A Novel Discovery: Holistic Efficacy at the Special Organ Level of Pungent Flavored Compounds from Pungent Traditional Chinese Medicine
Abstract
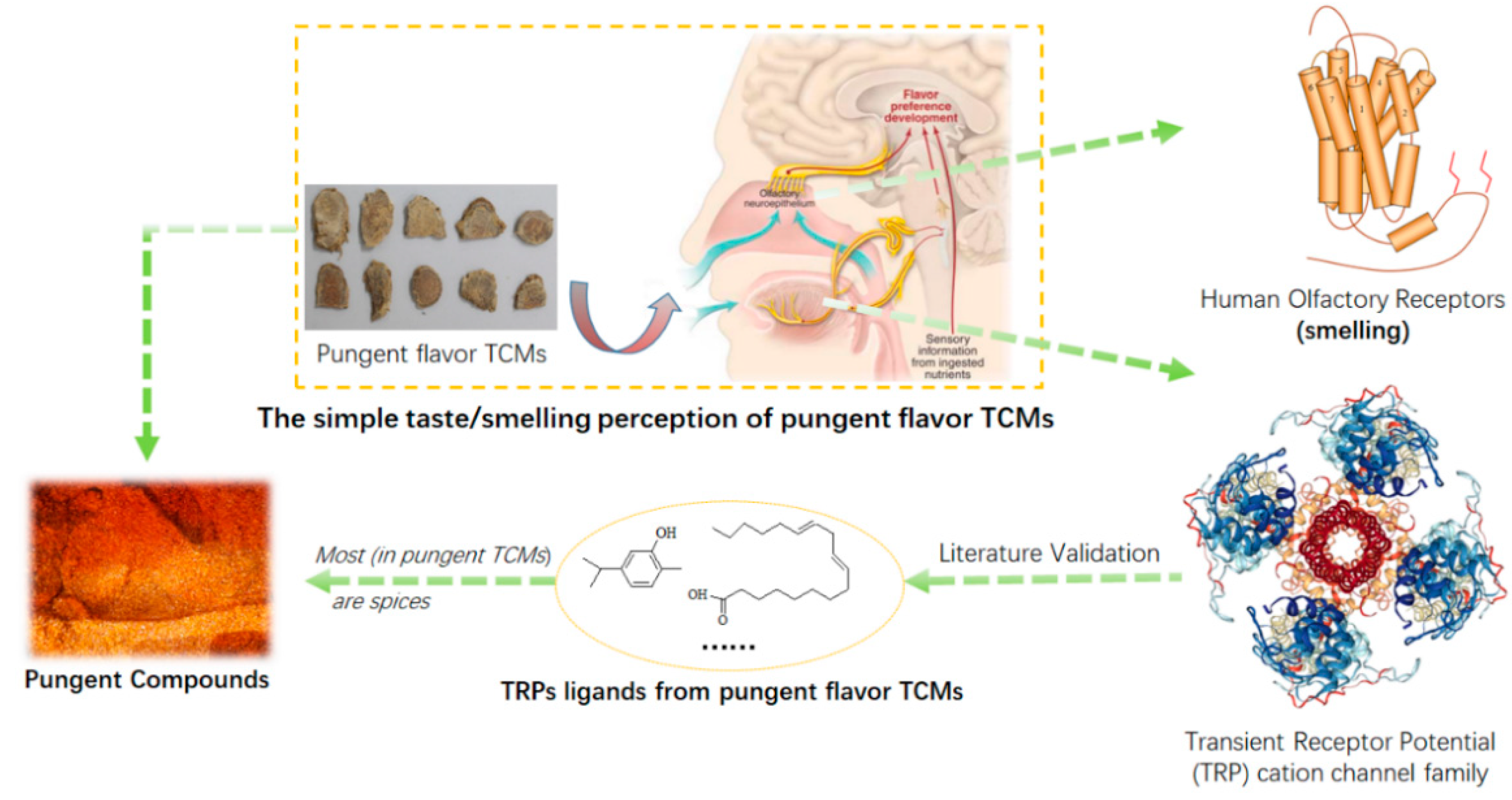
1. Introduction
2. Results
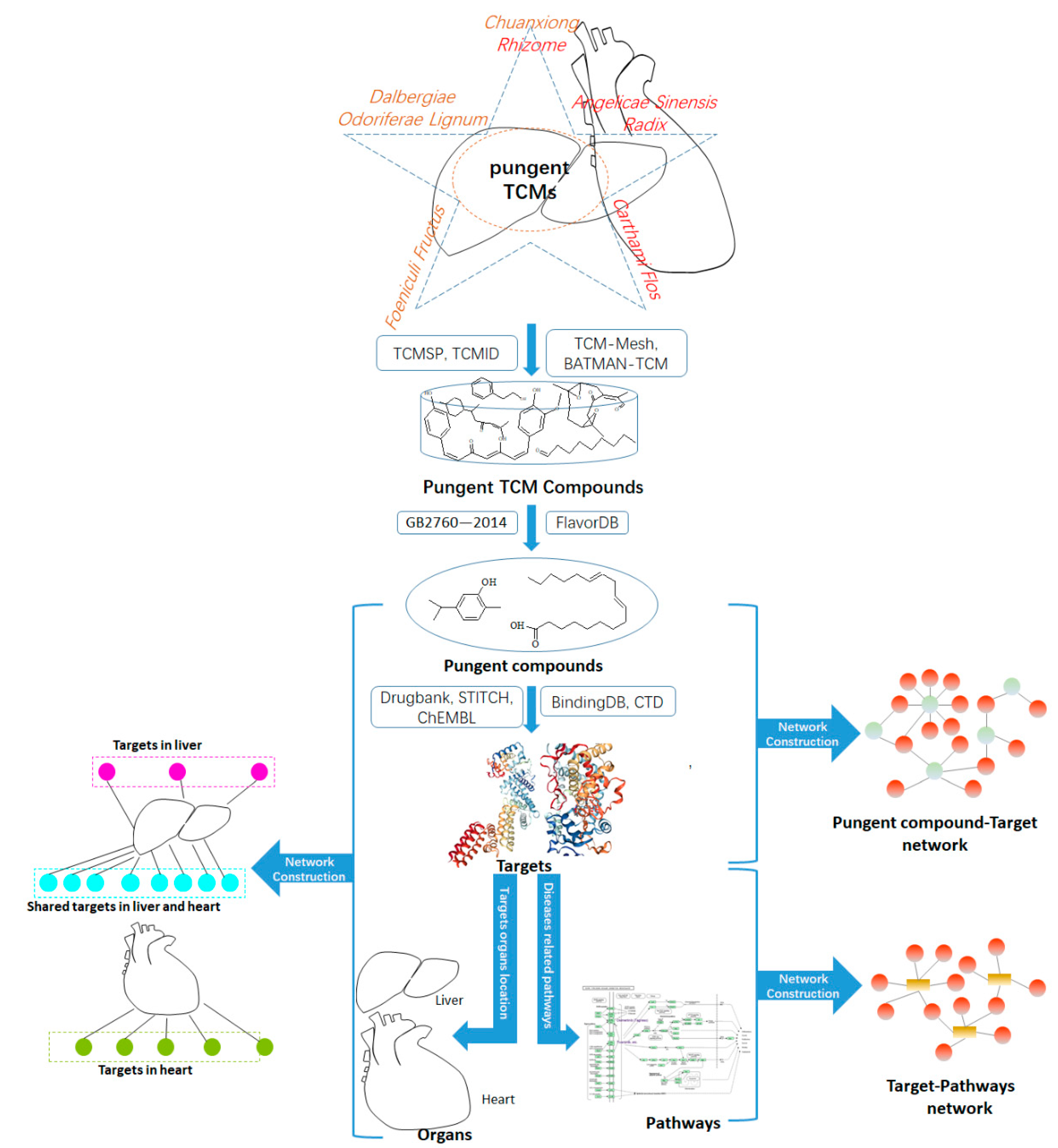
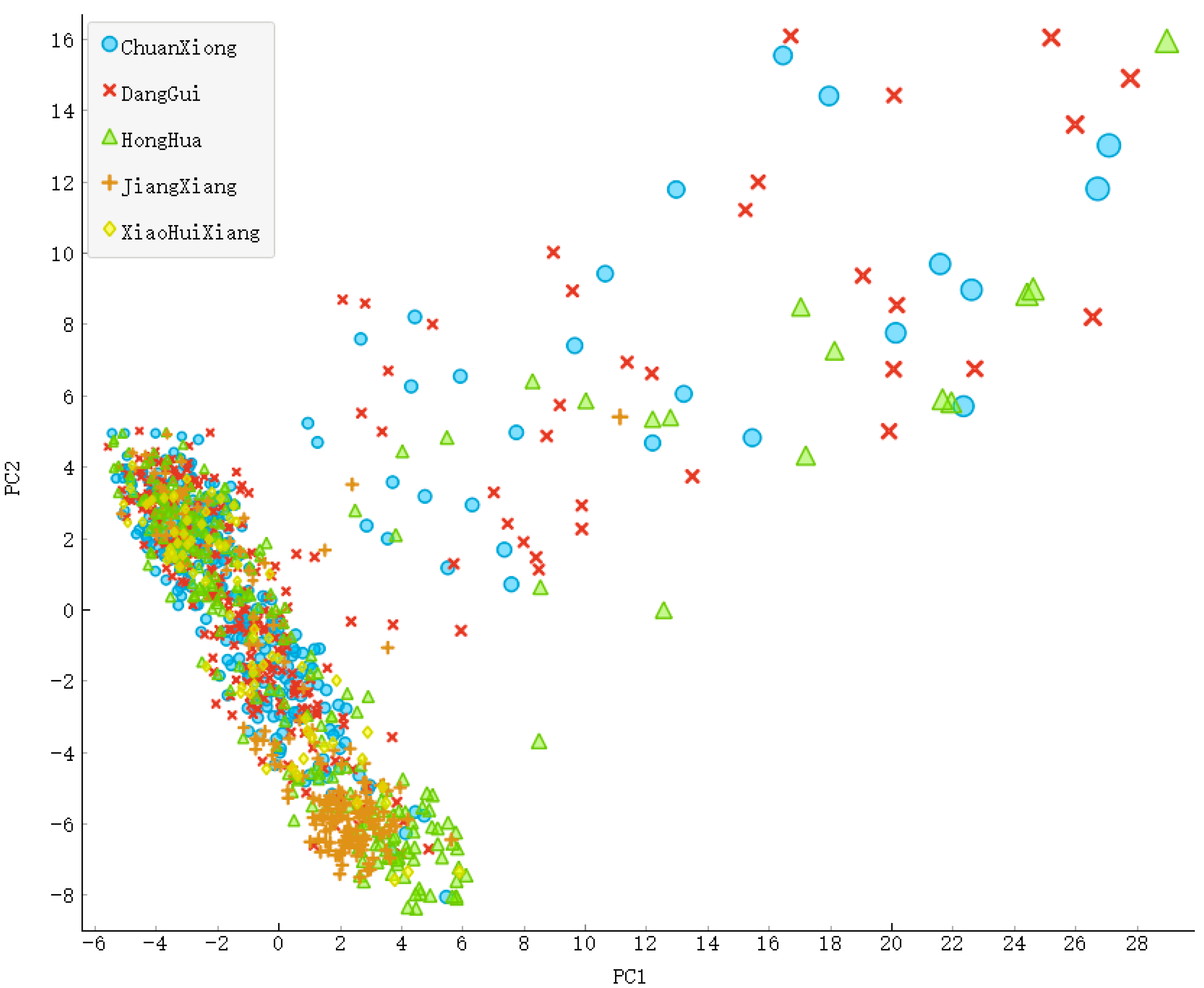
2.1. Collecting the Compounds of Pungent TCMs
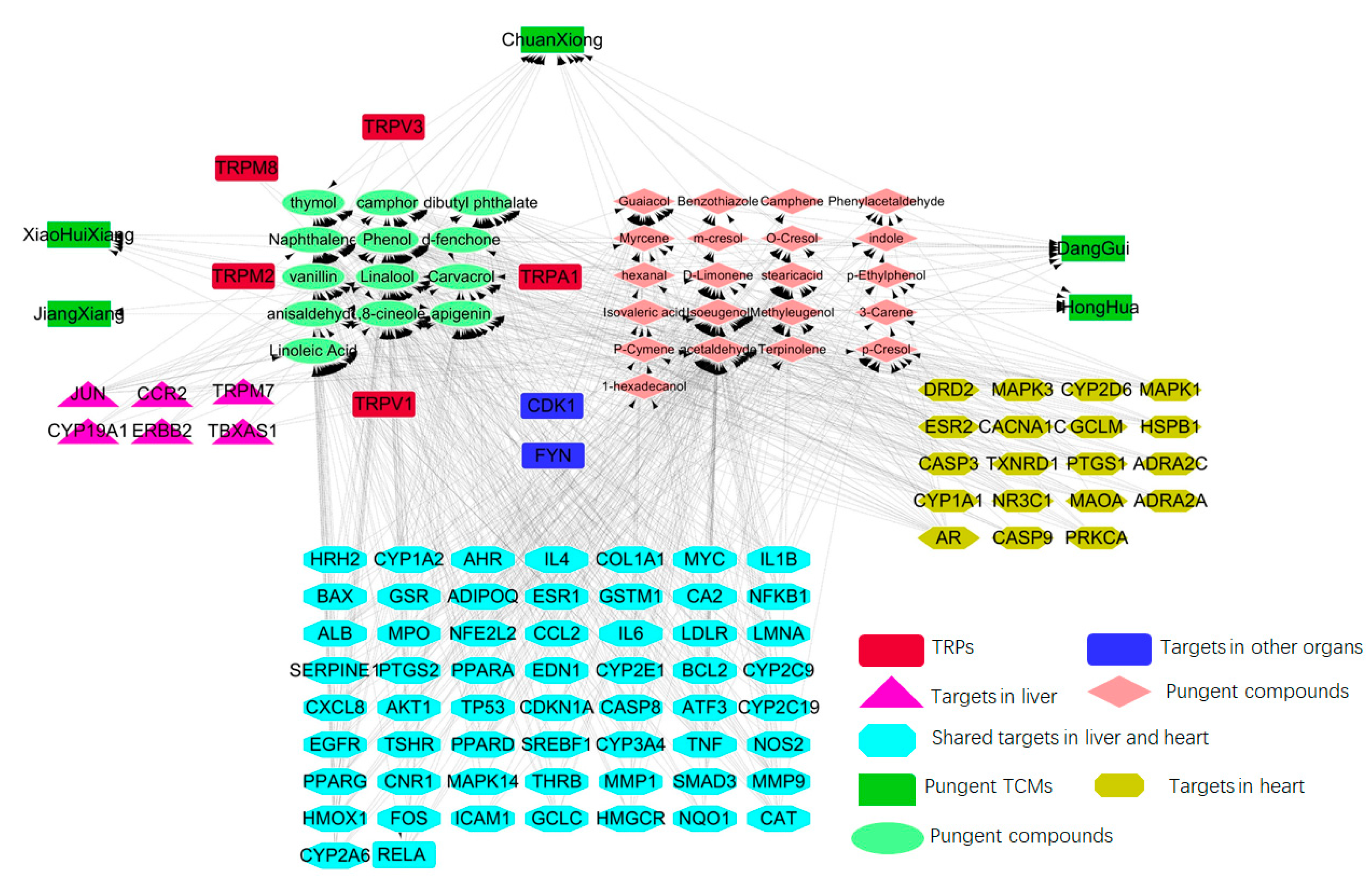
2.2. Pungent Flavor Compound-Target Network
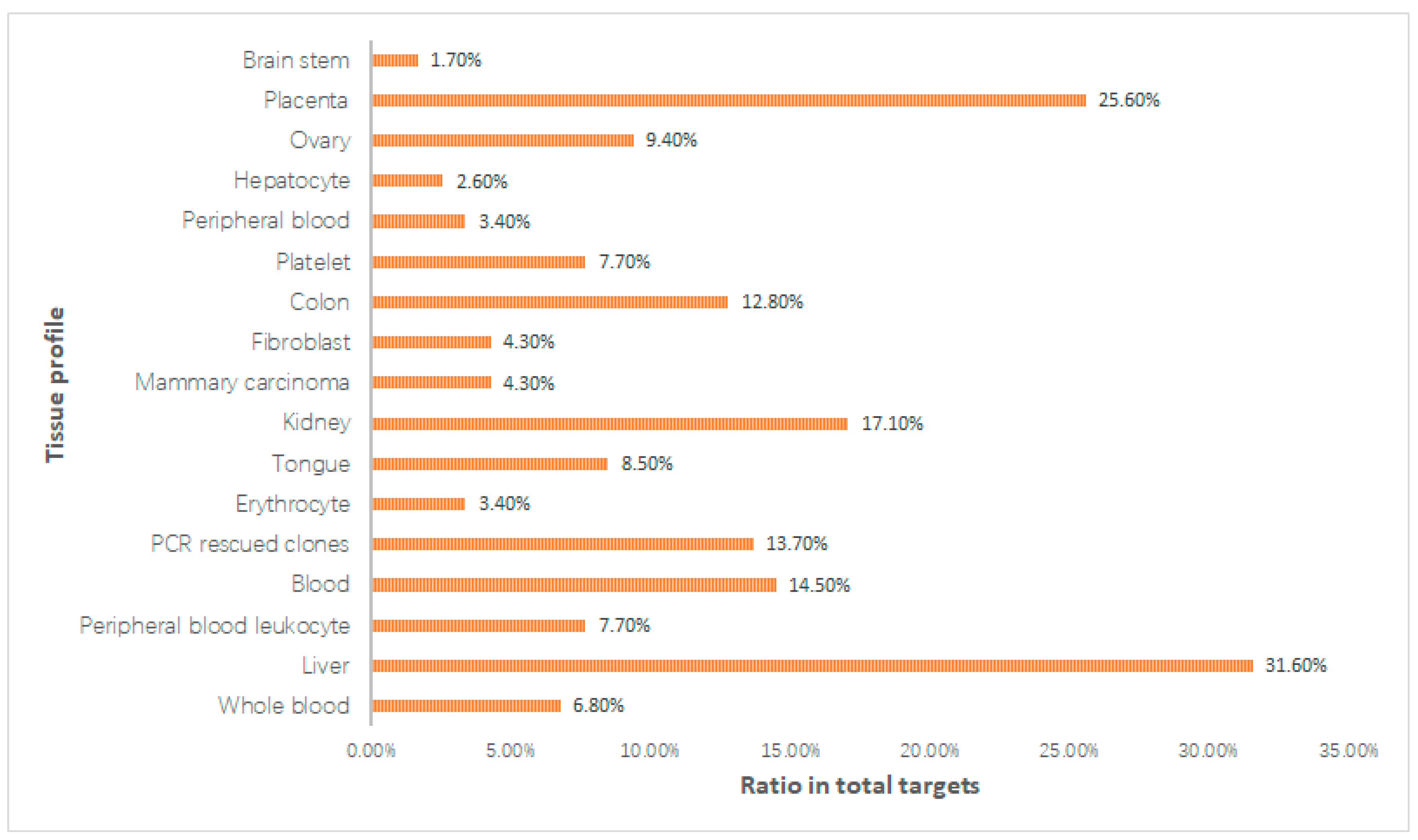
2.3. Organ Targets Location Map
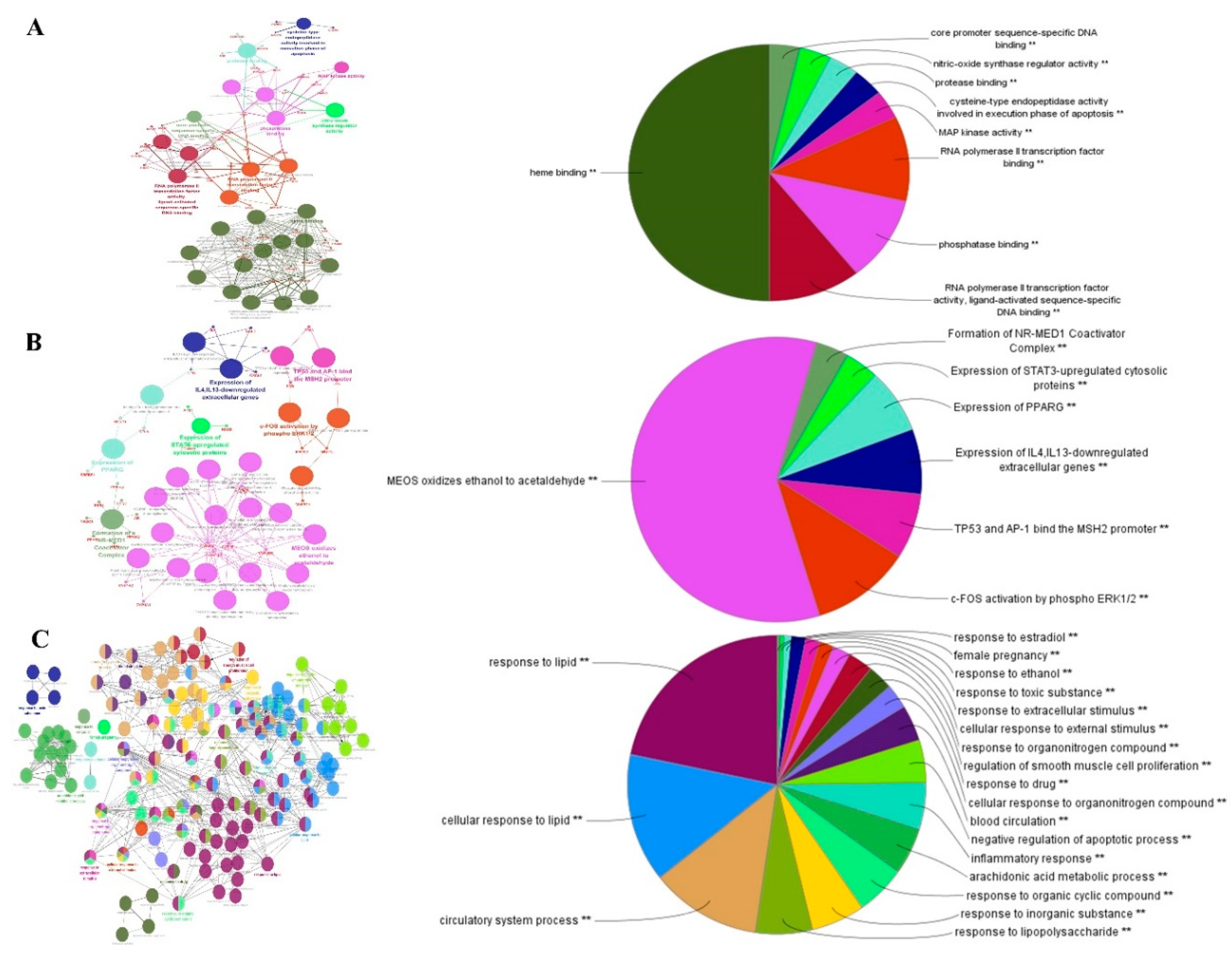
2.4. Target-Pathway Network
3. Discussion
3.1. The Material Base of Pungent Flavor of Pungent TCMs
3.2. Pungent Compounds Provide Support for the Treatment of Cardiovascular and Liver Diseases
3.3. Expression of Meridian Tropism at Level of Tissue and Organ
4. Materials and Methods
4.1. Date Collection and Visualization
4.2. The Gene Analysis of Liver and Heart Organ in Cardiovascular System
4.2.1. Identify the Pungent Flavored Compounds
4.2.2. Identify Organ Target Location with Gene Organizer
4.3. Network Construction
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| BASR | blood-activating and stasis-resolving |
| cGMP-PKG | cyclic guanosine monophosphate-dependent protein kinase |
| CNS | central nervous system |
| CVDs | cardiovascular diseases |
| CYP1A1 | cytochrome P450 1A1 |
| JUN | proto-oncogene c-JUN |
| KEGG | Kyoto Encyclopedia of Genes and Genomes |
| LDL | low-density lipoprotein |
| MACCS | molecular Access System |
| MDL | Molecular Design Limited |
| NAFLD | non-alcoholic fatty liver disease |
| NFE2L2 | nuclear factor erythroid 2-related factor 2 |
| NFKB1 | nuclear factor NF-kappa-B p105 subunit |
| PCA | Principal Component Analysis |
| PPARD | peroxisome proliferator-activated receptor delta, |
| PTGS2 | prostaglandin G/H synthase 2 |
| TCM | traditional Chinese medicine |
| TCM-HPs | traditional Chinese medicine herbal properties |
| TNF | tumor necrosis factor |
| TPAs | target-pathway association interactions |
| TRP | transient receptor potential |
| TRPA1 | transient receptor potential ankyrin 1 |
| TRPM7 | potential cation channel, subfamily M, member 7 |
| TRPM8 | potential cation channel, subfamily M, member 8 |
| TRPV1 | transient receptor potential cation channel subfamily V member 1 |
| TRPV3 | transient receptor potential cation channel subfamily V member 3 |
| VEGF | vascular endothelial growth factor |
References
- Kahleova, H.; Levin, S.; Barnard, N.D. Vegetarian dietary patterns and cardiovascular disease. Prog. Cardiovasc. Dis. 2018, 61, 54–61. [Google Scholar] [CrossRef] [PubMed]
- North, B.J.; Sinclair, D.A. The intersection between aging and cardiovascular disease. Circ. Res. 2012, 110, 1097–1108. [Google Scholar] [CrossRef] [PubMed]
- Lakatta, E.G.; Levy, D. Arterial and cardiac aging: Major shareholders in cardiovascular disease enterprises: Part I: Aging arteries: A “set up” for vascular disease. Circulation 2003, 107, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Humphries, K.H.; Izadnegahdar, M.; Sedlak, T.; Saw, J.; Johnston, N.; Schenck-Gustafsson, K.; Wei, J. Sex differences in cardiovascular disease-Impact on care and outcomes. Front. Neuroendocrinol. 2017, 46, 46–70. [Google Scholar] [CrossRef] [PubMed]
