Salivary Adipokine and Cytokine Levels as Potential Markers for the Development of Obesity and Metabolic Disorders
Abstract
:1. Introduction
2. The Role of Adipose Tissue in the Development of Obesity
3. Saliva as a Potential Material in the Diagnosis of Obesity and Its Associated Disorders
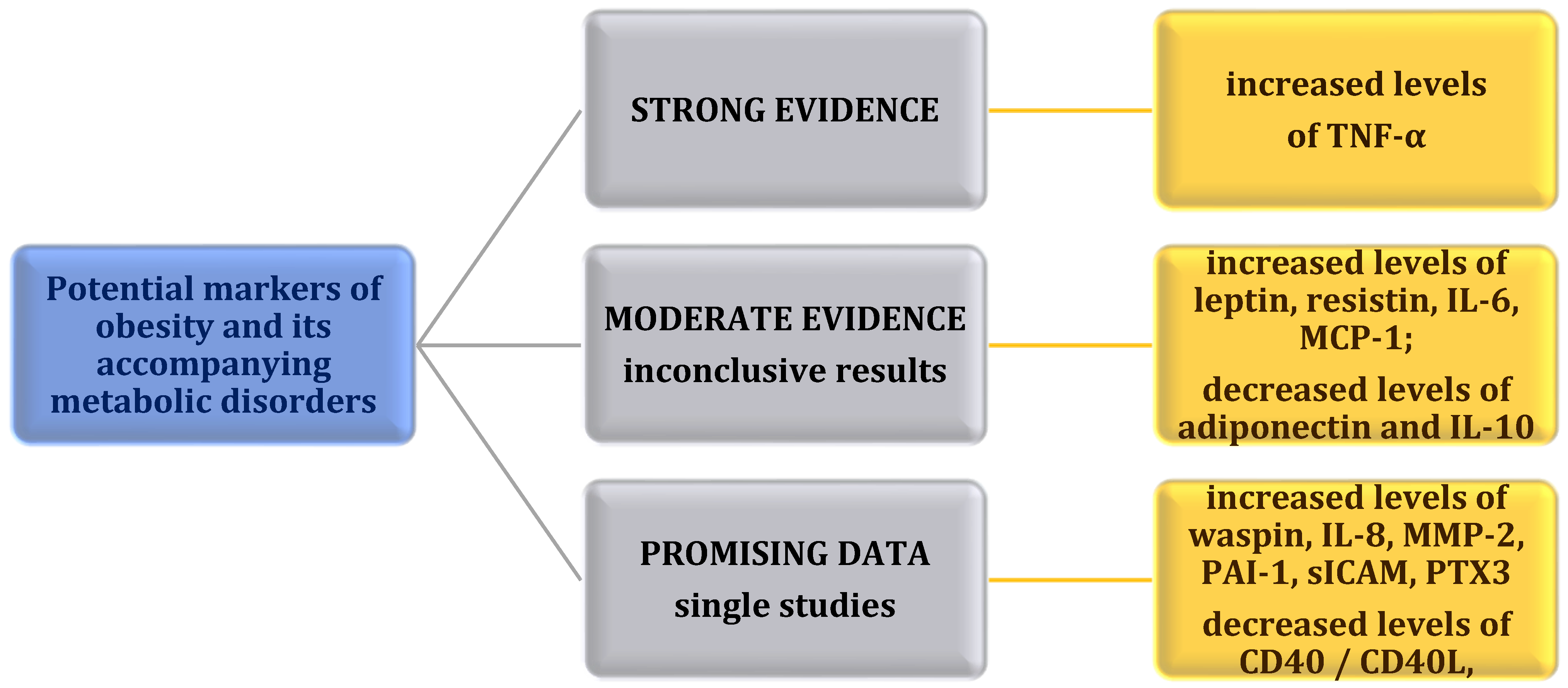
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organization. Preventing and Managing the Global Epidemic; World Health Organization: Geneva, Switzerland, 1998. [Google Scholar]
- World Health Organization. Obesity and Overweight. 2020. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 4 March 2021).
