Association of Hormone Receptor Expression with Survival in Ovarian Endometrioid Carcinoma: Biological Validation and Clinical Implications
Abstract
:1. Introduction
2. Results
2.1. Study Cohort and Patient Characteristics
2.2. Hormonal Receptor Expression
2.3. Hormonal Receptor Expression and Association with Survival
2.4. Clinical Utility of Hormone Receptor Expression Status
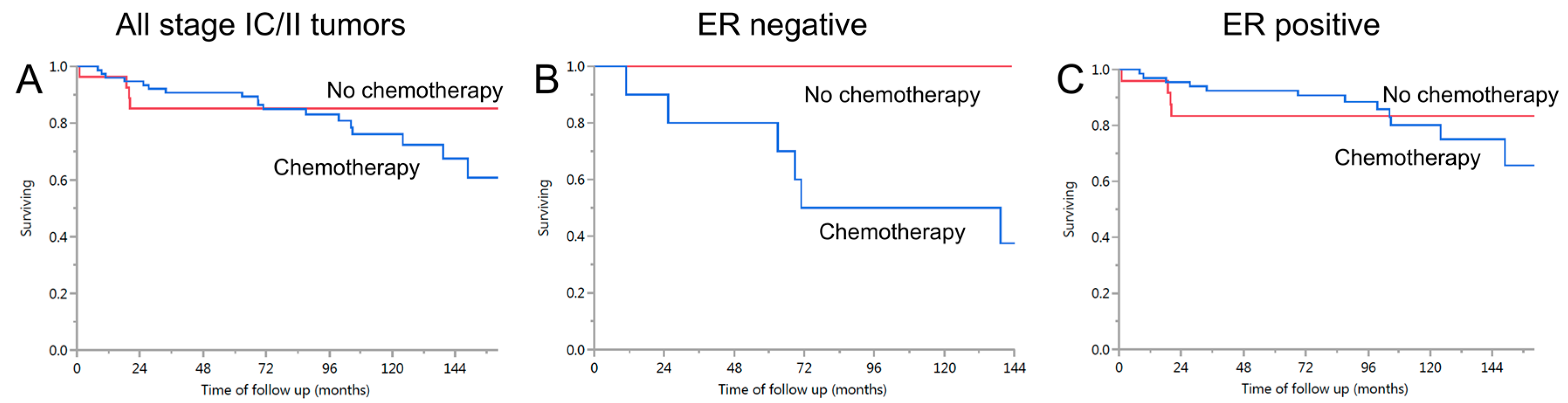
2.5. Exploring Whether Hormone Receptor Status Might Be an Effect Modifier of Chemotherapy Response within Stage IC/II
3. Discussion
4. Materials and Methods
4.1. Study Cohort
4.2. Clinical Information and Follow Up
4.3. Immunohistochemistry
4.4. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| EC | Endometrioid carcinoma |
| ER | Estrogen receptor |
| PR | Progesterone receptor |
| OCSS | Ovarian cancer specific survival |
References
- Köbel, M.; Kalloger, S.E.; Huntsman, D.G.; Santos, J.L.; Swenerton, K.D.; Seidman, J.D.; Gilks, C.B. Differences in tumor type in low-stage versus high-stage ovarian carcinomas. Int. J. Gynecol. Pathol. 2010, 29, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Köbel, M.; Kalloger, S.E.; Lee, S.; Duggan, M.A.; Kelemen, L.E.; Prentice, L.; Kalli, K.R.; Fridley, B.L.; Visscher, D.W.; Keeney, G.L.; et al. Biomarker-based ovarian carcinoma typing: A histologic investigation in the ovarian tumor tissue analysis consortium. Cancer Epidemiol. Biomark. Prev. 2013, 22, 1677–1686. [Google Scholar] [CrossRef] [PubMed]
- Köbel, M.; Kalloger, S.E.; Santos, J.L.; Huntsman, D.G.; Gilks, C.B.; Swenerton, K.D. Tumor type and substage predict survival in stage I and II ovarian carcinoma: Insights and implications. Gynecol. Oncol. 2010, 116, 50–56. [Google Scholar] [CrossRef] [PubMed]
- McConechy, M.K.; Ding, J.; Senz, J.; Yang, W.; Melnyk, N.; Tone, A.A.; Prentice, L.M.; Wiegand, K.C.; McAlpine, J.N.; Shah, S.P.; et al. Ovarian and endometrial endometrioid carcinomas have distinct ctnnb1 and pten mutation profiles. Mod. Pathol. 2014, 27, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Aysal, A.; Karnezis, A.; Medhi, I.; Grenert, J.P.; Zaloudek, C.J.; Rabban, J.T. Ovarian endometrioid adenocarcinoma: Incidence and clinical significance of the morphologic and immunohistochemical markers of mismatch repair protein defects and tumor microsatellite instability. Am. J. Surg. Pathol. 2012, 36, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Rambau, P.F.; Duggan, M.A.; Ghatage, P.; Warfa, K.; Steed, H.; Perrier, R.; Kelemen, L.E.; Kobel, M. Significant frequency of MSH2/MSH6 abnormality in ovarian endometrioid carcinoma supports histotype-specific lynch syndrome screening in ovarian carcinomas. Histopathology 2016, 69, 288–297. [Google Scholar] [CrossRef] [PubMed]
