Quality Evaluation of Ophiopogonis Radix from Two Different Producing Areas
Abstract
1. Introduction
2. Results
2.1. Optimization of Extraction Conditions
2.2. Optimization of Ultra-fast Performance Liquid Chromatography (UFLC) Conditions
2.3. Optimization of Mass Spectrometry (MS) Conditions
2.4. Method Validation
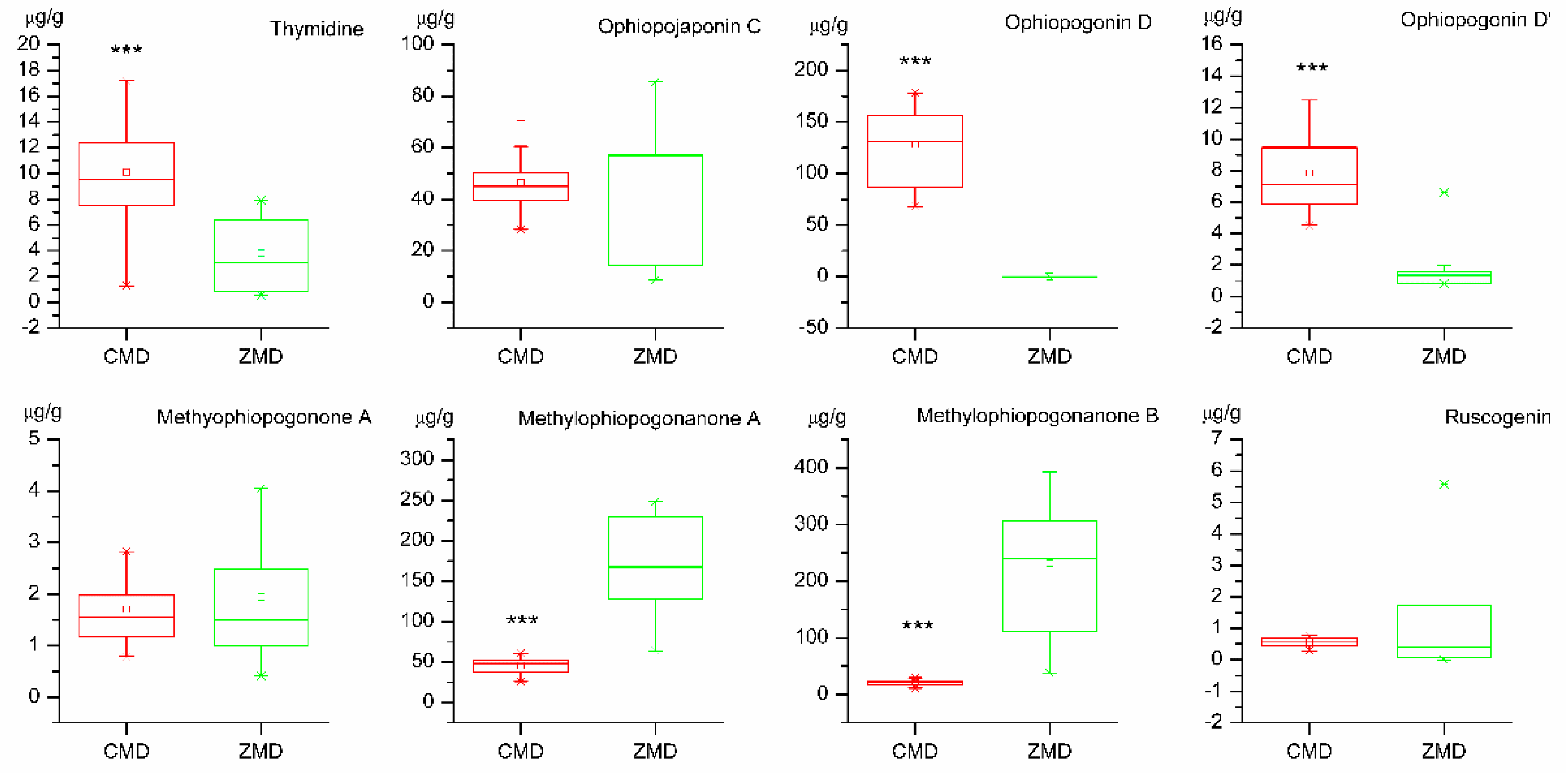
2.5. Quantitative Analysis of Samples
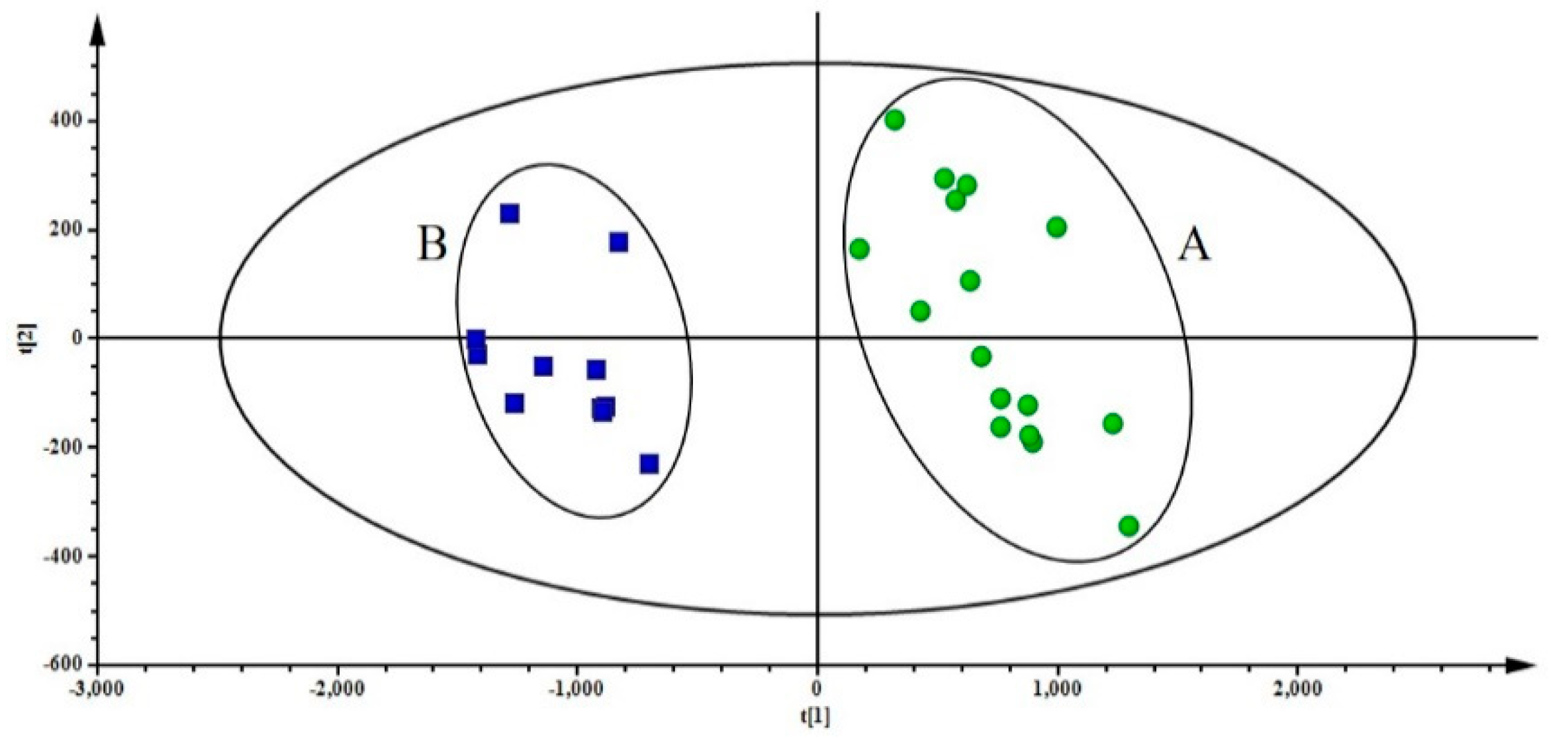
2.6. PCA of Samples
2.7. Partial Least Squares Discriminant Analysis (PLS-DA) of Samples
2.8. t-Test of Samples
2.9. Grey Relational Analysis (GRA) of Samples
3. Discussion
4. Materials and Methods
4.1. Plant Materials
4.2. Chemicals and Reagents
4.3. Preparation of Standard Solutions
4.4. Preparation of Sample Solutions
4.5. Chromatographic and Mass Spectrometric Conditions
4.6. Validation of the Method
4.7. Multivarite Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Chinese Pharmacopoeia Commission. Pharmacopoeia of the People’s Repulic of China. Part I; Chemical Industry Press: Beijing, China, 2015; p. 155. [Google Scholar]
- Chen, M.H.; Chen, X.J.; Wang, M.; Lin, L.G.; Wang, Y.T. Ophiopogon japonicus-A phytochemical, ethnomedicinal and pharmacological review. J. Ethnopharmacol. 2016, 181, 193–213. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Jiang, X.L.; Zhang, W.M.; Li, F.; Khan, A.A.; Liu, X.; Wang, M.K. Homo-aro-cholestane, furostane and spirostane saponins from the tubers of Ophiopogon japonicas. Phytochemistry 2017, 136, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Z.H.; Wu, T.; Li, L.Z.; Liu, N.; Yu, B.Y.; Xu, L.S. Studies on the liposoluble components from tuber of Ophiopogon japonicus. Chin. Pharmacol. J. 2005, 40, 337–341. [Google Scholar]
