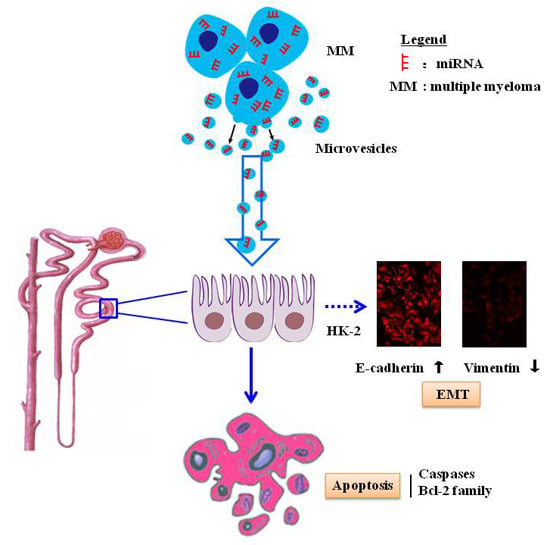
Tumor Cell-Derived Microvesicles Induced Not Epithelial-Mesenchymal Transition but Apoptosis in Human Proximal Tubular (HK-2) Cells: Implications for Renal Impairment in Multiple Myeloma
Abstract
:1. Introduction
2. Results
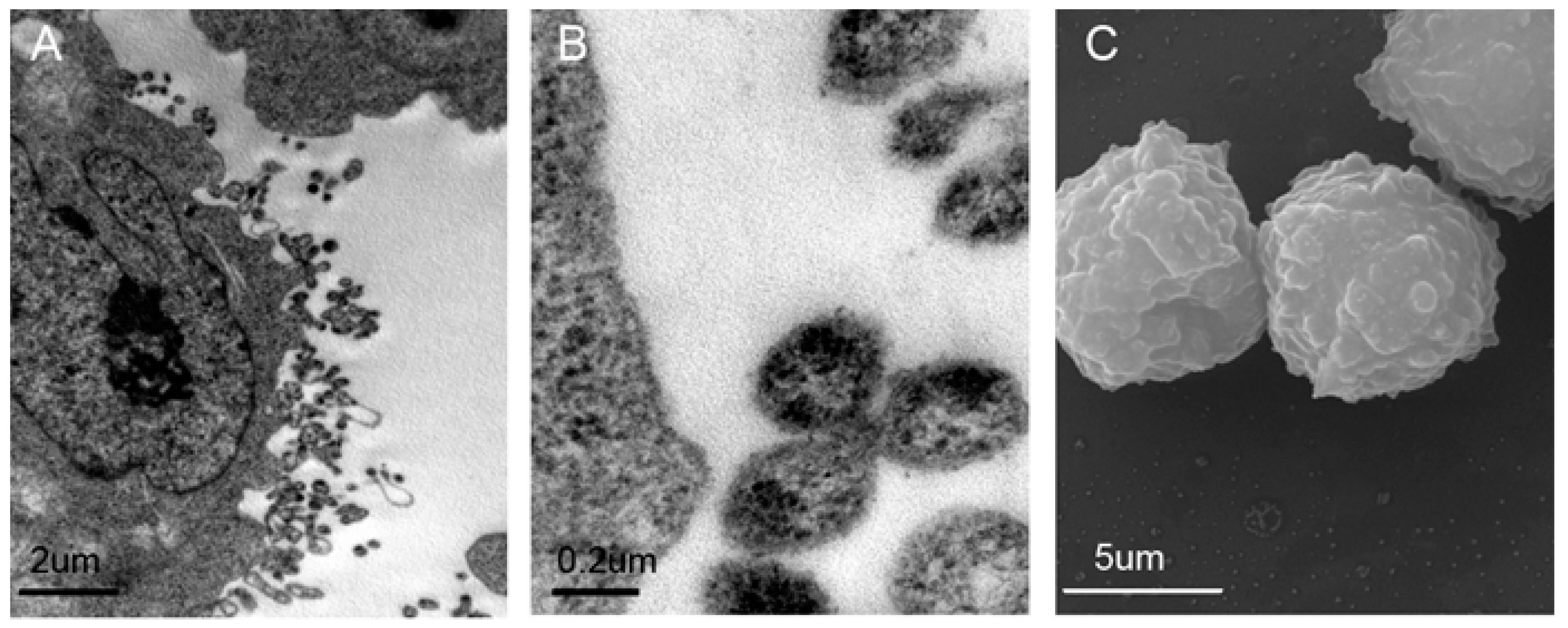
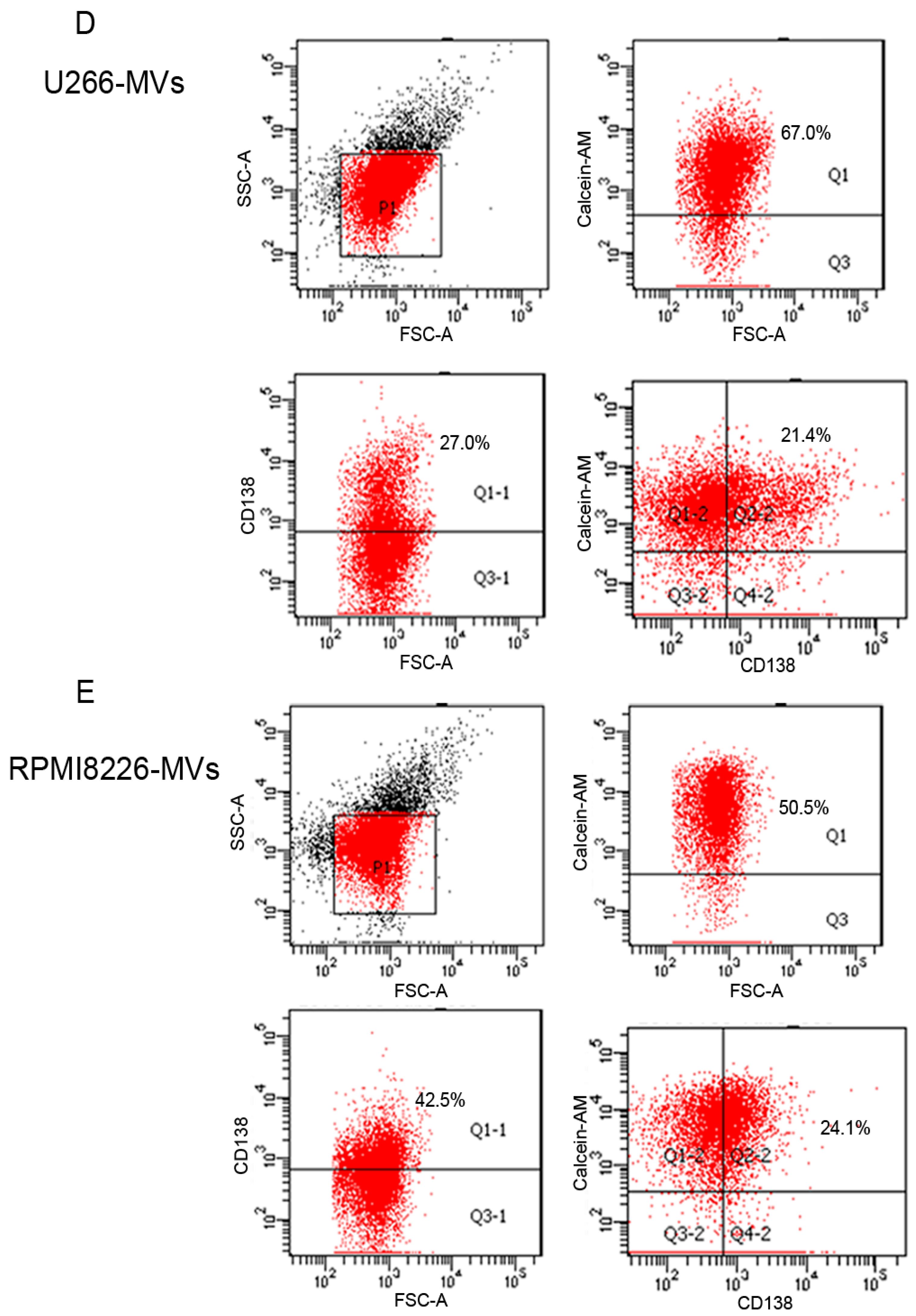
2.1. Validation and Characteristics of Myeloma Cell-Derived Microvesicles ( MM-MVs)
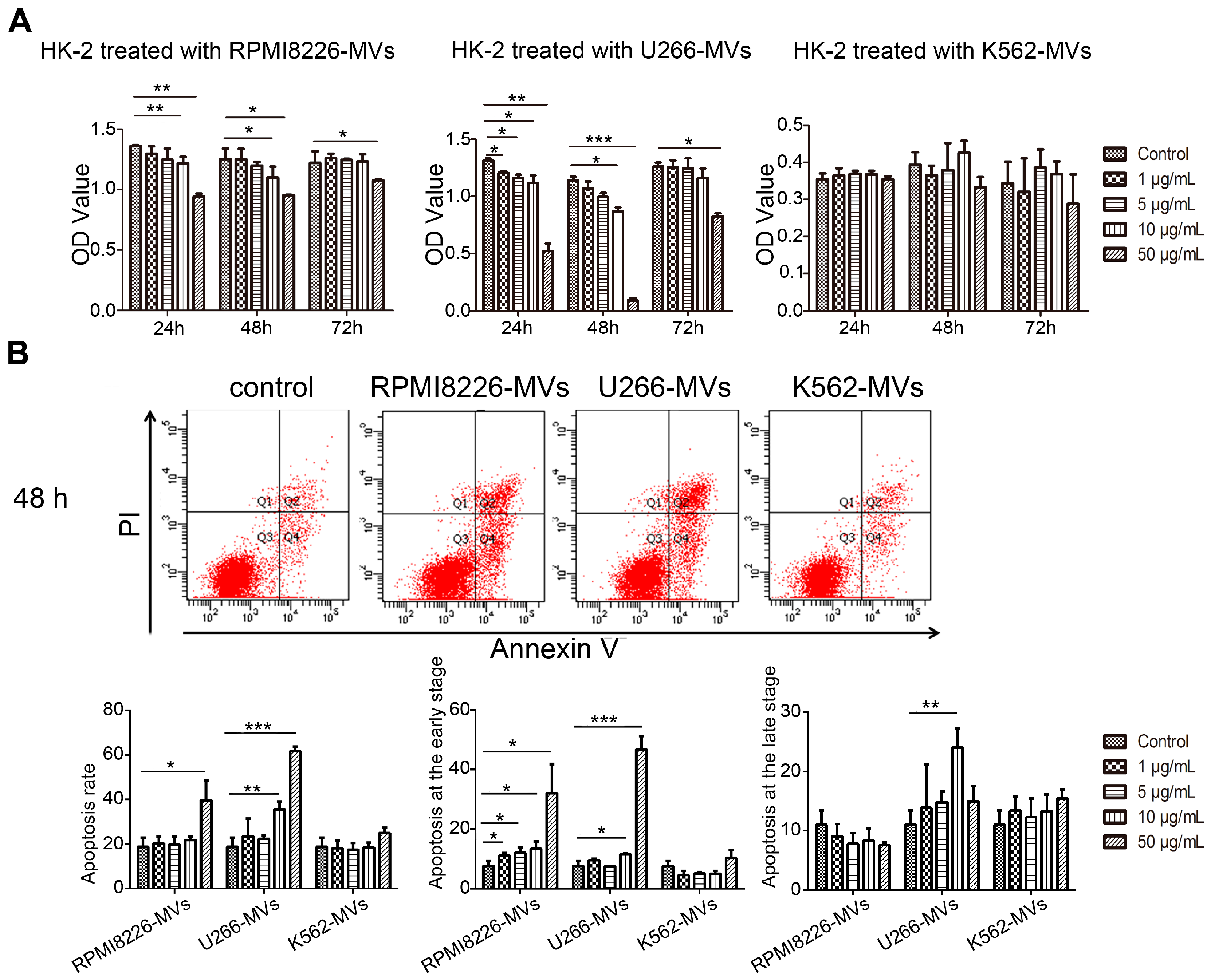
2.2. MM-MVs Inhibit Viability and Induce Apoptosis in Human Kidney-2 Cells (HK-2 Cells)
2.3. MM-MVs Activate Apoptic Pathways of Caspase-3, -8, -9 and Bcl-2 Family Members
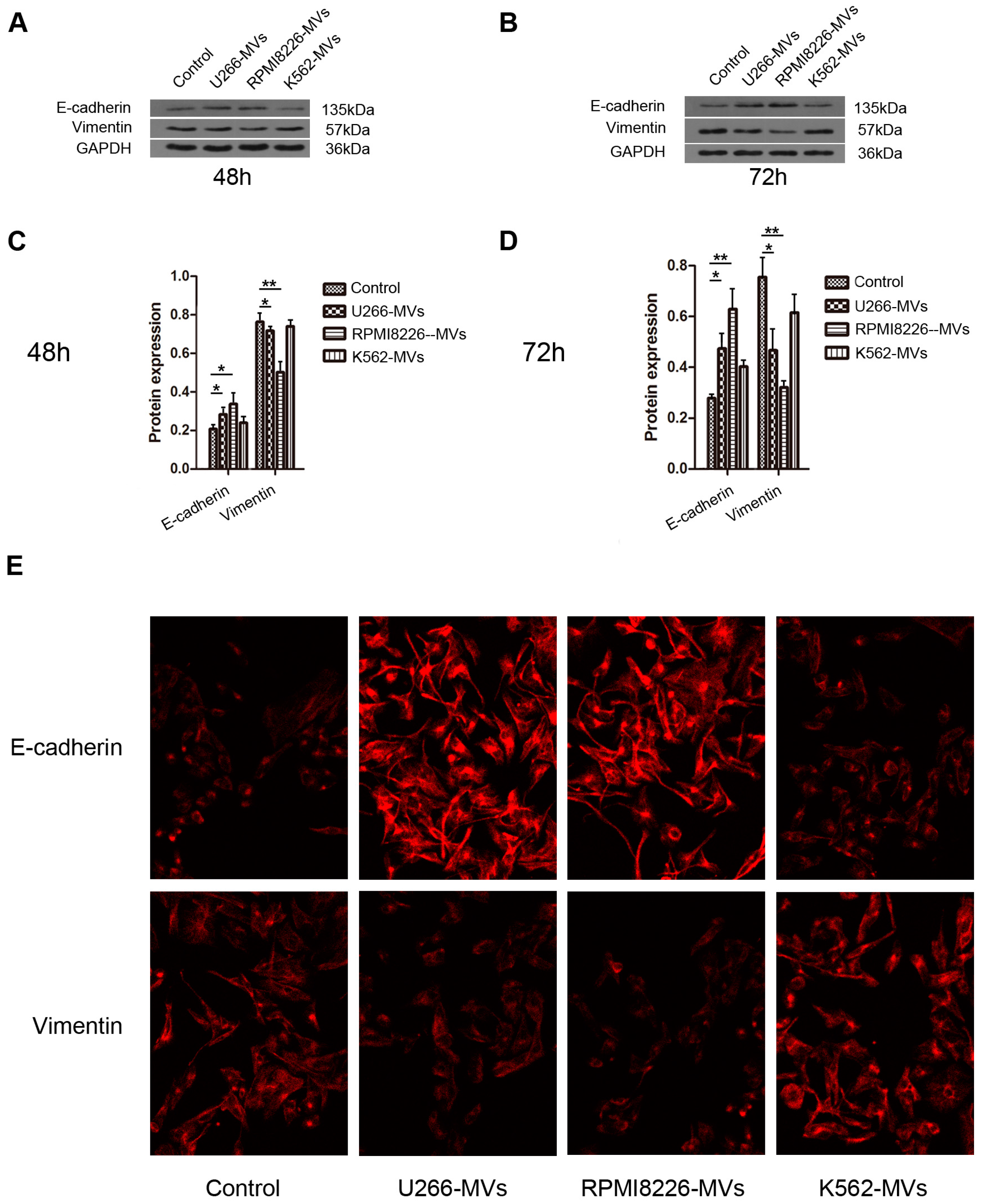
2.4. MM-MVs Up-Regulate E-Cadherin Protein and Down-Regulate Vimentin Protein in HK-2 Cells
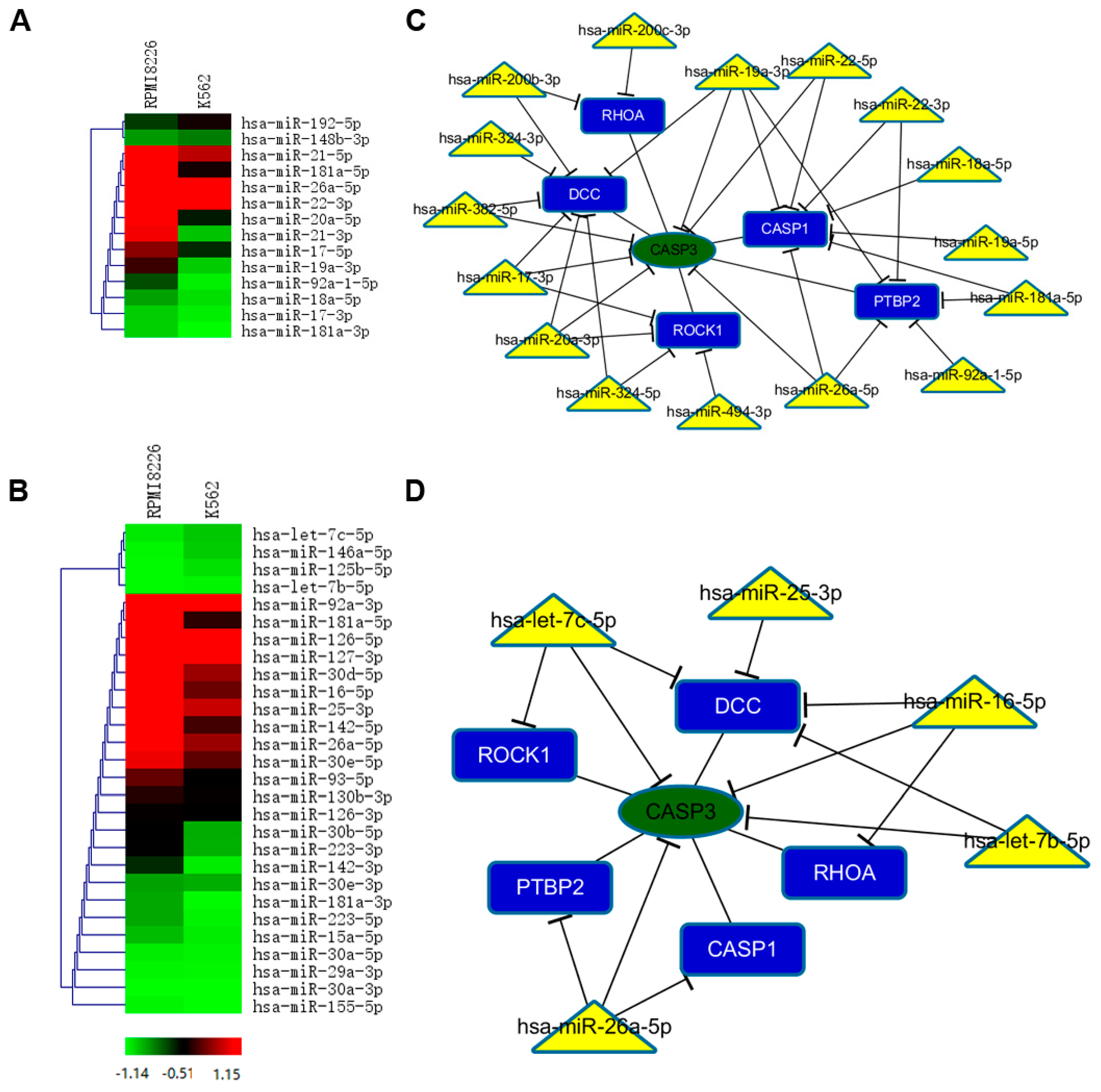
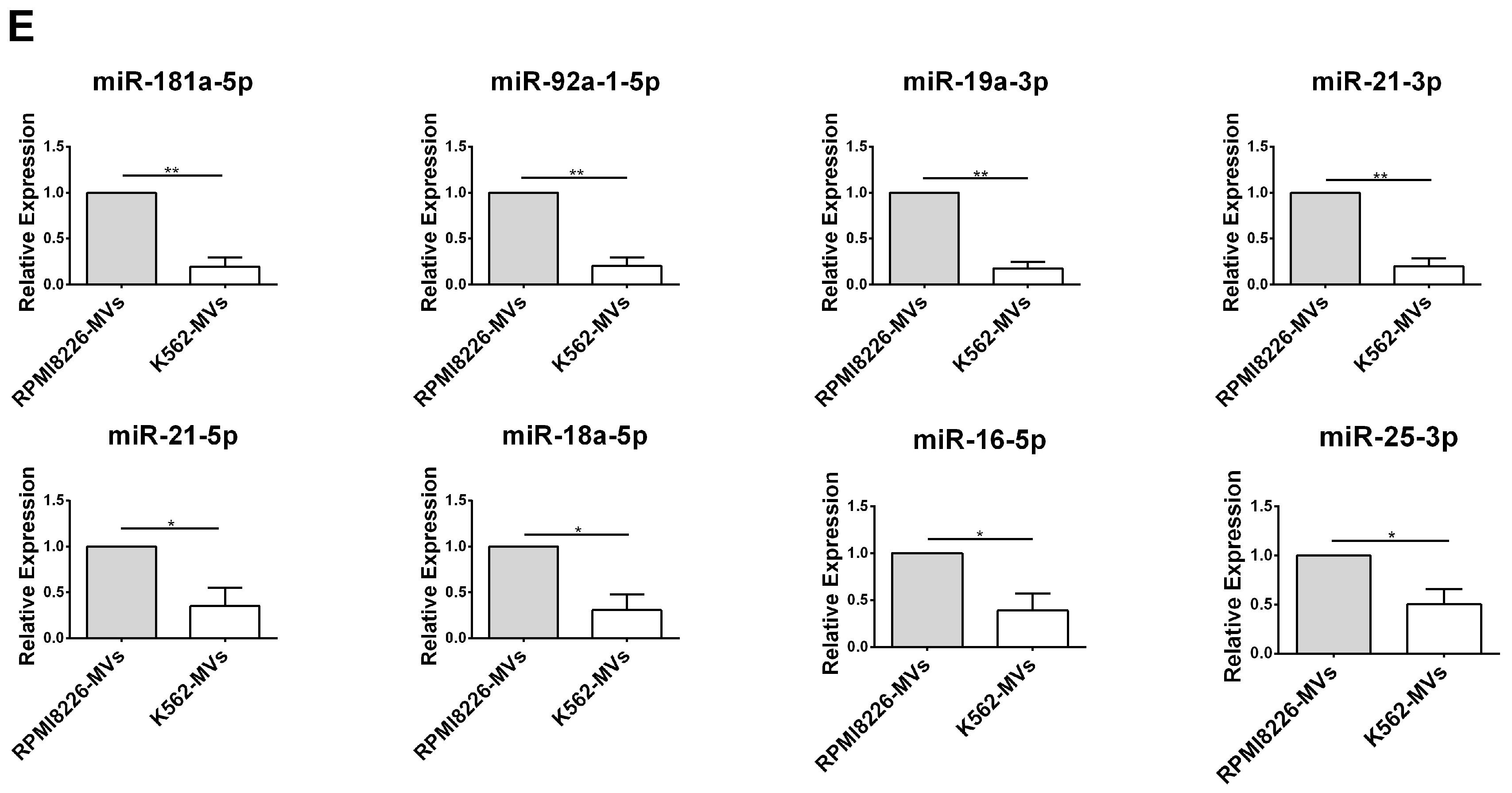
2.5. Selective miRNAs in MM-MVs that Confer Caspase-3-Induced Apoptosis in HK-2 Cells
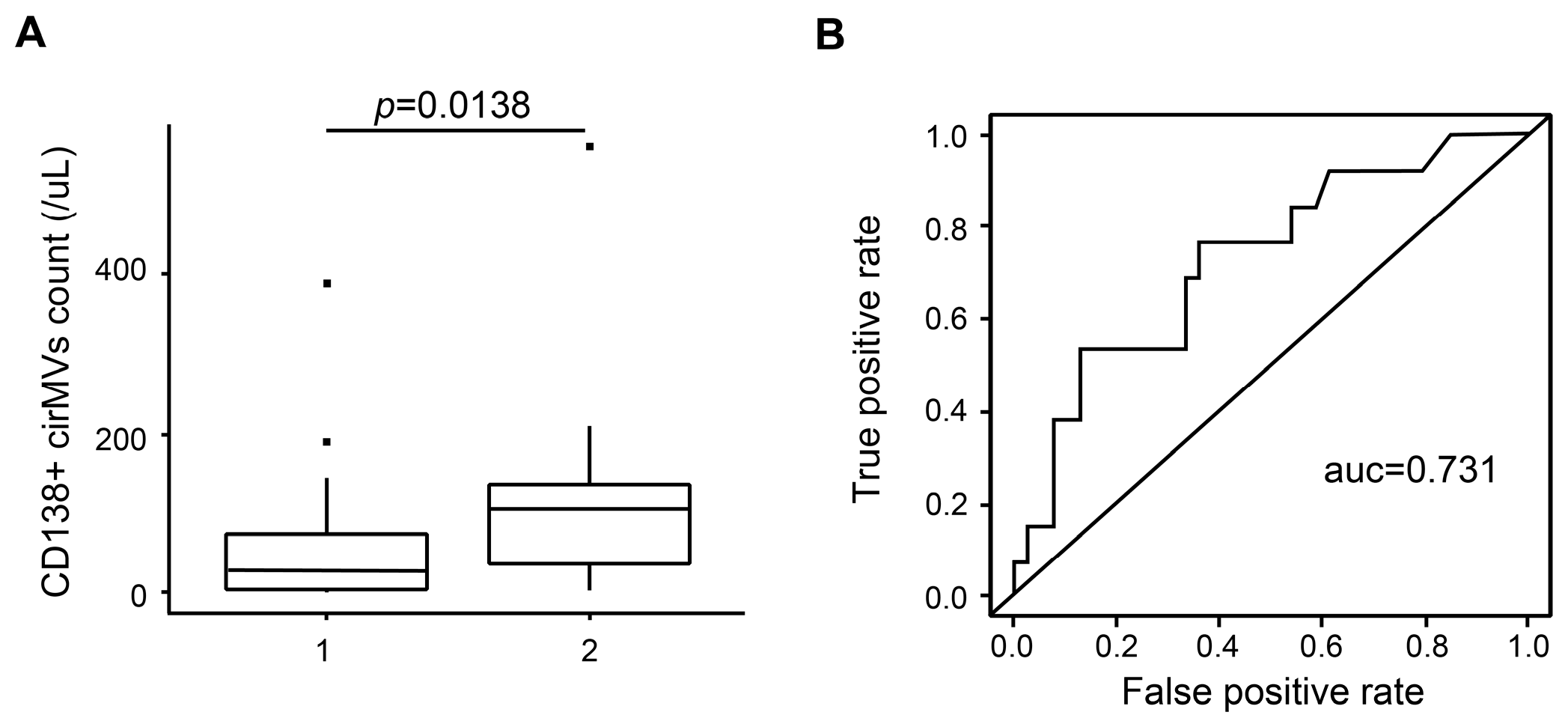
2.6. CD138+ Circulating MV (cirMV) Counts Positively Correlate with Renal Impairment
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. MV Isolation and Quantification
4.3. Transmission Electron Microscopy (TEM) and Scanning Electron Microscopy (SEM)
4.4. Small RNA Sequencing and Data Analyses
4.5. Cell Viability Detection
4.6. Apoptosis Detection
4.7. Western Blotting
4.8. Immunofluorescent Stainning
4.9. Construction of miRNA Regulatory Network
4.10. Realtime PCR
4.11. Clinical Samples Collection
4.12. Quantitative Detection of cirMVs
4.13. Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| RI | Renal Impairment |
| MM | Multiple Myeloma |
| MVs | Microvesicles |
