Effective Sequential Combined Chemotherapy with Trifluridine/Tipiracil and Regorafenib in Human Colorectal Cancer Cells
Abstract
:1. Introduction
2. Results
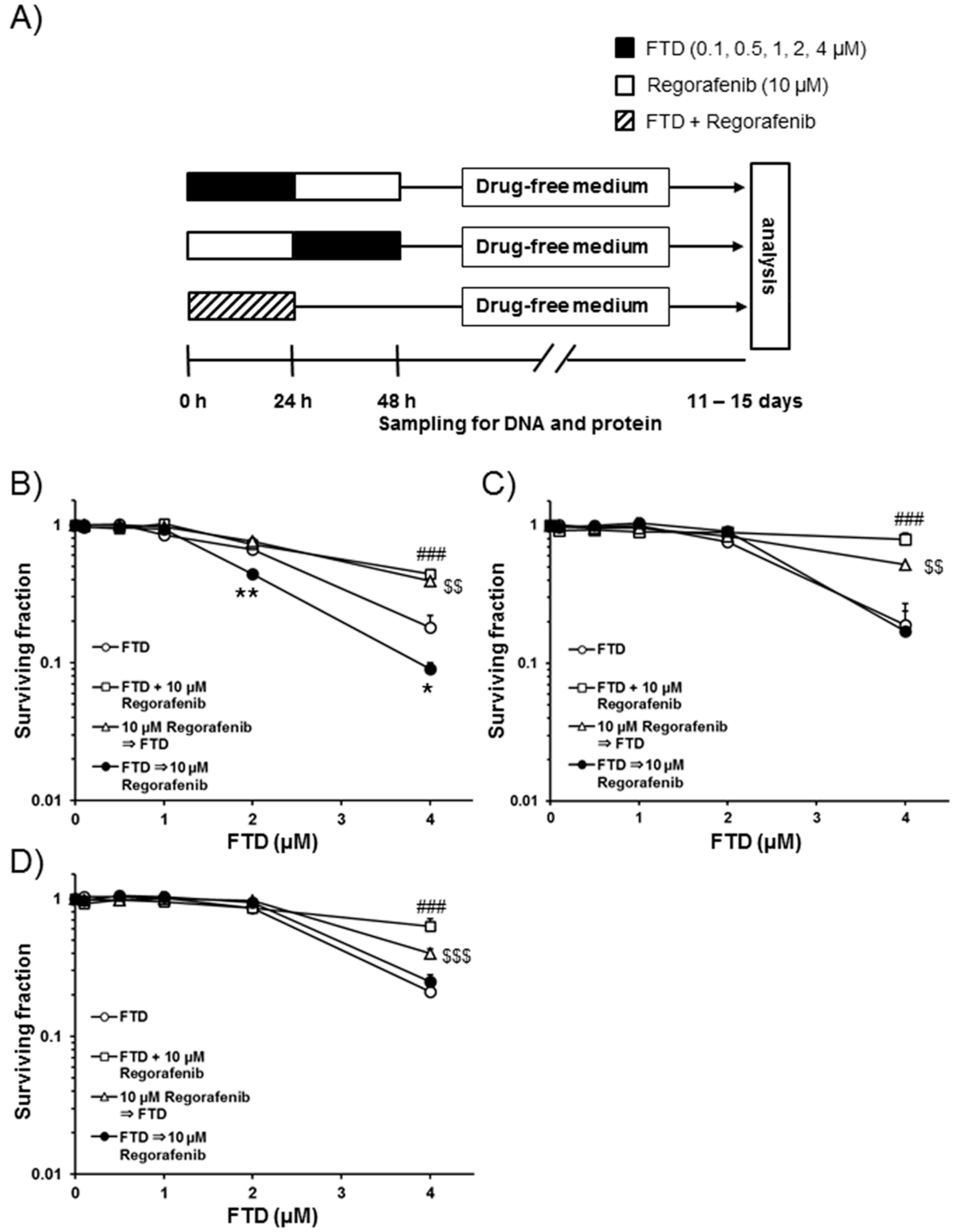
2.1. Cell Death after Sequential Administration of FTD Followed by Regorafenib
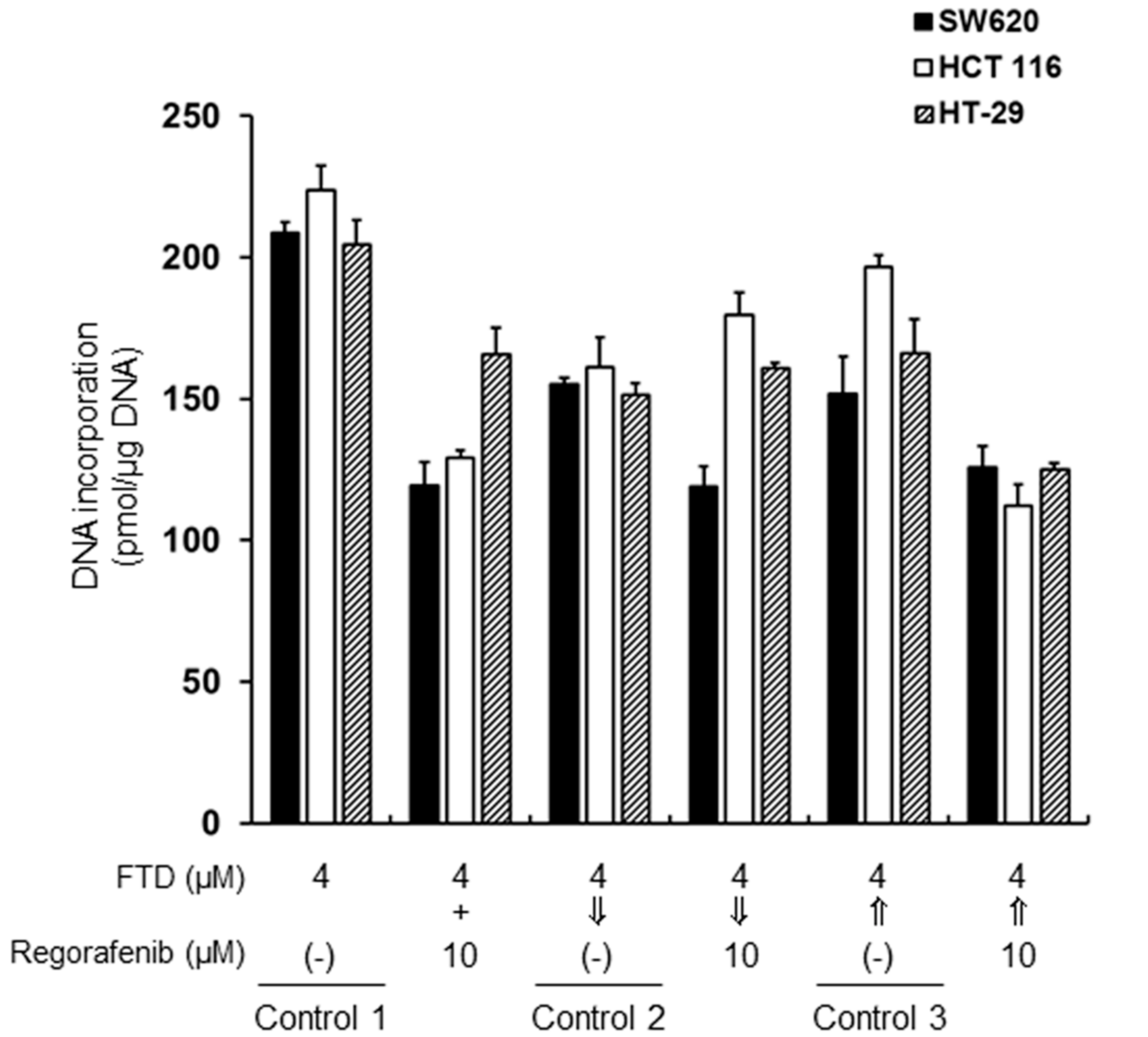
2.2. Incorporation of Trifluridine (FTD) into Genomic DNA
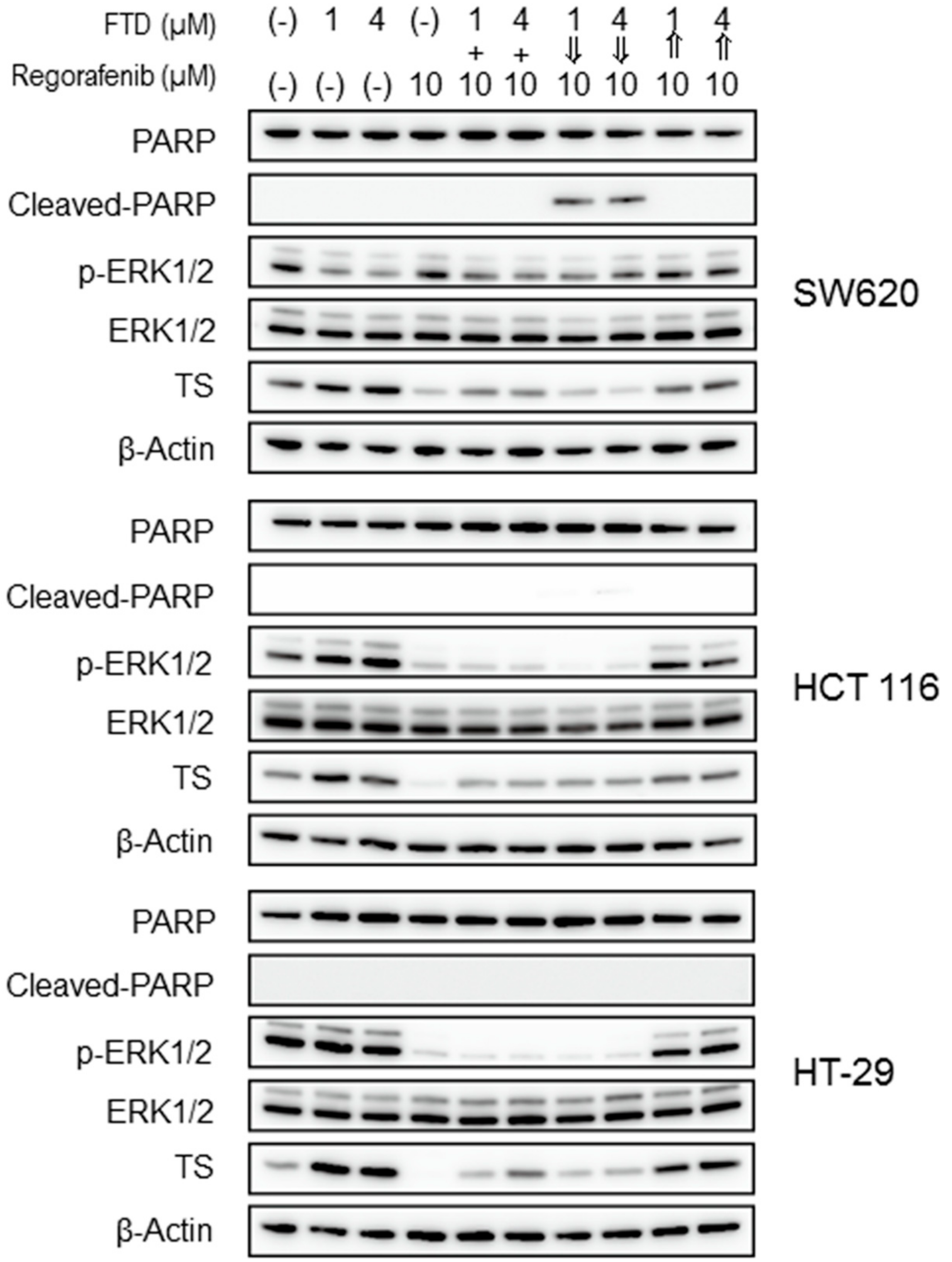
2.3. Drugs and Apoptosis-Related Protein Expression Following the Combination Treatment
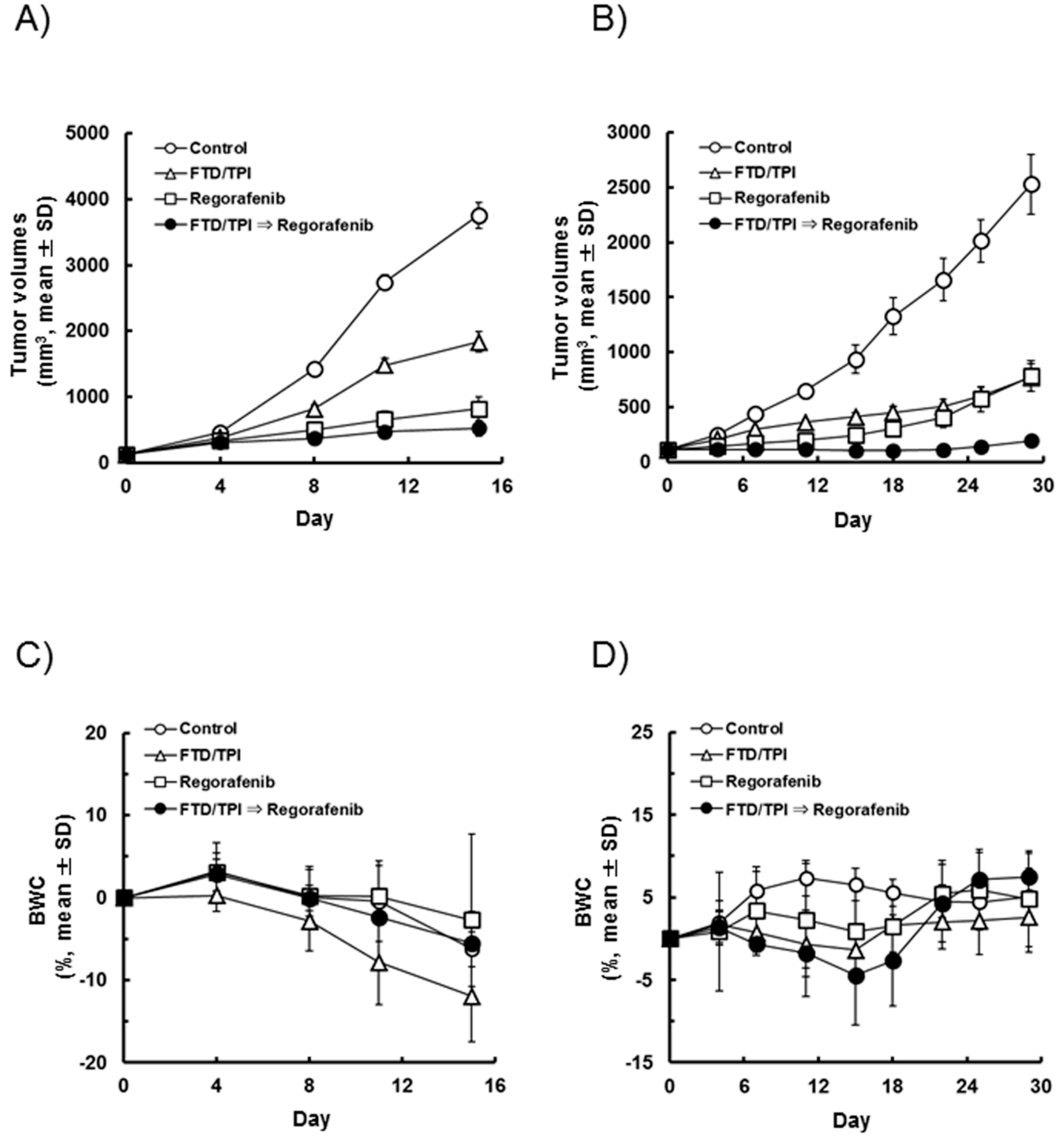
2.4. Administration of Regorafenib Subsequent to FTD/TPI Increases the Antitumor Effect
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Cancer Cell Lines
4.3. Animals
4.4. Clonogenic Cell Survival Assay
4.5. Determination of FTD Incorporation into DNA
4.6. Immunoblotting
4.7. Antitumor Activity In Vivo
4.8. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| BWC | Body weight change |
| CRC | Colorectal cancer |
| DMEM | Dulbecco’s modified Eagle medium |
| ERK | Extracellular signal-related kinase |
| FBS | Fetal bovine serum |
| FTD | Trifluridine |
| LV | Leucovorin |
| PARP | Poly(ADP-ribose) polymerase |
| PE | Plating efficiency |
| p-ERK1/2 | Phosphorylated extracellular signal-related kinase 1/2 |
| RIPA | Radioimmunoprecipitation assay |
| RTV | Relative tumor volume |
| SDS-PAGE | Sodium dodecyl sulfate polyacrylamide gel electrophoresis |
| SF | Surviving fractions |
| TGI | Tumor growth inhibition |
| TS | Thymidylate synthase |
| TPI | Tipiracil hydrochloride |
| 5-FU | 5-Fluorouracil |
