The Association between Hormone Replacement Therapy and Gastroparesis in Post-Menopausal Women: A Worldwide Database Analysis
Abstract
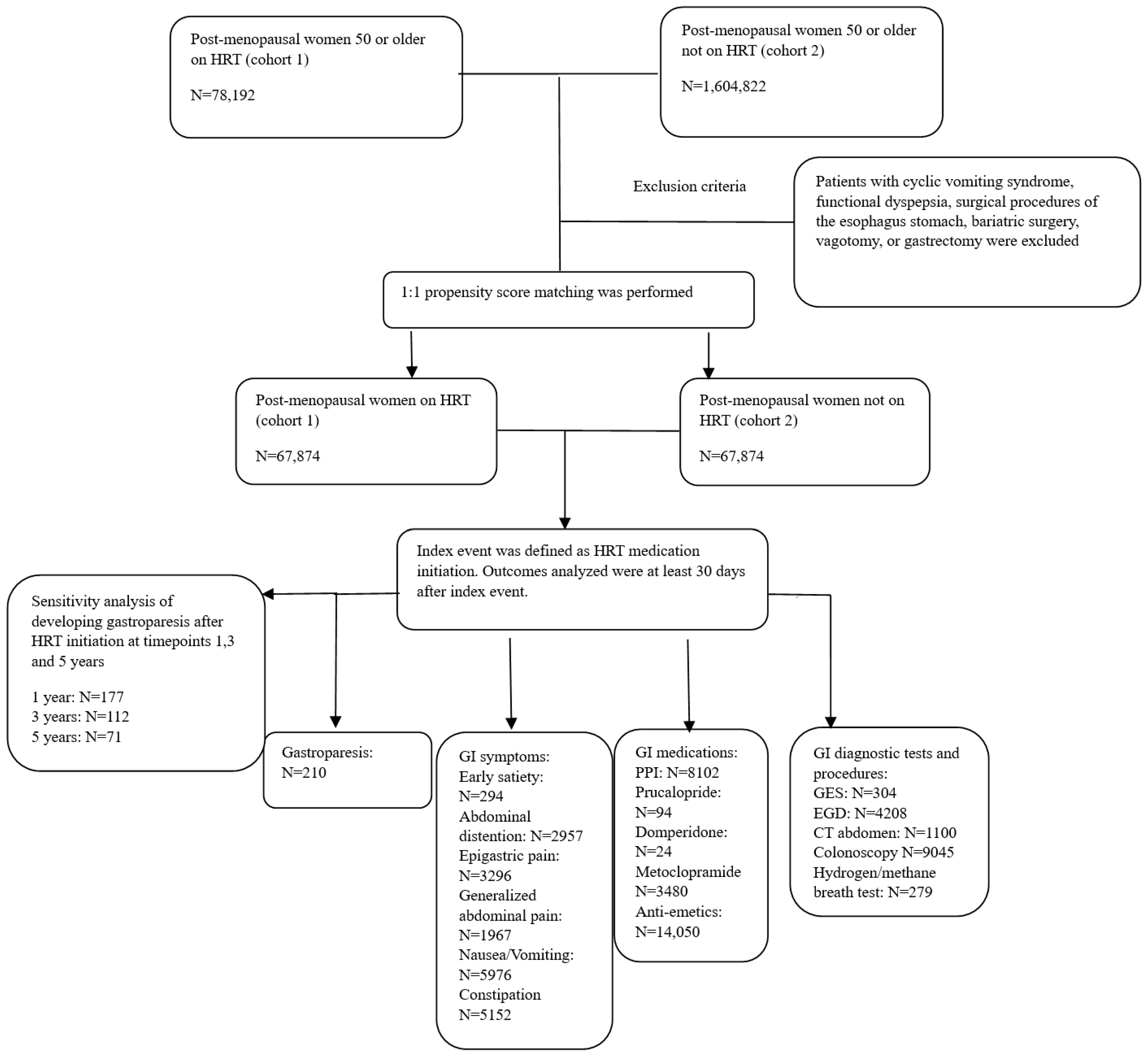
:1. Introduction
2. Materials and Methods
2.1. Data Source
2.2. Study Design
2.3. Institutional Review Board
2.4. Confounding Variables
2.5. Propensity Score Matching Details
2.6. Statistical Analysis
3. Results
3.1. Gastrointestinal Symptoms
3.2. Medications
3.3. Diagnostic Testing
4. Discussion
4.1. Hormone Replacement Therapy and Racial/Ethnic Implications
4.2. Hormone Replacement Therapy and GI Symptoms
4.3. Hormone Replacement Therapy and Medications
4.4. Hormone Replacement Therapy and Diagnostic Tests and Procedures
4.5. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ali, M.; Gonzalez, Z.; McCallum, R. Chapter 37-Female Predominance in Gastroparesis. In Gastroparesis; Academic Press: Cambridge, MA, USA, 2021; pp. 495–505. [Google Scholar]
- Camilleri, M.; Kuo, B.; Nguyen, L.; Vaughn, V.M.; Petrey, J.; Greer, K.; Yadlapati, R.; Abell, T.L. ACG Clinical Guideline: Gastroparesis. Am. J. Gastroenterol. 2022, 117, 1197–1220. [Google Scholar] [CrossRef]
- Langworthy, J.; Parkman, H.; Schey, R. Emerging Strategies for the Treatment of Gastroparesis. Expert Rev. Gastroenterol. Hepatol. 2016, 10, 817–825. [Google Scholar] [CrossRef]
- Oh, J.H.; Pasricha, P.J. Recent Advances in the Pathophysiology and Treatment of Gastroparesis. J. Neurogastroenterol. Motil. 2013, 19, 18–24. [Google Scholar] [CrossRef]
- Sato, H.; Grover, M. Gastroparesis and Functional Dyspepsia: Spectrum of Gastroduodenal Neuromuscular Disorders or Unique Entities? Gastro Hep Adv. 2023, 2, 438–448. [Google Scholar] [CrossRef]
- Zheng, T.; BouSaba, J.; Sanna, W.; Eckert, D.; Burton, D.; Camilleri, M. Comprehensive characterization of antral and pyloric contractions by high resolution manometry: Applied physiology in suspected gastroparesis. Am. J. Physiol.-Gastrointest. Liver Physiol. 2022, 323, G255–G264. [Google Scholar] [CrossRef]