| EMT | Epithelial-Mesenchymal Transition |
| RPM | Reads Per Million Mapped Reads |
References
- Dimopoulos, M.A.; Terpos, E.; Chanan-Khan, A.; Leung, N.; Ludwig, H.; Jagannath, S.; Niesvizky, R.; Giralt, S.; Fermand, J.P.; Blade, J.; et al. Renal impairment in patients with multiple myeloma: A consensus statement on behalf of the international myeloma working group. J. Clin. Oncol. 2010, 28, 4976–4984. [Google Scholar] [CrossRef] [PubMed]
- Knudsen, L.M.; Hippe, E.; Hjorth, M.; Holmberg, E.; Westin, J. Renal function in newly diagnosed multiple myeloma—A demographic study of 1353 patients. The nordic myeloma study group. Eur. J. Haematol. 1994, 53, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Yadav, P.; Cook, M.; Cockwell, P. Current trends of renal impairment in multiple myeloma. Kidney Dis. 2016, 1, 241–257. [Google Scholar] [CrossRef]
- Kyle, R.A.; Gertz, M.A.; Witzig, T.E.; Lust, J.A.; Lacy, M.Q.; Dispenzieri, A.; Fonseca, R.; Rajkumar, S.V.; Offord, J.R.; Larson, D.R.; et al. Review of 1027 patients with newly diagnosed multiple myeloma. Mayo Clin. Proc. 2003, 78, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Nordentoft, M. Prevention of suicide and attempted suicide in denmark. Epidemiological studies of suicide and intervention studies in selected risk groups. Dan. Med. Bull. 2007, 54, 306–369. [Google Scholar] [PubMed]
- Eleutherakis-Papaiakovou, V.; Bamias, A.; Gika, D.; Simeonidis, A.; Pouli, A.; Anagnostopoulos, A.; Michali, E.; Economopoulos, T.; Zervas, K.; Dimopoulos, M.A.; et al. Renal failure in multiple myeloma: Incidence, correlations, and prognostic significance. Leuk. Lymphoma 2007, 48, 337–341. [Google Scholar] [CrossRef] [PubMed]
