Multiple Sclerosis CD49d+CD154+ As Myelin-Specific Lymphocytes Induced During Remyelination
Abstract
1. Introduction
2. Materials and Methods
2.1. Human Subjects
2.2. EAE Mouse Model
2.3. Human Cell Model of Progenitor Cells Differentiation to Mature Myelin Producing Oligodendrocytes
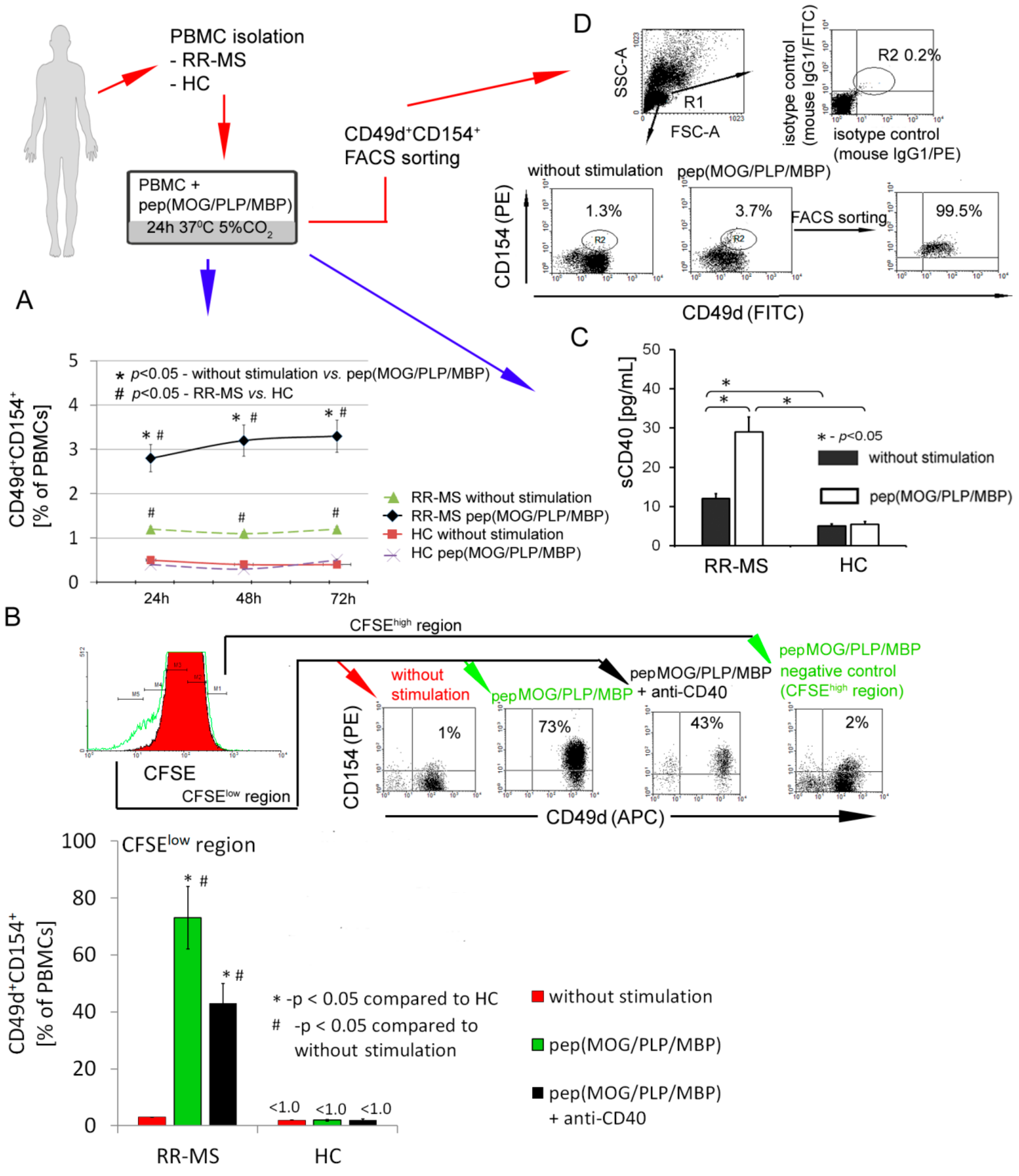
2.4. RR-MS CD49d+CD154+ Lymphocyte Sorting
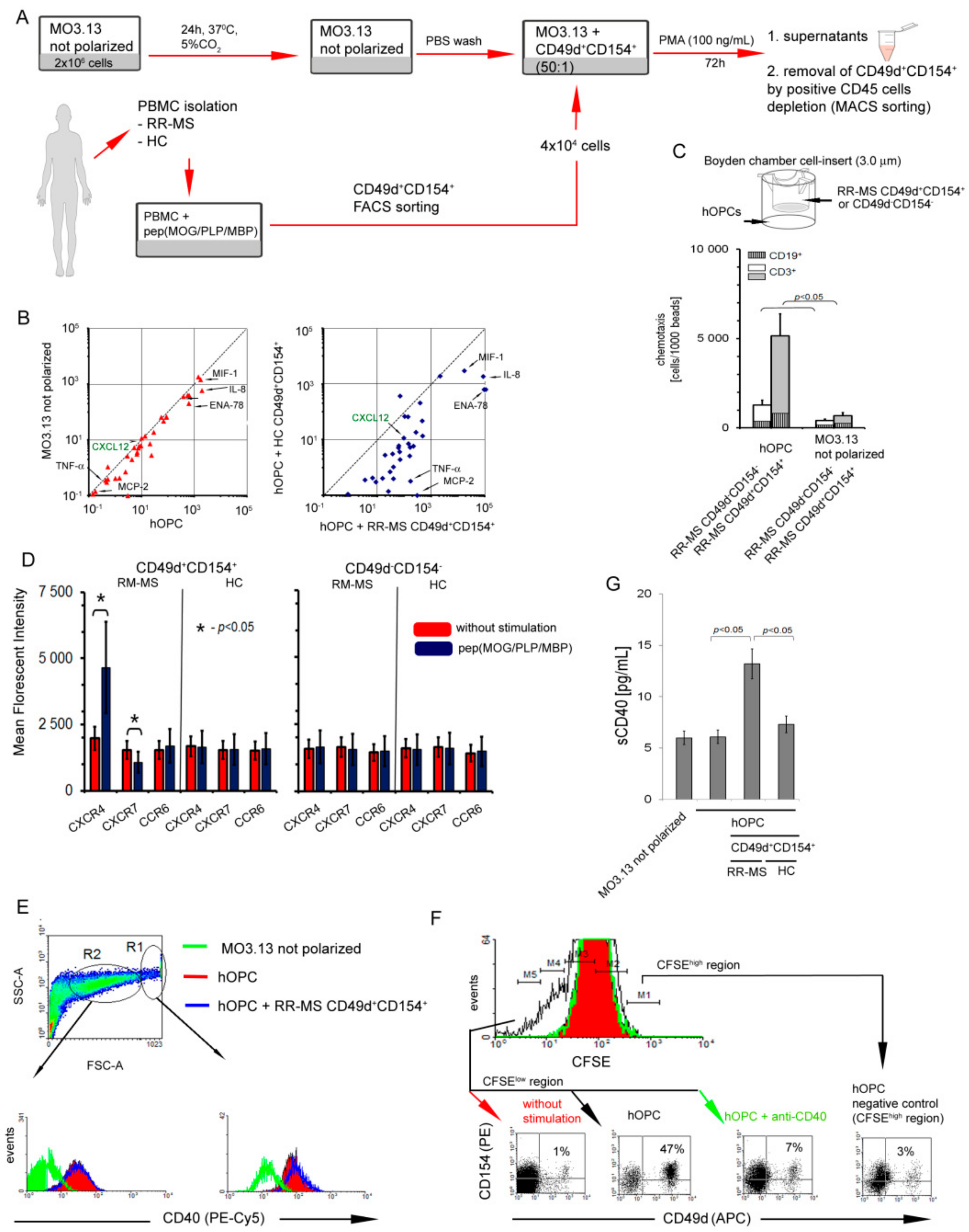
2.5. hOPC-Lymphocyte Co-Culture
2.6. Analysis of RR-MS Myelin-Specific CD49d+CD154+ Cell Proliferation using CFSE Method
2.7. Flow Cytometry Analysis of CXCR4, CXCR7, CCR6 Surface Expression on CD49d+CD154+ Lymphocytes, and CD40 on OPCs
2.8. Chemotaxis Assay
2.9. Human Chemokine Multiple Profiling Assays
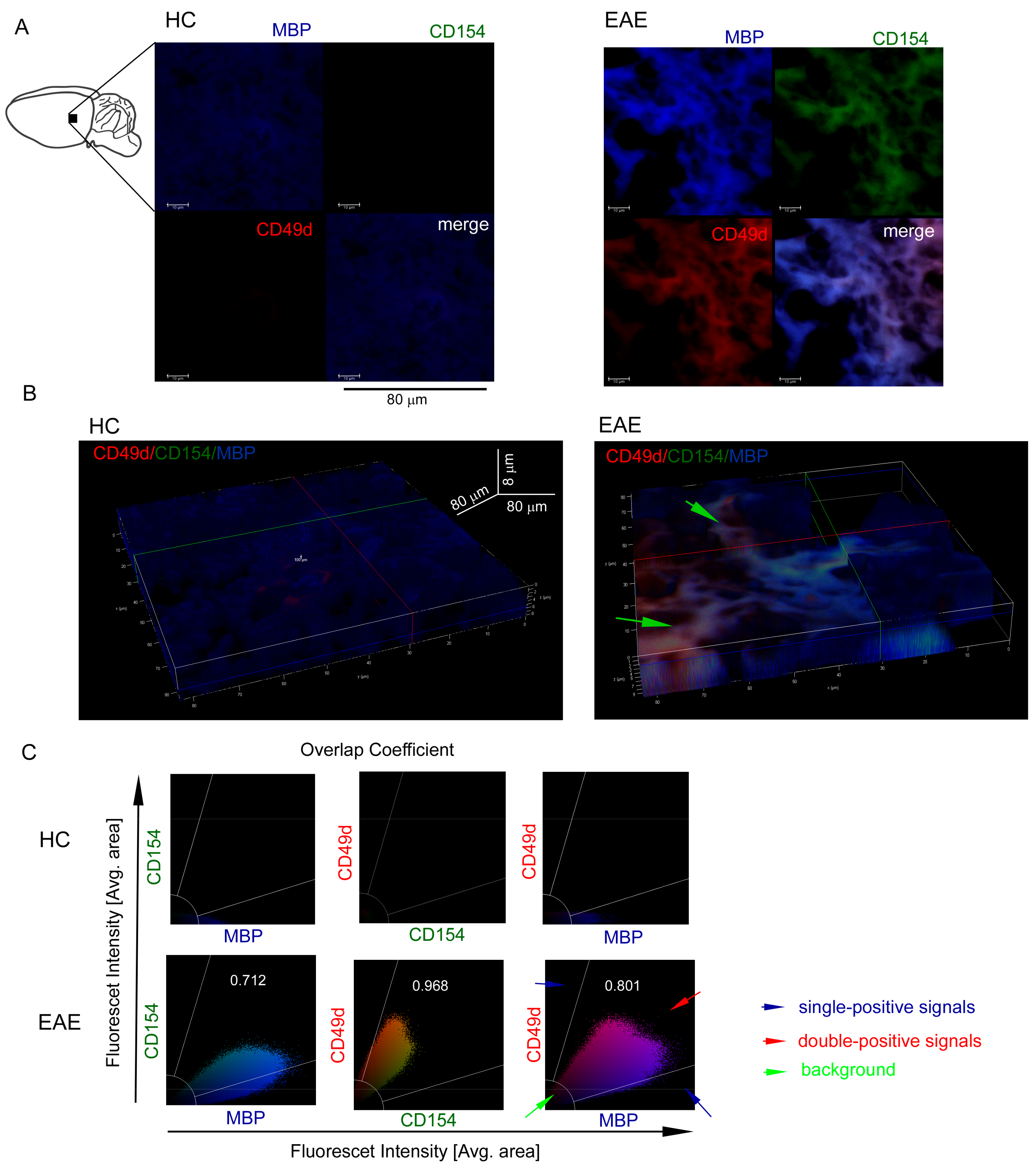
2.10. Mouse CNS Histopathological Examination
2.11. Immunohistochemistry (IHC)
2.12. Statistics
3. Results
3.1. CD49d+CD154+ Lymphocytes of RR-MS Patients Proliferate in Vitro in Response to Myelin Proteins
3.2. Interaction of RR-MS CD49d+CD154+ Lymphocytes with Maturing Human OPCs (hOPCs) Generates Positive Proinflammatory Feedback Loop
3.3. CD49d+CD154+ Lymphocytes are Present in the EAE Mouse Brain
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- McFarland, H.F.; Martin, R. Multiple sclerosis: A complicated picture of autoimmunity. Nat. Immunol. 2007, 8, 913–919. [Google Scholar] [CrossRef] [PubMed]
- Lublin, F.D.; Reingold, S.C.; Cohen, J.A.; Cutter, G.R.; Sørensen, P.S.; Thompson, A.J.; Wolinsky, J.S.; Balcer, L.J.; Banwell, B.; Barkhof, F.; et al. Defining the clinical course of multiple sclerosis: The 2013 revisions. Neurology 2014, 83, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Bramow, S.; Frischer, J.M.; Lassmann, H.; Koch-Henriksen, N.; Lucchinetti, C.F.; Sørensen, P.S.; Laursen, H. Demyelination versus remyelination in progressive multiple sclerosis. Brain 2010, 133, 2983–2998. [Google Scholar] [CrossRef] [PubMed]
- Chang, A.; Tourtellotte, W.W.; Rudick, R.; Trapp, B.D. Premyelinating oligodendrocytes in chronic lesions of multiple sclerosis. N. Engl. J. Med. 2002, 346, 165–173. [Google Scholar] [CrossRef]
