Biogeography of Selected Spring Endemics in Texas Interglacial-Drought Refugia with Unexpected Insights from a Spring-Dependent Nematode Parasite
Abstract
1. Introduction
Unexpected Distributional Discontinuities
2. Background Review and Proposed Model
- the geographical setting and the geological and chronological scopes of our study,
- the biocenotic communities and their crenal biotopes that led us to develop the proposed model, and
- the functional aspects of our proposed refugial model, the logic underpinning it, and the new name we have proposed for this unique category of evolutionary refugia.
2.1. Our Study
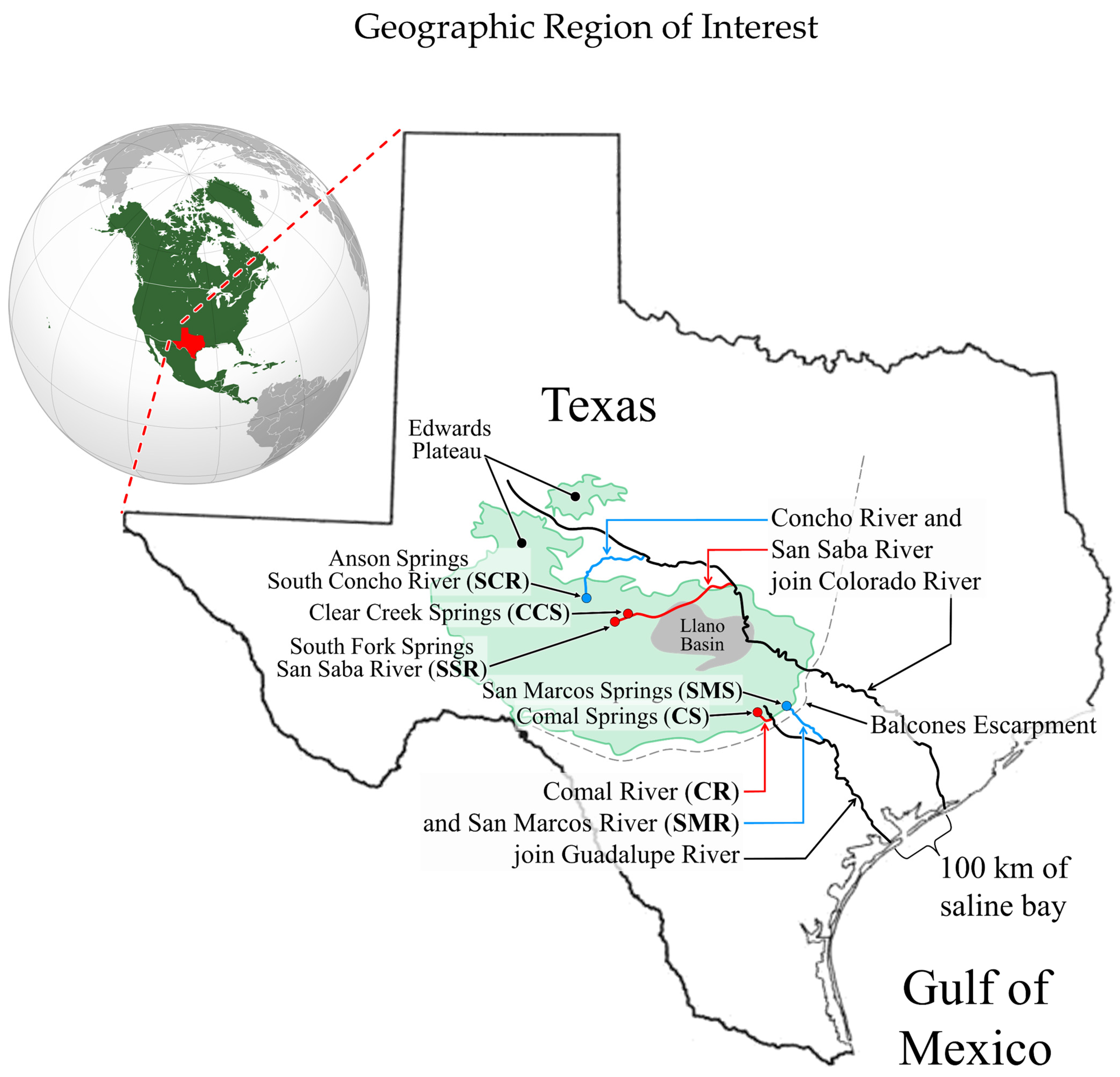