- Heher, E.C.; Rennke, H.G.; Laubach, J.P.; Richardson, P.G. Kidney disease and multiple myeloma. Clin. J. Am. Soc. Nephrol. 2013, 8, 2007–2017. [Google Scholar] [CrossRef] [PubMed]
- Kastritis, E.; Zervas, K.; Symeonidis, A.; Terpos, E.; Delimbassi, S.; Anagnostopoulos, N.; Michali, E.; Zomas, A.; Katodritou, E.; Gika, D.; et al. Improved survival of patients with multiple myeloma after the introduction of novel agents and the applicability of the international staging system (ISS): An analysis of the greek myeloma study group (GMSG). Leukemia 2009, 23, 1152–1157. [Google Scholar] [CrossRef] [PubMed]
- Tkach, M.; Thery, C. Communication by extracellular vesicles: Where we are and where we need to go. Cell 2016, 164, 1226–1232. [Google Scholar] [CrossRef] [PubMed]
- Van der Pol, E.; Boing, A.N.; Harrison, P.; Sturk, A.; Nieuwland, R. Classification, functions, and clinical relevance of extracellular vesicles. Pharmacol. Rev. 2012, 64, 676–705. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhu, X.J.; Zeng, C.; Wu, P.H.; Wang, H.X.; Chen, Z.C.; Li, Q.B. Microvesicles secreted from human multiple myeloma cells promote angiogenesis. Acta Pharmacol. Sin. 2014, 35, 230–238. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; You, Y.; Li, Q.; Zeng, C.; Fu, F.; Guo, A.; Zhang, H.; Zou, P.; Zhong, Z.; Wang, H.; et al. Bcr-abl1-positive microvesicles transform normal hematopoietic transplants through genomic instability: Implications for donor cell leukemia. Leukemia 2014, 28, 1666–1675. [Google Scholar] [CrossRef] [PubMed]