References
- Gustavsson, B.; Carlsson, G.; Machover, D.; Petrelli, N.; Roth, A.; Schmoll, H.J.; Tveit, K.M.; Gibson, F. A review of the evolution of systemic chemotherapy in the management of colorectal cancer. Clin. Colorectal Cancer 2015, 14, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Scheithauer, W.; Rosen, H.; Kornek, G.V.; Sebesta, C.; Depisch, D. Randomised comparison of combination chemotherapy plus supportive care with supportive care alone in patients with metastatic colorectal cancer. BMJ 1993, 306, 752–755. [Google Scholar] [CrossRef] [PubMed]
- Heidelberger, C.; Parsons, D.G.; Remy, D.C. Syntheses of 5-trifluoromethyluracil and 5-trifluoromethyl-2′-deoxyuridine. J. Med. Chem. 1964, 7, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, N.; Sakamoto, K.; Okabe, H.; Fujioka, A.; Yamamura, K.; Nakagawa, F.; Nagase, H.; Yokogawa, T.; Oguchi, K.; Ishida, K.; et al. Repeated oral dosing of TAS-102 confers high trifluridine incorporation into DNA and sustained antitumor activity in mouse models. Oncol. Rep. 2014, 32, 2319–2326. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matsuoka, K.; Iimori, M.; Niimi, S.; Tsukihara, H.; Watanabe, S.; Kiyonari, S.; Kiniwa, M.; Ando, K.; Tokunaga, E.; Saeki, H.; et al. Trifluridine induces p53-dependent sustained G2 phase arrest with its massive misincorporation into DNA and few DNA strand breaks. Mol. Cancer Ther. 2015, 14, 1004–1013. [Google Scholar] [CrossRef] [PubMed]
- Langenbach, R.J.; Danenberg, P.V.; Heidelberger, C. Thymidylate synthetase: Mechanism of inhibition by 5-fluoro-2′-deoxyuridylate. Biochem. Biophys. Res. Commun. 1972, 48, 1565–1571. [Google Scholar] [CrossRef]
