Predicting Distributions of Invasive Species. 2015. Available online: https://arxiv.org/ftp/arxiv/papers/1312/1312.0851.pdf (accessed on 10 January 2019).
- Fandohan, A.B.; Oduor, A.M.; Sodé, A.I.; Wu, L.; Cuni-Sanchez, A.; Assédé, E.; Gouwakinnou, G.N. Modeling vulnerability of protected areas to invasion by Chromolaena odorata under current and future climates. Ecosyst. Health Sustain. 2015, 1, 1–12. [Google Scholar] [CrossRef]
- Taylor, S.; Kumar, L. Potential distribution of an invasive species under climate change scenarios using CLIMEX and soil drainage: A case study of Lantana camara L. in Queensland, Australia. J. Environ. Manag. 2013, 114, 414–422. [Google Scholar] [CrossRef] [PubMed]
- Goodwin, B.J.; McAllister, A.J.; Fahrig, L. Predicting invasiveness of plant species based on biological information. Conserv. Biol. 1999, 13, 422–426. [Google Scholar] [CrossRef]
- Thuiller, W.; Richardson, D.M.; PYŠEK, P.; Midgley, G.F.; Hughes, G.O.; Rouget, M. Niche-based modelling as a tool for predicting the risk of alien plant invasions at a global scale. Glob. Chang. Biol. 2005, 11, 2234–2250. [Google Scholar] [CrossRef]
- Soberon, J.; Peterson, A.T. Interpretation of models of fundamental ecological niches and species’ distributional areas. Biodivers. Inform. 2005, 2. [Google Scholar] [CrossRef]
- Phillips, S.J. Transferability, sample selection bias and background data in presence-only modelling: A response to Peterson et al. (2007). Ecography 2008, 31, 272–278. [Google Scholar] [CrossRef]
- Pulliam, H.R. On the relationship between niche and distribution. Ecol. Lett. 2000, 3, 349–361. [Google Scholar] [CrossRef]
- Booth, T.H. Species distribution modelling tools and databases to assist managing forests under climate change. For. Ecol. Manag. 2018, 430, 196–203. [Google Scholar] [CrossRef]
- Yackulic, C.B.; Chandler, R.; Zipkin, E.F.; Royle, J.A.; Nichols, J.D.; Campbell Grant, E.H.; Veran, S. Presence-only modelling using MAXENT: When can we trust the inferences? Methods Ecol. Evol. 2013, 4, 236–243. [Google Scholar] [CrossRef]
- Dyderski, M.K.; Paź, S.; Frelich, L.E.; Jagodziński, A.M. How much does climate change threaten European forest tree species distributions? Glob. Chang. Biol. 2018, 24, 1150–1163. [Google Scholar] [CrossRef] [PubMed]
- MoMD&E. National Biodiversity Strategic Action Plan 2016–2022. Colombo, Sri Lanka; Biodiversity Secretariat, Ministry of Mahaweli Development and Environment: Battaramulla, Sri Lanka, 2016.
- MoFE. Biodiversity Conservation in Sri Lanka: A Framework for Action; Ministry of Forestry and Environment: Battaramulla, Sri Lanka, 1999.