- Xue, M.; Chen, K.; Yin, H. Relationship between platelet activation related factors and polymorphism of related genes in patients with coronary heart disease of blood-stasis syndrome. Chin. J. Integr. Med. 2008, 14, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Chiu, C.C.; Lan, C.Y.; Chang, Y.H. Objective assessment of blood stasis using computerized inspection of sublingual veins. Comput. Methods Progr. Biomed. 2002, 69, 1–12. [Google Scholar] [CrossRef]
- Keji, C. Cardiovascular Disease and Activating Blood Circulation to Remove Stasis; Beijing Science & Technology Press: Beijing, China, 2009. [Google Scholar]
- Lee, I.M.; Shiroma, E.J.; Lobelo, F.; Puska, P.; Blair, S.N.; Katzmarzyk, P.T. Lancet Physical Activity Series Working Group. Effect of physical inactivity on major non-communicable diseases worldwide: An analysis of burden of disease and life expectancy. Lancet 2012, 380, 219–229. [Google Scholar] [CrossRef]
- Gao, S.; Liu, Z.; Li, H.; Little, P.J.; Liu, P.; Xu, S. Cardiovascular actions and therapeutic potential of tanshinone IIA. Atherosclerosis 2012, 220, 3–10. [Google Scholar] [CrossRef]
- Liu, X.H.; Li, L.Q.; Lu, W.H.; Lai, C.L. Effect of safflow yellow treatment on ET, MMP-9, hs-CRP and platelet aggregation rate of patients with coronary heart disease. Chin. J. Integr. Med. Cardio-/Cerebrovasc. Dis. 2011, 9, 1036–1037. [Google Scholar]
- Tingmo, Z. Chinese Materia Medica; High Education Press: Beijing, China, 2008. [Google Scholar]
- Tu, Y.; Xue, Y.; Guo, D.; Sun, L.; Guo, M. Carthami flos: A review of its ethnopharmacology, pharmacology and clinical applications. Revista Brasileira de Farmacognosia 2015, 25, 553–566. [Google Scholar] [CrossRef]
- Ng, C.F.; Koon, C.M.; Cheung, D.W.S.; Lam, M.Y.; Leung, P.C.; Lau, C.B.S.; Fung, K.P. The anti-hypertensive effect of Danshen (Salvia miltiorrhiza) and Gegen (Pueraria lobata) formula in rats and its underlying mechanisms of vasorelaxation. J. Ethnopharmacol. 2011, 137, 1366–1372. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.; Zhao, Y.; Du, L. Emerging approaches of traditional Chinese medicine formulas for the treatment of hyperlipidemia. J. Ethnopharmacol. 2012, 140, 345–367. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Zhang, C.; Gao, F.; Fu, Q.; Fu, C.; He, Y.; Zhang, J. A systematic review on the rhizome of Ligusticum chuanxiong Hort. (Chuanxiong). Food Chem. Toxicol. 2018, 119, 309–325. [Google Scholar] [CrossRef] [PubMed]
- Mao, H.P.; Wang, X.Y.; Chang, Y.X.; Chen, L.; Niu, Z.C.; Ai, J.Q.; Gao, X.M. Danhong injection attenuates isoproterenol-induced cardiac hypertrophy by regulating p38 and NF-κb pathway. J. Ethnopharmacol. 2016, 186, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Crunkhorn, S. Cardiovascular disease: Spice might protect the heart. Nat. Rev. Drug Discov. 2008, 7, 389. [Google Scholar] [CrossRef]
- Cao, C.M.; Xia, Q.; Zhang, X.; Xu, W.H.; Jiang, H.D.; Chen, J. Salvia miltiorrhiza attenuates the changes in contraction and intracellular calcium induced by anoxia and reoxygenation in rat cardiomyocytes. Life Sci. 2003, 72, 2451–2463. [Google Scholar] [CrossRef]