- Yamada, N.; Tsujimura, N.; Kumazaki, M.; Shinohara, H.; Taniguchi, K.; Nakagawa, Y.; Naoe, T.; Akao, Y. Colorectal cancer cell-derived microvesicles containing microrna-1246 promote angiogenesis by activating smad 1/5/8 signaling elicited by pml down-regulation in endothelial cells. Biochim. Biophys. Acta 2014, 1839, 1256–1272. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Wang, H.X.; Zhu, X.J.; Wu, P.H.; Chen, W.Q.; Zou, P.; Li, Q.B.; Chen, Z.C. Serum deprivation elevates the levels of microvesicles with different size distributions and selectively enriched proteins in human myeloma cells in vitro. Acta Pharmacol. Sin. 2014, 35, 381–393. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Hering-Smith, K.S.; Simon, E.E.; Batuman, V. Myeloma light chains induce epithelial-mesenchymal transition in human renal proximal tubule epithelial cells. Nephrol. Dial. Transplant. 2008, 23, 860–870. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Liang, H.; Zhang, J.; Zen, K.; Zhang, C.Y. Secreted micrornas: A new form of intercellular communication. Trends Cell Biol. 2012, 22, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Pavkovic, M.; Vaidya, V.S. MicroRNAs and drug-induced kidney injury. Pharmacol. Ther. 2016, 163, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Trionfini, P.; Benigni, A.; Remuzzi, G. Micrornas in kidney physiology and disease. Nat. Rev. Nephrol. 2015, 11, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.G.; Kim, J.G.; Kim, H.J.; Kwon, H.K.; Cho, I.J.; Choi, D.W.; Lee, W.H.; Kim, W.D.; Hwang, S.J.; Choi, S.; et al. Discovery of an integrative network of micrornas and transcriptomics changes for acute kidney injury. Kidney Int. 2014, 86, 943–953. [Google Scholar] [CrossRef] [PubMed]