- Yang, X.; Guo, Y.; He, J.; Zhang, F.; Sun, X.; Yang, S.; Dong, H. Estrogen and Estrogen Receptors in the Modulation of Gastrointestinal Epithelial Secretion. Oncotarget 2017, 8, 97683–97692. [Google Scholar] [CrossRef]
- Gangula, P.R.; Mukhopadhyay, S.; Pasricha, P.J.; Ravella, K. Sepiapterin Reverses the Changes in Gastric nNOS Dimerization and Function in Diabetic Gastroparesis. Neurogastroenterol. Motil. 2010, 22, 1325–1331. [Google Scholar] [CrossRef]
- Jung, H.K.; Choung, R.S.; Locke, G.R.; Schleck, C.D.; Zinsmeister, A.R.; Szarka, L.A.; Mullan, B.; Talley, N.J. The Incidence, Prevalence, and Outcomes of Patients with Gastroparesis in Olmsted County, Minnesota, from 1996 to 2006. Gastroenterology 2009, 136, 1225–1233. [Google Scholar] [CrossRef]
- Ye, Y.; Jiang, B.; Manne, S.; Moses, P.L.; Almansa, C.; Bennett, D.; Dolin, P.; Ford, A.C. Epidemiology and outcomes of gastroparesis, as documented in general practice records, in the United Kingdom. Gut 2021, 70, 644–653. [Google Scholar] [CrossRef]
- Zia, J.K.; Heitkemper, M.M. Upper gastrointestinal tract motility disorders in women, gastroparesis, and gastroesophageal reflux disease. Gastroenterol. Clin. N. Am. 2016, 45, 239–251. [Google Scholar] [CrossRef]
- Ravella, K.; Al-Hendy, A.; Sharan, C.; Hale, A.B.; Channon, K.M.; Srinivasan, S.; Gangula, P.R. Chronic estrogen deficiency causes gastroparesis by altering neuronal nitric oxide synthase function. Dig. Dis. Sci. 2013, 58, 1507–1515. [Google Scholar] [CrossRef]
- Hutson, W.R.; Roehrkasse, R.L.; Wald, A. Influence of gender and menopause on gastric emptying and motility. Gastroenterology 1989, 96, 11–17. [Google Scholar] [CrossRef]
- Datz, F.; Christian, P.; Moore, J. Differences in gastric emptying rates between menstruating and postmenopausal women. J. Nucl. Med. 1987, 28, 604–605. [Google Scholar]
- Gangula, P.R.; Sekhar, K.R.; Mukhopadhyay, S. Gender bias in gastroparesis: Is nitric oxide the answer? Dig. Dis. Sci. 2011, 56, 2520–2527. [Google Scholar] [CrossRef]
- TriNetX, L. Volume 2023. 2023. Available online: https://TriNetX.com (accessed on 9 September 2023).