- Yin, Y.; Guan, Y.; Duan, J.; Wei, G.; Zhu, Y.; Quan, W.; Wen, A. Cardioprotective effect of Danshensu against myocardial ischemia/reperfusion injury and inhibits apoptosis of H9c2 cardiomyocytes via Akt and ERK1/2 phosphorylation. Eur. J. Pharmacol. 2013, 699, 219–226. [Google Scholar] [CrossRef]
- Ma, X.J.; Yin, S.J.; Jin, J.C.; Wu, C.F.; Huang, Y.; Shi, D.Z.; Yin, H.J. Synergistic protection of Danhong Injection and ischemic postconditioning on myocardial reperfusion injury in minipigs. Chin. J. Integr. Med. 2010, 16, 531–536. [Google Scholar] [CrossRef]
- Jia, Y.; Huang, F.; Zhang, S.; Leung, S.W. Is danshen (Salvia miltiorrhiza) dripping pill more effective than isosorbide dinitrate in treating angina pectoris? A systematic review of randomized controlled trials. Int. J. Cardiol. 2012, 157, 330–340. [Google Scholar] [CrossRef]
- Kang, D.G.; Yun, Y.G.; Ryoo, J.H.; Lee, H.S. Anti-hypertensive effect of water extract of danshen on renovascular hypertension through inhibition of the renin angiotensin system. Am. J. Chin. Med. 2002, 30, 87–93. [Google Scholar] [CrossRef]
- Fan, H.; Li, M.; Yu, L.; Jin, W.; Yang, J.; Zhang, Y.; Wan, H. Effects of Danhong injection on platelet aggregation in hyperlipidemia rats. J. Ethnopharmacol. 2018, 212, 67–73. [Google Scholar] [CrossRef]
- Marcellin, P.; Kutala, B.K. Liver diseases: A major, neglected global public health problem requiring urgent actions and large-scale screening. Liver Int. 2018, 38, 2–6. [Google Scholar] [CrossRef] [PubMed]
- Mokdad, A.A.; Lopez, A.D.; Shahraz, S.; Lozano, R.; Mokdad, A.H.; Stanaway, J.; Naghavi, M. Liver cirrhosis mortality in 187 countries between 1980 and 2010: A systematic analysis. BMC Med. 2014, 12, 145. [Google Scholar] [CrossRef]
- Murray, C.J.; Vos, T.; Lozano, R.; Naghavi, M.; Flaxman, A.D.; Michaud, C.; Aboyans, V. Disability-adjusted life years (DALYs) for 291 diseases and injuries in 21 regions, 1990–2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2197–2223. [Google Scholar] [CrossRef]
- Yang, S.S. Alcoholic Liver Disease in the Asian–Pacific Region with High Prevalence of Chronic Viral Hepatitis. J. Med. Ultrasound 2016, 24, 93–100. [Google Scholar] [CrossRef]
- Estes, C.; Anstee, Q.M.; Arias-Loste, M.T.; Bantel, H.; Bellentani, S.; Caballeria, J.; Geier, A. Modeling NAFLD Disease Burden in China, France, Germany, Italy, Japan, Spain, United Kingdom, and United States for the period 2016-2030. J. Hepatol. 2018, 69, 896–904. [Google Scholar] [CrossRef] [PubMed]
- Vernon, G.; Baranova, A.; Younossi, Z.M. Systematic review: The epidemiology and natural history of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in adults. Aliment. Pharmacol. Ther. 2011, 34, 274–285. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.G.; Kim, S.U.; Wong, V.W.S. New trends on obesity and NAFLD in Asia. J. Hepatol. 2017, 67, 862–873. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Obesity and Overweight. Available online: http://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 18 January 2019).