- Zhao, J.W.; Chen, D.S.; Deng, C.S.; Wang, Q.; Zhu, W.; Lin, L. Evaluation of anti-inflammatory activity of compounds isolated from the rhizome of Ophiopogon japonicas. BMC Complement. Altern. Med. 2017, 17, 7. [Google Scholar] [CrossRef] [PubMed]
- Ishibashi, H.; Mochidome, T.; Okai, J.; Ichiki, H.; Shimada, H.; Takahama, K. Activation of potassium conductance by ophiopogonin-D in acutely dissociated rat paratracheal neurons. British. J. Pharm. 2001, 132, 461–466. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Xu, B.L.; Chen, C.; Jia, H.J.; Wu, J.X.; Wang, X.C.; Sheng, J.L.; Huang, L.; Cheng, J. Methylophiopogonanone A suppresses ischemia/reperfusion-induced myocardial apoptosis in mice via activating PI3K/Akt/eNOS signaling pathway. Acta Pharmacol. Sin. 2016, 37, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.H.; Pang, X.C.; Wang, W.; Wang, Y.J.; Li, X.E.; Qin, M.J. Comparison on homoisoflavones extracted from Ophiopogon Japonicus in Sichuan and Zhejiang and their anti-oxidative activity. Chin. Tradit. Herbal. Drugs 2015, 46, 3091–3095. [Google Scholar]
- Wang, Y.C.; Liu, F.; Liang, Z.S.; Peng, L.; Wang, B.Q.; Yu, J.; Su, Y.Y.; Ma, C.D. Homoisoflavonoids and the antioxidant activity of Ophiopogon japonicus root. Iranian J. Pharm. Res. IJPR 2017, 16, 357–365. [Google Scholar]
- Chen, S.; Li, X.; Liu, L.; Liu, C.; Han, X. Ophiopogonin D alleviates high-fat diet-induced metabolic syndrome and changes the structure of gut microbiota in mice. FASEB J. 2018, 32, 1139–1153. [Google Scholar] [CrossRef]