- Gilks, C.B.; Ionescu, D.N.; Kalloger, S.E.; Kobel, M.; Irving, J.; Clarke, B.; Santos, J.; Le, N.; Moravan, V.; Swenerton, K.; et al. Tumor cell type can be reproducibly diagnosed and is of independent prognostic significance in patients with maximally debulked ovarian carcinoma. Hum. Pathol. 2008, 39, 1239–1251. [Google Scholar] [CrossRef] [PubMed]
- Köbel, M.; Bak, J.; Bertelsen, B.I.; Carpen, O.; Grove, A.; Hansen, E.S.; Levin Jakobsen, A.M.; Lidang, M.; Måsbäck, A.; Tolf, A.; et al. Ovarian carcinoma histotype determination is highly reproducible, and is improved through the use of immunohistochemistry. Histopathology 2014, 64, 1004–1013. [Google Scholar] [CrossRef] [PubMed]
- Köbel, M.; Kalloger, S.E.; Baker, P.M.; Ewanowich, C.A.; Arseneau, J.; Zherebitskiy, V.; Abdulkarim, S.; Leung, S.; Duggan, M.A.; Fontaine, D.; et al. Diagnosis of ovarian carcinoma cell type is highly reproducible: A transcanadian study. Am. J. Surg. Pathol. 2010, 34, 984–993. [Google Scholar] [CrossRef] [PubMed]
- Köbel, M.; Rahimi, K.; Rambau, P.F.; Naugler, C.; le Page, C.; Meunier, L.; de Ladurantaye, M.; Lee, S.; Leung, S.; Goode, E.L.; et al. An immunohistochemical algorithm for ovarian carcinoma typing. Int. J. Gynecol. Pathol. 2016, 35, 430–441. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Zhang, F.; Zhang, W.; He, J.; Zhao, Y.; Sun, J. Prognostic role of hormone receptors in ovarian cancer: A systematic review and meta-analysis. Int. J. Gynecol. Cancer 2013, 23, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Köbel, M.; Kalloger, S.E.; Boyd, N.; McKinney, S.; Mehl, E.; Palmer, C.; Leung, S.; Bowen, N.J.; Ionescu, D.N.; Rajput, A.; et al. Ovarian carcinoma subtypes are different diseases: Implications for biomarker studies. PLoS Med. 2008, 5, e232. [Google Scholar] [CrossRef] [PubMed]
- Sieh, W.; Köbel, M.; Longacre, T.A.; Bowtell, D.D.; deFazio, A.; Goodman, M.T.; Hogdall, E.; Deen, S.; Wentzensen, N.; Moysich, K.B.; et al. Hormone-receptor expression and ovarian cancer survival: An ovarian tumor tissue analysis consortium study. Lancet Oncol. 2013, 14, 853–862. [Google Scholar] [CrossRef]
- Kelemen, L.E.; Warren, G.W.; Koziak, J.M.; Kobel, M.; Steed, H. Smoking may modify the association between neoadjuvant chemotherapy and survival from ovarian cancer. Gynecol. Oncol. 2016, 140, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Rambau, P.; Kelemen, L.E.; Steed, H.; Quan, M.L.; Ghatage, P.; Köbel, M. University of Calgary, Calgary, Alberta, Canada, Kaplan-Meier Survival Curves for PR Status and ER/PR Status. Unpublished work. 2017. [Google Scholar]
- Altman, D.G.; McShane, L.M.; Sauerbrei, W.; Taube, S.E. Reporting recommendations for tumor marker prognostic studies (remark): Explanation and elaboration. PLoS Med. 2012, 9, e1001216. [Google Scholar] [CrossRef] [PubMed]