- Lebeaupin, C.; Vallée, D.; Hazari, Y.; Hetz, C.; Chevet, E.; Bailly-Maitre, B. Endoplasmic Reticulum stress signaling and the pathogenesis of Non-Alcoholic Fatty Liver Disease. J. Hepatol. 2018, 69, 927–947. [Google Scholar] [CrossRef]
- Chalasani, N.; Younossi, Z.; Lavine, J.E.; Charlton, M.; Cusi, K.; Rinella, M.; Sanyal, A.J. The diagnosis and management of nonalcoholic fatty liver disease: Practice guidance from the American Association for the Study of Liver Diseases. Hepatology 2017, 67, 328–357. [Google Scholar] [CrossRef]
- Younossi, Z.; Henry, L. Contribution of alcoholic and nonalcoholic fatty liver disease to the burden of liver-related morbidity and mortality. Gastroenterology 2016, 150, 1778–1785. [Google Scholar] [CrossRef]
- Younossi, Z.M.; Blissett, D.; Blissett, R.; Henry, L.; Stepanova, M.; Younossi, Y.; Beckerman, R. The economic and clinical burden of nonalcoholic fatty liver disease in the United States and Europe. Hepatology 2016, 64, 1577–1586. [Google Scholar] [CrossRef]
- European Association for the Study of the Liver; European Association for the Study of Diabetes (EASD). EASL-EASD-EASO Clinical Practice Guidelines for the management of non-alcoholic fatty liver disease. Obes. Facts 2016, 9, 65–90. [Google Scholar] [CrossRef]
- Angulo, P.; Kleiner, D.E.; Dam-Larsen, S.; Adams, L.A.; Bjornsson, E.S.; Charatcharoenwitthaya, P.; Haflidadottir, S. Liver fibrosis, but no other histologic features, is associated with long-term outcomes of patients with nonalcoholic fatty liver disease. Gastroenterology 2015, 149, 389–397. [Google Scholar] [CrossRef] [PubMed]
- Bhala, N.; Angulo, P.; van der Poorten, D.; Lee, E.; Hui, J.M.; Saracco, G.; Day, C.P. The natural history of nonalcoholic fatty liver disease with advanced fibrosis or cirrhosis: An international collaborative study. Hepatology 2011, 54, 1208–1216. [Google Scholar] [CrossRef] [PubMed]
- Ekstedt, M.; Hagström, H.; Nasr, P.; Fredrikson, M.; Stål, P.; Kechagias, S.; Hultcrantz, R. Fibrosis stage is the strongest predictor for disease-specific mortality in NAFLD after up to 33 years of follow-up. Hepatology 2015, 61, 1547–1554. [Google Scholar] [CrossRef] [PubMed]
- Hagström, H.; Nasr, P.; Ekstedt, M.; Hammar, U.; Stål, P.; Hultcrantz, R.; Kechagias, S. Fibrosis stage but not NASH predicts mortality and time to development of severe liver disease in biopsy-proven NAFLD. J. Hepatol. 2017, 67, 1265–1273. [Google Scholar] [CrossRef] [PubMed]
- Cheung, F. Made in china. Nature 2011, 480, S82–S83. [Google Scholar] [CrossRef] [PubMed]
- Qiu, J. China plans to modernize traditional medicine. Nature 2007, 446, 590–591. [Google Scholar] [CrossRef]
- Qiu, J. Traditional medicine: A culture in the balance. Nature 2007, 448, 126–128. [Google Scholar] [CrossRef]
- Science, A.A.F.T. The art and science of traditional medicine part 1: TCM today—A case for integration. Science 2014, 346, 1569. [Google Scholar]
- Xue, R.; Fang, Z.; Zhang, M.; Yi, Z.; Wen, C.; Shi, T. TCMID: Traditional Chinese medicine integrative database for herb molecular mechanism analysis. Nucleic Acids Res. 2012, 41, D1089–D1095. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Xie, D.; Yu, Y.; Liu, H.; Shi, Y.; Shi, T.; Wen, C. TCMID 2.0: A comprehensive resource for TCM. Nucleic Acids Res. 2018, 46, D1117–D1120. [Google Scholar] [CrossRef] [PubMed]
- Spices—A Survey of the World Market; Geneva, Switzerland, 1982; Volume 1, pp. 68–712.
- Billing, J.A.P.W. Antimicrobial functions of spices: Why some like it hot. Q. Rev. Biol. 1998, 73, 3–49. [Google Scholar] [CrossRef] [PubMed]
- Pavithra, G. Effect of Spices on Bacteria—A Short Review. J. Pharm. Sci. Res. 2014, 6, 268–270. [Google Scholar]
- Arora, D.S.; Kaur, J. Antimicrobial activity of spices. Int. J. Antimicrob. Agents 1999, 12, 257–262. [Google Scholar] [CrossRef]
- Srinivasan, K. Role of spices beyond food flavoring: Nutraceuticals with multiple health effects. Food Rev. Int. 2005, 21, 167–188. [Google Scholar] [CrossRef]
- Tapsell, L.C.; Hemphill, I.; Cobiac, L.; Patch, C.S.; Sullivan, D.R.; Fenech, M.; Roodenrys, S.; Keogh, J.B.; Clifton, P.M.; Williams, P.G.; et al. Health benefits of herbs and spices: The past, the present, the future. Med. J. Aust. 2006, 185, S4–24. [Google Scholar]
- Gore, J.M.; Dalen, J.E. Cardiovascular disease. JAMA-J. Am. Med. Assoc.-US Ed. 1994, 271, 1660–1661. [Google Scholar] [CrossRef]
- Warshafsky, S.; Kamer, R.S.; Sivak, S.L. Effect of garlic on total serum cholesterol: A meta-analysis. Ann. Intern. Med. 1993, 119, 599–605. [Google Scholar] [CrossRef]
- Sofos, J.N.; Beuchat, L.R.; Davidson, P.M.; Johnson, E.A. Naturally occurring antimicrobials in food. Regul. Toxicol. Pharmacol. 1998, 28, 71–72. [Google Scholar] [CrossRef] [PubMed]
- Beuchat, L.R. Antimicrobial properties of spices and their essential oils. Nat. Antimicrob. Syst. Food Preserv. 1994, 12, 257–262. [Google Scholar]
- Liu, Q.; Meng, X.; Li, Y.; Zhao, C.; Tang, G.; Li, H. Antibacterial and Antifungal Activities of Spices. Int. J. Mol. Sci. 2017, 18, 1283. [Google Scholar] [CrossRef] [PubMed]
- Tajkarimi, M.M.; Ibrahim, S.A.; Cliver, D.O. Antimicrobial herb and spice compounds in food. Food Control 2010, 21, 1199–1218. [Google Scholar] [CrossRef]
- Clapham, D.E. TRP channels as cellular sensors. Nature 2003, 426, 517. [Google Scholar] [CrossRef] [PubMed]
- Kichko, T.I.; Neuhuber, W.; Kobal, G.; Reeh, P.W. The roles of TRPV1, TRPA1 and TRPM8 channels in chemical and thermal sensitivity of the mouse oral mucosa. Eur. J. Neurosci. 2018, 47, 201–210. [Google Scholar] [CrossRef]
- Everaerts, W.; Gees, M.; Alpizar, Y.A.; Farre, R.; Leten, C.; Apetrei, A.; Nilius, B. The capsaicin receptor TRPV1 is a crucial mediator of the noxious effects of mustard oil. Curr. Biol. 2011, 21, 316–321. [Google Scholar] [CrossRef]
- Moparthi, L.; Kichko, T.I.; Eberhardt, M.; Högestätt, E.D.; Kjellbom, P.; Johanson, U.; Zygmunt, P.M. Human TRPA1 is a heat sensor displaying intrinsic U-shaped thermosensitivity. Sci. Rep.-UK 2016, 6, 28763. [Google Scholar] [CrossRef]
- Peier, A.M.; Moqrich, A.; Hergarden, A.C.; Reeve, A.J.; Andersson, D.A.; Story, G.M.; Patapoutian, A. A TRP channel that senses cold stimuli and menthol. Cell 2002, 108, 705–715. [Google Scholar] [CrossRef]
- Bandell, M.; Story, G.M.; Hwang, S.W.; Viswanath, V.; Eid, S.R.; Petrus, M.J.; Patapoutian, A. Noxious cold ion channel TRPA1 is activated by pungent compounds and bradykinin. Neuron 2004, 41, 849–857. [Google Scholar] [CrossRef]
- Bautista, D.M.; Movahed, P.; Hinman, A.; Axelsson, H.E.; Sterner, O.; Högestätt, E.D.; Zygmunt, P.M. Pungent products from garlic activate the sensory ion channel TRPA1. Proc. Natl. Acad. Sci. USA 2005, 102, 12248–12252. [Google Scholar] [CrossRef] [PubMed]
- Jordt, S.E.; Bautista, D.M.; Chuang, H.H.; McKemy, D.D.; Zygmunt, P.M.; Högestätt, E.D.; Julius, D. Mustard oils and cannabinoids excite sensory nerve fibres through the TRP channel ANKTM1. Nature 2004, 427, 260. [Google Scholar] [CrossRef] [PubMed]
- Macpherson, L.J.; Geierstanger, B.H.; Viswanath, V.; Bandell, M.; Eid, S.R.; Hwang, S.; Patapoutian, A. The pungency of garlic: Activation of TRPA1 and TRPV1 in response to allicin. Curr. Biol. 2005, 15, 929–934. [Google Scholar] [CrossRef] [PubMed]
- Salazar, H.; Llorente, I.; Jara-Oseguera, A.; García-Villegas, R.; Munari, M.; Gordon, S.E.; Islas, L.D.; Rosenbaum, T. A single N-terminal cysteine in TRPV1 determines activation by pungent compounds from onion and garlic. Nat. Neurosci. 2008, 11, 255–261. [Google Scholar] [CrossRef]
- Dhaka, A.; Viswanath, V.; Patapoutian, A. Trp ion channels and temperature sensation. Annu. Rev. Neurosci. 2006, 29, 135–161. [Google Scholar] [CrossRef]
- Chung, G.; Im, S.T.; Kim, Y.H.; Jung, S.J.; Rhyu, M.R.; Oh, S.B. Activation of transient receptor potential ankyrin 1 by eugenol. Neuroscience 2014, 261, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Brierley, S.M.; Kelber, O. Use of natural products in gastrointestinal therapies. Curr. Opin. Pharmacol. 2011, 11, 604–611. [Google Scholar] [CrossRef] [PubMed]
- Earley, S.; Gonzales, A.L.; Garcia, Z.I. A dietary agonist of TRPV3 elicits endothelium-dependent vasodilation. Mol. Pharmacol. 2010, 77, 612–620. [Google Scholar] [CrossRef]
