The Beneficial Effects of Essential Oils in Anti-Obesity Treatment
Abstract
1. Introduction
2. Essential Oils (EOs)
- –
- Carvacrol (2-methyl-5-[1-methylethyl] phenol), which is the main product of numerous aromatic plants including Origanum, Thymus, Satureja, and Thymbra. Several studies show that carvacrol has antimicrobial, anti-fungal, anti-inflammatory, antioxidant, and antiproliferative activities [16].
- –
- Limonene, which is the main constituent of EOs extracted from Citrus, but is also present in the resin of conifers, particularly Pinaceae. It has anti-inflammatory, antioxidant, and anticancer properties. It is commonly used as a natural food flavoring [17].
- –
- Trans-anethole (trans-1-methoxy-4-propenyl-benzene), which is the main component of EOs extracted from more than 20 species including fennel, anise, and star anise. It can have anti-inflammatory, anticancer and antidiabetic effects. It is used as a natural food flavoring [18].
- –
- Cinnamaldehyde (trans-cinnamic aldehyde), which is known to be the main component of cinnamon flavor. Several studies have highlighted the anti-inflammatory activity of this aldehyde; moreover, it has the following properties: anti-infective (antibacterial, antifungal, antiviral), antiseptic, mucolytic and expectorant, analgesic, and anti-edematous. It is commonly used as a natural food flavoring [19].
3. Adipose Tissue and Obesity
3.1. Regulation of Lipogenesis and Lipolysis
3.2. Metabolic Syndrome (MS)
4. Anti-Obesity Effect of EOs
5. Effect of EOs on Metabolic Syndrome (MS) and Related Pathologies
6. Effect of EOs on Microbiota
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ACC | acetyl-CoenzymeA carboxylase |
| AgRP | agouti-related peptide |
| AMPK | AMP-activated protein kinase |
| AT | adipose tissue |
| BAT | brown adipose tissue |
| BBB | blood brain barrier |
| BMI | Body Mass Index |
| CEO | cumin essential oils |
| ChREBP | carbohydrate response element binding protein nuclear transcription factor |
| CNS | central nervous system |
| COE | citronella essential oils |
| CiEO | cinnamon essential oils |
| CpEO | Campomanesia phaea (O.Berg) Landru essential oils |
| CYP2E1 | cytochrome P450 2E1 |
| DNL | de novo lipogenesis |
| EGIR | european group for study of insulin resistance |
| EOO | oregano essential oils |
| EOs | essential oils |
| FAs | fatty acids |
| FAS | fatty acid synthase |
| FBS | fasting blood glucose |
| FFA | free fatty acids |
| GEO | garlic essential oils |
| GFO | grapefruit essential oil |
| GH | growth hormone |
| GiEO | ginger essential oils |
| HbA1c | glycosylated hemoglobin |
| HFD | high fat content |
| HMGCR | 3-hydroxy-3-methylglutaryl-coenzyme A reductase |
| HSL | hormone-sensitive lipase |
| IL- 6 | interleukin 6 |
| IRS 1–4 | insulin receptor substrate 1–4 |
| JNK | Janus N-terminal kinase |
| LDs | lipid droplets |
| LPL | lipoprotein lipase |
| LPS | lipopolysaccharides |
| LXR | liver X receptors |
| MAPK | mitogen-activated protein kinase |
| MOEO | melissa officinalis essential oils |
| MS | metabolic syndrome |
| NAFLD | non-alcoholic fatty liver disease |
| NEFA | non esterified fatty acids |
| NF-kB | nuclear factor kB |
| NPY | neuropeptide Y |
| PAI-1 | plasminogen activator inhibitor-1 |
| PEO | patchouli essential oils |
| PKA | cAMP-dependent protein kinase A |
| POMC | pro-opiomelanocortin |
| PPAR γ | receptor-γ activated by peroxisome proliferators |
| RaEo | Rhaponticum acaule essential oils |
| ROS | reactive oxygen species |
| SAT | subcutaneous adipose tissue |
| SCFA | short-chain fatty acids |
| SEO | salvia officinalis essential oils |
| SHR | spontaneously hypertensive rats |
| SIRT1 | sirtuin1 |
| SOD | superoxide dismutase |
| SOEO | sweet orange essential oils |
| SREBP | sterol regulatory element binding protein |
| TGs | triacylglycerols |
| TLR | toll-like receptor |
| TNFα | tumor necrosis factor α |
| TRP | transient receptor potential |
| TRPA1 | transient potential of the ankyrin receptor 1 |
| TRPM8 | transient potential cation channel subfamily M (melastatin) member 8 |
| TRPV1 | transient vanilloid receptor type 1 |
| TSH | thyroid-stimulating hormone |
| UCP-1 | uncoupling protein-1 |
| VAT | visceral fat adipocytes |
| WAT | white adipose tissue |
| WC | waist circumference |
| WHO | world health organization |
| WJMSC | Wharton’s gelatin derived mesenchymal stem cells |
| ZFR | Zucker fatty rats |
| ZMEO | Zataria multiflora essential oils |
| α-MSH | α-melanocyte stimulating hormone |
References
- Apovian, C.M. Obesity: Definition, Comorbidities, Causes, and Burden. Am. J. Manag. Care 2016, 22, s176–s185. [Google Scholar]
- Kaur, Y.; de Souza, R.J.; Gibson, W.T.; Meyre, D. A Systematic Review of Genetic Syndromes with Obesity. Obes. Rev. 2017, 18, 603–634. [Google Scholar] [CrossRef] [PubMed]
- Christensen, S. Recognizing Obesity as a Disease. J. Am. Assoc. Nurse Pract. 2020, 32, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Wright, S.M.; Aronne, L.J. Causes of Obesity. Abdom. Imaging 2012, 37, 730–732. [Google Scholar] [CrossRef] [PubMed]
- Seidell, J.C.; Halberstadt, J. The Global Burden of Obesity and the Challenges of Prevention. Ann. Nutr. Metab. 2015, 66, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Lim, H. The Global Childhood Obesity Epidemic and the Association between Socio-Economic Status and Childhood Obesity. Int. Rev. Psychiatry 2012, 24, 176–188. [Google Scholar] [CrossRef]
- Leherbauer, I.; Stappen, I. Selected Essential Oils and Their Mechanisms for Therapeutic Use against Public Health Disorders. An Overview. Z. Gastroenterol. 2020, 75, 205–223. [Google Scholar] [CrossRef]