- Zhang, H.Q.; Liu, P.; Zhu, Z.H.; Dong, L.; Li, W.W.; Qian, D.W.; Duan, J.A. Analysis and evaluation of amino acids and nucleosides in different parts of ripe fruit of Trichosanthes rosthornii harm. Mod. Chin. Med. 2017, 19, 1683–1687. [Google Scholar]
- Zhang, H.Q.; Liu, P.; Duan, J.A.; Dong, L.; Shang, E.X.; Qian, D.W.; Zhu, Z.H.; Li, H.W.; Li, W.W. Comparative analysis of carbohydrates, nucleosides and amino acids in different parts of Trichosanthes kirilowii Maxim. by (Ultra) high-performance liquid chromatography coupled with tandem mass spectrometry and evaporative light scattering detector methods. Molecules 2019, 24, 1140. [Google Scholar]
- Lu, X.Y.; Tong, W.; Wang, S.F.; Li, J.H.; Zheng, J.; Fan, X.H.; Liu, L. Comparison of the chemical consituents and immunomodulatory activity of ophiopogonis radix from two different producing areas. J. Pharm. Biomed. Anal. 2016, 134, 60–70. [Google Scholar] [CrossRef] [PubMed]
- Li, X.E.; Wang, Y.X.; Sun, P.; Liao, D.Q. Determination of saponin content in hang maidong and chuan maidong via HPLC-ELSD analysis. J. Anal. Methods. Chem. 2016, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Dong, Z.J.; Wu, H.Z.; Ding, W.J.; Zhao, M.M.; Shi, Q.W.; Wang, Q. Comparative studies on Ophiopogonis and Liriopes based on the determination of 11 bioactive components using LC-MS/MS and hierarchical clustering analysis. Food Res. Int. 2014, 57, 15–25. [Google Scholar] [CrossRef]
- Jia, C.; Ye, Z.L.; Jiang, X.J.; Zhou, D.Z.; Li, D.K. Simultaneous determination of contents of three flavonoid ingredients in Radix Ophiopogonis by HPLC. Chin. Pharmacol. J. 2011, 46, 1209–1211. [Google Scholar]
- Jiang, Y.; Duan, C.Y.; Chai, X.Y.; Hua, H.M.; Tu, P.F. Studies on the chemical constituents of Ophiopogon japonicus. Chin. J. Chin. Mater. Med. 2007, 32, 1111–1114. [Google Scholar]