- Gunatilleke, N.; Pethiyagoda, R.; Gunatilleke, S. Biodiversity of Sri Lanka. J. Natl. Sci. Found. Sri Lanka 2017, 36, 25–62. [Google Scholar] [CrossRef]
- Mapa, R.; Kumaragamage, D.; Gunarathne, W.; Dassanayake, A. Land use in Sri Lanka: Past, present and the future. In Proceedings of the 17th World Congress of Social Science (WCSS), Bangkok, Thailand, 14–21 August 2002. [Google Scholar]
- Graham, C.H.; Hijmans, R.J. A comparison of methods for mapping species ranges and species richness. Glob. Ecol. Biogeogr. 2006, 15, 578–587. [Google Scholar] [CrossRef]
- Ramírez-Villegas, J.; Bueno Cabrera, A. Working with Climate Data and Niche Modeling: I. Creation of Bioclimatic Variables; International Center for Tropical Agriculture (CIAT): Cali, Colombia, 2009. [Google Scholar]
- Elith, J.; Graham, C.H.; Anderson, R.P.; Dudík, M.; Ferrier, S.; Guisan, A.; Hijmans, R.J.; Huettmann, F.; Leathwick, J.R.; Lehmann, A. Novel methods improve prediction of species’ distributions from occurrence data. Ecography 2006, 29, 129–151. [Google Scholar] [CrossRef]
- Dudik, M.; Phillips, S.J.; Schapire, R.E. Performance guarantees for regularized maximum entropy density estimation. In International Conference on Computational Learning Theory; Springer: Berlin, Germany, 2004; pp. 472–486. [Google Scholar]
- Glor, R.E.; Warren, D. Testing ecological explantions for biogeographic boundaries. Evolution 2010, 65, 673–683. [Google Scholar] [CrossRef] [PubMed]
- Lamsal, P.; Kumar, L.; Aryal, A.; Atreya, K. Invasive alien plant species dynamics in the Himalayan region under climate change. Ambio 2018, 47, 697–710. [Google Scholar] [CrossRef] [PubMed]
- Thapa, S.; Chitale, V.; Rijal, S.J.; Bisht, N.; Shrestha, B.B. Understanding the dynamics in distribution of invasive alien plant species under predicted climate change in Western Himalaya. PLoS ONE 2018, 13, e0195752. [Google Scholar] [CrossRef]
- Baldwin, R.A. Use of maximum entropy modeling in wildlife research. Entropy 2009, 11, 854–866. [Google Scholar] [CrossRef]
- Kittle, A.M.; Watson, A.C.; Cushman, S.A.; Macdonald, D.W. Forest cover and level of protection influence the island-wide distribution of an apex carnivore and umbrella species, the Sri Lankan leopard (Panthera pardus kotiya). Biodivers. Conserv. 2018, 27, 235–263. [Google Scholar] [CrossRef]
- MMD&E. Invasive Alien Species in Sri Lanka: Training Manual for Managers and Policymakers; Biodiversity Secretariat, Ministry of Mahaweli Development & Environment: Battaramulla, Sri Lanka, 2015.
- Carr, G.M.; Duthie, H.C.; Taylor, W.D. Models of aquatic plant productivity: A review of the factors that influence growth. Aquat. Bot. 1997, 59, 195–215. [Google Scholar] [CrossRef]
- Merow, C.; Smith, M.J.; Silander, J.A. A practical guide to MaxEnt for modeling species’ distributions: What it does, and why inputs and settings matter. Ecography 2013, 36, 1058–1069. [Google Scholar] [CrossRef]
- Boria, R.A.; Olson, L.E.; Goodman, S.M.; Anderson, R.P. Spatial filtering to reduce sampling bias can improve the performance of ecological niche models. Ecol. Model. 2014, 275, 73–77. [Google Scholar] [CrossRef]
- Mainali, K.P.; Warren, D.L.; Dhileepan, K.; McConnachie, A.; Strathie, L.; Hassan, G.; Karki, D.; Shrestha, B.B.; Parmesan, C. Projecting future expansion of invasive species: Comparing and improving methodologies for species distribution modeling. Glob. Chang. Biol. 2015, 21, 4464–4480. [Google Scholar] [CrossRef] [PubMed]
- CABI. CABI, Invasive Species Compendium; CAB International: Wallingford, UK, 2018. [Google Scholar]
- GBIF. GBIF Home Page. 2018. Available online: https://www.gbif.org (accessed on 13 August 2018).