- Rashed, A.; Nawi, N.; Sulaiman, K. Assessment of Essential Oil as a Potential Anti-Obesity Agent: A Narrative Review. J. Essent. Oil Res. 2016, 29, 1–10. [Google Scholar] [CrossRef]
- Villanueva-Millán, M.J.; Pérez-Matute, P.; Oteo, J.A. Gut Microbiota: A Key Player in Health and Disease. A Review Focused on Obesity. J. Physiol. Biochem. 2015, 71, 509–525. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.-J.; Li, S.; Gan, R.-Y.; Zhou, T.; Xu, D.-P.; Li, H.-B. Impacts of Gut Bacteria on Human Health and Diseases. Int. J. Mol. Sci. 2015, 16, 7493–7519. [Google Scholar] [CrossRef]
- Gérard, P. Gut Microbiota and Obesity. Cell. Mol. Life Sci. 2016, 73, 147–162. [Google Scholar] [CrossRef] [PubMed]
- Vigan, M. Essential Oils: Renewal of Interest and Toxicity. Eur. J. Dermatol. 2010, 20, 685–692. [Google Scholar] [CrossRef]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological Effects of Essential Oils—A Review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef] [PubMed]
- Aziz, Z.A.A.; Ahmad, A.; Setapar, S.H.M.; Karakucuk, A.; Azim, M.M.; Lokhat, D.; Rafatullah, M.; Ganash, M.; Kamal, M.A.; Ashraf, G.M. Essential Oils: Extraction Techniques, Pharmaceutical and Therapeutic Potential—A Review. Curr. Drug Metab. 2018, 19, 1100–1110. [Google Scholar] [CrossRef] [PubMed]
- Regnault-Roger, C.; Vincent, C.; Arnason, J.T. Essential Oils in Insect Control: Low-Risk Products in a High-Stakes World. Annu. Rev. Entomol. 2012, 57, 405–424. [Google Scholar] [CrossRef] [PubMed]
- Spalletta, S.; Flati, V.; Toniato, E.; di Gregorio, J.; Marino, A.; Pierdomenico, L.; Marchisio, M.; D’Orazi, G.; Cacciatore, I.; Robuffo, I. Carvacrol Reduces Adipogenic Differentiation by Modulating Autophagy and ChREBP Expression. PLoS ONE 2018, 13, e0206894. [Google Scholar] [CrossRef]
- Vieira, A.J.; Beserra, F.P.; Souza, M.C.; Totti, B.M.; Rozza, A.L. Limonene: Aroma of Innovation in Health and Disease. Chem. Biol. Interact. 2018, 283, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Kang, N.H.; Mukherjee, S.; Min, T.; Kang, S.C.; Yun, J.W. Trans-Anethole Ameliorates Obesity via Induction of Browning in White Adipocytes and Activation of Brown Adipocytes. Biochimie 2018, 151, 1–13. [Google Scholar] [CrossRef]
- Chung, J.; Kim, S.; Lee, H.A.; Park, M.H.; Kim, S.; Song, Y.R.; Na, H.S. Trans-Cinnamic Aldehyde Inhibits Aggregatibacter Actinomycetemcomitans-Induced Inflammation in THP-1–Derived Macrophages via Autophagy Activation. J. Periodontol. 2018, 89, 1262–1271. [Google Scholar] [CrossRef]
- Chiasson, H.; Bélanger, A.; Bostanian, N.; Vincent, C.; Poliquin, A. Acaricidal Properties of Artemisia Absinthium and Tanacetum Vulgare (Asteraceae) Essential Oils Obtained by Three Methods of Extraction. J. Econ. Entomol. 2001, 94, 167–171. [Google Scholar] [CrossRef]
- Park, A.; Kim, W.K.; Bae, K.-H. Distinction of White, Beige and Brown Adipocytes Derived from Mesenchymal Stem Cells. World J. Stem Cells 2014, 6, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Arner, P. Fat Tissue Growth and Development in Humans; Nestle Nutrition Institute Workshop Series; Nestlé Nutrition Institute: Basel, Switzerland, 2018; Volume 89, pp. 37–45. [Google Scholar] [CrossRef]
- Sun, K.; Kusminski, C.M.; Scherer, P.E. Adipose Tissue Remodeling and Obesity. J. Clin. Investig. 2011, 121, 2094–2101. [Google Scholar] [CrossRef] [PubMed]
- Wronska, A.; Kmiec, Z. Structural and Biochemical Characteristics of Various White Adipose Tissue Depots. Acta Physiol. 2012, 205, 194–208. [Google Scholar] [CrossRef]
- Heinonen, S.; Jokinen, R.; Rissanen, A.; Pietiläinen, K.H. White Adipose Tissue Mitochondrial Metabolism in Health and in Obesity. Obes. Rev. 2020, 21, e12958. [Google Scholar] [CrossRef] [PubMed]
- Walther, T.C.; Chung, J.; Farese, R.V. Lipid Droplet Biogenesis. Annu. Rev. Cell Dev. Biol. 2017, 33, 491–510. [Google Scholar] [CrossRef] [PubMed]
- Tansey, J.T.; Sztalryd, C.; Hlavin, E.M.; Kimmel, A.R.; Londos, C. The Central Role of Perilipin a in Lipid Metabolism and Adipocyte Lipolysis. IUBMB Life 2004, 56, 379–385. [Google Scholar] [CrossRef]
- Kershaw, E.E.; Flier, J.S. Adipose Tissue as an Endocrine Organ. J. Clin. Endocrinol. Metab. 2004, 89, 2548–2556. [Google Scholar] [CrossRef]
- Park, H.K.; Kwak, M.K.; Kim, H.J.; Ahima, R.S. Linking Resistin, Inflammation, and Cardiometabolic Diseases. Korean J. Intern. Med. 2017, 32, 239–247. [Google Scholar] [CrossRef]
- Balsan, G.A.; da Vieira, J.L.C.; de Oliveira, A.M.; Portal, V.L. Relationship between Adiponectin, Obesity and Insulin Resistance. Rev. Assoc. Med. Bras. 2015, 61, 72–80. [Google Scholar] [CrossRef]
- Wu, H.; Ballantyne, C.M. Skeletal Muscle Inflammation and Insulin Resistance in Obesity. J. Clin. Investig. 2017, 127, 43–54. [Google Scholar] [CrossRef]
- Patel, V.B.; Shah, S.; Verma, S.; Oudit, G.Y. Epicardial Adipose Tissue as a Metabolic Transducer: Role in Heart Failure and Coronary Artery Disease. Heart Fail. Rev. 2017, 22, 889–902. [Google Scholar] [CrossRef] [PubMed]
- Furuhashi, M.; Fuseya, T.; Murata, M.; Hoshina, K.; Ishimura, S.; Mita, T.; Watanabe, Y.; Omori, A.; Matsumoto, M.; Sugaya, T.; et al. Local Production of Fatty Acid-Binding Protein 4 in Epicardial/Perivascular Fat and Macrophages Is Linked to Coronary Atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2016, 36, 825–834. [Google Scholar] [CrossRef] [PubMed]
- Oelkrug, R.; Polymeropoulos, E.T.; Jastroch, M. Brown Adipose Tissue: Physiological Function and Evolutionary Significance. J. Comp. Physiol. B 2015, 185, 587–606. [Google Scholar] [CrossRef] [PubMed]