2.2. The Setting
2.3. Pre-Pleistocene Geological History of the Region
2.3.1. The Wisconsin and Holocene Climates
Climatological Effects on Local Aquatic Communities
2.3.2. The Chronology of Our Model
2.4. The Model System
2.4.1. The Clades of Focus
2.4.2. Overview of Corroborating Experiments
2.5. Our Research Hypothesis
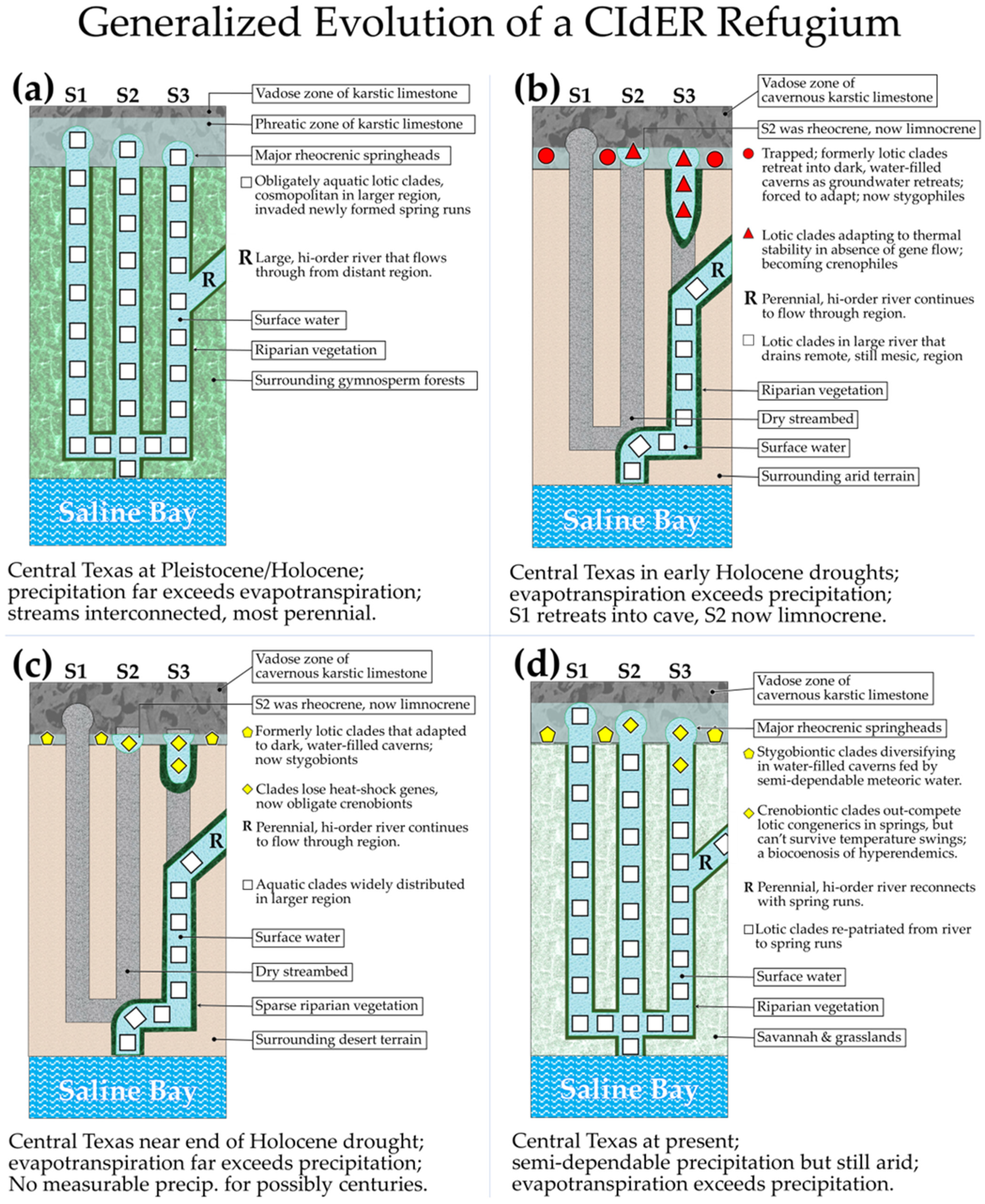
2.5.1. “Crenal Interglacial-drought Evolutionary Refugia” (CIdER)
2.5.2. Hypothetical Stages in the Development of a CIdER Refugium
- Members of the spring-endemic population are now crenobionts because they are unable to disperse into the connected ambient stream.