- Kariyawasam, C.; Kadupitiya, H.; Ratnayake, R.; Hettiarchchi, A.; Ratnayake, R. Identification of High-Risk Agro-Ecological Regions using Species Distribution Modeling of Priority Invasive Species in Sri Lanka. Indian J. Plant Genet. Resour. 2017, 30, 228–231. [Google Scholar] [CrossRef]
- Kirshanthan, S.; Jeyaeelan, E.C.; Nandakumar, J. Prevalence and distribution of Parthenium hysterophorus weed in Jaffna peninsula, Sri Lanka. Ann. Biol. Res. 2016, 7, 45–48. [Google Scholar]
- Kishojini, P.; Pakeerathan, K.; Mikunthan, G. GPS based density and distribution mapping and composting a sustainable approach for monitoring and managing Parthenium (Parthenium hysterophorus L.) in Northern Sri Lanka. Int. J. Agric. For. 2018, 8, 160–170. [Google Scholar]
- Adhikari, D.; Tiwary, R.; Barik, S.K. Modelling Hotspots for Invasive Alien Plants in India. PLoS ONE 2015, 10, e0134665. [Google Scholar] [CrossRef]
- Phillips, S.J.; Dudík, M. Modeling of species distributions with Maxent: New extensions and a comprehensive evaluation. Ecography 2008, 31, 161–175. [Google Scholar] [CrossRef]
- Hijmans, R.J.; Cameron, S.E.; Parra, J.L.; Jones, P.G.; Jarvis, A. Very high resolution interpolated climate surfaces for global land areas. Int. J. Climatol. 2005, 25, 1965–1978. [Google Scholar] [CrossRef]
- Watanabe, M.; Suzuki, T.; O’ishi, R.; Komuro, Y.; Watanabe, S.; Emori, S.; Takemura, T.; Chikira, M.; Ogura, T.; Sekiguchi, M. Improved climate simulation by MIROC5: Mean states, variability, and climate sensitivity. J. Clim. 2010, 23, 6312–6335. [Google Scholar] [CrossRef]
- Parolo, G.; Rossi, G.; Ferrarini, A. Toward improved species niche modelling: Arnica montana in the Alps as a case study. J. Appl. Ecol. 2008, 45, 1410–1418. [Google Scholar] [CrossRef]
- Dormann, C.F.; Elith, J.; Bacher, S.; Buchmann, C.; Carl, G.; Carré, G.; Marquéz, J.R.G.; Gruber, B.; Lafourcade, B.; Leitão, P.J. Collinearity: A review of methods to deal with it and a simulation study evaluating their performance. Ecography 2013, 36, 27–46. [Google Scholar] [CrossRef]
- Leroy, B.; Meynard, C.N.; Bellard, C.; Courchamp, F. virtualspecies, an R package to generate virtual species distributions. Ecography 2016, 39, 599–607. [Google Scholar] [CrossRef]
- Ibanez, I.; Silander, J., Jr.; Allen, J.M.; Treanor, S.A.; Wilson, A. Identifying hotspots for plant invasions and forecasting focal points of further spread. J. Appl. Ecol. 2009, 46, 1219–1228. [Google Scholar] [CrossRef]
- O’donnell, J.; Gallagher, R.V.; Wilson, P.D.; Downey, P.O.; Hughes, L.; Leishman, M.R. Invasion hotspots for non-native plants in Australia under current and future climates. Glob. Chang. Biol. 2012, 18, 617–629. [Google Scholar] [CrossRef]
- Boubli, J.; De Lima, M. Modeling the geographical distribution and fundamental niches of Cacajao spp. and Chiropotes israelita in Northwestern Amazonia via a maximum entropy algorithm. Int. J. Primatol. 2009, 30, 217–228. [Google Scholar] [CrossRef]
- Phillips, S.J.; Anderson, R.P.; Dudík, M.; Schapire, R.E.; Blair, M.E. Opening the black box: An open-source release of Maxent. Ecography 2017, 40, 887–893. [Google Scholar] [CrossRef]
- Wisz, M.S.; Hijmans, R.; Li, J.; Peterson, A.T.; Graham, C.; Guisan, A.; Group, N.P.S.D.W. Effects of sample size on the performance of species distribution models. Divers. Distrib. 2008, 14, 763–773. [Google Scholar] [CrossRef]
- Phillips, S.J.; Dudík, M.; Schapire, R.E. A maximum entropy approach to species distribution modeling. In Proceedings of the Twenty-First International Conference on Machine Learning, Banff, AB, Canada, 4–8 July 2004; p. 83. [Google Scholar]
- Hernandez, P.A.; Graham, C.H.; Master, L.L.; Albert, D.L. The effect of sample size and species characteristics on performance of different species distribution modeling methods. Ecography 2006, 29, 773–785. [Google Scholar] [CrossRef]
- Graham, C.H.; Elith, J.; Hijmans, R.J.; Guisan, A.; Townsend Peterson, A.; Loiselle, B.A.; The Nceas Predicting Species Distributions Working Group. The influence of spatial errors in species occurrence data used in distribution models. J. Appl. Ecol. 2008, 45, 239–247. [Google Scholar] [CrossRef]
- Elith, J.; Kearney, M.; Phillips, S. The art of modelling range-shifting species. Methods Ecol. Evol. 2010, 1, 330–342. [Google Scholar] [CrossRef]
- Keevers, T.L. Cross-Validation is Insufficient for Model Validation; Joint and Operations Analysis Division, Defence Science and Technology Group: Victoria, Australia, 2019.