- Pierson, E.R.; Stromnes, I.M.; Goverman, J.M. B cells promote induction of experimental autoimmune encephalomyelitis by facilitating reactivation of T cells in the central nervous system. J. Immunol. 2014, 192, 929–939. [Google Scholar] [CrossRef]
- Prineas, J.W.; Connell, F. Remyelination in multiple sclerosis. Ann. Neurol. 1979, 5, 22–31. [Google Scholar] [CrossRef]
- Gerritse, K.; Laman, J.D.; Noelle, R.J.; Aruffo, A.; Ledbetter, J.A.; Boersma, W.J.; Claassen, E. CD40-CD40 ligand interactions in experimental allergic encephalomyelitis and multiple sclerosis. Proc. Natl. Acad. Sci. USA 1996, 93, 2499–2504. [Google Scholar] [CrossRef]
- Issazadeh, S.; Navikas, V.; Schaub, M.; Sayegh, M.; Khoury, S. Kinetics of expression of costimulatory molecules and their ligands in murine relapsing experimental autoimmune encephalomyelitis in vivo. J. Immunol. 1998, 161, 1104–1112. [Google Scholar]
- Aarts, S.A.B.M.; Seijkens, T.T.P.; van Dorst, K.J.F.; Dijkstra, C.D.; Kooij, G.; Lutgens, E. The CD40–CD40L dyad in experimental autoimmune encephalomyelitis and multiple sclerosis. Front. Immunol. 2017, 12, 1791. [Google Scholar] [CrossRef]
- Grewal, I.S.; Foellmer, H.G.; Grewal, K.D.; Xu, J.; Hardardottir, F.; Baron, J.L.; Janeway, C.A., Jr.; Flavell, R.A. Requirement for CD40 ligand in costimulation induction, T cell activation, and experimental allergic encephalomyelitis. Science 1996, 273, 1864–1867. [Google Scholar] [CrossRef]
- Abromson-Leeman, S.; Maverakis, E.; Bronson, R.; Dorf, M.E. CD40-mediated activation of T cells accelerates, but is not required for, encephalitogenic potential of myelin basic protein-recognizing T cells in a model of progressive experimental autoimmune encephalomyelitis. Eur. J. Immunol. 2001, 31, 527–538. [Google Scholar] [CrossRef]