- Serino, G.; Sallustio, F.; Curci, C.; Cox, S.N.; Pesce, F.; de Palma, G.; Schena, F.P. Role of let-7b in the regulation of n-acetylgalactosaminyltransferase 2 in iga nephropathy. Nephrol. Dial. Transplant. 2015, 30, 1132–1139. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.H.; Lan, Y.F.; Li, H.F.; Cheng, C.F.; Lai, P.F.; Li, W.H.; Lin, H. Urinary mir-16 transactivated by C/EBPβ reduces kidney function after ischemia/reperfusion-induced injury. Sci. Rep. 2016, 6, 27945. [Google Scholar] [CrossRef] [PubMed]
- Rudnicki, M.; Beckers, A.; Neuwirt, H.; Vandesompele, J. RNA expression signatures and posttranscriptional regulation in diabetic nephropathy. Nephrol. Dial. Transplant. 2015, 30, iv35–iv42. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, S.R.; Luk, F.; Brown, R.D.; Suen, H.; Kwan, Y.; Bebawy, M. Isolation of human CD138+ microparticles from the plasma of patients with multiple myeloma. Neoplasia 2016, 18, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Nasr, S.H.; Valeri, A.M.; Sethi, S.; Fidler, M.E.; Cornell, L.D.; Gertz, M.A.; Lacy, M.; Dispenzieri, A.; Rajkumar, S.V.; Kyle, R.A.; et al. Clinicopathologic correlations in multiple myeloma: A case series of 190 patients with kidney biopsies. Am. J. Kidney Dis. 2012, 59, 786–794. [Google Scholar] [CrossRef] [PubMed]
- Heher, E.C.; Goes, N.B.; Spitzer, T.R.; Raje, N.S.; Humphreys, B.D.; Anderson, K.C.; Richardson, P.G. Kidney disease associated with plasma cell dyscrasias. Blood 2010, 116, 1397–1404. [Google Scholar] [CrossRef] [PubMed]