- Wu, F.M.; Cai, X.Y.; Wang, P.; Bao, X.H.; Li, M.; Zhou, J. HPLC simultaneous determination of contents of 5 saponin constituents in Ophiopogonis Radix. Chin. J. Chin. Mater. Med. 2015, 40, 4022–4025. [Google Scholar]
- Wu, F.M.; Yang, R.S.; Zhang, S.D.; Bao, X.H.; Li, M.; Zhou, J. Simultaneous determination of three flavone constituents in Ophiopogonis Radix by HPLC method. Chin. Pharm. J. 2016, 51, 655–658. [Google Scholar]
- Wang, S.N.; Hua, Y.J.; Xu, L.; Zou, L.S.; Liu, X.H.; Luo, Y.Y.; Liu, J.X.; Yan, Y. Quality evaluation of Scrophulariae Radix processed by different ‘Sweating’ methods based on simultaneous determination of multiple bioactive constituents combined with grey relational analysis. Molecules 2016, 21, 850. [Google Scholar] [CrossRef]
- Chen, C.H.; Liu, Z.X.; Zou, L.S.; Liu, X.H.; Chai, C.; Zhao, H.; Yan, Y.; Wang, C.C. Quality evaluation of Apocyni Veneti Folium from different habitats and commercial herbs based on simultaneous determination of multiple bioactive constituents combined with multivariate statistical analysis. Molecules 2018, 23, 573. [Google Scholar] [CrossRef]
- Yan, Y.; Zhao, H.; Chen, C.H.; Zou, L.S.; Liu, X.H.; Chai, C.; Wang, C.C.; Shi, J.J.; Chen, S.Y. Comparison of multiple bioactive constituents in different parts of Eucommia ulmoides based on UFLC-QTRAP-MS/MS combined with PCA. Molecules 2018, 23, 643. [Google Scholar]
- Chen, S.Y.; Shi, J.J.; Zou, L.S.; Liu, X.H.; Tang, R.M.; Ma, J.M.; Wang, C.C.; Tan, M.X.; Chen, J.L. Quality evaluation of wild and cultivated Schisandrae Chinensis Fructus based on simultaneous determination of multiple bioactive constituents combined with multivariate statistical analysis. Molecules 2019, 24, 1335. [Google Scholar] [CrossRef] [PubMed]