- Ueda, T.; Yamada, T.; Ugawa, S.; Ishida, Y.; Shimada, S. TRPV3, a thermosensitive channel is expressed in mouse distal colon epithelium. Biochem. Biophys. Res. Commun. 2009, 383, 130–134. [Google Scholar] [CrossRef]
- Vennekens, R.; Vriens, J.; Nilius, B. Herbal compounds and toxins modulating TRP channels. Curr. Neuropharmacol. 2008, 6, 79–96. [Google Scholar] [CrossRef]
- Baraldi, P.G.; Preti, D.; Materazzi, S.; Geppetti, P. Transient receptor potential ankyrin 1 (TRPA1) channel as emerging target for novel analgesics and anti-inflammatory agents. J. Med. Chem. 2010, 53, 5085–5107. [Google Scholar] [CrossRef] [PubMed]
- Hasan, R.; Leeson-Payne, A.T.S.; Jaggar, J.H.; Zhang, X. Calmodulin is responsible for Ca 2+-dependent regulation of TRPA1 Channels. Sci. Rep. 2017, 7, 45098. [Google Scholar] [CrossRef] [PubMed]
- Bae, C.Y.J.; Sun, H. Current understanding of TRPM7 pharmacology and drug development for stroke. Acta Pharmacol. Sin. 2013, 34, 10. [Google Scholar] [CrossRef] [PubMed]
- Kurzawski, M.; Dziedziejko, V.; Urasińska, E.; Post, M.; Wójcicki, M.; Miętkiewski, J.; Droździk, M. Nuclear factor erythroid 2-like 2 (Nrf2) expression in end-stage liver disease. Environ. Toxicol. Phar. 2012, 34, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Olagnier, D.; Brandtoft, A.M.; Gunderstofte, C.; Villadsen, N.L.; Krapp, C.; Thielke, A.L.; Thyrsted, J. Nrf2 negatively regulates STING indicating a link between antiviral sensing and metabolic reprogramming. Nat. Commun. 2018, 9, 3506. [Google Scholar] [CrossRef] [PubMed]
- Lamlé, J.; Marhenke, S.; Borlak, J.; Von Wasielewski, R.; Eriksson, C.P.; Geffers, R.; Vogel, A. Nuclear Factor-Eythroid 2–Related Factor 2 Prevents Alcohol-Induced Fulminant Liver Injury. Gastroenterology 2008, 134, 1159–1168. [Google Scholar] [CrossRef]
- Harrison, D.; Griendling, K.K.; Landmesser, U.; Hornig, B.; Drexler, H. Role of oxidative stress in atherosclerosis. Am. J. Cardiol. 2003, 91, 7–11. [Google Scholar] [CrossRef]
- Collins, A.R.; Gupte, A.A.; Ji, R.; Ramirez, M.R.; Minze, L.J.; Liu, J.Z.; Lyon, C.J. Myeloid deletion of nuclear factor erythroid 2−related factor 2 increases atherosclerosis and liver injury. Arterioscler. Thromb. Vasc. Biol. 2012, 32, 2839–2846. [Google Scholar] [CrossRef]
- Ruotsalainen, A.K.; Inkala, M.; Partanen, M.E.; Lappalainen, J.P.; Kansanen, E.; Mäkinen, P.I.; Hörkkö, S. The absence of macrophage Nrf2 promotes early atherogenesis. Cardiovasc. Res. 2013, 98, 107–115. [Google Scholar] [CrossRef]
- Harrison, D.G.; Gongora, M.C. Oxidative stress and hypertension. Med. Clin. 2009, 93, 621–635. [Google Scholar] [CrossRef]
- Cifuentes, M.E.; Pagano, P.J. Targeting reactive oxygen species in hypertension. Curr. Opin. Nephrol. Hypertens. 2006, 15, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Delles, C.; Miller, W.H.; Dominiczak, A.F. Targeting reactive oxygen species in hypertension. Antioxid. Redox Signal. 2008, 10, 1061–1078. [Google Scholar] [CrossRef] [PubMed]
- Mehta, P.K.; Griendling, K.K. Angiotensin II cell signaling: Physiological and pathological effects in the cardiovascular system. Am. J. Physiol.-Cell Physiol. 2007, 292, C82–C97. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Ichikawa, T.; Villacorta, L.; Janicki, J.S.; Brower, G.L.; Yamamoto, M.; Cui, T. Nrf2 protects against maladaptive cardiac responses to hemodynamic stress. Arterioscler. Thromb. Vasc. Biol. 2009, 29, 1843–1850. [Google Scholar] [CrossRef] [PubMed]
- Brewer, A.C.; Murray, T.V.; Arno, M.; Zhang, M.; Anilkumar, N.P.; Mann, G.E.; Shah, A.M. Nox4 regulates Nrf2 and glutathione redox in cardiomyocytes in vivo. Free Radic. Biol. Med. 2011, 51, 205–215. [Google Scholar] [CrossRef] [PubMed]
- Kuroda, J.; Ago, T.; Matsushima, S.; Zhai, P.; Schneider, M.D.; Sadoshima, J. NADPH oxidase 4 (Nox4) is a major source of oxidative stress in the failing heart. Proc. Natl. Acad. Sci. USA 2010, 107, 15565–15570. [Google Scholar] [CrossRef] [PubMed]
- Ichikawa, T.; Li, J.; Meyer, C.J.; Janicki, J.S.; Hannink, M.; Cui, T. Dihydro-CDDO-trifluoroethyl amide (dh404), a novel Nrf2 activator, suppresses oxidative stress in cardiomyocytes. PLoS ONE 2009, 4, e8391. [Google Scholar] [CrossRef]
- Bailey-Downs, L.C.; Mitschelen, M.; Sosnowska, D.; Toth, P.; Pinto, J.T.; Ballabh, P.; Bass, C. Liver-specific knockdown of IGF-1 decreases vascular oxidative stress resistance by impairing the Nrf2-dependent antioxidant response: A novel model of vascular aging. J. Gerontol. Ser. A Biomed. Sci. Med. Sci. 2011, 67, 313–329. [Google Scholar] [CrossRef]
- Dai, G.; Vaughn, S.; Zhang, Y.; Wang, E.T.; Garcia-Cardena, G.; Gimbrone, M.A. Biomechanical forces in atherosclerosis-resistant vascular regions regulate endothelial redox balance via phosphoinositol 3-kinase/Akt-dependent activation of Nrf2. Circ. Res. 2007, 101, 723–733. [Google Scholar] [CrossRef]
- Hosoya, T.; Maruyama, A.; Kang, M.I.; Kawatani, Y.; Shibata, T.; Uchida, K.; Yamamoto, M. Differential responses of the Nrf2-Keap1 system to laminar and oscillatory shear stresses in endothelial cells. J. Biol. Chem. 2005, 280, 27244–27250. [Google Scholar] [CrossRef]
- Jyrkkänen, H.K.; Kansanen, E.; Inkala, M.; Kivelä, A.M.; Hurttila, H.; Heinonen, S.E.; Oskolkova, O. Nrf2 regulates antioxidant gene expression evoked by oxidized phospholipids in endothelial cells and murine arteries in vivo. Circ. Res. 2008, 103, e1–e9. [Google Scholar] [CrossRef] [PubMed]
- Ungvari, Z.; Bagi, Z.; Feher, A.; Recchia, F.A.; Sonntag, W.E.; Pearson, K.; Csiszar, A. Resveratrol confers endothelial protection via activation of the antioxidant transcription factor Nrf2. Am. J. Physiol.-Heart Circ. 2010, 299, H18–H24. [Google Scholar] [CrossRef] [PubMed]
- Nigro, P.; Abe, J.I.; Berk, B.C. Flow shear stress and atherosclerosis: A matter of site specificity. Antioxid. Redox Signal. 2011, 15, 1405–1414. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.H.; Jin, S.W.; Kim, H.G.; Khanal, T.; Hwang, Y.P.; Lee, K.J.; Jeong, H.G. Platycodi Radix attenuates dimethylnitrosamine-induced liver fibrosis in rats by inducing Nrf2-mediated antioxidant enzymes. Food Chem. Toxicol. 2013, 56, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Hellerbrand, C.; Köhler, U.A.; Bugnon, P.; Kan, Y.W.; Werner, S.; Beyer, T.A. The Nrf2 transcription factor protects from toxin-induced liver injury and fibrosis. Lab Investig. 2008, 88, 1068. [Google Scholar] [CrossRef] [PubMed]
- Xing, Y.; Niu, T.; Wang, W.; Li, J.; Li, S.; Janicki, J.S.; Zhao, Y. Triterpenoid dihydro-CDDO-trifluoroethyl amide protects against maladaptive cardiac remodeling and dysfunction in mice: A critical role of Nrf2. PLoS ONE 2012, 7, e44899. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Shao, W.; Chiang, Y.; Foltz, W.; Zhang, Z.; Ling, W.; Jin, T. Oltipraz upregulates the nuclear respiratory factor 2 alpha subunit (NRF2) antioxidant system and prevents insulin resistance and obesity induced by a high-fat diet in C57BL/6J mice. Diabetologia 2011, 54, 922–934. [Google Scholar] [CrossRef]
- Migeon, B.R.; Brown, T.R.; Axelman, J.; Migeon, C.J. Studies of the locus for androgen receptor: Localization on the human X chromosome and evidence for homology with the Tfm locus in the mouse. Proc. Natl. Acad. Sci. USA 1981, 78, 6339–6343. [Google Scholar] [CrossRef]
- Gao, W.; Bohl, C.E.; Dalton, J.T. Chemistry and structural biology of androgen receptor. Chem. Rev. 2005, 105, 3352–3370. [Google Scholar] [CrossRef]
- Mangelsdorf, D.J.; Thummel, C.; Beato, M.; Herrlich, P.; Schütz, G.; Umesono, K.; Evans, R.M. The nuclear receptor superfamily: The second decade. Cell 1995, 83, 835–839. [Google Scholar] [CrossRef]
- Eisenfeld, A.J.; Aten, R.F. Estrogen receptors and androgen receptors in the mammalian liver. J. Steroid Biochem. 1987, 27, 1109–1118. [Google Scholar] [CrossRef]
- Winborn, W.B.; Sheridan, P.J.; McGill, H.C. Sex steroid receptors in the stomach, liver, pancreas, and gastrointestinal tract of the baboon. Gastroenterology 1987, 92, 23–32. [Google Scholar] [CrossRef]
- Keller, E.T.; Ershler, W.B.; Chang, C. The androgen receptor: A mediator of diverse responses. Front. Biosci. 1996, 1, d59-71. [Google Scholar] [PubMed]
- Zhu, D.; Hadoke, P.W.; Wu, J.; Vesey, A.T.; Lerman, D.A.; Dweck, M.R.; MacRae, V.E. Ablation of the androgen receptor from vascular smooth muscle cells demonstrates a role for testosterone in vascular calcification. Sci. Rep.-UK 2016, 6, 24807. [Google Scholar] [CrossRef] [PubMed]