- Fukushima, M.; Suzuki, N.; Emura, T.; Yano, S.; Kazuno, H.; Tada, Y.; Yamada, Y.; Asao, T. Structure and activity of specific inhibitors of thymidine phosphorylase to potentiate the function of antitumor 2′-deoxyribonucleosides. Biochem. Pharmacol. 2000, 59, 1227–1236. [Google Scholar] [CrossRef]
- Mayer, R.J.; Van Cutsem, E.; Falcone, A.; Yoshino, T.; Garcia-Carbonero, R.; Mizunuma, N.; Yamazaki, K.; Shimada, Y.; Tabernero, J.; Komatsu, Y.; et al. Randomized trial of TAS-102 for refractory metastatic colorectal cancer. N. Engl. J. Med. 2015, 372, 1909–1919. [Google Scholar] [CrossRef] [PubMed]
- Wilhelm, S.M.; Dumas, J.; Adnane, L.; Lynch, M.; Carter, C.A.; Schutz, G.; Thierauch, K.H.; Zopf, D. Regorafenib (BAY 73-4506): A new oral multikinase inhibitor of angiogenic, stromal and oncogenic receptor tyrosine kinases with potent preclinical antitumor activity. Int. J. Cancer 2011, 129, 245–255. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grothey, A.; Van Cutsem, E.; Sobrero, A.; Siena, S.; Falcone, A.; Ychou, M.; Humblet, Y.; Bouche, O.; Mineur, L.; Barone, C.; et al. Regorafenib monotherapy for previously treated metastatic colorectal cancer (CORRECT): An international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet 2013, 381, 303–312. [Google Scholar] [CrossRef]