- Laman, J.D.; Maassen, C.B.; Schellekens, M.M.; Visser, L.; Kap, M.; de Jong, E.; van Puijenbroek, M.; van Stipdonk, M.J.; van Meurs, M.; Schwärzler, C.; et al. Therapy with antibodies against CD40L (CD154) and CD44-variant isoforms reduces experimental autoimmune encephalomyelitis induced by a proteolipid protein peptide. Mult. Scler. J. 1998, 4, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Howard, L.M.; Miga, A.J.; Vanderlugt, C.L.; Dal Canto, M.C.; Laman, J.D.; Noelle, R.J.; Miller, S.D. Mechanisms of immunotherapeutic intervention by anti-CD40L (CD154) antibody in an animal model of multiple sclerosis. J. Clin. Invest 1999, 103, 281–290. [Google Scholar] [CrossRef] [PubMed]
- Becher, B.; Durell, B.G.; Miga, A.V.; Hickey, W.F.; Noelle, R.J. The clinical course of experimental autoimmune encephalomyelitis and inflammation is controlled by the expression of CD40 within the central nervous system. J. Exp. Med. 2001, 193, 967–974. [Google Scholar] [CrossRef] [PubMed]
- International Multiple Sclerosis Genetics Consortium (IMSGC); Beecham, A.H.; Patsopoulos, N.A.; Xifara, D.K.; Davis, M.F.; Kemppinen, A.; Cotsapas, C.; Shah, T.S.; Spencer, C.; Booth, D.; et al. Wellcome Trust Case Control Consortium 2 (WTCCC2). Analysis of immune-related loci identifies 48 new susceptibility variants for multiple sclerosis. Nat. Genet. 2013, 45, 1353–1360. [Google Scholar]
- Wagner, M.; Wisniewski, A.; Bilinska, M.; Pokryszko-Dragan, A.; Cyrul, M.; Kusnierczyk, P.; Jasek, M. Investigation of gene-gene interactions between CD40 and CD40L in Polish multiple sclerosis patients. Hum. Immunol. 2014, 75, 796–801. [Google Scholar] [CrossRef]
- Ding, C.; Ireland, S.J.; Remington, G.; Alvarez, E.; Racke, M.K.; Greenberg, B.; Frohman, E.M.; Monson, N.L. CD40-mediated NF-κB activation in B cells is increased in multiple sclerosis and modulated by therapeutics. J. Immunol. 2016, 197, 4257–4265. [Google Scholar]
- Rommer, P.S.; Dudesek, A.; Stüve, O.; Zettl, U.K. Monoclonal antibodies in treatment of multiple sclerosis. Clin. Exp. Immunol. 2014, 175, 373–384. [Google Scholar] [CrossRef]
- Elovaara, I.; Ukkonen, M.; Leppäkynnäs, M.; Lehtimäki, T.; Luomala, M.; Peltola, J.; Dastidar, P. Adhesion molecules in multiple sclerosis: Relation to subtypes of disease and methylprednisolone therapy. Arch. Neurol. 2000, 57, 546–551. [Google Scholar] [CrossRef]