- Wang, B.; Zhuang, R.; Luo, X.; Yin, L.; Pang, C.; Feng, T.; You, H.; Zhai, Y.; Ren, Y.; Zhang, L.; et al. Prevalence of Metabolically Healthy Obese and Metabolically Obese but Normal Weight in Adults Worldwide: A Meta-Analysis. Horm. Metab. Res. 2015, 47, 839–845. [Google Scholar] [CrossRef] [PubMed]
- Hill, H.; Solt, C.; Foster, M.T. Obesity associated disease risk: The role of inherent differences and location of adipose depots. Horm. Mol. Biol. Clin. Investig. 2018, 33. [Google Scholar] [CrossRef]
- Murawska-Ciałowicz, E. Tkanka tłuszczowa-charakterystyka morfologiczna i biochemiczna różnych depozytów. Postęp. Hig. Med. Dosw. 2017, 71, 466–484. [Google Scholar]
- European Commission CORDIS. Terapeutyczna Brunatna Tkanka Tłuszczowa w Walce z Otyłością. Available online: https://cordis.europa.eu/article/id/92120-therapeutic-brown-fat-to-tackle-obesity/pl (accessed on 19 September 2021).
- Bilir, B.E.; Güldiken, S.; Tunçbilek, N.; Demir, A.M.; Polat, A.; Bilir, B. The effects of fat distribution and some adipokines on insulin resistance in subjects with prediabetes. Endokrynol. Pol. 2016, 67, 277–282. [Google Scholar] [PubMed] [Green Version]
- Karpe, F.; Pinnick, K.E. Biology of upper-body and lower-body adipose tissue—Link to whole-body phenotypes. Nat. Rev. Endocrinol. 2015, 11, 90–100. [Google Scholar] [CrossRef]
- Rønn, P.F.; Andersen, G.S.; Lauritzen, T.; Christensen, D.L.; Aadahl, M.; Carstensen, B.; Grarup, N.; Jørgensen, M.E. Abdominal visceral and subcutaneous adipose tissue and associations with cardiometabolic risk in Inuit, Africans and Europeans: A cross-sectional study. BMJ Open 2020, 10, e038071. [Google Scholar] [CrossRef] [PubMed]
- Matsha, T.E.; Ismail, S.; Speelman, A.; Hon, G.M.; Davids, S.; Erasmus, R.T.; Kengne, A.P. Visceral and subcutaneous adipose tissue association with metabolic syndrome and its components in a South African population. Clin. Nutr. ESPEN 2019, 32, 76–81. [Google Scholar] [CrossRef]
- Lee, J.J.; Pedley, A.; Therkelsen, K.E.; Hoffmann, U.; Massaro, J.M.; Levy, D.; Long, M.T. Upper Body Subcutaneous Fat Is Associated with Cardiometabolic Risk Factors. Am. J. Med. 2017, 130, 958–966. [Google Scholar] [CrossRef] [Green Version]
- Kwon, H.; Kim, D.; Kim, J.S. Body Fat Distribution and the Risk of Incident Metabolic Syndrome: A Longitudinal Cohort Study. Sci. Rep. 2017, 7, 10955. [Google Scholar] [CrossRef] [Green Version]
- Cancello, R.; Zulian, A.; Gentilini, D.; Maestrini, S.; Barba, A.D.; Invitti, C.; Corà, D.; Caselle, M.; Liuzzi, A.; Di Blasio, A.M. Molecular and morphologic characterization of superficial-and deep-subcutaneous adipose tissue subdivisions in human obesity. Obesity 2013, 21, 2562–2570. [Google Scholar] [CrossRef]
- Neeland, I.J.; Ayers, C.R.; Rohatgi, A.K.; Turer, A.T.; Berry, J.D.; Das, S.R.; Vega, G.L.; Khera, A.; McGuire, D.K.; Grundy, S.M.; et al. Associations of visceral and abdominal subcutaneous adipose tissue with markers of cardiac and metabolic risk in obese adults. Obesity 2013, 21, E439–E447. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choi, J.; Joseph, L.; Pilote, L. Obesity and C-reactive protein in various populations: A systematic review and meta-analysis. Obes. Rev. 2013, 14, 232–244. [Google Scholar] [CrossRef]