- Barbet-Massin, M.; Rome, Q.; Villemant, C.; Courchamp, F. Can species distribution models really predict the expansion of invasive species? PLoS ONE 2018, 13, e0193085. [Google Scholar] [CrossRef]
- Hanley, J.A.; McNeil, B.J. The meaning and use of the area under a receiver operating characteristic (ROC) curve. Radiology 1982, 143, 29–36. [Google Scholar] [CrossRef]
- Manel, S.; Williams, H.C.; Ormerod, S.J. Evaluating presence–absence models in ecology: The need to account for prevalence. J. Appl. Ecol. 2001, 38, 921–931. [Google Scholar] [CrossRef]
- Allouche, O.; Tsoar, A.; Kadmon, R. Assessing the accuracy of species distribution models: Prevalence, kappa and the true skill statistic (TSS). J. Appl. Ecol. 2006, 43, 1223–1232. [Google Scholar] [CrossRef]
- Liu, C.; Berry, P.M.; Dawson, T.P.; Pearson, R.G. Selecting thresholds of occurrence in the prediction of species distributions. Ecography 2005, 28, 385–393. [Google Scholar] [CrossRef]
- Liu, C.; Newell, G.; White, M. On the selection of thresholds for predicting species occurrence with presence-only data. Ecol. Evol. 2016, 6, 337–348. [Google Scholar] [CrossRef]
- Liu, C.; White, M.; Newell, G. Selecting thresholds for the prediction of species occurrence with presence-only data. J. Biogeogr. 2013, 40, 778–789. [Google Scholar] [CrossRef]
- Rubio, G.D.; Acosta, L.E. Geographical distribution of the space-weaving spider, Chibchea salta, from northwestern Argentina: New records and bioclimatic modeling. J. Insect Sci. 2011, 11. [Google Scholar] [CrossRef]
- Phillips, S.J. A Brief Tutorial on Maxent. 2017. Available online: http://biodiversityinformatics.amnh.org/open_source/maxent/ (accessed on 10 September 2018).