- Polman, C.H.; Reingold, S.C.; Banwell, B.; Clanet, M.; Cohen, J.A.; Filippi, M.; Fujihara, K.; Havrdova, E.; Hutchinson, M.; Kappos, L.; et al. Diagnostic criteria for multiple sclerosis: Revisions to the McDonald criteria. Ann. Neurol. 2011, 69, 292–302. [Google Scholar] [CrossRef]
- Kilkenny, C.; Browne, W.; Cuthill, I.C.; Emerson, M.; Altman, D.G. Animal research: Repoting in vivo experiments: The ARRIVE guidelines. Br. J. Pharmacol. 2010, 160, 1577–1579. [Google Scholar] [CrossRef] [PubMed]
- Buntinx, M.; Vanderlocht, J.; Hellings, N.; Vandenabeele, F.; Lambrichts, I.; Raus, J.; Ameloot, M.; Stinissen, P.; Steels, P. Characterization of three human oligodendroglial cell lines as a model to study oligodendrocyte injury: Morphology and oligodendrocyte-specific gene expression. J. Neurocytol. 2003, 32, 25–38. [Google Scholar] [CrossRef] [PubMed]
- Givan, A.L. A flow cytometric assay for quantitation of rare antigen-specific T cells: Using cell-tracking dyes to calculate precursor frequencies for proliferation. Immunol. Invest 2007, 36, 563–580. [Google Scholar] [CrossRef] [PubMed]
- Lewkowicz, P.; Cwiklińska, H.; Mycko, M.P.; Cichalewska, M.; Domowicz, M.; Lewkowicz, N.; Jurewicz, A.; Selmaj, K.W. Dysregulated RNA-induced silencing complex (RISC) assembly within CNS corresponds with abnormal miRNA expression during autoimmune demyelination. J. Neurosci. 2015, 35, 7521–7537. [Google Scholar] [CrossRef]
- Clark, E.A.; Ledbetter, J.A. How B and T cells talk to each other. Nature 1994, 367, 425–428. [Google Scholar] [CrossRef]
- Chatzigeorgiou, A.; Lyberi, M.; Chatzilymperis, G.; Nezos, A.; Kamper, E. CD40/CD40L signaling and its implication in health and disease. Biofactors 2009, 35, 474–483. [Google Scholar] [CrossRef]
- Chu, T.; Shields, L.B.E.; Zhang, Y.P.; Feng, S.Q.; Shields, C.B.; Cai, J. CXCL12/CXCR4/CXCR7 chemokine axis in the central nervous system: Therapeutic targets for remyelination in demyelinating diseases. Neuroscientist 2017, 23, 627–648. [Google Scholar] [CrossRef]