- Tvarijonaviciute, A.; Martinez-Lozano, N.; Rios, R.; Marcilla de Teruel, M.C.; Garaulet, M.; Cerón, J.J. Saliva as a non-invasive tool for assessment of metabolic and inflammatory biomarkers in children. Clin. Nutr. 2020, 39, 2471–2478. [Google Scholar] [CrossRef]
- Góralska, M.; Majewska-Szczepanik, M.; Szczepanik, M. Mechanizmy immunologiczne towarzyszące otyłości i ich rola w zaburzeniach metabolizmu. Postep. Hig. Med. Dosw. 2015, 69, 1384–1404. [Google Scholar]
- Boden, G. Obesity, Insulin Resistance and Free Fatty Acids. Curr. Opin. Endocrinol. Diabetes Obes. 2011, 18, 139–143. [Google Scholar] [CrossRef] [Green Version]
- Pîrsean, C.; Neguț, C.; Stefan-van Staden, R.I.; Dinu-Pirvu, C.E.; Armean, P.; Udeanu, D.I. The salivary levels of leptin and interleukin-6 as potential inflammatory markers in children obesity. PLoS ONE 2019, 14, e0210288. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mamali, I.; Roupas, N.D.; Armeni, A.K.; Theodoropoulou, A.; Markou, K.B.; Georgopoulos, N.A. Measurement of salivary resistin, visfatin and adiponectin levels. Peptides 2012, 33, 120–124. [Google Scholar] [CrossRef]
- Thanakun, S.; Watanabe, H.; Thaweboon, S.; Izumi, Y. Comparison of salivary and plasma adiponectin and leptin in patients with metabolic syndrome. Diabetol. Metab. Syndr. 2014, 6, 19. [Google Scholar] [CrossRef] [Green Version]
- Achari, E.; Jain, S.K. Molecular Sciences Adiponectin, a Therapeutic Target for Obesity, Diabetes, and Endothelial Dysfunction. Int. J. Mol. Sci. 2017, 18, 1321. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Attlee, A.; Hasan, H.; AlQattan, A.; Sarhan, N.; Alshammari, R.; Ali, S.; Nabil, M.; Alattrash, A.; Raigangar, V.; Madkour, M.; et al. Relationship of salivary adipocytokines, diet quality, physical activity, and nutrition status in adult Emirati females in United Arab Emirates. Diabetes Metab. Syndr. Clin. Res. Rev. 2019, 13, 40–46. [Google Scholar] [CrossRef]
- Duffles, L.F.; Hermont, A.P.; Abreu, L.G.; Pordeus, I.A.; Silva, T.A. Association between obesity and adipokines levels in saliva and gingival crevicular fluid: A systematic review and meta-analysis. J. Evid.-Based Med. 2019, 12, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Kalyani, R.S.; Raghunath, V. Assessment of serum and salivary adiponectin levels in newly diagnosed Type II diabetes mellitus patients. J. Oral Maxillofac. Pathol. 2020, 24, 245–250. [Google Scholar] [PubMed]
- Goodson, J.M.; Kantarci, A.; Hartman, M.; Denis, G.V.; Stephens, D.; Hasturk, H.; Yaskell, T.; Vargas, J.; Wang, X.; Cugini, M.; et al. Metabolic disease risk in children by salivary biomarker analysis. PLoS ONE 2014, 9, e98799. [Google Scholar] [CrossRef] [PubMed]
- Escoté, X.; Gómez-Zorita, S.; López-Yoldi, M.; Milton-Laskibar, I.; Fernández-Quintela, A.; Martínez, J.A.; Moreno-Aliaga, M.J.; Portillo, M.P. Role of Omentin, Vaspin, Cardiotrophin-1, TWEAK and NOV/CCN3 in Obesity and Diabetes Development. Int. J. Mol. Sci. 2019, 18, 1770. [Google Scholar] [CrossRef] [Green Version]
- Lehmann-Kalata, A.; Miechowicz, I.; Korybalska, K.; Swora-Cwynar, E.; Czepulis, N.; Łuczak, J.; Orzechowska, Z.; Grzymisławski, M.; Surdacka, A.; Witowski, J. Salivary fingerprint of simple obesity. Cytokine 2018, 110, 174–180. [Google Scholar] [CrossRef] [PubMed]