- Fielding, A.H.; Bell, J.F. A review of methods for the assessment of prediction errors in conservation presence/absence models. Environ. Conserv. 1997, 24, 38–49. [Google Scholar] [CrossRef]
- Phillips, S.J.; Dudík, M.; Elith, J.; Graham, C.H.; Lehmann, A.; Leathwick, J.; Ferrier, S. Sample selection bias and presence-only distribution models: Implications for background and pseudo-absence data. Ecol. Appl. 2009, 19, 181–197. [Google Scholar] [CrossRef] [PubMed]
- Peterson, A.T.; Soberón, J.; Pearson, R.G.; Anderson, R.P.; Martínez-Meyer, E.; Nakamura, M.; Araújo, M.B. Ecological Niches and Geographic Distributions (MPB-49); Princeton University Press: Princeton, NJ, USA, 2011; Volume 56. [Google Scholar]
- Pearce, J.; Ferrier, S. Evaluating the predictive performance of habitat models developed using logistic regression. Ecol. Model. 2000, 133, 225–245. [Google Scholar] [CrossRef]
- Pramanik, M.; Paudel, U.; Mondal, B.; Chakraborti, S.; Deb, P. Predicting climate change impacts on the distribution of the threatened Garcinia indica in the Western Ghats, India. Clim. Risk Manag. 2018, 19, 94–105. [Google Scholar] [CrossRef]
- Kramer-Schadt, S.; Niedballa, J.; Pilgrim, J.D.; Schröder, B.; Lindenborn, J.; Reinfelder, V.; Stillfried, M.; Heckmann, I.; Scharf, A.K.; Augeri, D.M.; et al. The importance of correcting for sampling bias in MaxEnt species distribution models. Divers. Distrib. 2013, 19, 1366–1379. [Google Scholar] [CrossRef]
- Bezeng, B.S.; Morales-Castilla, I.; van der Bank, M.; Yessoufou, K.; Daru, B.H.; Davies, T.J. Climate change may reduce the spread of non-native species. Ecosphere 2017, 8, e01694. [Google Scholar] [CrossRef]
- Diez, J.M.; D’Antonio, C.M.; Dukes, J.S.; Grosholz, E.D.; Olden, J.D.; Sorte, C.J.; Blumenthal, D.M.; Bradley, B.A.; Early, R.; Ibáñez, I. Will extreme climatic events facilitate. Biol. Invasions Front. Ecol. Environ. 2012, 10, 249–257. [Google Scholar] [CrossRef]
- Taylor, S.; Kumar, L.; Reid, N.; Kriticos, D.J. Climate change and the potential distribution of an invasive shrub, Lantana camara L. PLoS ONE 2012, 7, e35565. [Google Scholar] [CrossRef]
- Romero-Alvarez, D.; Escobar, L.E.; Varela, S.; Larkin, D.J.; Phelps, N.B. Forecasting distributions of an aquatic invasive species (Nitellopsis obtusa) under future climate scenarios. PLoS ONE 2017, 12, e0180930. [Google Scholar] [CrossRef]
- Taylor, S.; Kumar, L. Will climate change impact the potential distribution of a native vine (Merremia peltata) which is behaving invasively in the Pacific region? Ecol. Evol. 2016, 6, 742–754. [Google Scholar] [CrossRef]
- Kriticos, D.; Watt, M.; Potter, K.; Manning, L.; Alexander, N.; Tallent-Halsell, N. Managing invasive weeds under climate change: Considering the current and potential future distribution of Buddleja davidii. Weed Res. 2011, 51, 85–96. [Google Scholar] [CrossRef]
- Gillard, M.; Thiébaut, G.; Deleu, C.; Leroy, B. Present and future distribution of three aquatic plants taxa across the world: Decrease in native and increase in invasive ranges. Biol. Invasions 2017, 19, 2159–2170. [Google Scholar] [CrossRef]
- Shrestha, U.B.; Sharma, K.P.; Devkota, A.; Siwakoti, M.; Shrestha, B.B. Potential impact of climate change on the distribution of six invasive alien plants in Nepal. Ecol. Indic. 2018, 95, 99–107. [Google Scholar] [CrossRef]