- Terry, J.; Torlakovic, E.E.; Garratt, J.; Miller, D.; Kobel, M.; Cooper, J.; Bahzad, S.; Pilavdzic, D.; O’Malley, F.; O’Brien, A.E.; et al. Implementation of a canadian external quality assurance program for breast cancer biomarkers: An initiative of canadian quality control in immunohistochemistry (CIQC) and canadian association of pathologists (CAP) national standards committee/immunohistochemistry. Appl. Immunohistochem. Mol. Morphol. 2009, 17, 375–382. [Google Scholar] [PubMed]
- Kumar, A.; Le, N.; Tinker, A.V.; Santos, J.L.; Parsons, C.; Hoskins, P.J. Early-stage endometrioid ovarian carcinoma: Population-based outcomes in british columbia. Int. J. Gynecol. Cancer 2014, 24, 1401–1405. [Google Scholar] [CrossRef] [PubMed]
- Karnezis, A.N.; Hoang, L.N.; Coatham, M.; Ravn, S.; Almadani, N.; Tessier-Cloutier, B.; Irving, J.; Meng, B.; Li, X.; Chow, C.; et al. Loss of switch/sucrose non-fermenting complex protein expression is associated with dedifferentiation in endometrial carcinomas. Mod. Pathol. 2016, 29, 302–314. [Google Scholar] [CrossRef] [PubMed]
- Anglesio, M.S.; Wiegand, K.C.; Melnyk, N.; Chow, C.; Salamanca, C.; Prentice, L.M.; Senz, J.; Yang, W.; Spillman, M.A.; Cochrane, D.R.; et al. Type-specific cell line models for type-specific ovarian cancer research. PLoS ONE 2013, 8, e72162. [Google Scholar] [CrossRef]
- Bernardini, M.Q.; Gien, L.T.; Lau, S.; Altman, A.D.; Gilks, B.; Ferguson, S.E.; Kobel, M.; Samouelian, V.; Wesa, M.; Cameron, A.; et al. Treatment related outcomes in high-risk endometrial carcinoma: Canadian high risk endometrial cancer consortium (Chrec). Gynecol. Oncol. 2016, 141, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, H.; Russell, I.A.; Stark, R.; Rueda, O.M.; Hickey, T.E.; Tarulli, G.A.; Serandour, A.A.; Birrell, S.N.; Bruna, A.; Saadi, A.; et al. Progesterone receptor modulates eralpha action in breast cancer. Nature 2015, 523, 313–317. [Google Scholar] [CrossRef]
- Rose, P.G. Endometrial carcinoma. N. Engl. J. Med. 1996, 335, 640–649. [Google Scholar] [CrossRef] [PubMed]
- Garrett, A.; Quinn, M.A. Hormonal therapies and gynaecological cancers. Best. Pract. Res. Clin. Obstet. Gynaecol. 2008, 22, 407–421. [Google Scholar] [CrossRef] [PubMed]
- Hatch, K.D.; Beecham, J.B.; Blessing, J.A.; Creasman, W.T. Responsiveness of patients with advanced ovarian carcinoma to tamoxifen. A gynecologic oncology group study of second-line therapy in 105 patients. Cancer 1991, 68, 269–271. [Google Scholar] [CrossRef]
- Perez-Gracia, J.L.; Carrasco, E.M. Tamoxifen therapy for ovarian cancer in the adjuvant and advanced settings: Systematic review of the literature and implications for future research. Gynecol. Oncol. 2002, 84, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Sommeijer, D.W.; Sjoquist, K.M.; Friedlander, M. Hormonal treatment in recurrent and metastatic gynaecological cancers: A review of the current literature. Curr. Oncol. Rep. 2013, 15, 541–548. [Google Scholar] [CrossRef] [PubMed]