- Saraiva, M.; Vieira, P.; O’garra, A. Biology and therapeutic potential of interleukin-10. J. Exp. Med. 2020, 217, e20190418. [Google Scholar] [CrossRef] [Green Version]
- Chauhan, A.; Yadav, S.S.; Dwivedi, P.; Lal, N.; Usman, K.; Khattri, S. Correlation of Serum and Salivary Cytokines Level With Clinical Parameters in Metabolic Syndrome With Periodontitis. J. Clin. Lab. Anal. 2016, 30, 649–655. [Google Scholar] [CrossRef] [Green Version]
- Selvaraju, V.; Babu, J.R.; Geetha, T. Association of salivary C-reactive protein with the obesity measures and markers in children. Diabetes Metab. Syndr. Obes. 2019, 12, 1239–1247. [Google Scholar] [CrossRef] [Green Version]
- Lehmann, A.P.; Nijakowski, K.; Swora-Cwynar, E.; Łuczak, J.; Czepulis, N.; Surdacka, A. Characteristics of salivary inflammation in obesity. Polish Arch. Intern. Med. 2020, 130, 297–303. [Google Scholar]
- Landecho, M.F.; Tuero, C.; Valentí, V.; Bilbao, I.; De La Higuera, M.; Frühbeck, G. Relevance of Leptin and Other Adipokines in Obesity-Associated Cardiovascular Risk. Nutrients 2019, 11, 2664. [Google Scholar] [CrossRef] [Green Version]
- Martin, S.; Blaha, M.; Muse, E.D.; Qasim, A.N.; Reilly, M.P.; Blumenthal, R.S.; Nasir, K.; Criqui, M.H.; McClelland, R.L.; Hughes-Austin, J.M.; et al. Leptin and incident cardiovascular disease: The Multi-ethnic Study of Atherosclerosis (MESA). Atherosclerosis 2015, 239, 67–72. [Google Scholar] [CrossRef] [Green Version]
- Jayachandran, T.; Srinivasan, B.; Padmanabhan, S. Salivary leptin levels in normal weight and overweight individuals and their correlation with orthodontic tooth movement. Angle Orthod. 2017, 87, 739–744. [Google Scholar] [CrossRef] [Green Version]
- Park, H.K.; Kwak, M.K.; Kim, H.J.; Ahima, R.S. Linking resistin, inflammation, and cardiometabolic diseases. Korean J. Intern. Med. 2017, 32, 239–247. [Google Scholar] [CrossRef]
- Al-Rawi, N.; Al-Marzooq, F. The Relation between Periodontopathogenic Bacterial Levels and Resistin in the Saliva of Obese Type 2 Diabetic Patients. J. Diabetes Res. 2017, 2017, 2643079. [Google Scholar] [CrossRef] [Green Version]
- Doğusal, G.; Afacan, B.; Bozkurt, E.; Sönmez, I. Gingival crevicular fluid and salivary resistin and tumor necrosis factor-alpha levels in Obese children with gingivitis. J. Periodontol. 2018, 89, 973–982. [Google Scholar] [CrossRef] [PubMed]
- Akash, M.S.H.; Rehman, K.; Liaqat, A. Tumor Necrosis Factor-Alpha: Role in Development of Insulin Resistance and Pathogenesis of Type 2 Diabetes Mellitus. J. Cell. Biochem. 2018, 119, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Ostrowska, L.; Gornowicz, A.; Pietraszewska, B.; Bielawski, K.; Bielawska, A. Which salivary components can differentiate metabolic obesity? PLoS ONE 2020, 15, e0235358. [Google Scholar] [CrossRef]
- Han, M.S.; White, A.; Perry, R.; Camporez, J.P.; Hidalgo, J.; Shulman, G.I.; Davis, R.J. Regulation of adipose tissue inflammation by interleukin 6. Proc. Natl. Acad. Sci. USA 2020, 117, 2751–2760. [Google Scholar] [CrossRef] [Green Version]
- Makki, K.; Froguel, P.; Wolowczuk, I. Adipose Tissue in Obesity-Related Inflammation and Insulin Resistance: Cells, Cytokines, and Chemokines. 2013. Available online: https://downloads.hindawi.com/archive/2013/139239.pdf (accessed on 9 July 2021).