- Čuda, J.; Skálová, H.; Janovský, Z.; Pyšek, P. Competition among native and invasive Impatiens species: The roles of environmental factors, population density and life stage. AoB Plants 2015, 7. [Google Scholar] [CrossRef] [PubMed]
- Hayes, K.R.; Barry, S.C. Are there any consistent predictors of invasion success? Biol. Invasions 2008, 10, 483–506. [Google Scholar] [CrossRef]
- Gallardo, B.; Aldridge, D.C.; González-Moreno, P.; Pergl, J.; Pizarro, M.; Pyšek, P.; Thuiller, W.; Yesson, C.; Vilà, M. Protected areas offer refuge from invasive species spreading under climate change. Glob. Chang. Biol. 2017, 23, 5331–5343. [Google Scholar] [CrossRef]
- Marchese, C. Biodiversity hotspots: A shortcut for a more complicated concept. Glob. Ecol. Conserv. 2015, 3, 297–309. [Google Scholar] [CrossRef]
- Goberville, E.; Beaugrand, G.; Hautekèete, N.C.; Piquot, Y.; Luczak, C. Uncertainties in the projection of species distributions related to general circulation models. Ecol. Evol. 2015, 5, 1100–1116. [Google Scholar] [CrossRef]
- Sharmila, S.; Joseph, S.; Sahai, A.; Abhilash, S.; Chattopadhyay, R. Future projection of Indian summer monsoon variability under climate change scenario: An assessment from CMIP5 climate models. Glob. Planet. Chang. 2015, 124, 62–78. [Google Scholar] [CrossRef]
- Mishra, V.; Kumar, D.; Ganguly, A.R.; Sanjay, J.; Mujumdar, M.; Krishnan, R.; Shah, R.D. Reliability of regional and global climate models to simulate precipitation extremes over India. J. Geophys. Res. Atmos. 2014, 119, 9301–9323. [Google Scholar] [CrossRef]
- Sperber, K.R.; Annamalai, H.; Kang, I.-S.; Kitoh, A.; Moise, A.; Turner, A.; Wang, B.; Zhou, T. The Asian summer monsoon: An intercomparison of CMIP5 vs. CMIP3 simulations of the late 20th century. Clim. Dyn. 2013, 41, 2711–2744. [Google Scholar] [CrossRef]
- Su, J.; Aryal, A.; Nan, Z.; Ji, W. Climate change-induced range expansion of a subterranean rodent: Implications for rangeland management in Qinghai-Tibetan Plateau. PLoS ONE 2015, 10, e0138969. [Google Scholar] [CrossRef] [PubMed]
- Aryal, A.; Shrestha, U.B.; Ji, W.; Ale, S.B.; Shrestha, S.; Ingty, T.; Maraseni, T.; Cockfield, G.; Raubenheimer, D. Predicting the distributions of predator (snow leopard) and prey (blue sheep) under climate change in the Himalaya. Ecol. Evol. 2016, 6, 4065–4075. [Google Scholar] [CrossRef] [PubMed]
- Thuiller, W.; Guéguen, M.; Renaud, J.; Karger, D.N.; Zimmermann, N.E. Uncertainty in ensembles of global biodiversity scenarios. Nat. Commun. 2019, 10, 1446. [Google Scholar] [CrossRef] [PubMed]
- Norberg, A.; Abrego, N.; Blanchet, F.G.; Adler, F.R.; Anderson, B.J.; Anttila, J.; Araújo, M.B.; Dallas, T.; Dunson, D.; Elith, J. A comprehensive evaluation of predictive performance of 33 species distribution models at species and community levels. Ecol. Monogr. 2019, e01370. [Google Scholar] [CrossRef]
- Araújo, M.B.; New, M. Ensemble forecasting of species distributions. Trends Ecol. Evol. 2007, 22, 42–47. [Google Scholar] [CrossRef]
- Elith, J.; Phillips, S.J.; Hastie, T.; Dudík, M.; Chee, Y.E.; Yates, C.J. A statistical explanation of MaxEnt for ecologists. Divers. Distrib. 2011, 17, 43–57. [Google Scholar] [CrossRef]