- Kato, I.; Toniolo, P.; Akhmedkhanov, A.; Koenig, K.L.; Shore, R.; Zeleniuch-Jacquotte, A. Prospective study of factors influencing the onset of natural menopause. J. Clin. Epidemiol. 1998, 51, 1271–1276. [Google Scholar] [CrossRef]
- Von Elm, E.; Altman, D.; Egger, M.; Pocock, S.; Gotzsche, P.; Vandenbroucke, J. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. Lancet 2007, 370, 1453–1457. [Google Scholar] [CrossRef]
- Austin, P.C. An introduction to propensity score methods for reducing the effects of confounding in observational studies. Multivar. Behav. Res. 2011, 46, 399–424. [Google Scholar] [CrossRef]
- Hadi, Y.; Lakhani, D.; Naqvi, S.; Fatima, N.; Sarwari, A. Outcomes of SARS-CoV-2 infection in patients with cystic fibrosis: A multicenter retrospective research network study. Respir. Med. 2021, 188, 106606. [Google Scholar] [CrossRef]
- Hadi, Y.B.; Naqvi, S.F.Z.; Kupec, J.T.; Sarwari, A.R. Characteristics and outcomes of COVID-19 in patients with HIV: A multicentre research network study. AIDS 2020, 34, F3–F8. [Google Scholar] [CrossRef]
- Hadi, Y.B.; Naqvi, S.F.Z.; Kupec, J.T.; Sofka, S.; Sarwari, A. Outcomes of COVID-19 in solid organ transplant recipients: A propensity-matched analysis of a large research network. Transplantation 2021, 105, 1365–1371. [Google Scholar] [CrossRef]
- Hadi, Y.B.; Lakhani, D.A.; Naqvi, S.F.Z.; Singh, S.; Kupec, J.T. Outcomes of SARS-CoV-2 infection in patients with pulmonary sarcoidosis: A multicenter retrospective research network study. Respir. Med. 2021, 187, 106538. [Google Scholar] [CrossRef]
- Hallan, D.R.; Freedman, Z.; Rizk, E. Obesity Paradox and Surgical Evacuation for Chronic Subdural Hematoma. Cureus 2022, 14, e24002. [Google Scholar] [CrossRef]
- Haukoos, J.S.; Lewis, R.J. The Propensity Score. JAMA 2015, 314, 1637–1638. [Google Scholar] [CrossRef]
- Parkman, H.P.; Harris, A.D.; Miller, M.A.; Fisher, R.S. Influence of age, gender, and menstrual cycle on the normal electrogastrogram. Am. J. Gastroenterol. 1996, 91, 127–133. [Google Scholar]
- Parkman, H.P.; Yamada, G.; Van Natta, M.L.; Yates, K.; Hasler, W.L.; Sarosiek, I.; Grover, M.; Schey, R.; Abell, T.L.; Koch, K.L.; et al. Ethnic, Racial, and Sex Differences in Etiology, Symptoms, Treatment, and Symptom Outcomes of Patients With Gastroparesis. Clin. Gastroenterol. Hepatol. 2018, 17, 1489–1499.e8. [Google Scholar] [CrossRef]
- Pershad, A.; Morris, J.; Khanna, P. Racial and Ethnic Disparities in Menopausal Hormone Therapy Acceptance [A134]. Obstet. Gynecol. 2022, 139, 39S. [Google Scholar] [CrossRef]
- Revicki, D.A.; Rentz, A.M.; Dubois, D.; Kahrilas, P.; Stanghellini, V.; Talley, N.J.; Tack, J. Development and validation of a patient-assessed gastroparesis symptom severity measure: The Gastroparesis Cardinal Symptom Index. Aliment. Pharmacol. Ther. 2003, 18, 141–150. [Google Scholar] [CrossRef]
- Hasler, W.L.; Parkman, H.P.; Wilson, L.A.; Pasricha, P.J.; Koch, K.L.; Abell, T.L.; Snape, W.J.; Farrugia, G.; Lee, L.; Tonascia, J.; et al. Psychological dysfunction is associated with symptom severity but not disease etiology or degree of gastric retention in patients with gastroparesis. Am. J. Gastroenterol. 2010, 105, 2357–2367. [Google Scholar] [CrossRef]