- Reboldi, A.; Coisne, C.; Baumjohann, D.; Benvenuto, F.; Bottinelli, D.; Lira, S.; Uccelli, A.; Lanzavecchia, A.; Engelhardt, B.; Sallusto, F. C-C chemokine receptor 6-regulated entry of TH-17 cells into the CNS through the choroid plexus is required for the initiation of EAE. Nat. Immunol. 2009, 10, 514–523. [Google Scholar] [CrossRef]
- Falcão, A.M.; van Bruggen, D.; Marques, S.; Meijer, M.; Jäkel, S.; Agirre, E.; Floriddia, E.M.; Vanichkina, D.P.; ffrench-Constant, C.; Williams, A.; et al. Disease-specific oligodendrocyte lineage cells arise in multiple sclerosis. Nat Med. 2018, 24, 1837–1844. [Google Scholar] [CrossRef]
- Franklin, R.J.M.; ffrench-Constant, C. Regenerating CNS myelin—From mechanisms to experimental medicines. Nat. Rev. Neurosci. 2017, 18, 753–769. [Google Scholar] [CrossRef]
- Goldman, S.A.; Nedergaard, M.; Windrem, M.S. Glial progenitor cell-based treatment and modeling of neurological disease. Science 2012, 338, 491–495. [Google Scholar] [CrossRef] [PubMed]
- Calderon, T.M.; Eugenin, E.A.; Lopez, L.; Kumar, S.S.; Hesselgesser, J.; Raine, C.S.; Berman, J.W. A role for CXCL12 (SDF-1α) in the pathogenesis of multiple sclerosis: Regulation of CXCL12 expression in astrocytes by soluble myelin basic protein. J. Neuroimmunol. 2006, 177, 27–39. [Google Scholar] [CrossRef] [PubMed]
- Moll, N.M.; Cossoy, M.B.; Fisher, E.; Staugaitis, S.M.; Tucky, B.H.; Rietsch, A.M.; Chang, A.; Fox, R.J.; Trapp, B.D.; Ransohoff, R.M. Imaging correlates of leukocyte accumulation and CXCR4/CXCL12 in multiple sclerosis. Arch. Neurol. 2009, 66, 44–53. [Google Scholar] [CrossRef] [PubMed]
- McCandless, E.E.; Zhang, B.; Diamond, M.S.; Klein, R.S. CXCR4 antagonism increases T cell trafficking in the central nervous system and improves survival from West Nile virus encephalitis. Proc. Natl. Acad. Sci. USA 2008, 105, 11270–11275. [Google Scholar] [CrossRef]
- Balabanian, K.; Lagane, B.; Infantino, S.; Chow, K.Y.; Harriague, J.; Moepps, B.; Arenzana-Seisdedos, F.; Thelen, M.; Bachelerie, F. The chemokine SDF-1/CXCL12 binds to and signals through the orphan receptor RDC1 in T lymphocytes. J. Biol. Chem. 2005, 280, 35760–35766. [Google Scholar] [CrossRef]