- Mod, H.K.; Scherrer, D.; Luoto, M.; Guisan, A. What we use is not what we know: Environmental predictors in plant distribution models. J. Veg. Sci. 2016, 27, 1308–1322. [Google Scholar] [CrossRef]
- Lahoz-Monfort, J.J.; Guillera-Arroita, G.; Wintle, B.A. Imperfect detection impacts the performance of species distribution models. Glob. Ecol. Biogeogr. 2014, 23, 504–515. [Google Scholar] [CrossRef]
- Miller, D.A.; Nichols, J.D.; McClintock, B.T.; Grant, E.H.C.; Bailey, L.L.; Weir, L.A. Improving occupancy estimation when two types of observational error occur: Non-detection and species misidentification. Ecology 2011, 92, 1422–1428. [Google Scholar] [CrossRef]
- Chen, G.; Kéry, M.; Plattner, M.; Ma, K.; Gardner, B. Imperfect detection is the rule rather than the exception in plant distribution studies. J. Ecol. 2013, 101, 183–191. [Google Scholar] [CrossRef]
- Monk, J. How long should we ignore imperfect detection of species in the marine environment when modelling their distribution? Fish Fish. 2014, 15, 352–358. [Google Scholar] [CrossRef]
- Comte, L.; Grenouillet, G. Species distribution modelling and imperfect detection: Comparing occupancy versus consensus methods. Divers. Distrib. 2013, 19, 996–1007. [Google Scholar] [CrossRef]
- Guisan, A.; Tingley, R.; Baumgartner, J.B.; Naujokaitis-Lewis, I.; Sutcliffe, P.R.; Tulloch, A.I.; Regan, T.J.; Brotons, L.; McDonald-Madden, E.; Mantyka-Pringle, C. Predicting species distributions for conservation decisions. Ecol. Lett. 2013, 16, 1424–1435. [Google Scholar] [CrossRef] [PubMed]
- Soberón, J.; Nakamura, M. Niches and distributional areas: Concepts, methods, and assumptions. Proc. Natl. Acad. Sci. USA 2009, 106, 19644–19650. [Google Scholar] [CrossRef] [PubMed]

| No | Species | Family | Common Name | Life Form | Year of Introduction | Affected Climatic Zones/Habitats | No. of Occurrences |
|---|---|---|---|---|---|---|---|
| 1 | Alstonia macrophylla Wall. | Apocynaceae | Hard milkwood | Tree | unknown | Wet zone | 116 |
| 2 | Annona glabra L. | Annonaceae | Pond apple | Tree | unknown | Wet zone (e.g., Coastal wetlands) | 69 |
| 3 | Austroeupatorium inulifolium (H.B.K.) R. M. King & H. Rob | Asteraceae | Austroeupatorium | Shrub | unknown | Montane zone (e.g., Knuckles Conservation Forest) | 60 |
| 4 | Clidemia hirta (L.) D. Don | Melastomataceae | Soapbush, Koster’s curse | Herb | 1894 | Wet zone/Lowland wet zone forests (e.g., Sinharaja forest) | 80 |
| 5 | Dillenia suffruticosa (Griff ex Hook.f. & Thomson) Martelli | Dilleniaceae | Shrubby Dillenia | Tree | 1882 | Lowland wet zone | 68 |
| 6 | Lantana camara L. | Verbenaceae | Lantana | Bush | 1826 | Intermediate zone (e.g., Udawalawa National Park) | 253 |
| 7 | Leucaena leucocephala (Lam.) de Wit | Fabaceae | White lead tree | Shrub/Tree | 1980 | Dry and intermediate zones | 151 |
| 8 | Mimosa pigra L. | Fabaceae | Giant Mimosa | Bush | 1980 | Intermediate zone | 36 |
| 9 | Opuntia dillenii (Ker-Gawl.) Haw | Cactaceae | Prickly pear cactus | Cactus | unknown | Dry zone (e.g., Bundala National Park) | 25 |
| 10 | Panicum maximum Jacq. | Poaceae | Guinea grass | Grass | 1801-1802 | All zones | 323 |
| 11 | Parthenium hysterophorus L. | Asteraceae | Parthenium | Herb | 1980 | Dry zone | 169 |
| 12 | Prosopis juliflora (Sw.) DC. | Fabaceae | Mesquite | Tree | 1880 | Dry Zone (e.g., Bundala National Park) | 48 |
