Genomic and Molecular Landscapes of Cardiac Remodeling and Heart Failure
A special issue of Genes (ISSN 2073-4425). This special issue belongs to the section "Genetic Diagnosis".
Deadline for manuscript submissions: closed (20 April 2026) | Viewed by 1020
Special Issue Editor
Special Issue Information
Dear Colleagues,
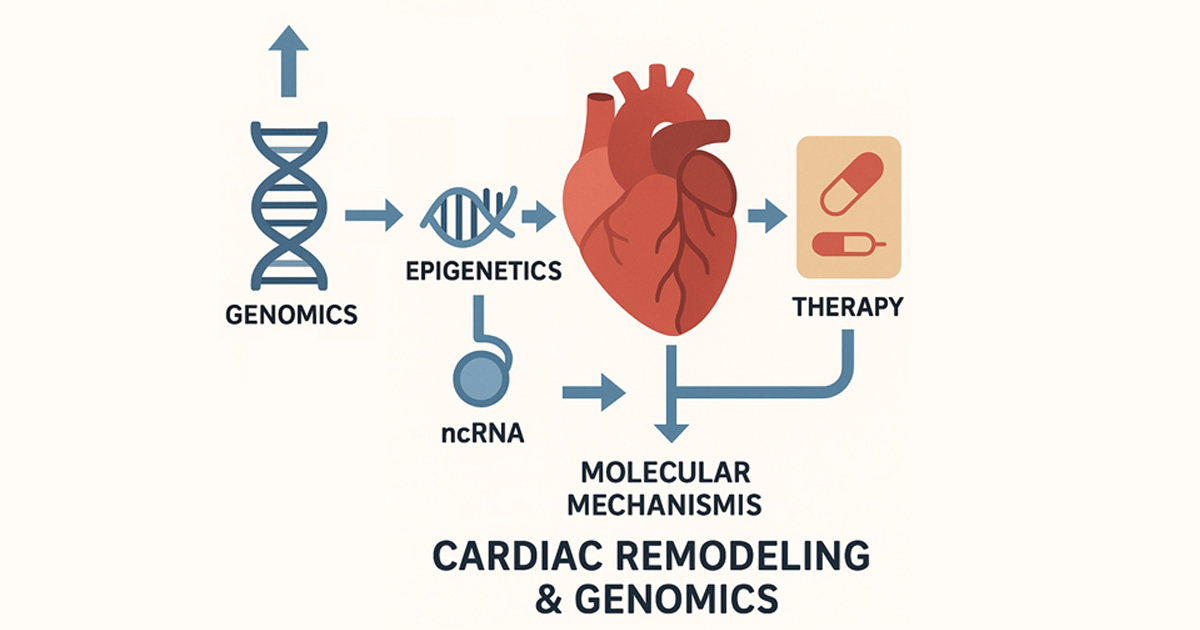
Heart failure remains one of the leading causes of morbidity and mortality worldwide, with myocardial remodeling and fibrosis being central hallmarks of disease progression. Advances in genomics, transcriptomics, epigenetics, and non-coding RNAs are providing new insights into the molecular pathways underlying cardiac dysfunction, while translational approaches offer novel opportunities for early diagnosis and therapy.
This Special Issue of Genes aims to provide a broad and interdisciplinary platform for studies addressing the genetic, molecular, and cellular mechanisms of heart failure, highlighting discoveries that bridge basic research with clinical application. We welcome the submission of original research, reviews, and methodological papers focused on, but not limited to, the following:
- Genomic and transcriptomic profiling in heart failure and cardiomyopathies;
- Epigenetic regulation and chromatin remodeling in cardiac disease;
- Non-coding RNAs (miRNA, lncRNA, circRNA) in cardiac pathophysiology;
- Molecular and cellular mechanisms of myocardial fibrosis and regeneration;
- Multiomics, bioinformatics, and AI-driven approaches to cardiac genomics;
- Translational perspectives and therapeutic applications of genomic discoveries.
The goal of this Special Issue is to explore the genomic and molecular signatures shaping cardiac remodeling, identify novel biomarkers and therapeutic targets, and contribute to the development of precision cardiology.
Dr. Dorota Ciołczyk-Wierzbicka
Guest Editor
Manuscript Submission Information
Manuscripts should be submitted online at www.mdpi.com by registering and logging in to this website. Once you are registered, click here to go to the submission form. Manuscripts can be submitted until the deadline. All submissions that pass pre-check are peer-reviewed. Accepted papers will be published continuously in the journal (as soon as accepted) and will be listed together on the special issue website. Research articles, review articles as well as short communications are invited. For planned papers, a title and short abstract (about 250 words) can be sent to the Editorial Office for assessment.
Submitted manuscripts should not have been published previously, nor be under consideration for publication elsewhere (except conference proceedings papers). All manuscripts are thoroughly refereed through a single-blind peer-review process. A guide for authors and other relevant information for submission of manuscripts is available on the Instructions for Authors page. Genes is an international peer-reviewed open access monthly journal published by MDPI.
Please visit the Instructions for Authors page before submitting a manuscript. The Article Processing Charge (APC) for publication in this open access journal is 2600 CHF (Swiss Francs). Submitted papers should be well formatted and use good English. Authors may use MDPI's English editing service prior to publication or during author revisions.
Keywords
- heart failure
- genomics
- transcriptomics
- epigenetics/chromatin remodeling
- non-coding RNAs
- myocardial fibrosis
- cardiac regeneration
- multi-omics & AI-driven bioinformatics
- precision cardiology
- translational therapeutics
Benefits of Publishing in a Special Issue
- Ease of navigation: Grouping papers by topic helps scholars navigate broad scope journals more efficiently.
- Greater discoverability: Special Issues support the reach and impact of scientific research. Articles in Special Issues are more discoverable and cited more frequently.
- Expansion of research network: Special Issues facilitate connections among authors, fostering scientific collaborations.
- External promotion: Articles in Special Issues are often promoted through the journal's social media, increasing their visibility.
- Reprint: MDPI Books provides the opportunity to republish successful Special Issues in book format, both online and in print.
Further information on MDPI's Special Issue policies can be found here.