- Fenzl, A.; Kiefer, F.W. Brown Adipose Tissue and Thermogenesis. Horm. Mol. Biol. Clin. Investig. 2014, 19, 25–37. [Google Scholar] [CrossRef]
- Chouchani, E.T.; Kazak, L.; Spiegelman, B.M. New Advances in Adaptive Thermogenesis: UCP1 and Beyond. Cell. Metab. 2019, 29, 27–37. [Google Scholar] [CrossRef]
- Desjardins, E.M.; Steinberg, G.R. Emerging Role of AMPK in Brown and Beige Adipose Tissue (BAT): Implications for Obesity, Insulin Resistance, and Type 2 Diabetes. Curr. Diab. Rep. 2018, 18, 80. [Google Scholar] [CrossRef] [PubMed]
- Giralt, M.; Villarroya, F. White, Brown, Beige/Brite: Different Adipose Cells for Different Functions? Endocrinology 2013, 154, 2992–3000. [Google Scholar] [CrossRef]
- Wang, W.; Seale, P. Control of Brown and Beige Fat Development. Nat. Rev. Mol. Cell Biol. 2016, 17, 691–702. [Google Scholar] [CrossRef]
- Wu, J.; Boström, P.; Sparks, L.M.; Ye, L.; Choi, J.H.; Giang, A.-H.; Khandekar, M.; Virtanen, K.A.; Nuutila, P.; Schaart, G.; et al. Beige Adipocytes Are a Distinct Type of Thermogenic Fat Cell in Mouse and Human. Cell 2012, 150, 366–376. [Google Scholar] [CrossRef]
- Ikeda, K.; Maretich, P.; Kajimura, S. The Common and Distinct Features of Brown and Beige Adipocytes. Trends Endocrinol. Metab. 2018, 29, 191–200. [Google Scholar] [CrossRef]
- Cammisotto, P.G.; Levy, E.; Bukowiecki, L.J.; Bendayan, M. Cross-Talk between Adipose and Gastric Leptins for the Control of Food Intake and Energy Metabolism. Prog. Histochem. Cytochem. 2010, 45, 143–200. [Google Scholar] [CrossRef] [PubMed]
- Klok, M.D.; Jakobsdottir, S.; Drent, M.L. The Role of Leptin and Ghrelin in the Regulation of Food Intake and Body Weight in Humans: A Review. Obes. Rev. 2007, 8, 21–34. [Google Scholar] [CrossRef] [PubMed]
- Timper, K.; Brüning, J.C. Hypothalamic Circuits Regulating Appetite and Energy Homeostasis: Pathways to Obesity. Dis. Model. Mech. 2017, 10, 679–689. [Google Scholar] [CrossRef] [PubMed]
- Zhan, C. POMC Neurons: Feeding, Energy Metabolism, and Beyond. Adv. Exp. Med. Biol. 2018, 1090, 17–29. [Google Scholar] [CrossRef]
- Hynes, G.R.; Jones, P.J. Leptin and Its Role in Lipid Metabolism. Curr. Opin. Lipidol. 2001, 12, 321–327. [Google Scholar] [CrossRef]
- Friedman, J.M. Leptin and the Endocrine Control of Energy Balance. Nat. Metab. 2019, 1, 754–764. [Google Scholar] [CrossRef] [PubMed]
- Wada, N.; Hirako, S.; Takenoya, F.; Kageyama, H.; Okabe, M.; Shioda, S. Leptin and Its Receptors. J. Chem. Neuroanat. 2014, 61–62, 191–199. [Google Scholar] [CrossRef]
- Harrison, L.; Schriever, S.C.; Feuchtinger, A.; Kyriakou, E.; Baumann, P.; Pfuhlmann, K.; Messias, A.C.; Walch, A.; Tschöp, M.H.; Pfluger, P.T. Fluorescent Blood-Brain Barrier Tracing Shows Intact Leptin Transport in Obese Mice. Int. J. Obes. 2019, 43, 1305–1318. [Google Scholar] [CrossRef]
- Zieba, D.A.; Biernat, W.; Barć, J. Roles of Leptin and Resistin in Metabolism, Reproduction, and Leptin Resistance. Domest. Anim. Endocrinol. 2020, 73, 106472. [Google Scholar] [CrossRef]
- Ozcan, L.; Ergin, A.S.; Lu, A.; Chung, J.; Sarkar, S.; Nie, D.; Myers, M.G.; Ozcan, U. Endoplasmic Reticulum Stress Plays a Central Role in Development of Leptin Resistance. Cell. Metab. 2009, 9, 35–51. [Google Scholar] [CrossRef]
- Gruzdeva, O.; Borodkina, D.; Uchasova, E.; Dyleva, Y.; Barbarash, O. Leptin Resistance: Underlying Mechanisms and Diagnosis. Diabetes Metab. Syndr. Obes. 2019, 12, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Myers, M.G.; Cowley, M.A.; Münzberg, H. Mechanisms of Leptin Action and Leptin Resistance. Annu. Rev. Physiol. 2008, 70, 537–556. [Google Scholar] [CrossRef] [PubMed]
- Al Massadi, O.; Nogueiras, R.; Dieguez, C.; Girault, J.-A. Ghrelin and Food Reward. Neuropharmacology 2019, 148, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Makris, M.C.; Alexandrou, A.; Papatsoutsos, E.G.; Malietzis, G.; Tsilimigras, D.I.; Guerron, A.D.; Moris, D. Ghrelin and Obesity: Identifying Gaps and Dispelling Myths. A Reappraisal. Vivo 2017, 31, 1047–1050. [Google Scholar] [CrossRef]
- Parker, J.A.; Bloom, S.R. Hypothalamic Neuropeptides and the Regulation of Appetite. Neuropharmacology 2012, 63, 18–30. [Google Scholar] [CrossRef]
- Duncan, R.E.; Ahmadian, M.; Jaworski, K.; Sarkadi-Nagy, E.; Sul, H.S. Regulation of Lipolysis in Adipocytes. Annu. Rev. Nutr. 2007, 27, 79–101. [Google Scholar] [CrossRef]
- McMurray, R.G.; Hackney, A.C. Interactions of Metabolic Hormones, Adipose Tissue and Exercise. Sports Med. 2005, 35, 393–412. [Google Scholar] [CrossRef]
- Yang, A.; Mottillo, E.P. Adipocyte Lipolysis: From Molecular Mechanisms of Regulation to Disease and Therapeutics. Biochem. J. 2020, 477, 985–1008. [Google Scholar] [CrossRef]
- Engin, A.B. What Is Lipotoxicity? Adv. Exp. Med. Biol. 2017, 960, 197–220. [Google Scholar] [CrossRef] [PubMed]
- Carmen, G.-Y.; Víctor, S.-M. Signalling Mechanisms Regulating Lipolysis. Cell. Signal. 2006, 18, 401–408. [Google Scholar] [CrossRef]
- Wang, Q.; Liu, S.; Zhai, A.; Zhang, B.; Tian, G. AMPK-Mediated Regulation of Lipid Metabolism by Phosphorylation. Biol. Pharm. Bull. 2018, 41, 985–993. [Google Scholar] [CrossRef]
- Yin, C.; Liu, W.H.; Liu, Y.; Wang, L.; Xiao, Y. PID1 Alters the Antilipolytic Action of Insulin and Increases Lipolysis via Inhibition of AKT/PKA Pathway Activation. PLoS ONE 2019, 14, e0214606. [Google Scholar] [CrossRef]
- Yang, Q.; Vijayakumar, A.; Kahn, B.B. Metabolites as Regulators of Insulin Sensitivity and Metabolism. Nat. Rev. Mol. Cell Biol. 2018, 19, 654–672. [Google Scholar] [CrossRef] [PubMed]
- Song, Z.; Xiaoli, A.M.; Yang, F. Regulation and Metabolic Significance of De Novo Lipogenesis in Adipose Tissues. Nutrients 2018, 10, 1383. [Google Scholar] [CrossRef]