- Jelcic, I.; Al Nimer, F.; Wang, J.; Lentsch, V.; Planas, R.; Jelcic, I.; Madjovski, A.; Ruhrmann, S.; Faigle, W.; Frauenknecht, K.; et al. Memory B cells activate brain-homing, autoreactive CD4+ T cells in multiple sclerosis. Cell 2018, 175, 85–100. [Google Scholar] [CrossRef]
- Mues, M.; Bartholomaus, I.; Thestrup, T.; Griesbeck, O.; Wekerle, H.; Kawakami, N.; Krishnamoorthy, G. Real-time in vivo analysis of T cell activation in the central nervous system using a genetically encoded calcium indicator. Nat. Med. 2013, 19, 778–783. [Google Scholar] [CrossRef]
- Piatek, P.; Namiecinska, M.; Domowicz, M.; Przygodzka, P.; Wieczorek, M.; Michlewska, S.; Lewkowicz, N.; Tarkowski, M.; Lewkowicz, P. MS CD49d+CD154+ lymphocytes reprogram oligodendrocytes into immune reactive cells affecting CNS regeneration. Cells 2019, 8, 1508. [Google Scholar] [CrossRef]

| PBMC (2 × 106 cells/mL) | ||
|---|---|---|
| RR-MS (n = 10) | HC (n = 10) | |
| CCL1 (I-309) | 22 ± 14.2 # | 6.8 ± 2.99 |
| CCL2 (MCP-1) | 552 ± 177.2 # | 66 ± 25.9 |
| CCL3 (MIP-1α) | <3.1 | <3.1 |
| CCL7 (MCP-3) | 134 ± 69.2 # | <3.2 |
| CCL8 (MCP-2) | 57 ± 23.5 # | <0.6 |
| CCL11(Eotaxin) | 34.8 ± 18.66 | 35.4 ± 13.33 |
| CCL13 (MCP-4) | <9.4 | <9.4 |
| CCL15 (MIP-1D) | <3.1 | <3.1 |
| CCL17 (TARC) | 27 ± 10.9 # | <3.4 |
| CCL19 (MIP-3β) | <9.1 | <9.1 |
| CCL20 (MIP-3α) | 26 ± 9.3 # | <8.7 |
| CCL21 (6Ckine) | 69 ± 24.9 # | <1.2 |
| CCL22 (MDC) | 124 ± 58.3 # | <5.9 |
| CCL23 (MPIF-1) | <3.5 | <3.5 |
| CCL24(Eotaxin-2) | 369. ± 81.1 # | 218 ± 67.1 |
| CCL25 (TECK) | 127 ± 39.0 # | 50.8 ± 25.1 |
| CCL26 (Eotaxin-3) | 9.2 ± 2.19 # | <1.4 |
| CCL27 (CTACK) | 39.4 ± 8.31 # | 0.8 ± 0.42 |
| CX3CL1 (Factalkine) | 109 ± 31.7 # | <9.6 |
| CXCL12 (SDFα+β) | 33 ± 12.0 # | <5.3 |
| CXCL1 (Gro-α) | 456 ± 119.34 # | 46.1 ± 27.11 |
| CXCL2 (Gro-β) | 153 ± 33.9 # | 12 ± 7.8 |
| CXCL5 (ENA-78) | 6205 ± 1842.3 # | 676 ± 227.4 |
| CXCL6 (GCP-2) | 18.9 ± 10.34 # | <3.1 |
| CXCL8 (IL-8) | 11206 ± 909.4 # | 867 ± 299.7 |
| CXCL9 (MIG) | 87 ± 44.7 # | <4.9 |
| CXCL10 (IP-10) | <2.6 | <2.6 |
| CXCL11 (I-TAC) | 59 ± 17.9 # | <2.9 |