- Qu, C.; Pu, Z.J.; Zhou, G.S.; Wang, J.; Zhu, Z.H.; Yue, S.J.; Li, J.P.; Shang, L.L.; Tang, Y.P.; Shi, X.Q.; et al. Comparative analysis of main bio-active components in the herb pair Danshen-Honghua and its single herbs by ultra-high performance liguid chromatography coupled to triple quadrupole tandem mass spectrometry. J. Sep. Sci. 2017, 40, 3392–3401. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |
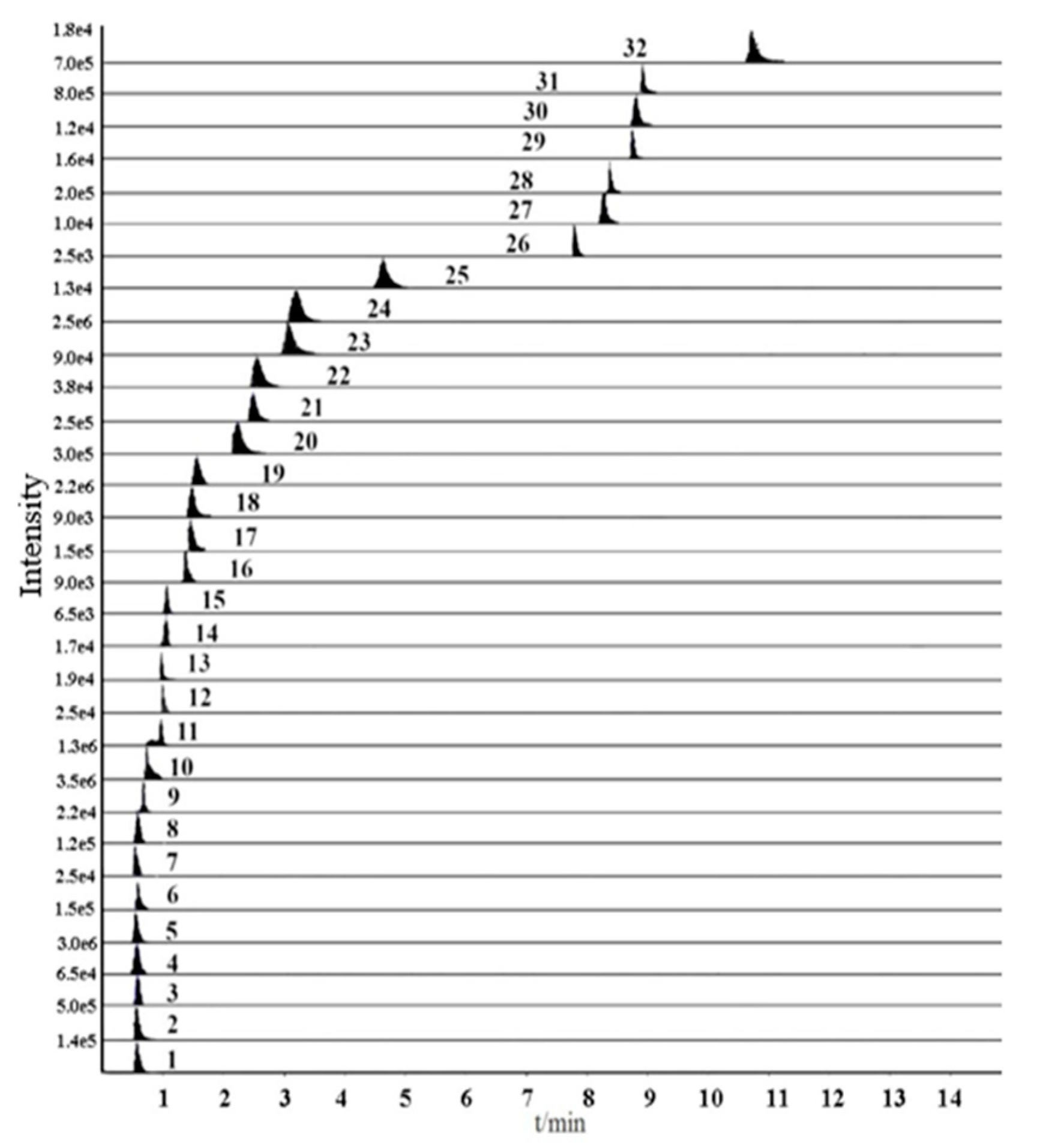



| No. | Compounds | tR (min) | Precursor Ion (m/z) | Product Ion (m/z) | Declustering Potentional (DP)(V) | Collision Energy (CE)(eV) |
|---|---|---|---|---|---|---|
| 1 | Glutamic acid | 0.52 | 147.08 | 83.92 | 83 | 14 |
| 2 | Lysine | 0.52 | 147.11 | 83.91 | 66 | 14 |
| 3 | Histidine | 0.53 | 156.08 | 110.03 | 95 | 16 |
| 4 | Arginine | 0.55 | 175.12 | 70.02 | 88 | 18 |
| 5 | Threonine | 0.57 | 120.07 | 74.00 | 93 | 20 |
| 6 | Aspartic acid | 0.57 | 134.05 | 87.96 | 59 | 10 |
| 7 | Serine | 0.57 | 106.05 | 59.99 | 67 | 8 |
| 8 | Alanine | 0.58 | 90.06 | 44.02 | 79 | 10 |
| 9 | Proline | 0.65 | 116.07 | 70.02 | 68 | 10 |
| 10 | Valine | 0.71 | 118.09 | 72.06 | 54 | 10 |
| 11 | Cytidine | 0.93 | 244.09 | 112.00 | 61 | 10 |
| 12 | Uracil | 0.98 | 113.04 | 70.00 | 111 | 21 |
| 13 | Guanine | 0.98 | 152.00 | 135.00 | 51 | 25 |
| 14 | Methionine | 1.02 | 150.06 | 104.03 | 91 | 10 |
| 15 | 2′-deoxycytidine | 1.07 | 228.20 | 112.05 | 76 | 13 |
| 16 | Isoleucine | 1.36 | 132.10 | 86.05 | 64 | 10 |
| 17 | Uridine | 1.41 | 244.90 | 113.00 | 103 | 13 |
| 18 | Leucine | 1.48 | 132.11 | 86.05 | 98 | 10 |