- Crabbe, P.; Bogaert, V.; De Bacquer, D.; Goemaere, S.T.E.F.; Zmierczak, H.; Kaufman, J.M. Part of the interindividual variation in serum testosterone levels in healthy men reflects differences in androgen sensitivity and feedback set point: Contribution of the androgen receptor polyglutamine tract polymorphism. J. Clin. Endocrinol. Metab. 2007, 92, 3604–3610. [Google Scholar] [CrossRef] [PubMed]
- Page, S.T.; Kupelian, V.; Bremner, W.J.; McKinlay, J.B. The androgen receptor gene CAG repeat polymorphism does not predict increased risk of heart disease: Longitudinal results from the Massachusetts Male Ageing Study. Clin. Endocrinol. 2006, 65, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Guengerich, F.P.; Shimada, T. Oxidation of toxic and carcinogenic chemicals by human cytochrome P-450 enzymes. Chem. Res. Toxicol. 1991, 4, 391–407. [Google Scholar] [CrossRef] [PubMed]
- Wrighton, S.A.; Stevens, J.C. The human hepatic cytochromes P450 involved in drug metabolism. Crit. Rev. Toxicol. 1992, 22, 1–21. [Google Scholar] [CrossRef]
- Preissner, S.C.; Hoffmann, M.F.; Preissner, R.; Dunkel, M.; Gewiess, A.; Preissner, S. Polymorphic cytochrome P450 enzymes (CYPs) and their role in personalized therapy. PLoS ONE 2013, 8, e82562. [Google Scholar] [CrossRef]
- Culhane, A.C.; Schwarzl, T.; Sultana, R.; Picard, K.C.; Picard, S.C.; Lu, T.H.; Quackenbush, J. GeneSigDB—A curated database of gene expression signatures. Nucleic Acids Res. 2009, 38, D716–D725. [Google Scholar] [CrossRef]
- Thum, T.; Borlak, J. Gene expression in distinct regions of the heart. Lancet 2000, 355, 979–983. [Google Scholar] [CrossRef]
- Gebremichael, A.; Chang, A.M.; Buckpitt, A.R.; Plopper, C.G.; Pinkerton, K.E. Postnatal development of cytochrome P4501A1 and 2B1 in rat lung and liver: Effect of aged and diluted sidestream cigarette smoke. Toxicol. Appl. Pharm. 1995, 135, 246–253. [Google Scholar] [CrossRef] [PubMed]
- Uno, S.; Nebert, D.W.; Makishima, M. Cytochrome P450 1A1 (CYP1A1) protects against nonalcoholic fatty liver disease caused by Western diet containing benzo [a] pyrene in mice. Food Chem. Toxicol. 2018, 113, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Ozeki, J.; Uno, S.; Ogura, M.; Choi, M.; Maeda, T.; Sakurai, K.; Makishima, M. Aryl hydrocarbon receptor ligand 2, 3, 7, 8-tetrachlorodibenzo-p-dioxin enhances liver damage in bile duct-ligated mice. Toxicology 2011, 280, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, Y.; Hashida, H.; Arita, A.; Hamaguchi, K.; Shimura, F. High dose of commercial products of kava (Piper methysticum) markedly enhanced hepatic cytochrome P450 1A1 mRNA expression with liver enlargement in rats. Food Chem. Toxicol. 2008, 46, 3732–3738. [Google Scholar] [CrossRef] [PubMed]
- Harrigan, J.A.; McGarrigle, B.P.; Sutter, T.R.; Olson, J.R. Tissue specific induction of cytochrome P450 (CYP) 1A1 and 1B1 in rat liver and lung following in vitro (tissue slice) and in vivo exposure to benzo (a) pyrene. Toxicol. In Vitro 2006, 20, 426–438. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.K.; Yeung, H.Y.; Woo, P.S.; Wong, M.H. Specific expression of cytochrome P4501A1 gene in gill, intestine and liver of tilapia exposed to coastal sediments. Aquat. Toxicol. 2001, 54, 69–80. [Google Scholar] [CrossRef]
- Wong, C.K.C.; Yeung, H.Y.; Cheung, R.Y.H.; Yung, K.K.L.; Wong, M.H. Ecotoxicological assessment of persistent organic and heavy metal contamination in Hong Kong coastal sediment. Arch. Environ. Contam. Toxicol. 2000, 38, 486–493. [Google Scholar] [CrossRef]
- White, R.D.; Shea, D.; Coloww, A.R.; Stegeman, J.J. Induction and post-transcriptional suppression of hepatic cytochrome P450 1A1 by 3, 3′, 4, 4′-tetrachlorobiphenyl. Biochem. Pharmacol. 1997, 53, 1029–1040. [Google Scholar] [CrossRef]
- Almli, B.; Egaas, E.; Christiansen, A.; Eklo, O.M.; Lode, O.; Källqvist, T. Effects of three fungicides alone and in combination on glutathione S-transferase activity (GST) and cytochrome P-450 (CYP 1A1) in the liver and gill of brown trout (Salmo trutta). Mar. Environ. Res. 2002, 54, 237–240. [Google Scholar] [CrossRef]
- Petkam, R.; Renaud, R.L.; Leatherland, J.F. The role of CYP 1A1 in the in vitro metabolism of pregnenolone by the liver of rainbow trout embryos. Comp. Bioch. Physiol. Part C Toxicol. Pharmacol. 2003, 135, 277–284. [Google Scholar] [CrossRef]
- Stiborová, M.; Martínek, V.; Rýdlová, H.; Koblas, T.; Hodek, P. Expression of cytochrome P450 1A1 and its contribution to oxidation of a potential human carcinogen 1-phenylazo-2-naphthol (Sudan I) in human livers. Cancer Lett. 2005, 220, 145–154. [Google Scholar] [CrossRef] [PubMed]
- Huang, P.; Ceccatelli, S.; Rannug, A. A study on diurnal mRNA expression of CYP1A1, AHR, ARNT, and PER2 in rat pituitary and liver. Environ. Toxicol. Pharmacol. 2002, 11, 119–126. [Google Scholar] [CrossRef]
- Badawi, A.F.; Cavalieri, E.L.; Rogan, E.G. Role of human cytochrome P450 1A1, 1A2, 1B1, and 3A4 in the 2-, 4-, and 16 [alpha]-hydroxylation of 17 [beta]-estradiol. Metabolism 2001, 50, 1001–1003. [Google Scholar] [CrossRef]
- Bindea, G.; Mlecnik, B.; Hackl, H.; Charoentong, P.; Tosolini, M.; Kirilovsky, A.; Galon, J. ClueGO: A Cytoscape plug-in to decipher functionally grouped gene ontology and pathway annotation networks. Bioinformatics 2009, 25, 1091–1093. [Google Scholar] [CrossRef]
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 2008, 4, 44. [Google Scholar] [CrossRef]
- Wu, C.; MacLeod, I.; Su, A.I. BioGPS and MyGene. info: Organizing online, gene-centric information. Nucleic Acids Res. 2012, 41, D561–D565. [Google Scholar] [CrossRef]
- Wu, C.; Orozco, C.; Boyer, J.; Leglise, M.; Goodale, J.; Batalov, S.; Su, A.I.B.A. BioGPS: An extensible and customizable portal for querying and organizing gene annotation resources. Genome Biol. 2009, 10, R130. [Google Scholar] [CrossRef]
- Park, J.; Lee, D.S.; Christakis, N.A.; Barabási, A.L. The impact of cellular networks on disease comorbidity. Mol. Syst. Biol. 2009, 5, 262. [Google Scholar] [CrossRef]
- Gokhman, D.; Kelman, G.; Amartely, A.; Gershon, G.; Tsur, S.; Carmel, L. Gene ORGANizer: Linking genes to the organs they affect. Nucleic Acids Res. 2017, 45, W138–W145. [Google Scholar] [CrossRef]
- Zhang, W.; Tao, Q.; Guo, Z.; Fu, Y.; Chen, X.; Shar, P.A.; Wu, Z.; Wang, Z. Systems Pharmacology Dissection of the Integrated Treatment for Cardiovascular and Gastrointestinal Disorders by Traditional Chinese Medicine. Sci. Rep.-UK 2016, 6, 32400. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.; Wang, X.; Niu, Y.; Duan, D.; Yang, X.; Hao, J.; Zhu, C.; Chen, D.; Wang, K.; Qin, X.; Wu, X. Molecular targets of Chinese herbs: A clinical study of hepatoma based on network pharmacology. Sci. Rep.-UK 2016, 6, 24944. [Google Scholar] [CrossRef] [PubMed]
- Kanehisa, M.; Goto, S. KEGG: Kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 2000, 28, 27–30. [Google Scholar] [CrossRef] [PubMed]
- Marks, A.R. Ryanodine receptors/calcium release channels in heart failure and sudden cardiac death. J. Mol. Cell Cardiol. 2001, 33, 615–624. [Google Scholar] [CrossRef] [PubMed]
- Bradshaw, R.A.; Dennis, E.A.E. Handbook of Cell Signaling; Academic Press: New York, NY, USA, 2009. [Google Scholar]
- Birschmann, I.; Walter, U. Physiology and pathophysiology of vascular signaling controlled by guanosine 3′, 5′-cyclic monophosphate-dependent protein kinase. Acta Biochim. Pol. 2004, 51, 397–404. [Google Scholar] [PubMed]
- Kong, Q.; Blanton, R.M. Protein kinase GI and heart failure: Shifting focus from vascular unloading to direct myocardial antiremodeling effects. Circ. Heart Fail. 2013, 6, 1268–1283. [Google Scholar] [CrossRef] [PubMed]
- Taylor, R.N.; Roberts, J.M.; Cunningham, F.G.; Lindheimer, M.D.E. Chesley’s Hypertensive Disorders in Pregnancy; Elsevier: Amsterdam, The Netherlands, 2014. [Google Scholar]
- Meyer, J.; Lejmi, E.; Fontana, P.; Morel, P.; Gonelle-Gispert, C.; Bühler, L. A focus on the role of platelets in liver regeneration: Do platelet-endothelial cell interactions initiate the regenerative process? J. Hepatol. 2015, 63, 1263–1271. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, E.; Gibbins, J.M.; Holbrook, L.M.; Palomo, I. Nadph Oxidase 2 (Nox2): A Key Target of Oxidative Stress-Mediated Platelet Activation and Thrombosis. Trends Cardiovasc. Med. 2018, 28, 429–434. [Google Scholar] [CrossRef] [PubMed]