- Williams, C.; Simera, I.; Bryant, A. Tamoxifen for relapse of ovarian cancer. Cochrane Database Syst. Rev. 2010. [Google Scholar] [CrossRef]
- Kuhnel, R.; Delemarre, J.F.; Rao, B.R.; Stolk, J.G. Correlation of aromatase activity and steroid receptors in human ovarian carcinoma. Anticancer Res. 1986, 6, 889–892. [Google Scholar] [PubMed]
- Pan, Y.; Kao, M.S. Endometrioid ovarian carcinoma benefits from aromatase inhibitors: Case report and literature review. Curr. Oncol. 2010, 17, 82–85. [Google Scholar] [CrossRef] [PubMed]
- Rendina, G.M.; Donadio, C.; Giovannini, M. Steroid receptors and progestinic therapy in ovarian endometrioid carcinoma. Eur. J. Gynaecol. Oncol. 1982, 3, 241–246. [Google Scholar] [PubMed]
- Smyth, J.F.; Gourley, C.; Walker, G.; MacKean, M.J.; Stevenson, A.; Williams, A.R.; Nafussi, A.A.; Rye, T.; Rye, R.; Stewart, M.; et al. Antiestrogen therapy is active in selected ovarian cancer cases: The use of letrozole in estrogen receptor-positive patients. Clin. Cancer Res. 2007, 13, 3617–3622. [Google Scholar] [CrossRef] [PubMed]
- Gershenson, D.M.; Sun, C.C.; Iyer, R.B.; Malpica, A.L.; Kavanagh, J.J.; Bodurka, D.C.; Schmeler, K.; Deavers, M. Hormonal therapy for recurrent low-grade serous carcinoma of the ovary or peritoneum. Gynecol. Oncol. 2012, 125, 661–666. [Google Scholar] [CrossRef] [PubMed]
- De Bastos, M.; Stegeman, B.H.; Rosendaal, F.R.; van Hylckama Vlieg, A.; Helmerhorst, F.M.; Stijnen, T.; Dekkers, O.M. Combined oral contraceptives: Venous thrombosis. Cochrane Database Syst. Rev. 2014, CD010813. [Google Scholar] [CrossRef]
- Fleming, G.F.; Filiaci, V.L.; Marzullo, B.; Zaino, R.J.; Davidson, S.A.; Pearl, M.; Makker, V.; Burke, J.J.; Zweizig, S.L.; van Le, L.; et al. Temsirolimus with or without megestrol acetate and tamoxifen for endometrial cancer: A gynecologic oncology group study. Gynecol. Oncol. 2014, 132, 585–592. [Google Scholar] [CrossRef] [PubMed]
- Kilickap, S.; Tural, D. Does adjuvant hormone therapy improve survival in epithelial ovarian cancer? J. Clin. Oncol. 2016, 34, 2069. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.C.; Lomo, L.; Baak, J.P.; Eng, C.; Ince, T.A.; Crum, C.P.; Mutter, G.L. Squamous morules are functionally inert elements of premalignant endometrial neoplasia. Mod. Pathol. 2009, 22, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Horn, L.C.; Faber, S.; Bilek, K.; Leo, C. Expression of COX-2 and HER-2/neu and estrogen and progesterone receptor in primary squamous cell carcinomas of the endometrium. J. Cancer Res. Clin. Oncol. 2005, 131, 226–228. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhao, D.; Gong, C.; Zhang, F.; He, J.; Zhang, W.; Zhao, Y.; Sun, J. Prognostic role of hormone receptors in endometrial cancer: A systematic review and meta-analysis. World J. Surg. Oncol. 2015, 25, 208. [Google Scholar] [CrossRef] [PubMed]