- Janssens, J.; Peeters, T.L.; Vantrappen, G.; Tack, J.; Urbain, J.L.; De Roo, M.; Muls, E.; Bouillon, R. Improvement of gastric emptying in diabetic gastroparesis by erythromycin. Preliminary studies. N. Engl. J. Med. 1990, 322, 1028–1031. [Google Scholar] [CrossRef]
- Khoo, J.; Rayner, C.K.; Jones, K.L.; Horowitz, M. Pathophysiology and management of gastroparesis. Expert Rev. Gastroenterol. Hepatol. 2009, 3, 167–181. [Google Scholar] [CrossRef]
- Parkman, H.P.; Hasler, W.L.; Fisher, R.S. American Gastroenterological Association technical review on the diagnosis and treatment of gastroparesis. Gastroenterology 2004, 127, 1592–1622. [Google Scholar] [CrossRef]
- Tonini, M.; Cipollina, L.; Poluzzi, E.; Crema, F.; Corazza, G.R.; De Ponti, F. Review article: Clinical implications of enteric and central D2 receptor blockade by antidopaminergic gastrointestinal prokinetics. Aliment. Pharmacol. Ther. 2004, 19, 379–390. [Google Scholar] [CrossRef]
- Dumitrascu, D.L.; Weinbeck, M. Domperidone versus metoclopramide in the treatment of diabetic gastroparesis. Am. J. Gastroenterol. 2000, 95, 316–317. [Google Scholar] [CrossRef] [PubMed]
- Quigley, E.M.M. Prucalopride: Safety, efficacy and potential applications. Ther. Adv. Gastroenterol. 2012, 5, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.; Ramsey, F.V.; Norton, W.F.; Norton, N.; Schneck, S.; Gaetano, T.; Parkman, H.P. The Burdens, Concerns, and Quality of Life of Patients with Gastroparesis. Dig. Dis. Sci. 2017, 62, 879–893. [Google Scholar] [CrossRef]
- Reddymasu, S.C.; McCallum, R.W. Small Intestinal Bacterial Overgrowth in Gastroparesis: Are There Any Predictors? J. Clin. Gastroenterol. 2010, 44, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Rezaie, A.; Buresi, M.; Lembo, A.; Lin, H.; McCallum, R.; Rao, S.; Schmulson, M.; Valdovinos, M.; Zakko, S.; Pimentel, M. Hydrogen and Methane-Based Breath Testing in Gastrointestinal Disorders: The North American Consensus. Am. J. Gastroenterol. 2017, 112, 775–784. [Google Scholar] [CrossRef]
| Cohort 1 (N = 78,192) and Cohort 2 (N = 1,604,822) before Propensity Matching | Cohort 1 (N = 67,874) and Cohort 2 (N = 67,874) after Propensity Matching | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Cohort | Mean ± SD | Patients | %Cohort | p-Value | SD | Mean ± SD | Patients | %Cohort | p-Value | SD | |
| Age at Index | 1 | 57.9 ± 6.7 | 67,874 | 100% | <0.001 | 0.735 | 57.9 ± 6.7 | 67,874 | 100% | 0.992 | <0.001 |
| 2 | 63.7 ± 8.9 | 1,578,637 | 100% | 57.9 ± 6.7 | 67,874 | 100% | |||||
| Female | 1 | 67,874 | 100% | - | - | 67,874 | 100% | - | - | ||
| 2 | 1,578,637 | 100% | 67,874 | 100% | |||||||
| White | 1 | 54,710 | 80.6% | <0.001 | 0.202 | 54,710 | 80.6% | 1 | <0.001 | ||
| 2 | 1,137,542 | 72.1% | 54,710 | 80.6% | |||||||
| Not Hispanic/Latino | 1 | 51,502 | 75.9% | <0.001 | 0.189 | 51,502 | 75.9% | 1 | <0.001 | ||
| 2 | 1,064,194 | 67.4% | 51,502 | 75.9% | |||||||
| Black or African American | 1 | 3815 | 5.6% | <0.001 | 0.198 | 3815 | 5.6% | 0.962 | <0.001 | ||
| 2 | 174,524 | 11.1% | 3819 | 5.6% | |||||||
| Hispanic or Latino | 1 | 2860 | 4.2% | <0.001 | 0.141 | 2860 | 4.2% | 0.925 | 0.001 | ||