| 19 | Tyrosine | 1.54 | 182.16 | 136.00 | 46 | 17 |
| 20 | Adenosine | 2.20 | 268.10 | 136.07 | 86 | 23 |
| 21 | Inosine | 2.47 | 269.00 | 137.07 | 46 | 15 |
| 22 | Guanosine | 2.52 | 284.30 | 152.00 | 62 | 15 |
| 23 | Phenylalanine | 3.04 | 166.10 | 120.05 | 56 | 14 |
| 24 | 2′-deoxyguanosine | 3.16 | 268.10 | 152.10 | 61 | 15 |
| 25 | Thymidine | 4.57 | 243.10 | 127.07 | 61 | 13 |
| 26 | Ophiopojaponin C | 7.74 | 887.40 | 393.30 | 110 | 45 |
| 27 | Ophiopogonin D | 8.23 | 855.60 | 287.30 | 30 | 38 |
| 28 | Ophiopogonin D’ | 8.30 | 855.60 | 253.30 | 130 | 45 |
| 29 | Methylophiopogonone A | 8.71 | 339.13 | 130.92 | −135 | −42 |
| 30 | Methylophiopogonanone A | 8.76 | 341.10 | 178.00 | −110 | −46 |
| 31 | Methylophiopogonanone B | 8.90 | 327.10 | 178.00 | −90 | −41 |
| 32 | Ruscogenin | 10.65 | 431.40 | 287.10 | 130 | 47 |
| No. | ri | Ranking | No. | ri | Ranking |
|---|---|---|---|---|---|
| S1 | 0.5096 | 11 | S15 | 0.6414 | 2 |
| S2 | 0.5614 | 8 | S16 | 0.6152 | 4 |
| S3 | 0.6262 | 3 | S17 | 0.3649 | 21 |
| S4 | 0.4180 | 16 | S18 | 0.3539 | 23 |
| S5 | 0.4642 | 15 | S19 | 0.3664 | 20 |
| S6 | 0.4918 | 13 | S20 | 0.3420 | 24 |
| S7 | 0.5229 | 10 | S21 | 0.3965 | 17 |
| S8 | 0.4833 | 14 | S22 | 0.3767 | 18 |
| S9 | 0.5629 | 7 | S23 | 0.3223 | 26 |
| S10 | 0.6513 | 1 | S24 | 0.2860 | 27 |
| S11 | 0.5085 | 12 | S25 | 0.3264 | 25 |
| S12 | 0.5940 | 6 | S26 | 0.3718 | 19 |
| S13 | 0.6074 | 5 | S27 | 0.3631 | 22 |
| S14 | 0.5578 | 9 |
| Samples | No. | Habitats | Samples | No. | Habitats |
|---|---|---|---|---|---|
| CMD | S1 | Guanghui, Sichuan | ZMD | S17 | Lishui, Zhejiang |
| S2 | Yongan, Sichuan | S18 | Lishui, Zhejiang | ||