- Masuishi, T.; Taniguchi, H.; Hamauchi, S.; Komori, A.; Kito, Y.; Narita, Y.; Tsushima, T.; Ishihara, M.; Todaka, A.; Tanaka, T.; et al. Regorafenib versus trifluridine/tipiracil for refractory metastatic colorectal cancer: A retrospective comparison. Clin. Colorectal Cancer 2017, 16, e15–e22. [Google Scholar] [CrossRef] [PubMed]
- Moriwaki, T.; Fukuoka, S.; Taniguchi, H.; Takashima, A.; Kumekawa, Y.; Kajiwara, T.; Yamazaki, K.; Esaki, T.; Makiyama, C.; Denda, T.; et al. Propensity score analysis of regorafenib versus trifluridine/tipiracil in patients with metastatic colorectal cancer refractory to standard chemotherapy (REGOTAS): A Japanese Society for Cancer of the Colon and Rectum Multicenter Observational Study. Oncologist 2018, 23, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Schmieder, R.; Hoffmann, J.; Becker, M.; Bhargava, A.; Muller, T.; Kahmann, N.; Ellinghaus, P.; Adams, R.; Rosenthal, A.; Thierauch, K.H.; et al. Regorafenib (BAY 73-4506): Antitumor and antimetastatic activities in preclinical models of colorectal cancer. Int. J. Cancer 2014, 135, 1487–1496. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bijnsdorp, I.V.; Kruyt, F.A.; Fukushima, M.; Smid, K.; Gokoel, S.; Peters, G.J. Molecular mechanism underlying the synergistic interaction between trifluorothymidine and the epidermal growth factor receptor inhibitor erlotinib in human colorectal cancer cell lines. Cancer Sci. 2010, 101, 440–447. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baba, Y.; Tamura, T.; Satoh, Y.; Gotou, M.; Sawada, H.; Ebara, S.; Shibuya, K.; Soeda, J.; Nakamura, K. Panitumumab interaction with TAS-102 leads to combinational anticancer effects via blocking of EGFR-mediated tumor response to trifluridine. Mol. Oncol. 