- Collino, F.; Bruno, S.; Incarnato, D.; Dettori, D.; Neri, F.; Provero, P.; Pomatto, M.; Oliviero, S.; Tetta, C.; Quesenberry, P.J.; et al. Aki recovery induced by mesenchymal stromal cell-derived extracellular vesicles carrying microRNAs. J. Am. Soc. Nephrol. 2015, 26, 2349–2360. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Xu, H.; Xu, W.; Wang, B.; Wu, H.; Tao, Y.; Zhang, B.; Wang, M.; Mao, F.; Yan, Y.; et al. Exosomes released by human umbilical cord mesenchymal stem cells protect against cisplatin-induced renal oxidative stress and apoptosis in vivo and in vitro. Stem Cell Res. Ther. 2013, 4, 34. [Google Scholar] [CrossRef] [PubMed]
- Jia, P.; Teng, J.; Zou, J.; Fang, Y.; Wu, X.; Liang, M.; Ding, X. Xenon protects against septic acute kidney injury via mir-21 target signaling pathway. Crit. Care Med. 2015, 43, e250–e259. [Google Scholar] [CrossRef] [PubMed]
- Belkaya, S.; Silge, R.L.; Hoover, A.R.; Medeiros, J.J.; Eitson, J.L.; Becker, A.M.; de la Morena, M.T.; Bassel-Duby, R.S.; van Oers, N.S. Dynamic modulation of thymic microRNAs in response to stress. PLoS ONE 2011, 6, e27580. [Google Scholar] [CrossRef] [PubMed]
- Marrone, A.K.; Stolz, D.B.; Bastacky, S.I.; Kostka, D.; Bodnar, A.J.; Ho, J. MicroRNA-17~92 is required for nephrogenesis and renal function. J. Am. Soc. Nephrol. 2014, 25, 1440–1452. [Google Scholar] [CrossRef] [PubMed]
- Thornberry, N.A.; Lazebnik, Y. Caspases: Enemies within. Science 1998, 281, 1312–1316. [Google Scholar] [CrossRef] [PubMed]
- Mrvar-Brecko, A.; Sustar, V.; Jansa, V.; Stukelj, R.; Jansa, R.; Mujagic, E.; Kruljc, P.; Iglic, A.; Hagerstrand, H.; Kralj-Iglic, V. Isolated microvesicles from peripheral blood and body fluids as observed by scanning electron microscope. Blood Cells Mol. Dis. 2010, 44, 307–312. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.M.; Li, Q.; Zhu, X.; Liu, W.; Hu, H.; Liu, T.; Cheng, F.; You, Y.; Zhong, Z.; Zou, P.; et al. Mir-146b-5p within bcr-abl1-positive microvesicles promotes leukemic transformation of hematopoietic cells. Cancer Res. 2016, 76, 2901–2911. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Durbin, R. Fast and accurate short read alignment with burrows-wheeler transform. Bioinformatics 2009, 25, 1754–1760. [Google Scholar] [CrossRef] [PubMed]
- Kozomara, A.; Griffiths-Jones, S. Mirbase: Annotating high confidence micrornas using deep sequencing data. Nucleic Acids Res. 2014, 42, D68–D73. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.C.; Lin, F.M.; Chang, W.C.; Lin, K.Y.; Huang, H.D.; Lin, N.S. Mirexpress: Analyzing high-throughput sequencing data for profiling microRNA expression. BMC Bioinform. 2009, 10, 328. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, V.; Bell, G.W.; Nam, J.W.; Bartel, D.P. Predicting effective microRNA target sites in mammalian mrnas. eLife 2015, 4, e05005. [Google Scholar] [CrossRef] [PubMed]
- Betel, D.; Wilson, M.; Gabow, A.; Marks, D.S.; Sander, C. The microRNA.Org resource: Targets and expression. Nucleic Acids Res. 2008, 36, D149–D153. [Google Scholar] [CrossRef] [PubMed]
- Shannon, P.; Markiel, A.; Ozier, O.; Baliga, N.S.; Wang, J.T.; Ramage, D.; Amin, N.; Schwikowski, B.; Ideker, T. Cytoscape: A software environment for integrated models of biomolecular interaction networks. Genome Res. 2003, 13, 2498–2504. [Google Scholar] [CrossRef] [PubMed]
- Kong, F.; Zhang, L.; Wang, H.; Yuan, G.; Guo, A.; Li, Q.; Chen, Z. Impact of collection, isolation and storage methodology of circulating microvesicles on flow cytometric analysis. Exp. Ther. Med. 2015, 10, 2093–2101. [Google Scholar] [CrossRef] [PubMed]