- Bing, C. Is interleukin-1b a culprit in macrophage-adipocyte crosstalk in obesity? Adipocyte 2015, 4, 149–152. [Google Scholar] [CrossRef] [Green Version]
- Bianconi, V.; Sahebkar, A.; Atkin, S.L.; Pirro, M. The regulation and importance of monocyte chemoattractant protein-1. Curr. Opin. Hematol. 2018, 25, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Miksztowicz, V.; Machulsky, N.F.; Lucero, D.; Fassio, E.; Schreier, L.; Berg, G. Adiponectin predicts MMP-2 activity independently of obesity. Eur. J. Clin. Invest. 2014, 44, 951–957. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Yan, J.; Liu, P.; Wang, Z.; Wang, C. Plasminogen activator inhibitor links obesity and thrombotic cerebrovascular diseases: The roles of PAI-1 and obesity on stroke. Metab. Brain Dis. 2017, 32, 667–673. [Google Scholar] [CrossRef] [PubMed]
- Neyraud, E.; Cabaret, S.; Brignot, H.; Chabanet, C.; Labouré, H.; Guichard, E.; Berdeaux, O. The basal free fatty acid concentration in human saliva is related to salivary lipolytic activity. Sci. Rep. 2017, 7, 5969. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Diagnostic Parameters of Obesity and Metabolic Disorders | The Levels of Adipokine/Cytokine in Saliva | |||||||
|---|---|---|---|---|---|---|---|---|
| Adiponectin | IL-10 | Resistin | TNF-α | IL-8 | MMP-2 | Study Cited | Age Group | |
| ↑ BMI [kg/m2] | ↓ * ▪▪ | ↓ ▪ | ↑ * ▪▪ | Attlee et al. [23] | A | |||
| ↑ * (no data) | Al-Rawi et al. [37] | A | ||||||
| ↑ * ▪▪▪▪ | ↑ * ▪▪▪ | ↑ * ▪▪▪▪▪ | Ostrowska et al. [40] | A | ||||
| ↑ WC [cm] | ↓ ▪▪ | ↑ * ▪▪ | Attlee et al. [23] | A | ||||
| ↑ * ▪▪▪ | ↑ * ▪▪▪▪ | Ostrowska et al. [40] | A | |||||
| ↑ BFM [kg] | ↑ * ▪▪▪ | ↑ * ▪▪▪ | ↑ * ▪▪▪▪▪ | Ostrowska et al. [40] | A | |||
| ↑ PBF [%] | ↑ * ▪▪▪ | ↑ * ▪▪▪▪ | Ostrowska et al. [40] | A | ||||
| ↑ VAT [cm3] | ↑ * ▪▪▪ | ↑ * ▪▪▪▪ | Ostrowska et al. [40] | A | ||||
| ↑ VAT/SAT ratio | ↑ * ▪▪▪▪ | ↑ * ▪▪▪▪ | Ostrowska et al. [40] | A | ||||
| ↑ FPG [mg/dl] | ↑ * ▪▪▪ | Chauhan et al. [30] | A | |||||
| ↑ PPBS [mg/dl] | ↓ * ▪▪▪ | Kalyani et al. [25] | A | |||||
| ↑ TG [mg/dl] | ↓ * ▪▪ | ↑ * ▪▪▪ | Chauhan et al. [30] | A | ||||
| ↓ HDL [mg/dl] | ↓ * ▪▪▪ | Chauhan et al. [30] | A | |||||
| Salivary TNF-α levels | ↓ * ▪▪ | Attlee et al. [23] | A | |||||
| ↑ * ▪▪▪ | Dogusal et al. [38] | Ch | ||||||
| The Levels of Adipokine/Cytokine in Saliva | Study Cited | Age Group | Differences between the Study Group and the Control Group | p-Value |
|---|---|---|---|---|
| Adipone-ctin | Kaluani et al. [25] | A | decreased adiponectin levels in the group with newly diagnosed type 2 diabetes as compared to group without diabetes; | 0.04 * |
| Goodson et al. [26] | Ch | decreased adiponectin levels among obese group without elevated CRP levels (relative to normal-weight group); | <0.0004 * | |
| Waspin (serpin A12) | Lehmann-Kalata et al. [28] | A | higher waspin levels among obese group without comorbidities compared to overweight and normal weight people; | 0.000004 * |
| IL-10 | Chauhan et al. [30] | A | decreased IL-10 levels in patients diagnosed with metabolic syndrome compared to controls; | >0.05 |
| Goodson et al. [26] | Ch | decreased IL-10 levels among obese group without elevated CRP levels (relative to normal-weight group); | <0.0004 * | |
| Selvaraju et al. [31] | Ch | 7.6-fold lower IL-10 concentration in overweight or obese group compared to normal weight group; | >0.05 | |
| CD40/CD40L | Lehmann et al. [32] | A | lower sCD40L concentrations in obese adults compared to normal weight subjects; | <0.001 * |
| Leptin | Jayachan-dran et al. [35] | A | higher leptin concentrations in overweight group; | <0.01 * |
| Goodson et al. [26] | Ch | elevated leptin levels in obese group with coexisting elevated serum CRP levels compared to normal weight group without elevated CRP; | <0.0004 * | |
| Pirsean et al. [19] | Ch | 3-fold higher leptin levels in overweight or obese group compared to normal weight group; | <0.01 * | |