2017, 11, 1065–1077. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peters, G.J.; Backus, H.H.; Freemantle, S.; van Triest, B.; Codacci-Pisanelli, G.; van der Wilt, C.L.; Smid, K.; Lunec, J.; Calvert, A.H.; Marsh, S.; et al. Induction of thymidylate synthase as a 5-fluorouracil resistance mechanism. Biochim. Biophys. Acta 2002, 1587, 194–205. [Google Scholar] [CrossRef]
- Kitao, H.; Morodomi, Y.; Niimi, S.; Kiniwa, M.; Shigeno, K.; Matsuoka, K.; Kataoka, Y.; Iimori, M.; Tokunaga, E.; Saeki, H.; et al. The antibodies against 5-bromo-2′-deoxyuridine specifically recognize trifluridine incorporated into DNA. Sci. Rep. 2016, 6, 25286. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, W.J.; Li, Y.; Wei, M.N.; Chen, Y.; Qiu, J.G.; Jiang, Q.W.; Yang, Y.; Zheng, D.W.; Qin, W.M.; Huang, J.R.; et al. Synergistic antitumor activity of regorafenib and lapatinib in preclinical models of human colorectal cancer. Cancer Lett. 2017, 386, 100–109. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, A.; Shiota, M.; Tatsugami, K.; Yokomizo, A.; Eto, M.; Inokuchi, J.; Kuroiwa, K.; Kiyoshima, K.; Naito, S. Sorafenib augments cytotoxic effect of S-1 in vitro and in vivo through TS suppression. Cancer Chemother. Pharmacol. 2011, 68, 1557–1564. [Google Scholar] [CrossRef] [PubMed]
- Zhai, J.M.; Yin, X.Y.; Lai, Y.R.; Hou, X.; Cai, J.P.; Hao, X.Y.; Liang, L.J.; Zhang, L.J. Sorafenib enhances the chemotherapeutic efficacy of S-1 against hepatocellular carcinoma through downregulation of transcription factor E2F-1. Cancer Chemother. Pharmacol. 2013, 71, 1255–1264. [Google Scholar] [CrossRef] [PubMed]