- Köbel, M.; Atenafu, E.G.; Rambau, P.F.; Ferguson, S.E.; Nelson, G.S.; Ho, T.C.; Panzarella, T.; McAlpine, J.N.; Gilks, C.B.; Clarke, B.A.; et al. Progesterone receptor expression is associated with longer overall survival within high-grade histotypes of endometrial carcinoma: A Canadian high risk endometrial cancer consortium (CHREC) study. Gynecol. Oncol. 2016, 141, 559–563. [Google Scholar] [CrossRef] [PubMed]
- Mackenzie, R.; Talhouk, A.; Eshragh, S.; Lau, S.; Cheung, D.; Chow, C.; Le, N.; Cook, L.S.; Wilkinson, N.; McDermott, J.; et al. Morphologic and molecular characteristics of mixed epithelial ovarian cancers. Am. J. Surg. Pathol. 2015, 39, 1548–1557. [Google Scholar] [CrossRef] [PubMed]

| Parameter | n (%) | Five-Year Ovarian Cancer Specific Survival |
|---|---|---|
| Centre | ||
| Calgary | 93 (51.1%) | 85.60% |
| Edmonton | 89 (48.9%) | 86.40% |
| Stage | ||
| IA/IB | 46 (25.3%) | 97.80% |
| IC | 40 (22.0%) | 90.00% |
| II | 64 (35.1%) | 87.50% |
| III | 21 (11.5%) | 59.60% |
| IV | 2 (1.1%) | 0 |
| Not documented | 9 (5.0%) | 76.20% |
| Residual tumor | ||
| Absent | 94 (51.6%) | 89.00% |
| Present | 15 (8.2%) | 36.60% |
| Not documented | 73 (40.1%) | 91.80% |
| Grade | ||
| 1 | 125 (68.7%) | 89.40% |
| 2 | 23 (12.6%) | 82.60% |
| 3 | 34 (18.7%) | 76.40% |
| Variable | ER Absent | ER Present | p | PR Absent | PR Present | p |
|---|---|---|---|---|---|---|
| Tumor Expression, n (%) | 23 (12.7) | 158 (87.3) | 24 (13.3) | 157 (86.7) | ||
| Age at diagnosis, Mean (SE) | 52.8 (2.6) | 54.1 (1.0) | 0.63 | 54.6 (2.6) | 53.8 (1.0) | 0.79 |
| BMI 1, Mean (SE) | 25.8 (1.8) | 29.9 (0.7) | 0.040 | 25.8 (1.9) | 29.8 (0.7) | 0.051 |
| CA125, Mean (SE) | 428 (274) | 549 (101) | 0.68 | 748 (274) | 505 (101) | 0.40 |
| Endometriosis, n (%) | 0.42 | 0.54 | ||||
| Present | 7 (30.4) | 66 (41.8) | 8 (33.3) | 65 (41.4) | ||
| Absent | 16 (69.6) | 90 (60.1) | 16 (66.7) | 90 (57.3) | ||
| Unknown | 0 | 2 (1.3%) | 0 | 2 (1.3%) | ||
| Synchronous endometrial endometrioid carcinoma, n (%) | 0.45 | 0.38 | ||||
| Present | 5 (21.4) | 55 (34.8) | 5 (20.8) | 55 (35.0) | ||
| Absent | 11 (47.8) | 61 (38.6) | 11 (45.8) | 61 (38.9) | ||
| Unknown | 7 (30.4) | 42 (26.6) | 8 (33.3) | 41 (26.1) | ||
| Stage, n (%) | 0.57 | 0.70 | ||||
| IA/IB | 4 (17.4) | 41 (26.0) | 5 (20.8) | 40 (25.5) | ||
| IC/II | 13 (56.5) | 91 (57.6) | 15 (62.5) | 89 (56.7) | ||
| III | 3 (13.0) | 18 (11.4) | 2 (8.3) | 19 (12.1) | ||
| IV | 1 (4.4) | 1 (0.6) | 1 (4.2) | 1 (0.6) | ||
| Unknown | 2 (8.7) | 7 (4.4) | 1 (4.2) | 8 (5.1) | ||
| FIGO grade, n (%) | 0.0075 | <0.0001 | ||||
| 1 | 9 (39.1) | 115 (72.3) | 7 (29.2) | 117 (74.5) | ||
| 2 | 6 (26.1) | 17 (10.8) | 9 (37.5) | 14 (8.9) | ||
| 3 | 8 (34.5) | 26 (16.5) | 8 (33.3) | 26 (16.6) | ||