- Faggio, C.; Sureda, A.; Morabito, S.; Sanches-Silva, A.; Mocan, A.; Nabavi, S.F.; Nabavi, S.M. Flavonoids and platelet aggregation: A brief review. Eur. J. Pharmacol. 2017, 807, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Bodin, S.; Tronchère, H.; Payrastre, B. Lipid rafts are critical membrane domains in blood platelet activation processes. Biochim. Biophys. Acta (BBA)-Biomembr. 2003, 1610, 247–257. [Google Scholar] [CrossRef]
- Touyz, R.M.; Herrmann, S.; Herrmann, J. Vascular toxicities with VEGF inhibitor therapies-Focus on Hypertension and Arterial Thrombotic Events. J. Am. Soc. Hypertens. 2018, 12, 409–425. [Google Scholar] [CrossRef] [PubMed]
- Xin, H.; Zhong, C.; Nudleman, E.; Ferrara, N. Evidence for pro-angiogenic functions of VEGF-Ax. Cell 2016, 167, 275–284. [Google Scholar] [CrossRef]
- Roskoski, R., Jr. Vascular endothelial growth factor (VEGF) and VEGF receptor inhibitors in the treatment of renal cell carcinomas. Pharmacol. Res. 2017, 120, 116–132. [Google Scholar] [CrossRef]
- Teschke, R.; Xuan, T.D. A Contributory Role of Shell Ginger (Alpinia zerumbet) for Human Longevity in Okinawa, Japan? Nutrients 2018, 10, 166. [Google Scholar] [CrossRef] [PubMed]
- Ung, C.Y.; Li, H.; Kong, C.Y.; Wang, J.F.; Chen, Y.Z. Usefulness of traditionally defined herbal properties for distinguishing prescriptions of traditional Chinese medicine from non-prescription recipes. J. Ethnopharmacol. 2007, 1, 21–28. [Google Scholar] [CrossRef]
- Chen, Z.; Cao, Y.; He, S.; Qiao, Y. Development of models for classification of action between heat-clearing herbs and blood-activating stasis-resolving herbs based on theory of traditional Chinese medicine. Chin. Med.-UK 2018, 13, 12. [Google Scholar] [CrossRef] [PubMed]
- Unschuld, P.U. Huang Di Nei Jing Su Wen: Nature, Knowledge, Imagery in an Ancient Chinese Medical Text: With an Appendix: The Doctrine of the Five Periods and Six Qi in the Huang Di Nei Jing Su Wen; University of California Press: Berkeley, CA, USA, 2003. [Google Scholar]
- Li-Ling, J. Human phenome based on Traditional Chinese Medicine—A solution to congenital syndromology. Am. J. Chin. Med. 2003, 31, 991–1000. [Google Scholar] [CrossRef]
- Member of the Chinese Pharmacopoeia. Pharmacopoeia of the People’s Republic of China; Subdivision of 2015 edition; China Medical Science and Technology Press: Beijing, China, 2015; pp. 274–275. [Google Scholar]
- Ru, J.; Li, P.; Wang, J.; Zhou, W.; Li, B.; Huang, C.; Li, P.; Guo, Z.; Tao, W.; Yang, Y.; et al. TCMSP: A database of systems pharmacology for drug discovery from herbal medicines. J. Cheminform. 2014, 6, 13. [Google Scholar] [CrossRef]
- Liu, Z.; Guo, F.; Wang, Y.; Li, C.; Zhang, X.; Li, H.; Diao, L.; Gu, J.; Wang, W.; Li, D.; et al. BATMAN-TCM: A Bioinformatics Analysis Tool for Molecular mechANism of Traditional Chinese Medicine. Sci. Rep.-UK 2016, 6, 21146. [Google Scholar] [CrossRef]
- Zhang, R.; Yu, S.; Bai, H.; Ning, K. TCM-Mesh: The database and analytical system for network pharmacology analysis for TCM preparations. Sci. Rep.-UK 2017, 7, 2821. [Google Scholar] [CrossRef]
- Chen, C.Y.C. TCM Database@ Taiwan: The world’s largest traditional Chinese medicine database for drug screening in silico. PLoS ONE 2011, 6, e15939. [Google Scholar] [CrossRef] [PubMed]
- O’Boyle, N.M. Towards a Universal SMILES representation-A standard method to generate canonical SMILES based on the InChI. J. Cheminform. 2012, 4, 22. [Google Scholar] [CrossRef] [PubMed]
- Yap, C.W. PaDEL-descriptor: An open source software to calculate molecular descriptors and fingerprints. J. Comput. Chem. 2011, 32, 1466–1474. [Google Scholar] [CrossRef] [PubMed]
- Wishart, D.S.; Feunang, Y.D.; Guo, A.C.; Lo, E.J.; Marcu, A.; Grant, J.R.; Assempour, N. DrugBank 5.0: A major update to the DrugBank database for 2018. Nucleic Acids Res. 2017, 46, D1074–D1082. [Google Scholar] [CrossRef] [PubMed]
- Knox, C.; Law, V.; Jewison, T.; Liu, P.; Ly, S.; Frolkis, A.; Pon, A.; Banco, K.; Mak, C.; Neveu, V.; et al. DrugBank 3.0: A comprehensive resource for ‘Omics’ research on drugs. Nucleic Acids Res. 2010, 39, D1035–D1041. [Google Scholar] [CrossRef] [PubMed]
- Szklarczyk, D.; Santos, A.; von Mering, C.; Jensen, L.J.; Bork, P.; Kuhn, M. STITCH 5: Augmenting protein–chemical interaction networks with tissue and affinity data. Nucleic Acids Res. 2016, 44, D380–D384. [Google Scholar] [CrossRef]
- Gaulton, A.; Bellis, L.J.; Bento, A.P.; Chambers, J.; Davies, M.; Hersey, A.; Light, Y.; McGlinchey, S.; Michalovich, D.; Al-Lazikani, B.; et al. ChEMBL: A large-scale bioactivity database for drug discovery. Nucleic Acids Res. 2011, 40, D1100–D1107. [Google Scholar] [CrossRef]
- Liu, T.; Lin, Y.; Wen, X.; Jorissen, R.N.; Gilson, M.K. BindingDB: A web-accessible database of experimentally determined protein–ligand binding affinities. Nucleic Acids Res. 2006, 35, D198–D201. [Google Scholar] [CrossRef]
- Gilson, M.K.; Liu, T.; Baitaluk, M.; Nicola, G.; Hwang, L.; Chong, J. BindingDB in 2015: A public database for medicinal chemistry, computational chemistry and systems pharmacology. Nucleic Acids Res. 2015, 44, D1045–D1053. [Google Scholar] [CrossRef]
- National Health and Family Planning Commission of PRC. Food safety national standard for use of food additives. In GB2760—2014; National Health and Family Planning Commission of PRC, Ed.; Beijing, China, 2014; p. 279. [Google Scholar]
- Adams, T.B.; Cohen, S.M.; Doull, J.; Feron, V.J.; Goodman, J.I.; Marnett, L.J.; Wagner, B.M. The FEMA GRAS assessment of cinnamyl derivatives used as flavor ingredients. Food Chem. Toxicol. 2004, 42, 157–185. [Google Scholar] [CrossRef]
- Garg, N.; Sethupathy, A.; Tuwani, R.; Dokania, S.; Iyer, A.; Gupta, A.; Badhwar, R. FlavorDB: A database of flavor molecules. Nucleic Acids Res. 2017, 46, D1210–D1216. [Google Scholar] [CrossRef] [PubMed]
- John, R.; Owen, I.T.N.J. Visualization of Molecular Fingerprints. J. Chem. Inf. Model. 2011, 51, 1552–1563. [Google Scholar]
- Monev, V. Introduction to similarity searching in chemistry. MATCH Commun. Math. Comput. Chem. 2004, 51, 7–38. [Google Scholar]
- Zhu, F.; Shi, Z.; Qin, C.; Tao, L.; Liu, X.; Xu, F.; Han, B. Therapeutic target database update 2012: A resource for facilitating target-oriented drug discovery. Nucleic Acids Res. 2011, 40, D1128–D1136. [Google Scholar] [CrossRef]
- Chen, X.; Ji, Z.L.; Chen, Y.Z. TTD: Therapeutic target database. Nucleic Acids Res. 2002, 30, 412–415. [Google Scholar] [CrossRef] [PubMed]
- Davis, A.P.; Grondin, C.J.; Johnson, R.J.; Sciaky, D.; McMorran, R.; Wiegers, J.; Mattingly, C.J. The comparative toxicogenomics database: Update 2019. Nucleic Acids Res. 2018, 47, D948–D954. [Google Scholar] [CrossRef]
- Kanehisa, M.; Goto, S.; Sato, Y.; Furumichi, M.; Tanabe, M. KEGG for integration and interpretation of large-scale molecular data sets. Nucleic Acids Res. 2011, 40, D109–D114. [Google Scholar] [CrossRef]
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Bioinformatics enrichment tools: Paths toward the comprehensive functional analysis of large gene lists. Nucleic Acids Res. 2009, 37, 1–13. [Google Scholar] [CrossRef]
- Shannon, P.; Markiel, A.; Ozier, O.; Baliga, N.S.; Wang, J.T.; Ramage, D.; Ideker, T. Cytoscape: A software environment for integrated models of biomolecular interaction networks. Genome Res. 2003, 13, 2498–2504. [Google Scholar] [CrossRef]
- Scardoni, G.; Petterlini, M.; Laudanna, C. Analyzing biological network parameters with CentiScaPe. Bioinformatics 2009, 25, 2857–2859. [Google Scholar] [CrossRef]
 : molecular interaction or relation (inhibition).
: molecular interaction or relation (inhibition).  : Phosphorylation,
: Phosphorylation,  : dephosphorylation.
: dephosphorylation.
 : molecular interaction or relation (inhibition).
: molecular interaction or relation (inhibition).  : Phosphorylation,
: Phosphorylation,  : dephosphorylation.
: dephosphorylation.
 : molecular interaction or relation (inhibition).
: molecular interaction or relation (inhibition).  : Phosphorylation,
: Phosphorylation,  : dephosphorylation.
: dephosphorylation.
 : molecular interaction or relation (inhibition).
: molecular interaction or relation (inhibition).  : Phosphorylation,
: Phosphorylation,  : dephosphorylation.