- Matsuoka, K.; Nakagawa, F.; Kobunai, T.; Takechi, T. Trifluridine/tipiracil overcomes the resistance of human gastric 5-fluorouracil-refractory cells with high thymidylate synthase expression. Oncotarget 2018, 9, 13438–13450. [Google Scholar] [CrossRef] [PubMed]
- Nukatsuka, M.; Saito, H.; Nakagawa, F.; Tsujimoto, H.; Sakamoto, K.; Tsukioka, S.; Uchida, J.; Kiniwa, M.; Kobunai, T.; Takechi, T. Combination therapy using oral S-1 and targeted agents against human tumor xenografts in nude mice. Exp. Ther. Med. 2012, 3, 755–762. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Emura, T.; Nakagawa, F.; Fujioka, A.; Ohshimo, H.; Yokogawa, T.; Okabe, H.; Kitazato, K. An optimal dosing schedule for a novel combination antimetabolite, TAS-102, based on its intracellular metabolism and its incorporation into DNA. Int. J. Mol. Med. 2004, 13, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Emura, T.; Suzuki, N.; Fujioka, A.; Ohshimo, H.; Fukushima, M. Potentiation of the antitumor activity of alpha, alpha, alpha-trifluorothymidine by the co-administration of an inhibitor of thymidine phosphorylase at a suitable molar ratio in vivo. Int. J. Oncol. 2005, 27, 449–455. [Google Scholar] [CrossRef] [PubMed]
- Bauer, P.; Röhmel, J.; Maurer, W.; Hothorn, L. Testing strategies in multi-dose experiments including active control. Stat. Med. 1998, 17, 2133–2146. [Google Scholar] [CrossRef]
| Group | Dose (mg/kg/day) | Treatment | n | RTV(mean ± SD) | TGI(%) |
|---|---|---|---|---|---|