| Squamous differentiation, n (%) | 0.0058 | 0.014 | ||||
| Absent | 18 (78.3) | 76 (48.4) | 18 (75.0) | 76 (48.7) | ||
| Present | 5 (21.7) | 81 (51.6) | 6 (25.0) | 80 (51/3) | ||
| Treatment, n (%) | ||||||
| Residual tumor present | 2 (8.7) | 13 (8.2) | 0.56 | 2 (8.3) | 13 (8.3) | 0.76 |
| Radiation received | 2 (8.7) | 16 (10.1) | 0.64 | 4 (16.7) | 14 (8.9) | 0.36 |
| Chemotherapy received | 16 (69.5) | 99 (63.1) | 0.65 | 17 (70.8) | 98 (62.8) | 0.59 |
| MMR 2, n (%) | 0.69 | 0.90 | ||||
| Deficient | 2 (8.7) | 23 (14.6) | 3 (12.5) | 22 (14.0) | ||
| Proficient | 21 (91.3) | 134 (84.8) | 21 (87.5) | 134 (85.4) | ||
| Missing | 0 | 1 (0.6) | 0 | 1 (0.6) |
| Model | ER HR (95% CI) | PR HR (95% CI) | ER + PR HR (95% CI) |
|---|---|---|---|
| Model 1 a | n = 172 | n = 172 | |
| Marker absent | 1.0 (Reference) | 1.0 (Reference) | Not performed |
| Marker focal | 0.230 (0.0713–0.685) | 0.256 (0.079–0.780) | |
| Marker diffuse | 0.160 (0.07–0.403) | 0.209 (0.089–0.517) | |
| p | 0.0003 | 0.0018 | |
| Model 2 a | n = 172 | n = 172 | n = 160 * |
| Markers absent | 1.0 (Reference) | 1.0 (Reference) | 1.0 (Reference) |
| Markers present | 0.175 (0.076–0.425) | 0.218 (0.096–0.527) | 0.123 (0.066–0.428) |
| p | 0.0002 | 0.0011 | 0.0001 |
| Model 3 b Stage I/II | n = 149 | n = 149 | n = 138 * |
| Markers absent | 1.0 (Reference) | 1.0 (Reference) | 1.0 (Reference) |
| Markers present | 0.351 (0.137–0.976) | 0.400 (0.151–1.176) | 0.257 (0.079–0.946) |
| p | 0.0452 | 0.0932 | 0.0417 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rambau, P.; Kelemen, L.E.; Steed, H.; Quan, M.L.; Ghatage, P.; Köbel, M. Association of Hormone Receptor Expression with Survival in Ovarian Endometrioid Carcinoma: Biological Validation and Clinical Implications. Int. J. Mol. Sci. 2017, 18, 515. https://doi.org/10.3390/ijms18030515
Rambau P, Kelemen LE, Steed H, Quan ML, Ghatage P, Köbel M. Association of Hormone Receptor Expression with Survival in Ovarian Endometrioid Carcinoma: Biological Validation and Clinical Implications. International Journal of Molecular Sciences. 2017; 18(3):515. https://doi.org/10.3390/ijms18030515
Chicago/Turabian StyleRambau, Peter, Linda E. Kelemen, Helen Steed, May Lynn Quan, Prafull Ghatage, and Martin Köbel. 2017. "Association of Hormone Receptor Expression with Survival in Ovarian Endometrioid Carcinoma: Biological Validation and Clinical Implications" International Journal of Molecular Sciences 18, no. 3: 515. https://doi.org/10.3390/ijms18030515
APA StyleRambau, P., Kelemen, L. E., Steed, H., Quan, M. L., Ghatage, P., & Köbel, M. (2017). Association of Hormone Receptor Expression with Survival in Ovarian Endometrioid Carcinoma: Biological Validation and Clinical Implications. International Journal of Molecular Sciences, 18(3), 515. https://doi.org/10.3390/ijms18030515