| CXCL13 (BCA-1) | <0.1 | <0.1 |
| CXCL16 (SCYB16) | 76 ± 17.8 # | <13.4 |
| TNF-α | 39 ± 16.6 # | <3.1 |
| IFN-γ | 28.1 ± 3.2 # | <0.1 |
| IL-1β | 14 ± 3.9 # | 5 ± 3.7 |
| IL-2 | 3.0 ± 1.96 # | <0.4 |
| IL-4 | 7.1 ± 5.77 | 5.9 ± 3.08 |
| IL-6 | 28 ± 8.1 # | 8 ± 3.2 |
| MIF1 | 1071 ± 377.0 # | 408 ± 144.2 |
| IL-10 | <5.1 | <5.1 |
| IL-16 | 219 ± 83.1 | 213 ± 57.2 |
| GM-CSF | <1.5 | <1.5 |
| MO3.13 (2 × 106 cells/mL) | ||||
|---|---|---|---|---|
| Not Polarized | Polarized to OLs | Polarized to OLs + RR-MS CD49d+CD154+ | Polarized to OLs + HC CD49d+CD154+ | |
| CCL1 (I-309)) | 3.6 ± 2.21 | 6.8 ± 2.99 | 68.1 ± 18.06 * § | 10.4 ± 4.94 |
| CCL2 (MCP-1) | 391.6 ± 94.32 | 895 ± 209.9† | 977 ± 494.1 | 927 ± 200.3 |
| CCL3 (MIP-1α) | <3.1 | <3.1 | <3.1 | <3.1 |
| CCL7 (MCP-3) | <3.2 | <3.2 | <3.2 | <3.2 |
| CCL8 (MCP-2) | <0.6 | <0.6 | 62 ± 15.9 * § | <0.6 |
| CCL11(Eotaxin) | 64.7 ± 19.57 | 95.6 ± 26.47 | 137.0 ± 31.59 * § | 81.8 ± 21.60 |
| CCL13 (MCP-4) | <9.4 | <9.4 | <9.4 | <9.4 |
| CCL15 (MIP-1D) | <3.1 | <3.1 | <3.1 | <3.1 |
| CCL17 (TARC) | <3.4 | <3.4 | <3.4 | <3.4 |
| CCL19 (MIP-3β) | <9.1 | <9.1 | <9.1 | <9.1 |
| CCL20 (MIP-3α) | <8.7 | <8.7 | 99 ± 18.7 * § | <8.7 |
| CCL21 (6Ckine) | 18 ± 4.9 | 29 ± 10.7 | 76.5 ± 18.2 * § | 26 ± 12.94 |
| CCL22 (MDC) | <5.9 | <5.9 | 23 ± 12.1 * § | <5.9 |
| CCL23 (MPIF-1) | <3.5 | <3.5 | <3.5 | <3.5 |
| CCL24(Eotaxin-2) | <1.1 | <1.1 | 71 ± 27.1 * § | 12 ± 11.9* |
| CCL25 (TECK) | 46 ± 18.5 | 69 ± 23.3 | 263 ± 83.3 * § | 79. ± 20.8 |
| CCL26 (Eotaxin-3) | <1.4 | <1.4 | 16.8 ± 2.60 * § | <1.4 |
| CCL27 (CTACK) | 0.4 ± 0.33 | 0.5 ± 0.37 | 29.5 ± 15.19 * § | 1.5 ± 1.25 |
| CX3CL1 (Factalkine) | 57 ± 10.48 | 89 ± 11.8† | 506 ± 46.4 * § | 79 ± 30.9 |
| CXCL12 (SDFα+β) | <5.3 | <5.3 | 264 ± 59.2 * § | <5.3 |
| CXCL1 (Gro-α) | 208.1 ± 79.34 | 347.5 ± 127.78† | 1533.4 ± 301.68 * § | 365.9 ± 195.19 |
| CXCL2 (Gro-β) | 7 ± 3.9 | 21 ± 8.8† | 217.9 ± 185.66 * § | 33 ± 13.5 |
| CXCL5 (ENA-78) | 296. ± 164.8 | 862 ± 227.4† | 10,432 ± 1129.3 * § | 985 ± 395.2 |
| CXCL6 (GCP-2) | 64.8 ± 23.95 | 54.9 ± 27.33 | 329.9 ± 105.99 * § | 64.0 ± 28.58 |