| Control | - | - | 6 | 29.94 ± 3.40 | - |
| FTD/TPI | 150 | Day 1–14 (b.i.d.) | 6 | 14.57 ± 1.44 a | 51.3 |
| Regorafenib | 10 | Day 1–14 (q.d.) | 6 | 6.55 ± 1.34 a | 78.1 |
| FTD/TPI followed by Regorafenib | 150 + 10 | - | 6 | 4.07 ± 0.56 a,b | 86.4 |
| Group | Dose (mg/kg/day) | Treatment | n | RTV(mean ± SD) | TGI(%) |
|---|---|---|---|---|---|
| Control | - | - | 6 | 22.83 ± 2.07 | - |
| FTD/TPI | 150 | Day 1–14 (b.i.d.) | 6 | 7.02 ± 1.02 a | 69.3 |
| Regorafenib | 10 | Day 1–14 (q.d.) | 6 | 7.09 ± 1.05 a | 68.9 |
| FTD/TPI followed by Regorafenib | 150 + 10 | - | 6 | 1.76 ± 0.29 a,b | 92.3 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matsuoka, K.; Nakagawa, F.; Tanaka, N.; Okabe, H.; Matsuo, K.; Takechi, T. Effective Sequential Combined Chemotherapy with Trifluridine/Tipiracil and Regorafenib in Human Colorectal Cancer Cells. Int. J. Mol. Sci. 2018, 19, 2915. https://doi.org/10.3390/ijms19102915
Matsuoka K, Nakagawa F, Tanaka N, Okabe H, Matsuo K, Takechi T. Effective Sequential Combined Chemotherapy with Trifluridine/Tipiracil and Regorafenib in Human Colorectal Cancer Cells. International Journal of Molecular Sciences. 2018; 19(10):2915. https://doi.org/10.3390/ijms19102915
Chicago/Turabian StyleMatsuoka, Kazuaki, Fumio Nakagawa, Nozomu Tanaka, Hiroyuki Okabe, Kenichi Matsuo, and Teiji Takechi. 2018. "Effective Sequential Combined Chemotherapy with Trifluridine/Tipiracil and Regorafenib in Human Colorectal Cancer Cells" International Journal of Molecular Sciences 19, no. 10: 2915. https://doi.org/10.3390/ijms19102915