: dephosphorylation.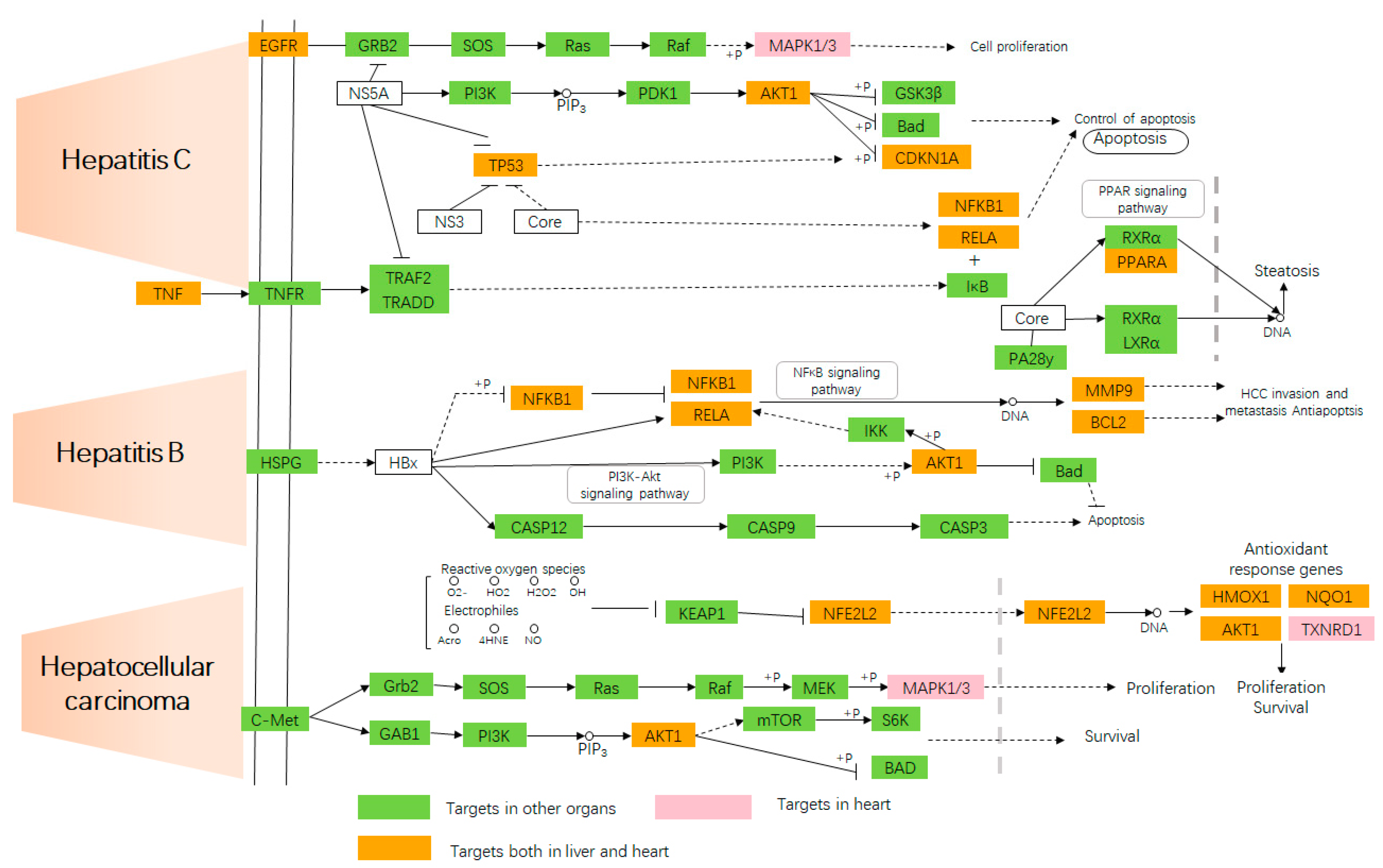
| Latin Name | English Name | Chinese Name | Flavor |
|---|---|---|---|
| Piper Nigrum | Black pepper | Heihujiao | Fragrant smelling, taste pungent |
| Syzygium aromaticum | Cloves | Dingxiang | Strong fragrant smelling, taste pungent and feeling on tongue |
| Trigonella foenum-graecum L. | Fenugreek | Huluba | Sweet smelling aroma; fragrant |
| Menthae Haplocalycis Herba | Peppermint | Bohe | Cool and fragrant smelling, taste pungent |
| Zanthoxyli Pericarpium | Pepper | Huajiao | Fragrant smelling; taste sweet and pungent |
| Myristicae Semen | Nutmeg | Roudoukou | Strong fragrant smelling, taste pungent |
| Aucklandiae Radix | Common vladimiria root | Muxiang | Specific fragrant smelling, taste slightly bitter |
| Origanum vulgare L. | Oregano | Niuzhi | Taste pungent and slightly bitter |
| Thymus mongolicusRonn | Thyme | Bailixiang | Taste pungent |
| Cinnamomt Cortex | Cinnamon | Rougui | Strong fragrant smelling, taste sweet and pungent |
| Rosmarinus officinalis | Rosemary | Midiexiang | Pungent |
| Allii Sativi Bulbus | Garlic | Dasuan | Specific smelling, taste pungent |
| Zingiberis Rhizoma Recens | Ginger | Shengjiang | Specific fragrant smelling, taste pungent |
| Alpiniae Officinarum Rhizoma | Galangal | Gaoliangjiang | Fragrant smelling, taste pungent |
| Latin Name | Chinese Herb | Diversity | No. Entries | Descriptor |
|---|---|---|---|---|
| Chuanxiong Rhizome | ChuanXiong | 0.6678 | 352 | TCMID, TCMSP, BATMAN-TCM, TCM Database@Taiwan, TCM-Mesh |
| Carthami Flos | HongHua | 0.7011 | 272 | |
| Dalbergiae Odoriferae Lignum | JiangXiang | 0.5255 | 169 | |
| Foeniculi Fructus | XiaoHuiXiang | 0.6495 | 74 | |
| Angelicae Sinensis Radix | DangGui | 0.7116 | 292 |
| Latin Name | Herbs | Molecule Name | FEMA or PubChem ID |
|---|---|---|---|
| Chuanxiong Rhizome | ChuanXiong | Ethylpalmitate | 2451 |
| Trimethylamine | 3241 | ||
| Vanillin | 3107 | ||
| Nonanal | 2782 | ||
| 1,8-cineole | 2465 | ||
| Myrcene | 2762 | ||
| Methyleugenol | 2475 | ||
| hexanal | 2557 | ||
| thymol | 3066 | ||
| trans-2-Nonen-1-ol | 3379 | ||
| P-Cymene | 2356 | ||
| Borneol | 2157 | ||
| Naphthalene | pubchem_931 | ||
| Phenylacetaldehyde | 2874 | ||
| O-Cresol | 3480 | ||
| Linalool | 2635 | ||
| Camphene | 2229 | ||
| Linoleic Acid | 3380 | ||
| 3-Carene | 3821 | ||
| 2-pentylfuran | 3317 | ||
| Angelicae Sinensis Radix | DangGui | Camphene | 2229 |
| Carvacrol | 2245 | ||
| o-Cresol | 3480 | ||
| p-Cresol | 2337 | ||
| p-Ethylphenol | 3156 | ||
| Guaiacol | 2532 | ||
| Isoeugenol | 2468 | ||
| Phenol | 3223 | ||
| Vanillin | 3107 | ||
| Nonanal | 2782 | ||
| Myrcene | 2762 | ||
| naphthalene | pubchem_931 | ||
| 1-hexadecanol | 2554 | ||
| m-cresol | 3530 | ||
| 3-carene | 3821 | ||
| Carthami Flos | HongHua | Benzothiazole | 3256 |
| 3-Hexanol | 3351 | ||
| Phenylacetaldehyde | 2874 | ||
| Nonanal | 2782 | ||
| Methylcinnamate | 2698 | ||
| carvacrol | 2245 | ||
| Isovalericacid | 3102 | ||
| stearicacid | 3035 | ||
| Dalbergiae Odoriferae Lignum | JiangXiang | 1,8-cineole | 2465 |
| indole | 2593 | ||
| Ethylpalmitate | 2451 | ||
| Foeniculi Fructus | XiaoHuiXiang | D-Limonene | 2633 |
| α-Terpinene | 3558 | ||
| Terpinolene | 3046 | ||
| 1,8-cineole | 2465 | ||
| Myrcene | 2762 | ||
| acetaldehyde | 2003 | ||
| d-fenchone | 2479 |
| Target | UniProt ID | Gene Name | Related Diseases | Degree |
|---|---|---|---|---|
| NFE2L2 | Q16236 | Nuclear factor erythroid 2-related factor 2 | Atherosclerosis, hypertension, Heart failure | 16 |
| AR | P10275 | Androgen Receptor | Atherosclerosis, Coronary artery disease, Hypertension, Myocardial infarction | 15 |
| PTGS2 | P35354 | Prostaglandin G/H synthase 2 | Pain, Coronary heart disease, Myocardial infarction, Vascular lesion regression | 13 |
| TNF | P01375 | Tumor necrosis factor | Heart failure, Hepatitis C infection, Hypertension, Coronary atherosclerosis, Cirrhosis | 13 |
| NFKB1 | P19838 | Nuclear factor NF-kappa-B p105 subunit | Atherosclerosis, Cancer, Type 2 diabetes, Hepatitis C, Chronic Liver cirrhosis | 12 |
| PPARD | Q03181 | Peroxisome proliferator-activated receptor delta | Central nervous system disease, Metabolic disorders, Atherosclerosis, Coronary; Coronary heart disease | 12 |
| CYP1A1 | P04798 | Cytochrome P450 1A1 | Liver cancer; Liver disease, Coronary artery disease, Hypertension | 12 |
| JUN | P05412 | Proto-oncogene c-JUN | Heart failure, Breast cancer | 11 |
| CYP2E1 | P05181 | Cytochrome P450 2E1 | Liver cancer; Liver disease | 10 |
| AHR | P35869 | Aryl hydrocarbon receptor | Multiple myeloma, HIV infections, Myocardial infarction | 10 |
| CAT | P04040 | Catalase | Skin burns, Heart failure | 10 |
| ALB | P02768 | Serum albumin | Hemophilia’ Visualizing lesions with abnormal blood brain barrier | 10 |
| PTGS1 | P23219 | Cyclooxygenase-1 | Platelet function, Myocardial Infarction, Acute coronary syndrome | 9 |
| CXCL8 | P10145 | Interleukin-8 | Melanoma. Autoimmune diabetes | 9 |
| MMP9 | P14780 | Matrix metalloproteinase 9 | Atherosclerosis, Carotid, Colorectal cancer | 9 |
| HMOX1 | P09601 | Heme oxygenase 1 | Myocardial infarction, Atherosclerosis, Coronary, Cardiovascular disease, Ischemic injury of the liver, Inflammation, Cerebral vasospasm | 9 |
| IL1B | P01584 | Interleukin-1 β | Coronary disease, Hypertension, Chronic hepatitis C | 8 |
| IL6 | P05231 | Interleukin-6 | Arteriosclerosis, Heart valve diseases, Hepatitis B | 8 |
| ACE | P22303 | Acetylcholinesterase | Chronic heart failure, Hypertension | 8 |
| HSPB1 | P04792 | Heat shock protein β-1 | Breast cancer. Ovarian cancer, Bladder cancer, Prostate cancer, Lung cancer, Arthritis | 8 |
| Latin Name | Chinese Herb | TCM-HPs |
|---|---|---|
| Chuanxiong Rhizome | ChuanXiong | Warm, pungent, liver, gallbladder, pericardium meridian tropism |
| Carthami Flus | HongHua | Warm, pungent, liver, heart meridian tropism |
| Dalbergiae Odoriferae Lignum | JiangXiang | Warm, pungent, liver, spleen meridian tropism |
| Foeniculi Fructus | XiaoHuiXiang | Warm, pungent, liver, kidney, spleen, stomach meridian tropism |
| Angelicae Sinensis Radix | DangGui | Warm, pungent, liver, heart, spleen meridian tropism |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, Z.; Cao, Y.; Zhang, Y.; Qiao, Y. A Novel Discovery: Holistic Efficacy at the Special Organ Level of Pungent Flavored Compounds from Pungent Traditional Chinese Medicine. Int. J. Mol. Sci. 2019, 20, 752. https://doi.org/10.3390/ijms20030752
Chen Z, Cao Y, Zhang Y, Qiao Y. A Novel Discovery: Holistic Efficacy at the Special Organ Level of Pungent Flavored Compounds from Pungent Traditional Chinese Medicine. International Journal of Molecular Sciences. 2019; 20(3):752. https://doi.org/10.3390/ijms20030752
Chicago/Turabian StyleChen, Zhao, Yanfeng Cao, Yanling Zhang, and Yanjiang Qiao. 2019. "A Novel Discovery: Holistic Efficacy at the Special Organ Level of Pungent Flavored Compounds from Pungent Traditional Chinese Medicine" International Journal of Molecular Sciences 20, no. 3: 752. https://doi.org/10.3390/ijms20030752
APA StyleChen, Z., Cao, Y., Zhang, Y., & Qiao, Y. (2019). A Novel Discovery: Holistic Efficacy at the Special Organ Level of Pungent Flavored Compounds from Pungent Traditional Chinese Medicine. International Journal of Molecular Sciences, 20(3), 752. https://doi.org/10.3390/ijms20030752