- Smith, U.; Kahn, B.B. Adipose Tissue Regulates Insulin Sensitivity: Role of Adipogenesis, de Novo Lipogenesis and Novel Lipids. J. Intern. Med. 2016, 280, 465–475. [Google Scholar] [CrossRef] [PubMed]
- Vijayakumar, A.; Aryal, P.; Wen, J.; Syed, I.; Vazirani, R.P.; Moraes-Vieira, P.M.; Camporez, J.P.; Gallop, M.R.; Perry, R.J.; Peroni, O.D.; et al. Absence of Carbohydrate Response Element Binding Protein in Adipocytes Causes Systemic Insulin Resistance and Impairs Glucose Transport. Cell Rep. 2017, 21, 1021–1035. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Gurmaches, J.; Tang, Y.; Jespersen, N.Z.; Wallace, M.; Martinez Calejman, C.; Gujja, S.; Li, H.; Edwards, Y.J.K.; Wolfrum, C.; Metallo, C.M.; et al. Brown Fat AKT2 Is a Cold-Induced Kinase That Stimulates ChREBP-Mediated De Novo Lipogenesis to Optimize Fuel Storage and Thermogenesis. Cell. Metab. 2018, 27, 195–209.e6. [Google Scholar] [CrossRef]
- Sae-Lee, C.; Moolsuwan, K.; Chan, L.; Poungvarin, N. ChREBP Regulates Itself and Metabolic Genes Implicated in Lipid Accumulation in β-Cell Line. PLoS ONE 2016, 11, e0147411. [Google Scholar] [CrossRef]
- Cornier, M.-A.; Dabelea, D.; Hernandez, T.L.; Lindstrom, R.C.; Steig, A.J.; Stob, N.R.; Van Pelt, R.E.; Wang, H.; Eckel, R.H. The Metabolic Syndrome. Endocr. Rev. 2008, 29, 777–822. [Google Scholar] [CrossRef]
- Engin, A. The Definition and Prevalence of Obesity and Metabolic Syndrome. Adv. Exp. Med. Biol. 2017, 960, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Sáez-Lara, M.J.; Robles-Sanchez, C.; Ruiz-Ojeda, F.J.; Plaza-Diaz, J.; Gil, A. Effects of Probiotics and Synbiotics on Obesity, Insulin Resistance Syndrome, Type 2 Diabetes and Non-Alcoholic Fatty Liver Disease: A Review of Human Clinical Trials. Int. J. Mol. Sci. 2016, 17, 928. [Google Scholar] [CrossRef] [PubMed]
- Saklayen, M.G. The Global Epidemic of the Metabolic Syndrome. Curr. Hypertens. Rep. 2018, 20, 12. [Google Scholar] [CrossRef]
- Bays, H.E.; González-Campoy, J.M.; Bray, G.A.; Kitabchi, A.E.; Bergman, D.A.; Schorr, A.B.; Rodbard, H.W.; Henry, R.R. Pathogenic Potential of Adipose Tissue and Metabolic Consequences of Adipocyte Hypertrophy and Increased Visceral Adiposity. Expert Rev. Cardiovasc. Ther. 2008, 6, 343–368. [Google Scholar] [CrossRef]
- Lafontan, M.; Langin, D. Lipolysis and Lipid Mobilization in Human Adipose Tissue. Prog. Lipid. Res. 2009, 48, 275–297. [Google Scholar] [CrossRef] [PubMed]
- Hellmér, J.; Marcus, C.; Sonnenfeld, T.; Arner, P. Mechanisms for Differences in Lipolysis between Human Subcutaneous and Omental Fat Cells. J. Clin. Endocrinol. Metab. 1992, 75, 15–20. [Google Scholar] [CrossRef] [PubMed]
- Ciccarelli, M.; Santulli, G.; Pascale, V.; Trimarco, B.; Iaccarino, G. Adrenergic Receptors and Metabolism: Role in Development of Cardiovascular Disease. Front. Physiol. 2013, 4, 265. [Google Scholar] [CrossRef] [PubMed]
- He, P.; Hou, B.; Li, Y.; Xu, C.; Ma, P.; Lam, S.M.; Gil, V.; Yang, X.; Yang, X.; Zhang, L.; et al. Lipid Profiling Reveals Browning Heterogeneity of White Adipose Tissue by Β3-Adrenergic Stimulation. Biomolecules 2019, 9, 444. [Google Scholar] [CrossRef]
- Boucher, J.; Kleinridders, A.; Kahn, C.R. Insulin Receptor Signaling in Normal and Insulin-Resistant States. Cold Spring Harb. Perspect. Biol. 2014, 6, a009191. [Google Scholar] [CrossRef]
- Shaw, L.M. The Insulin Receptor Substrate (IRS) Proteins: At the Intersection of Metabolism and Cancer. Cell Cycle 2011, 10, 1750–1756. [Google Scholar] [CrossRef]
- Capurso, C.; Capurso, A. From Excess Adiposity to Insulin Resistance: The Role of Free Fatty Acids. Vascul. Pharmacol. 2012, 57, 91–97. [Google Scholar] [CrossRef]
- Gao, Z.; Zhang, X.; Zuberi, A.; Hwang, D.; Quon, M.J.; Lefevre, M.; Ye, J. Inhibition of Insulin Sensitivity by Free Fatty Acids Requires Activation of Multiple Serine Kinases in 3T3-L1 Adipocytes. Mol. Endocrinol. 2004, 18, 2024–2034. [Google Scholar] [CrossRef] [PubMed]
- Mraz, M.; Lacinova, Z.; Drapalova, J.; Haluzikova, D.; Horinek, A.; Matoulek, M.; Trachta, P.; Kavalkova, P.; Svacina, S.; Haluzik, M. The Effect of Very-Low-Calorie Diet on MRNA Expression of Inflammation-Related Genes in Subcutaneous Adipose Tissue and Peripheral Monocytes of Obese Patients with Type 2 Diabetes Mellitus. J. Clin. Endocrinol. Metab. 2011, 96, E606–E613. [Google Scholar] [CrossRef]
- Donath, M.Y.; Shoelson, S.E. Type 2 Diabetes as an Inflammatory Disease. Nat. Rev. Immunol. 2011, 11, 98–107. [Google Scholar] [CrossRef] [PubMed]
- Tilg, H.; Moschen, A.R. Inflammatory Mechanisms in the Regulation of Insulin Resistance. Mol. Med. 2008, 14, 222–231. [Google Scholar] [CrossRef]
- Baker, R.G.; Hayden, M.S.; Ghosh, S. NF-ΚB, Inflammation, and Metabolic Disease. Cell. Metab. 2011, 13, 11–22. [Google Scholar] [CrossRef]
- Hur, M.; Kim, C.; Kim, C.-H.; Ahn, H.-C.; Ahn, H.-Y. The effects of inhalation of essential oils on the body weight, food efficiency rate and serum leptin of growing SD rats. Taehan Kanho Hakhoe Chi 2006, 36, 236–243. [Google Scholar] [CrossRef]
- Shen, J.; Niijima, A.; Tanida, M.; Horii, Y.; Maeda, K.; Nagai, K. Olfactory Stimulation with Scent of Grapefruit Oil Affects Autonomic Nerves, Lipolysis and Appetite in Rats. Neurosci. Lett. 2005, 380, 289–294. [Google Scholar] [CrossRef]
- Schwartz, M.W.; Woods, S.C.; Porte, D.; Seeley, R.J.; Baskin, D.G. Central Nervous System Control of Food Intake. Nature 2000, 404, 661–671. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, T.; Inui, T.; Tsuji, T. The Odor of Osmanthus Fragrans Attenuates Food Intake. Sci. Rep. 2013, 3, 1518. [Google Scholar] [CrossRef] [PubMed]