| Clinical Characteristics | Patient Number (n) |
|---|---|
| Total | 61 |
| Sex | |
| Male | 40 |
| Female | 21 |
| Median age, years (range) | 59 (40–82) |
| Durie-Salmon stage | |
| I | 3 |
| II | 11 |
| III | 38 |
| International staging system | |
| I | 6 |
| II | 25 |
| III | 27 |
| Type of monoclonal Ig | |
| IgG | 27 |
| IgA | 16 |
| IgD | 3 |
| LC a only | 8 |
| Renal function | |
| Group 1 (SCr b < 2 mg/dL) | 45 |
| Group 2 (SCr ≥ 2 mg/dL) | 16 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, A.; Kong, F.; Liu, C.-J.; Yan, G.; Gao, F.; Guo, H.; Guo, A.-Y.; Chen, Z.; Li, Q. Tumor Cell-Derived Microvesicles Induced Not Epithelial-Mesenchymal Transition but Apoptosis in Human Proximal Tubular (HK-2) Cells: Implications for Renal Impairment in Multiple Myeloma. Int. J. Mol. Sci. 2017, 18, 513. https://doi.org/10.3390/ijms18030513
Zhao A, Kong F, Liu C-J, Yan G, Gao F, Guo H, Guo A-Y, Chen Z, Li Q. Tumor Cell-Derived Microvesicles Induced Not Epithelial-Mesenchymal Transition but Apoptosis in Human Proximal Tubular (HK-2) Cells: Implications for Renal Impairment in Multiple Myeloma. International Journal of Molecular Sciences. 2017; 18(3):513. https://doi.org/10.3390/ijms18030513
Chicago/Turabian StyleZhao, Aiqi, Fancong Kong, Chun-Jie Liu, Guoxin Yan, Fei Gao, Hao Guo, An-Yuan Guo, Zhichao Chen, and Qiubai Li. 2017. "Tumor Cell-Derived Microvesicles Induced Not Epithelial-Mesenchymal Transition but Apoptosis in Human Proximal Tubular (HK-2) Cells: Implications for Renal Impairment in Multiple Myeloma" International Journal of Molecular Sciences 18, no. 3: 513. https://doi.org/10.3390/ijms18030513