| 13 | Sphagneticola trilobata (L.) Pruski | Asteraceae | Creeping ox-eye | Herb | unknown | Wet zone | 47 |
| 14 | Ulex europaeus L. | Fabaceae | Gorse | Bush | 1888 | Montane zone/Wet Patana grassland (e.g., Horton Plains National Park) | 15 |
| No | Variable | Abbreviation | Unit |
|---|---|---|---|
| 1 | Annual mean diurnal temperature range | bio2 | °C |
| 2 | Maximum temperature of warmest month | bio5 | °C |
| 3 | Minimum temperature of coldest month | bio6 | °C |
| 4 | Annual precipitation | bio12 | mm |
| 5 | Precipitation of driest month | bio14 | mm |
| 6 | Precipitation seasonality | bio15 | % |
| 7 | Precipitation of coldest quarter | bio19 | mm |
| IAPS Class | Suitable Area (km2) under RCP 4.5 | Suitable Area (km2) under RCP 8.5 | ||||
|---|---|---|---|---|---|---|
| 2050 (Relevant to Current Climate) | 2070 (Relevant to Current Climate) | 2070 (Relevant to 2050) | 2050 (Relevant to Current Climate) | 2070 (Relevant to Current Climate) | 2070 (Relevant to 2050) | |
| Very Low | ||||||
| Contraction | 21,181 (79) | 21,700 (81) | 2682 (31) | 23,617 (88) | 25,103 (93) | 3055 (54) |
| Expansion | 3019 (11) | 3087 (11) | 2231 (25) | 2354 (9) | 1847 (7) | 1062 (19) |
| Unchanged | 5738 (21) | 5218 (19) | 6074 (69) | 3301 (12) | 1816 (7) | 2601 (46) |
| Low | ||||||
| Contraction | 9746 (58) | 10,259 (61) | 9170 (36) | 11,677 (69) | 13,177 (70) | 11,943 (63) |
| Expansion | 18,323 (109) | 16,424 (97) | 6757 (27) | 13,693 (81) | 10,482 (55) | 7232 (38) |
| Unchanged | 7134 (42) | 6620 (39) | 16,287 (64) | 5203 (31) | 3703 (20) | 6953 (37) |
| Moderate | ||||||
| Contraction | 7247 (65) | 6608 (60) | 7649 (30) | 7258 (66) | 6443 (58) | 5465 (18) |
| Expansion | 21,574 (195) | 20,624 (186) | 7338 (29) | 26,609 (240) | 37,439 (338) | 17,109 (56) |
| Unchanged | 3823 (35) | 4462 (40) | 17,748 (70) | 3811 (34) | 4626 (42) | 24,956 (82) |
| High | ||||||
| Contraction | 4429 (54) | 4374 (53) | 1992 (37) | 3352 (41) | 5988 (60) | 7003 (71) |
| Expansion | 1614 (20) | 4151 (50) | 4584 (84) | 5015 (61) | 2852 (29) | 2205 (22) |
| Unchanged | 3823 (46) | 3878 (47) | 3444 (63) | 4899 (59) | 2264 (23) | 2911 (29) |
| Very High | ||||||
| Contraction | 2112 (100) | 2065 (97) | 105 (55) | 2111 (100) | 2109 (99) | 354 (100) |
| Expansion | 184 (9) | 720 (34) | 688 (358) | 344 (16) | 200 (9) | 211 (60) |
| Unchanged | 9 (0) | 55 (3) | 88 (46) | 9 (0) | 11(1) | 0 (0) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kariyawasam, C.S.; Kumar, L.; Ratnayake, S.S. Invasive Plant Species Establishment and Range Dynamics in Sri Lanka under Climate Change. Entropy 2019, 21, 571. https://doi.org/10.3390/e21060571
Kariyawasam CS, Kumar L, Ratnayake SS. Invasive Plant Species Establishment and Range Dynamics in Sri Lanka under Climate Change. Entropy. 2019; 21(6):571. https://doi.org/10.3390/e21060571
Chicago/Turabian StyleKariyawasam, Champika S., Lalit Kumar, and Sujith S. Ratnayake. 2019. "Invasive Plant Species Establishment and Range Dynamics in Sri Lanka under Climate Change" Entropy 21, no. 6: 571. https://doi.org/10.3390/e21060571
APA StyleKariyawasam, C. S., Kumar, L., & Ratnayake, S. S. (2019). Invasive Plant Species Establishment and Range Dynamics in Sri Lanka under Climate Change. Entropy, 21(6), 571. https://doi.org/10.3390/e21060571