| Resistin | Al.-Rawi et al. [37] | A | higher resistin levels in obese patients (both in the diabetic and non-diabetic groups) compared to those without diabetes and obesity; | 0.01 * |
| Selvaraju et al. [31] | Ch | 2.4-fold higher resistin levels in overweight and obese subjects compared to normal weight subjects; | <0.0001 * | |
| Lehmann-Kalata et al. [28] | A | higher resistin levels in subjects with BMI > 30 kg/m2 compared to subjects with BMI < 30 kg/m2; | 0.013 * | |
| TNF-α | Lehmann-Kalata et al. [28] | A | higher TNF-α levels and higher TNF-α-R1 levels in subjects with BMI > 30 kg/m2 compared to subjects with BMI < 30 kg/m2; | 0.002 * 0.0003 * |
| Lehmann et al. [32] | A | higher TNF-α receptors concentrations in obese subjects compared to normal weight subjects; | <0.001 * TNF-α-R1 0.02 * TNF-α-R2 | |
| Chauhan et al. [30] | A | higher TNF-α levels in patients with metabolic syndrome than in controls; increase in TNF-α levels with an increase in the number of components of the metabolic syndrome; | <0.05 * | |
| Selvaraju et al. [31] | Ch | 1.5-fold higher TNF-α levels in overweight and obese children compared to normal weight children; | <0.001 * | |
| IL-6 | Pirsean et al. [19] | Ch | 4.5-fold higher IL-6 levels in overweight or obese children compared to normal weight children; the highest IL-6 concentrations (98 ng/mL) in the entire study group in the child with the highest degree of obesity (>99th percentile); | <0.01 * |
| Selvaraju et al. [31] | Ch | 3.4-fold higher IL-6 levels in overweight and obese children compared to normal weight children; | <0.001 * | |
| IL-8 | Ostrowska et al. [40] | A | higher IL-8 levels among obese women compared to normal weight women; | 0.042 * |
| IL-1β | Tvarijona-viciute et al. [16] | Ch | 2.6-fold higher IL-1β levels in overweight/obese children compared to normal weight children; | 0.028 * |
| MCP-1/CCL2 | Lehmann-Kalata et al. [28] | A | higher MCP-1 concentrations in obese adults without comorbidities compared to overweight and normal weight subjects; | 0.032 * |
| Lehmann et al. [32] | A | higher MCP-1 concentrations in obese adults without comorbidities compared to normal weight subjects; | <0.001 * | |
| Selvaraju et al. [31] | Ch | 4.4-fold higher MCP-1 levels in overweight and obese subjects compared to normal weight subjects; | <0.0001 * | |
| MMP-2 | Ostrowska et al. [40] | A | higher MMP-2 levels among obese women compared to normal weight women; | 0.005 * |
| PAI-1 | Lehmann-Kalata et al. [28] | A | higher PAI-1 levels in people with a BMI > 30 kg/m2 and no comorbidities compared with those with BMI < 30 kg/m2; | 0.007 * |
| sICAM | Lehmann-Kalata et al. [28] | A | higher sICAM levels of obese subjects compared to overweight and normal weight subjects; | 0.007 * |
| Lehmannet al. [32] | A | higher sICAM levels of obese subjects compared to normal weight subjects; | <0.001 | |
| PTX3 | Lehmann-Kalata et al. [28] | A | higher PTX3 levels in obese subjects compared to overweight and normal weight subjects; | 0.021 * |
| Lehmannet al. [32] | A | higher PTX3 levels in obese subjects compared to normal weight subjects; | 0.006 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zyśk, B.; Ostrowska, L.; Smarkusz-Zarzecka, J. Salivary Adipokine and Cytokine Levels as Potential Markers for the Development of Obesity and Metabolic Disorders. Int. J. Mol. Sci. 2021, 22, 11703. https://doi.org/10.3390/ijms222111703
Zyśk B, Ostrowska L, Smarkusz-Zarzecka J. Salivary Adipokine and Cytokine Levels as Potential Markers for the Development of Obesity and Metabolic Disorders. International Journal of Molecular Sciences. 2021; 22(21):11703. https://doi.org/10.3390/ijms222111703
Chicago/Turabian StyleZyśk, Beata, Lucyna Ostrowska, and Joanna Smarkusz-Zarzecka. 2021. "Salivary Adipokine and Cytokine Levels as Potential Markers for the Development of Obesity and Metabolic Disorders" International Journal of Molecular Sciences 22, no. 21: 11703. https://doi.org/10.3390/ijms222111703