| CXCL8 (IL-8) | 913. ± 209.0 | 3235 ± 998.6† | 16,896 ± 5981.1 * § | 3626 ± 672.1 |
| CXCL9 (MIG) | <4.9 | <4.9 | 69 ± 13.9 * § | <4.9 |
| CXCL10 (IP-10) | <2.6 | <2.6 | <2.6 | <2.6 |
| CXCL11 (I-TAC) | <2.9 | <2.9 | <2.9 | <2.9 |
| CXCL13 (BCA-1) | <0.1 | <0.1 | <0.1 | <0.1 |
| CXCL16 (SCYB16) | 360 ± 178.2 | 383 ± 166.1 | 388 ± 258.7 | 342 ± 187.0 |
| TNF-α | <3.1 | <3.1 | 36.5 ± 12.22 * § | <3.1 |
| IFN-γ | <0.1 | <0.1 | <0.1 | <0.1 |
| IL-1β | <0.3 | <0.3 | 25.8 ± 12.33* § | <0.3 |
| IL-2 | <0.4 | <0.4 | <0.4 | <0.4 |
| IL-4 | <0.4 | <0.4 | <0.4 | <0.4 |
| IL-6 | 3 ± 1.8 | 24 ± 9.8† | 259 ± 13.2 * § | 27 ± 10.2 |
| MIF1 | 1931 ± 822.2 | 2735 ± 767.9† | 3687 ± 872.8 * § | 2708. ± 772.4 |
| IL-10 | <5.1 | <5.1 | <5.1 | <5.1 |
| IL-16 | <13.7 | <13.7 | <13.7 | <13.7 |
| GM-CSF | <1.5 | <1.5 | <1.5 | <1.5 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piatek, P.; Namiecinska, M.; Domowicz, M.; Wieczorek, M.; Michlewska, S.; Matysiak, M.; Lewkowicz, N.; Tarkowski, M.; Lewkowicz, P. Multiple Sclerosis CD49d+CD154+ As Myelin-Specific Lymphocytes Induced During Remyelination. Cells 2020, 9, 15. https://doi.org/10.3390/cells9010015
Piatek P, Namiecinska M, Domowicz M, Wieczorek M, Michlewska S, Matysiak M, Lewkowicz N, Tarkowski M, Lewkowicz P. Multiple Sclerosis CD49d+CD154+ As Myelin-Specific Lymphocytes Induced During Remyelination. Cells. 2020; 9(1):15. https://doi.org/10.3390/cells9010015
Chicago/Turabian StylePiatek, Paweł, Magdalena Namiecinska, Małgorzata Domowicz, Marek Wieczorek, Sylwia Michlewska, Mariola Matysiak, Natalia Lewkowicz, Maciej Tarkowski, and Przemysław Lewkowicz. 2020. "Multiple Sclerosis CD49d+CD154+ As Myelin-Specific Lymphocytes Induced During Remyelination" Cells 9, no. 1: 15. https://doi.org/10.3390/cells9010015
APA StylePiatek, P., Namiecinska, M., Domowicz, M., Wieczorek, M., Michlewska, S., Matysiak, M., Lewkowicz, N., Tarkowski, M., & Lewkowicz, P. (2020). Multiple Sclerosis CD49d+CD154+ As Myelin-Specific Lymphocytes Induced During Remyelination. Cells, 9(1), 15. https://doi.org/10.3390/cells9010015