| 2 | 118,797 | 7.5% | 2853 | 4.2% | |||||||
| Asian | 1 | 1626 | 2.4% | <0.001 | 0.082 | 1626 | 2.4% | 1 | <0.001 | ||
| 2 | 60,399 | 3.8% | 1626 | 2.4% | |||||||
| American Indian/Alaska Native | 1 | 141 | 0.2% | 0.373 | 0.004 | 141 | 0.2% | 0.764 | 0.002 | ||
| 2 | 3540 | 0.2% | 136 | 0.2% | |||||||
| Native Hawaiian/Pacific Islander | 1 | 79 | 0.1% | <0.001 | 0.052 | 79 | 0.1% | 1 | <0.001 | ||
| 2 | 5892 | 0.4% | 79 | 0.1% | |||||||
| BMI | 1 | 26.8 ± 5.8 | 32,415 | 47.8% | <0.001 | 0.402 | 26.8 ± 5.8 | 32,424 | 47.8% | 0.987 | <0.001 |
| 2 | 29.3 ±6.8 | 395,212 | 25.0% | 28.9 ± 6.8 | 32,421 | 47.8% | |||||
| Hgb A1c | 1 | 5.7 ± 1.1 | 17,710 | 26.1% | <0.001 | 0.405 | 5.7 ± 1.1 | 17,711 | 26.1% | 0.985 | 0.172 |
| 2 | 6.3 ± 1.6 | 415,386 | 26.3% | 6.0 ± 1.4 | 17,708 | 26.1% | |||||
| Diabetes Mellitus | 1 | 5051 | 7.4% | <0.001 | 0.256 | 5051 | 7.4% | 0.967 | <0.001 | ||
| 2 | 245,285 | 15.5% | 5047 | 7.4% | |||||||
| OR | 95% CI | p-Value | |
|---|---|---|---|
| Year 1 | 1.44 | 1.144–1.814 | 0.0018 |
| Year 3 | 1.602 | 1.188–2.159 | 0.0018 |
| Year 5 | 1.653 | 1.132–2.414 | 0.0086 |
| OR | 95% CI | p-Value | %RISK | |
|---|---|---|---|---|
| Early satiety | 1.227 | [1.034–1.456] | 0.0187 | 0.435% |
| Abdominal distention | 1.441 | [1.362–1.526] | <0.0001 | 4.59% |
| Epigastric pain | 1.187 | [1.127–1.249] | <0.0001 | 5.166% |
| Generalized abdominal pain | 1.203 | [1.126–1.285] | <0.0001 | 3.008% |
| Nausea and vomiting | 1.138 | [1.095–1.183] | <0.0001 | 9.849% |
| Constipation | 1.219 | [1.169–1.271] | <0.0001 | 8.484% |
| Weight loss | 1.057 | [0.986–1.132] | 0.1180 | 2.564% |
| OR | 95% CI | p-Value | %Risk | |
|---|---|---|---|---|
| PPI | 1.14 | [1.102–1.179] | <0.0001 | 16.421% |
| Prucalopride | 3.765 | [2.422–5.853] | <0.0001 | 0.139% |
| Domperidone | 2.402 | [1.148–5.023] | 0.0163 | 0.035% |
| Metoclopramide | 1.112 | [1.059–1.169] | <0.0001 | 5.487% |
| Antiemetics | 1.21 | [1.176–1.245] | <0.0001 | 29.704% |
| OR | 95% CI | p-Value | %risk | |
|---|---|---|---|---|
| GES | 1.674 | [1.393–2.013] | <0.0001 | 0.45% |
| EGD | 1.199 | [1.145–1.255] | <0.0001 | 6.624% |
| CT abdomen | 1.25 | [1.143–1.366] | <0.0001 | 1.678% |
| Colonoscopy | 1.204 | [1.166–1.244] | <0.0001 | 16.343% |
| Hydrogen/Methane breath test | 1.783 | [1.466–2.169] | <0.0001 | 0.413% |
| MRI abodomen | 1.007 | [0.931–1.09] | 0.8580 | 1.875% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khalil, J.; Hill, H.; Kaelber, D.; Song, G. The Association between Hormone Replacement Therapy and Gastroparesis in Post-Menopausal Women: A Worldwide Database Analysis. J. Pers. Med. 2024, 14, 275. https://doi.org/10.3390/jpm14030275
Khalil J, Hill H, Kaelber D, Song G. The Association between Hormone Replacement Therapy and Gastroparesis in Post-Menopausal Women: A Worldwide Database Analysis. Journal of Personalized Medicine. 2024; 14(3):275. https://doi.org/10.3390/jpm14030275
Chicago/Turabian StyleKhalil, Jacqueline, Hannah Hill, David Kaelber, and Gengqing Song. 2024. "The Association between Hormone Replacement Therapy and Gastroparesis in Post-Menopausal Women: A Worldwide Database Analysis" Journal of Personalized Medicine 14, no. 3: 275. https://doi.org/10.3390/jpm14030275