- Belgardt, B.F.; Brüning, J.C. CNS Leptin and Insulin Action in the Control of Energy Homeostasis. Ann. N. Y. Acad. Sci. 2010, 1212, 97–113. [Google Scholar] [CrossRef] [PubMed]
- Nagai, K.; Niijima, A.; Horii, Y.; Shen, J.; Tanida, M. Olfactory Stimulatory with Grapefruit and Lavender Oils Change Autonomic Nerve Activity and Physiological Function. Auton. Neurosci. 2014, 185, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Niijima, A.; Nagai, K. Effect of Olfactory Stimulation with Flavor of Grapefruit Oil and Lemon Oil on the Activity of Sympathetic Branch in the White Adipose Tissue of the Epididymis. Exp. Biol. Med. 2003, 228, 1190–1192. [Google Scholar] [CrossRef] [PubMed]
- Provensi, G.; Coccurello, R.; Umehara, H.; Munari, L.; Giacovazzo, G.; Galeotti, N.; Nosi, D.; Gaetani, S.; Romano, A.; Moles, A.; et al. Satiety Factor Oleoylethanolamide Recruits the Brain Histaminergic System to Inhibit Food Intake. Proc. Natl. Acad. Sci. USA 2014, 111, 11527–11532. [Google Scholar] [CrossRef] [PubMed]
- Musilli, C.; de Siena, G.; Manni, M.E.; Logli, A.; Landucci, E.; Zucchi, R.; Saba, A.; Donzelli, R.; Passani, M.B.; Provensi, G.; et al. Histamine Mediates Behavioural and Metabolic Effects of 3-Iodothyroacetic Acid, an Endogenous End Product of Thyroid Hormone Metabolism. Br. J. Pharmacol. 2014, 171, 3476–3484. [Google Scholar] [CrossRef]
- Batubara, I.; Suparto, I.H.; Sa’diah, S.; Matsuoka, R.; Mitsunaga, T. Effects of Inhaled Citronella Oil and Related Compounds on Rat Body Weight and Brown Adipose Tissue Sympathetic Nerve. Nutrients 2015, 7, 1859–1870. [Google Scholar] [CrossRef]
- Swamy, M.K.; Sinniah, U.R. A Comprehensive Review on the Phytochemical Constituents and Pharmacological Activities of Pogostemon Cablin Benth.: An Aromatic Medicinal Plant of Industrial Importance. Molecules 2015, 20, 8521–8547. [Google Scholar] [CrossRef]
- Zhang, R.; Yan, P.; Li, Y.; Xiong, L.; Gong, X.; Peng, C. A Pharmacokinetic Study of Patchouli Alcohol after a Single Oral Administration of Patchouli Alcohol or Patchouli Oil in Rats. Eur. J. Drug Metab. Pharmacokinet. 2016, 41, 441–448. [Google Scholar] [CrossRef]
- Hong, S.J.; Cho, J.; Boo, C.G.; Youn, M.Y.; Pan, J.H.; Kim, J.K.; Shin, E.-C. Inhalation of Patchouli (Pogostemon Cablin Benth.) Essential Oil Improved Metabolic Parameters in Obesity-Induced Sprague Dawley Rats. Nutrients 2020, 12, 2077. [Google Scholar] [CrossRef]
- Leyva-López, N.; Gutiérrez-Grijalva, E.P.; Vazquez-Olivo, G.; Heredia, J.B. Essential Oils of Oregano: Biological Activity beyond Their Antimicrobial Properties. Molecules 2017, 22, 989. [Google Scholar] [CrossRef] [PubMed]
- Diniz do Nascimento, L.; de Moraes, A.A.B.; da Costa, K.S.; Pereira Galúcio, J.M.; Taube, P.S.; Costa, C.M.L.; Neves Cruz, J.; de Aguiar Andrade, E.H.; de Faria, L.J.G. Bioactive Natural Compounds and Antioxidant Activity of Essential Oils from Spice Plants: New Findings and Potential Applications. Biomolecules 2020, 10, 988. [Google Scholar] [CrossRef]
- Moghrovyan, A.; Sahakyan, N.; Babayan, A.; Chichoyan, N.; Petrosyan, M.; Trchounian, A. Essential Oil and Ethanol Extract of Oregano (Origanum Vulgare L.) from Armenian Flora as a Natural Source of Terpenes, Flavonoids and Other Phytochemicals with Antiradical, Antioxidant, Metal Chelating, Tyrosinase Inhibitory and Antibacterial Activity. Curr. Pharm. Des. 2019, 25, 1809–1816. [Google Scholar] [CrossRef]
- Cho, S.; Choi, Y.; Park, S.; Park, T. Carvacrol Prevents Diet-Induced Obesity by Modulating Gene Expressions Involved in Adipogenesis and Inflammation in Mice Fed with High-Fat Diet. J. Nutr. Biochem. 2012, 23, 192–201. [Google Scholar] [CrossRef] [PubMed]
- Balusamy, S.R.; Perumalsamy, H.; Huq, M.A.; Balasubramanian, B. Anti-Proliferative Activity of Origanum Vulgare Inhibited Lipogenesis and Induced Mitochondrial Mediated Apoptosis in Human Stomach Cancer Cell Lines. Biomed. Pharmacother. 2018, 108, 1835–1844. [Google Scholar] [CrossRef]
- Li, D.; Wu, H.; Dou, H. Weight Loss Effect of Sweet Orange Essential Oil Microcapsules on Obese SD Rats Induced by High-Fat Diet. Biosci. Biotechnol. Biochem. 2019, 83, 923–932. [Google Scholar] [CrossRef]
- Jing, L.; Zhang, Y.; Fan, S.; Gu, M.; Guan, Y.; Lu, X.; Huang, C.; Zhou, Z. Preventive and Ameliorating Effects of Citrus D-Limonene on Dyslipidemia and Hyperglycemia in Mice with High-Fat Diet-Induced Obesity. Eur. J. Pharmacol. 2013, 715, 46–55. [Google Scholar] [CrossRef]
- Derbenev, A.V.; Zsombok, A. Potential Therapeutic Value of TRPV1 and TRPA1 in Diabetes Mellitus and Obesity. Semin. Immunopathol. 2016, 38, 397–406. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.; Zhai, M.; Yan, D.; Li, D.; Li, C.; Zhang, Y.; Xiao, L.; Xiong, D.; Deng, Q.; Sun, W. Dietary Menthol-Induced TRPM8 Activation Enhances WAT “Browning” and Ameliorates Diet-Induced Obesity. Oncotarget 2017, 8, 75114–75126. [Google Scholar] [CrossRef]
- Ali-Shtayeh, M.S.; Jamous, R.M.; Abu-Zaitoun, S.Y.; Khasati, A.I.; Kalbouneh, S.R. Biological Properties and Bioactive Components of Mentha Spicata L. Essential Oil: Focus on Potential Benefits in the Treatment of Obesity, Alzheimer’s Disease, Dermatophytosis, and Drug-Resistant Infections. Evid.-Based Complement. Alternat. Med. 2019, 2019, 3834265. [Google Scholar] [CrossRef] [PubMed]
- Terada, Y.; Yamashita, R.; Ihara, N.; Yamazaki-Ito, T.; Takahashi, Y.; Masuda, H.; Sakuragawa, S.; Ito, S.; Ito, K.; Watanabe, T. Human TRPA1 Activation by Terpenes Derived from the Essential Oil of Daidai, Citrus Aurantium L. Var. Daidai Makino. Biosci. Biotechnol. Biochem. 2019, 83, 1721–1728. [Google Scholar] [CrossRef] [PubMed]