| S3 | Zengsheng, Sichuan | S19 | Lishui, Zhejiang | ||
| S4 | Yongming, Sichuan | S20 | Lishui, Zhejiang | ||
| S5 | Baiqing, Sichuan | S21 | Lishui, Zhejiang | ||
| S6 | Lingxing, Sichuan | S22 | Cixi, Zhejiang | ||
| S7 | Xinde, Sichuan | S23 | Cixi, Zhejiang | ||
| S8 | Huanyuan, Sichuan | S24 | Cixi, Zhejiang | ||
| S9 | Guangming, Sichuan | S25 | Cixi, Zhejiang | ||
| S10 | Jianshe, Sichuan | S26 | Cixi, Zhejiang | ||
| S11 | Songya, Sichuan | S27 | Cixi, Zhejiang | ||
| S12 | Licheng, Sichuan | ||||
| S13 | Liuying, Sichuan | ||||
| S14 | Laoma, Sichuan | ||||
| S15 | Sanyuan, Sichuan | ||||
| S16 | Shimiao, Sichuan |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tan, M.; Chen, J.; Wang, C.; Zou, L.; Chen, S.; Shi, J.; Mei, Y.; Wei, L.; Liu, X. Quality Evaluation of Ophiopogonis Radix from Two Different Producing Areas. Molecules 2019, 24, 3220. https://doi.org/10.3390/molecules24183220
Tan M, Chen J, Wang C, Zou L, Chen S, Shi J, Mei Y, Wei L, Liu X. Quality Evaluation of Ophiopogonis Radix from Two Different Producing Areas. Molecules. 2019; 24(18):3220. https://doi.org/10.3390/molecules24183220
Chicago/Turabian StyleTan, Mengxia, Jiali Chen, Chengcheng Wang, Lisi Zou, Shuyu Chen, Jingjing Shi, Yuqi Mei, Lifang Wei, and Xunhong Liu. 2019. "Quality Evaluation of Ophiopogonis Radix from Two Different Producing Areas" Molecules 24, no. 18: 3220. https://doi.org/10.3390/molecules24183220
APA StyleTan, M., Chen, J., Wang, C., Zou, L., Chen, S., Shi, J., Mei, Y., Wei, L., & Liu, X. (2019). Quality Evaluation of Ophiopogonis Radix from Two Different Producing Areas. Molecules, 24(18), 3220. https://doi.org/10.3390/molecules24183220