- Legrand, C.; Merlini, J.M.; de Senarclens-Bezençon, C.; Michlig, S. New Natural Agonists of the Transient Receptor Potential Ankyrin 1 (TRPA1) Channel. Sci. Rep. 2020, 10, 11238. [Google Scholar] [CrossRef] [PubMed]
- Camacho, S.; Michlig, S.; de Senarclens-Bezençon, C.; Meylan, J.; Meystre, J.; Pezzoli, M.; Markram, H.; le Coutre, J. Anti-Obesity and Anti-Hyperglycemic Effects of Cinnamaldehyde via Altered Ghrelin Secretion and Functional Impact on Food Intake and Gastric Emptying. Sci. Rep. 2015, 5, 7919. [Google Scholar] [CrossRef] [PubMed]
- Cheng, B.-H.; Sheen, L.-Y.; Chang, S.-T. Hypolipidemic Effects of S-(+)-Linalool and Essential Oil from Cinnamomum Osmophloeum Ct. Linalool Leaves in Mice. J. Tradit. Complement. Med. 2018, 8, 46–52. [Google Scholar] [CrossRef]
- Kaur, N.; Chugh, H.; Tomar, V.; Sakharkar, M.K.; Dass, S.K.; Chandra, R. Cinnamon Attenuates Adiposity and Affects the Expression of Metabolic Genes in Diet-Induced Obesity Model of Zebrafish. Artif. Cells Nanomed. Biotechnol. 2019, 47, 2930–2939. [Google Scholar] [CrossRef]
- Tan, C.Y.; Ishikawa, K.; Virtue, S.; Vidal-Puig, A. Brown Adipose Tissue in the Treatment of Obesity and Diabetes: Are We Hot Enough? J. Diabetes Investig. 2011, 2, 341–350. [Google Scholar] [CrossRef] [PubMed]
- Claussnitzer, M.; Dankel, S.N.; Kim, K.-H.; Quon, G.; Meuleman, W.; Haugen, C.; Glunk, V.; Sousa, I.S.; Beaudry, J.L.; Puviindran, V.; et al. FTO Obesity Variant Circuitry and Adipocyte Browning in Humans. N. Engl. J. Med. 2015, 373, 895–907. [Google Scholar] [CrossRef]
- Ricquier, D. Uncoupling Protein 1 of Brown Adipocytes, the Only Uncoupler: A Historical Perspective. Front. Endocrinol (Lausanne) 2011, 2, 85. [Google Scholar] [CrossRef]
- Kaur, I.P.; Deol, P.K.; Kondepudi, K.K.; Bishnoi, M. Anticancer Potential of Ginger: Mechanistic and Pharmaceutical Aspects. Curr. Pharm. Des. 2016, 22, 4160–4172. [Google Scholar] [CrossRef]
- Wang, J.; Ke, W.; Bao, R.; Hu, X.; Chen, F. Beneficial Effects of Ginger Zingiber Officinale Roscoe on Obesity and Metabolic Syndrome: A Review. Ann. N. Y. Acad. Sci. 2017, 1398, 83–98. [Google Scholar] [CrossRef] [PubMed]
- Emílio-Silva, M.T.; Rodrigues, V.P.; Bueno, G.; Ohara, R.; Martins, M.G.; Horta-Júnior, J.A.C.; Branco, L.G.S.; Rocha, L.R.M.; Hiruma-Lima, C.A. Hypothermic Effect of Acute Citral Treatment during LPS-Induced Systemic Inflammation in Obese Mice: Reduction of Serum TNF-α and Leptin Levels. Biomolecules 2020, 10, 1454. [Google Scholar] [CrossRef] [PubMed]
- Lai, Y.-S.; Lee, W.-C.; Lin, Y.-E.; Ho, C.-T.; Lu, K.-H.; Lin, S.-H.; Panyod, S.; Chu, Y.-L.; Sheen, L.-Y. Ginger Essential Oil Ameliorates Hepatic Injury and Lipid Accumulation in High Fat Diet-Induced Nonalcoholic Fatty Liver Disease. J. Agric. Food Chem. 2016, 64, 2062–2071. [Google Scholar] [CrossRef]
- Belhadj, S.; Hentati, O.; Hammami, M.; Ben Hadj, A.; Boudawara, T.; Dammak, M.; Zouari, S.; El Feki, A. Metabolic Impairments and Tissue Disorders in Alloxan-Induced Diabetic Rats Are Alleviated by Salvia officinalis L. Essential Oil. Biomed. Pharmacother. 2018, 108, 985–995. [Google Scholar] [CrossRef] [PubMed]
- Behradmanesh, S.; Derees, F.; Rafieian-Kopaei, M. Effect of Salvia officinalis on Diabetic Patients. J. Renal. Inj. Prev. 2013, 2, 51–54. [Google Scholar] [CrossRef] [PubMed]
- Lima, C.F.; Azevedo, M.F.; Araujo, R.; Fernandes-Ferreira, M.; Pereira-Wilson, C. Metformin-like Effect of Salvia officinalis (Common Sage): Is It Useful in Diabetes Prevention? Br. J. Nutr. 2006, 96, 326–333. [Google Scholar] [CrossRef] [PubMed]
- Luca, T.; Napoli, E.; Privitera, G.; Musso, N.; Ruberto, G.; Castorina, S. Antiproliferative Effect and Cell Cycle Alterations Induced by Salvia officinalis Essential Oil and Its Three Main Components in Human Colon Cancer Cell Lines. Chem. Biodivers. 2020, 17, e2000309. [Google Scholar] [CrossRef] [PubMed]
- Russo, A.; Formisano, C.; Rigano, D.; Senatore, F.; Delfine, S.; Cardile, V.; Rosselli, S.; Bruno, M. Chemical Composition and Anticancer Activity of Essential Oils of Mediterranean Sage (Salvia Officinalis L.) Grown in Different Environmental Conditions. Food Chem. Toxicol. 2013, 55, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Koubaa, F.G.; Chaâbane, M.; Turki, M.; Ayadi, F.M.; El Feki, A. Anti-Oxidant and Hepatoprotective Effects of Salvia officinalis Essential Oil against Vanadium-Induced Oxidative Stress and Histological Changes in the Rat Liver. Environ. Sci. Pollut. Res. Int. 2021, 28, 11001–11015. [Google Scholar] [CrossRef]
- Koubaa-Ghorbel, F.; Chaâbane, M.; Turki, M.; Makni-Ayadi, F.; El Feki, A. The Protective Effects of Salvia officinalis Essential Oil Compared to Simvastatin against Hyperlipidemia, Liver, and Kidney Injuries in Mice Submitted to a High-Fat Diet. J. Food Biochem. 2020, 44, e13160. [Google Scholar] [CrossRef]
- Jafari, S.; Sattari, R.; Ghavamzadeh, S. Evaluation the Effect of 50 and 100 Mg Doses of Cuminum Cyminum Essential Oil on Glycemic Indices, Insulin Resistance and Serum Inflammatory Factors on Patients with Diabetes Type II: A Double-Blind Randomized Placebo-Controlled Clinical Trial. J. Tradit. Complement. Med. 2017, 7, 332–338. [Google Scholar] [CrossRef]
- Jafari, T.; Mahmoodnia, L.; Tahmasebi, P.; Memarzadeh, M.R.; Sedehi, M.; Beigi, M.; Fallah, A.A. Effect of Cumin (Cuminum Cyminum) Essential Oil Supplementation on Metabolic Profile and Serum Leptin in Pre-Diabetic Subjects: A Randomized Double-Blind Placebo-Controlled Clinical Trial. J. Funct. Foods 2018, 47, 416–422. [Google Scholar] [CrossRef]
- Taghizadeh, M.; Memarzadeh, M.R.; Asemi, Z.; Esmaillzadeh, A. Effect of the Cumin cyminum L. Intake on Weight Loss, Metabolic Profiles and Biomarkers of Oxidative Stress in Overweight Subjects: A Randomized Double-Blind Placebo-Controlled Clinical Trial. Ann. Nutr. Metab. 2015, 66, 117–124. [Google Scholar] [CrossRef]
- Kalaivani, P.; Saranya, R.B.; Ramakrishnan, G.; Ranju, V.; Sathiya, S.; Gayathri, V.; Thiyagarajan, L.K.; Venkhatesh, J.R.; Babu, C.S.; Thanikachalam, S. Cuminum Cyminum, a Dietary Spice, Attenuates Hypertension via Endothelial Nitric Oxide Synthase and NO Pathway in Renovascular Hypertensive Rats. Clin. Exp. Hypertens 2013, 35, 534–542. [Google Scholar] [CrossRef]
- Hadi, A.; Mohammadi, H.; Hadi, Z.; Roshanravan, N.; Kafeshani, M. Cumin (Cuminum Cyminum L.) Is a Safe Approach for Management of Lipid Parameters: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Phytother. Res. 2018, 32, 2146–2154. [Google Scholar] [CrossRef]
- Mosbah, H.; Chahdoura, H.; Kammoun, J.; Hlila, M.B.; Louati, H.; Hammami, S.; Flamini, G.; Achour, L.; Selmi, B. Rhaponticum Acaule (L) DC Essential Oil: Chemical Composition, in Vitro Antioxidant and Enzyme Inhibition Properties. BMC Complement. Altern. Med. 2018, 18, 79. [Google Scholar] [CrossRef] [PubMed]
- Chan, D.C.; Barrett, P.H.R.; Watts, G.F. The Metabolic and Pharmacologic Bases for Treating Atherogenic Dyslipidaemia. Best Pract. Res. Clin. Endocrinol. Metab. 2014, 28, 369–385. [Google Scholar] [CrossRef] [PubMed]
- Jun, H.-J.; Lee, J.H.; Jia, Y.; Hoang, M.-H.; Byun, H.; Kim, K.H.; Lee, S.-J. Melissa Officinalis Essential Oil Reduces Plasma Triglycerides in Human Apolipoprotein E2 Transgenic Mice by Inhibiting Sterol Regulatory Element-Binding Protein-1c-Dependent Fatty Acid Synthesis. J. Nutr. 2012, 142, 432–440. [Google Scholar] [CrossRef][Green Version]
- Yen, H.-F.; Hsieh, C.-T.; Hsieh, T.-J.; Chang, F.-R.; Wang, C.-K. In Vitro Anti-Diabetic Effect and Chemical Component Analysis of 29 Essential Oils Products. J. Food Drug Anal. 2015, 23, 124–129. [Google Scholar] [CrossRef]
- Talpur, N.; Echard, B.; Ingram, C.; Bagchi, D.; Preuss, H. Effects of a Novel Formulation of Essential Oils on Glucose-Insulin Metabolism in Diabetic and Hypertensive Rats: A Pilot Study. Diabetes Obes. Metab. 2005, 7, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Broadhurst, C.L.; Polansky, M.M.; Anderson, R.A. Insulin-like Biological Activity of Culinary and Medicinal Plant Aqueous Extracts in Vitro. J. Agric. Food Chem. 2000, 48, 849–852. [Google Scholar] [CrossRef]
- Puri, D. Therapeutic Potentials of Fenugreek. Indian J. Physiol. Pharmacol. 1998, 42, 423–424. [Google Scholar] [PubMed]
- Puri, D. Fenugreek in Diabetes Mellitus. J. Assoc. Physicians India 1999, 47, 255–256. [Google Scholar] [PubMed]
- Imparl-Radosevich, J.; Deas, S.; Polansky, M.M.; Baedke, D.A.; Ingebritsen, T.S.; Anderson, R.A.; Graves, D.J. Regulation of PTP-1 and Insulin Receptor Kinase by Fractions from Cinnamon: Implications for Cinnamon Regulation of Insulin Signalling. Horm. Res. 1998, 50, 177–182. [Google Scholar] [CrossRef]
- Lefranc, C.; Friederich-Persson, M.; Palacios-Ramirez, R.; Nguyen Dinh Cat, A. Mitochondrial Oxidative Stress in Obesity: Role of the Mineralocorticoid Receptor. J. Endocrinol. 2018, 238, R143–R159. [Google Scholar] [CrossRef] [PubMed]
- Lorençoni, M.F.; Figueira, M.M.; Toledo E Silva, M.V.; Pimentel Schmitt, E.F.; Endringer, D.C.; Scherer, R.; Barth, T.; Vilela Bertolucci, S.K.; Fronza, M. Chemical Composition and Anti-Inflammatory Activity of Essential Oil and Ethanolic Extract of Campomanesia Phaea (O. Berg.) Landrum Leaves. J. Ethnopharmacol. 2020, 252, 112562. [Google Scholar] [CrossRef]
- Donado-Pestana, C.M.; Pessoa, É.V.M.; Rodrigues, L.; Rossi, R.; Moura, M.H.C.; dos Santos-Donado, P.R.; Castro, É.; Festuccia, W.T.; Genovese, M.I. Polyphenols of Cambuci (Campomanesia Phaea (O. Berg.)) Fruit Ameliorate Insulin Resistance and Hepatic Steatosis in Obese Mice. Food Chem. 2021, 340, 128169. [Google Scholar] [CrossRef] [PubMed]
- Lai, Y.-S.; Chen, W.-C.; Ho, C.-T.; Lu, K.-H.; Lin, S.-H.; Tseng, H.-C.; Lin, S.-Y.; Sheen, L.-Y. Garlic Essential Oil Protects against Obesity-Triggered Nonalcoholic Fatty Liver Disease through Modulation of Lipid Metabolism and Oxidative Stress. J. Agric. Food Chem. 2014, 62, 5897–5906. [Google Scholar] [CrossRef]
- Luc, K.; Schramm-Luc, A.; Guzik, T.J.; Mikolajczyk, T.P. Oxidative Stress and Inflammatory Markers in Prediabetes and Diabetes. J. Physiol. Pharmacol. 2019, 70, 809–824. [Google Scholar] [CrossRef]
- Salehi, F.; Behboudi, H.; Kavoosi, G.; Ardestani, S.K. Incorporation of Zataria Multiflora Essential Oil into Chitosan Biopolymer Nanoparticles: A Nanoemulsion Based Delivery System to Improve the in-Vitro Efficacy, Stability and Anticancer Activity of ZEO against Breast Cancer Cells. Int. J. Biol. Macromol. 2020, 143, 382–392. [Google Scholar] [CrossRef]
- Karimi, A.; Krähmer, A.; Herwig, N.; Schulz, H.; Hadian, J.; Meiners, T. Variation of Secondary Metabolite Profile of Zataria Multiflora Boiss. Populations Linked to Geographic, Climatic, and Edaphic Factors. Front. Plant. Sci. 2020, 11, 969. [Google Scholar] [CrossRef] [PubMed]
- Aminizadeh, M.; Kavoosi, G.; Kariminia, A. In Vitro and Ex Vivo Anti-Diabetic and Anti-Hyperglycemic Properties of Zataria Multiflora Essential Oil. Mol. Biol. Rep. 2020, 47, 7805–7813. [Google Scholar] [CrossRef]
- Lozupone, C.A.; Stombaugh, J.I.; Gordon, J.I.; Jansson, J.K.; Knight, R. Diversity, Stability and Resilience of the Human Gut Microbiota. Nature 2012, 489, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Adak, A.; Khan, M.R. An Insight into Gut Microbiota and Its Functionalities. Cell. Mol. Life Sci. 2019, 76, 473–493. [Google Scholar] [CrossRef]
- Marchesi, J.R.; Adams, D.H.; Fava, F.; Hermes, G.D.A.; Hirschfield, G.M.; Hold, G.; Quraishi, M.N.; Kinross, J.; Smidt, H.; Tuohy, K.M.; et al. The Gut Microbiota and Host Health: A New Clinical Frontier. Gut 2016, 65, 330–339. [Google Scholar] [CrossRef] [PubMed]
- Bäckhed, F.; Ding, H.; Wang, T.; Hooper, L.V.; Koh, G.Y.; Nagy, A.; Semenkovich, C.F.; Gordon, J.I. The Gut Microbiota as an Environmental Factor That Regulates Fat Storage. Proc. Natl. Acad. Sci. USA 2004, 101, 15718–15723. [Google Scholar] [CrossRef] [PubMed]
- Le Chatelier, E.; Nielsen, T.; Qin, J.; Prifti, E.; Hildebrand, F.; Falony, G.; Almeida, M.; Arumugam, M.; Batto, J.-M.; Kennedy, S.; et al. Richness of Human Gut Microbiome Correlates with Metabolic Markers. Nature 2013, 500, 541–546. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Wu, H.; Dou, H.; Guo, L.; Huang, W. Microcapsule of Sweet Orange Essential Oil Changes Gut Microbiota in Diet-Induced Obese Rats. Biochem. Biophys. Res. Commun. 2018, 505, 991–995. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhang, Y.; Fan, G.; Ren, J.-N.; Zhang, L.-L.; Pan, S.-Y. Effects of Orange Essential Oil on Intestinal Microflora in Mice. J. Sci. Food Agric. 2019, 99, 4019–4028. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.-A.; Gu, W.; Lee, I.-A.; Joh, E.-H.; Kim, D.-H. High Fat Diet-Induced Gut Microbiota Exacerbates Inflammation and Obesity in Mice via the TLR4 Signaling Pathway. PLoS ONE 2012, 7, e47713. [Google Scholar] [CrossRef]
- Li, A.-L.; Ni, W.-W.; Zhang, Q.-M.; Li, Y.; Zhang, X.; Wu, H.-Y.; Du, P.; Hou, J.-C.; Zhang, Y. Effect of Cinnamon Essential Oil on Gut Microbiota in the Mouse Model of Dextran Sodium Sulfate-Induced Colitis. Microbiol. Immunol. 2020, 64, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Leong, W.; Huang, G.; Khan, I.; Xia, W.; Li, Y.; Liu, Y.; Li, X.; Han, R.; Su, Z.; Hsiao, W.L.W. Patchouli Essential Oil and Its Derived Compounds Revealed Prebiotic-Like Effects in C57BL/6J Mice. Front. Pharmacol. 2019, 10, 1229. [Google Scholar] [CrossRef]
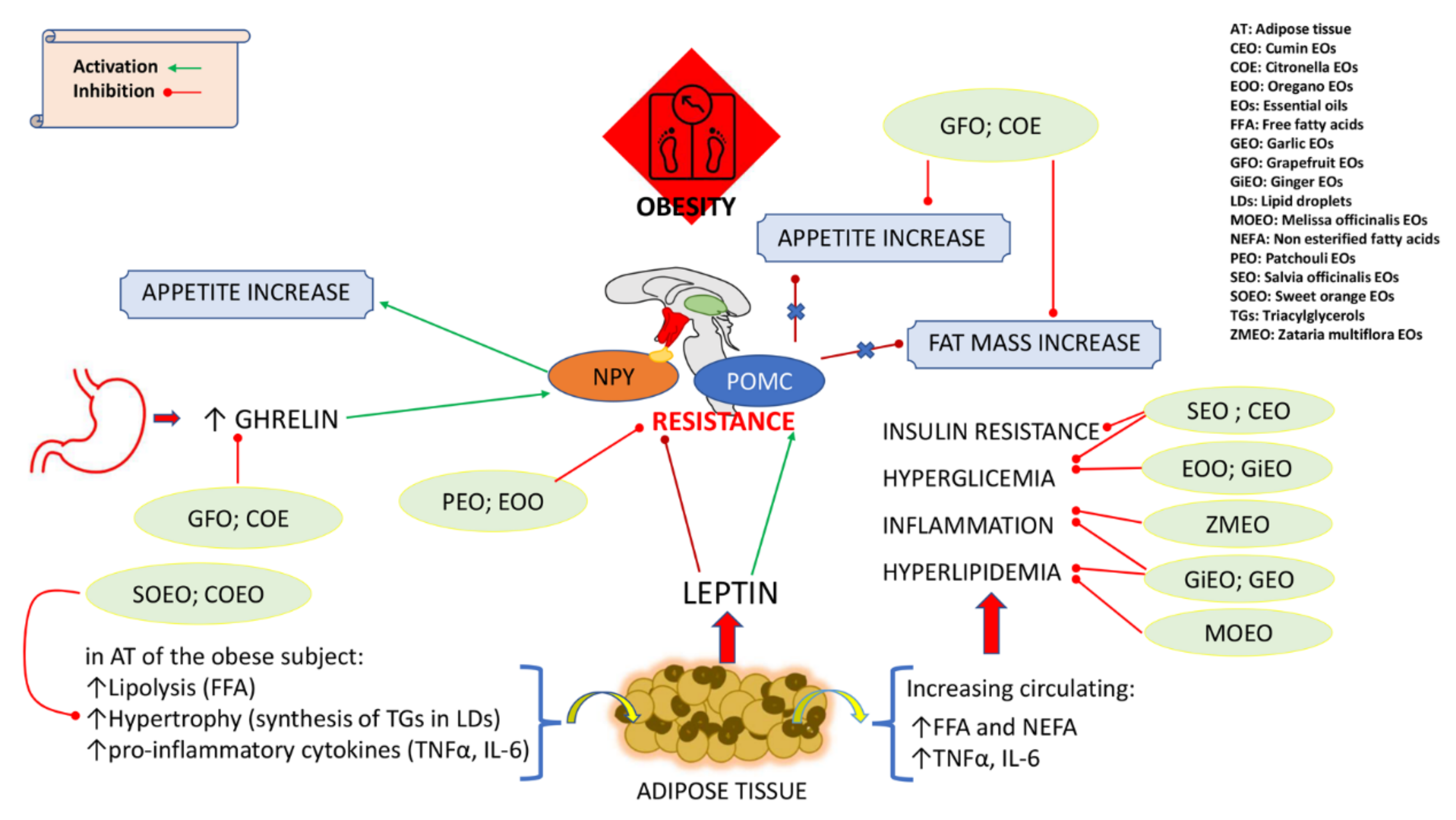
| Effect of Essential Oils (Eos) on: | |||||
|---|---|---|---|---|---|
| EOs | Obesity | Metabolic Syndrome and Related Pathologies | Microbiota | Model System | Ref |
| Campomanesia EOs (CpEO) | Antioxidant and anti-inflammatory effects; ↓insulin resistance and hepatic steatosis | Obese C57BL/6J mice; RAW 264.7 macrophages | [145,146] | ||
| Cinnamon EOs (CiEO) | ↓body weight | ↓hyperlipidemia | Probiotic effect; ↓Helicobacter and Bacteroides | Six-week-old male ICR mice; APOE2 transgenic mice | [113,114,159] |
| Citronella EOs (COE) | ↓appetite and body weight | Sprague Dawley adult male rats | [96] | ||
| Cumin EOs (CEO) | ↓body weight | ↓fasting blood glucose, glycosilated hemoglobin, insulin, inflammation, hypertension | In vivo models (diabetic patients); hypertensive rats | [129,130,131] | |
| Garlic EOs (GEO) | ↑lipolysis | ↓NAFLD | C57BL/6 mice | [146] | |
| Ginger EOs (GiEO) | ↓FFA and inflammation | Obese mice | [120,121] | ||
| Grapefruit EOs (GFO) | ↑lipolysis and thermogenesis; ↓appetite | ↓hyperlipidemia and cardiovascular risk | Rats and mice | [88,92,93,94,95] | |
| Melissa officinalis EOs MOEO | ↓ plasma TG and cholesterol levels | APOE2 transgenic mice; APOE2 transgenic mice | [136,137] | ||
| Oregano EOs (EOO) | ↓adipogenesis | ↓hypercholesterolemia and inflammation | Cell lines; C57BL/6 mice | [16,100,101,102,103] | |
| Patchouli Eos (PEO) | ↓leptin resistance and food intake | Prebiotic effect | Sprague Dawley male rats; C57BL/6 mice | [97,98,99,160] | |
| Rhaponticum acaule EOs (RaEO) | Antioxidant; antidiabetic | Enzyme kinetic studies | [134] | ||
| Salvia EOs (SEO) | ↓body weight | ↓hyperglycemia and hyperlipipidemia; antioxidant effects | Hyperlipidemic mice; diabetic rats; in vivo models (diabetic patients) | [122,123,124,129] | |
| Sweet Orange EOs (SOEO) | ↑lipolysis; ↓lipogenesis and body weight | ↓dysplipidemia and hyperglycemia | ↑protection of gut barrier; ↑Bifidobacterium and Lactobacillus; ↓endotoxin | Obese SD rats; mice | [105,106,156,157] |
| Zataria multiflora EOs (ZMEO) | Antioxidant | Mouse macrophages (J774 A.1) | [149,150] | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Blasio, A.; D’Anneo, A.; Lauricella, M.; Emanuele, S.; Giuliano, M.; Pratelli, G.; Calvaruso, G.; Carlisi, D. The Beneficial Effects of Essential Oils in Anti-Obesity Treatment. Int. J. Mol. Sci. 2021, 22, 11832. https://doi.org/10.3390/ijms222111832
De Blasio A, D’Anneo A, Lauricella M, Emanuele S, Giuliano M, Pratelli G, Calvaruso G, Carlisi D. The Beneficial Effects of Essential Oils in Anti-Obesity Treatment. International Journal of Molecular Sciences. 2021; 22(21):11832. https://doi.org/10.3390/ijms222111832
Chicago/Turabian StyleDe Blasio, Anna, Antonella D’Anneo, Marianna Lauricella, Sonia Emanuele, Michela Giuliano, Giovanni Pratelli, Giuseppe Calvaruso, and Daniela Carlisi. 2021. "The Beneficial Effects of Essential Oils in Anti-Obesity Treatment" International Journal of Molecular Sciences 22, no. 21: 11832. https://doi.org/10.3390/ijms222111832
APA StyleDe Blasio, A., D’Anneo, A., Lauricella, M., Emanuele, S., Giuliano, M., Pratelli, G., Calvaruso, G., & Carlisi, D. (2021). The Beneficial Effects of Essential Oils in Anti-Obesity Treatment. International Journal of Molecular Sciences, 22(21), 11832. https://doi.org/10.3390/ijms222111832