- The cosmopolitan congener (repatriated sister group) does not introgressively swamp the endemic population because of apparent reproductive incompatibilities.
- The widespread congener is apparently unable to displace the endemic population from the spring by out-competing it.
- Both populations remain in graduated sympatry [63] in the eucrenal and/or the spring-influenced upper reach of the spring run.
- Our team is unable to determine by mere inspection whether the observed endemism of the crenobionts had originated through sympatric or allopatric processes.
- Our team is also unable to determine by inspection what factors are limiting the down-stream dispersal of the crenobionts.
- Thus, if an endemic species occurs in a patch of habitat that is far from other such patches, that alone is insufficient evidence that the endemism developed in allopatry; likewise, when an endemic species occurs sympatrically with a closely related congener, that observation is not necessarily evidence that the endemism had developed in sympatry. Therefore, while speculation might be useful for developing testable hypotheses, the development of a definitive and supportable narrative that parsimoniously explains the cause of contemporary endemism should be based on multiple lines of reasoning that are corroborated by pertinent data and, where possible, experimental findings.
2.6. The Need for Rigor
3. Methods, Results and Discussion
3.1. Q1: Is There Evidence That SMS Has Continuously Maintained Groundwater at the Surface since the Last Glacial Maximum?
3.1.1. An isolated Huffmanela Clade Was Extirpated from CS by a Historic 4.5-Month Cessation of Spring Discharge
| Spring Name | References | Coords. (Deg W, Deg N) | Crenobiontic Huffmanela spp. | Crenobiontic Hyalella spp. |
|---|---|---|---|---|
| Barton Springs | [47] | 30.263759, −97.770876 | N | ? |
| Blue Springs | [48] | 29.893691, −100.994661 | N | N |
| Caroline Spring | [48] | 30.469016, −101.803561 | N | N |
| Clear Creek Springs | [48] | 30.907044, −99.960929 | Y | Y |
| Comal Springs (CS) | [45] (fish from [5]) | 29.714441, −98.135296 | Y 1 | N 2 |
| Comal Springs (CS) | [47,48,74,75,76] | 29.714441, −98.135296 | N 3 | N |
| Fessenden Springs | [47,48] | 30.166927, −99.342635 | N | N |
| Finnegan Springs | [48] | 29.901371, −100.999576 | N | N |
| Hueco Springs | [47] | 29.759169, −98.140878 | N | ? |
| Las Moras Springs | [48] | 29.309747, −100.420961 | N | N |
| San Felipe Springs | [48] | 29.373565, −100.885139 | N | N |
| San Marcos Springs (SMS) | [48,75,76,77] | 29.893931, −97.930088 | Y | Y |
| San Saba Springs | [48] | 30.825901, −100.119022 | Y | Y |
| South Concho Springs | [49] | 31.135639, −100.493499 | Y | N |
3.1.2. More Unexpected Contributions from Texas Wild Rice (Zizania texana)
3.2. Q2: Can the Occurrence of Endemic Hyalellids Similar to the SMS Hyalella in Other Springs Reveal Other CIdER Refugia Harboring Huffmanela?
3.3. Q3: Are the Downstream Distributional Limits of Crenobionts in the SMR Determined by Change in Some Aspect of Spring Physicochemistry?
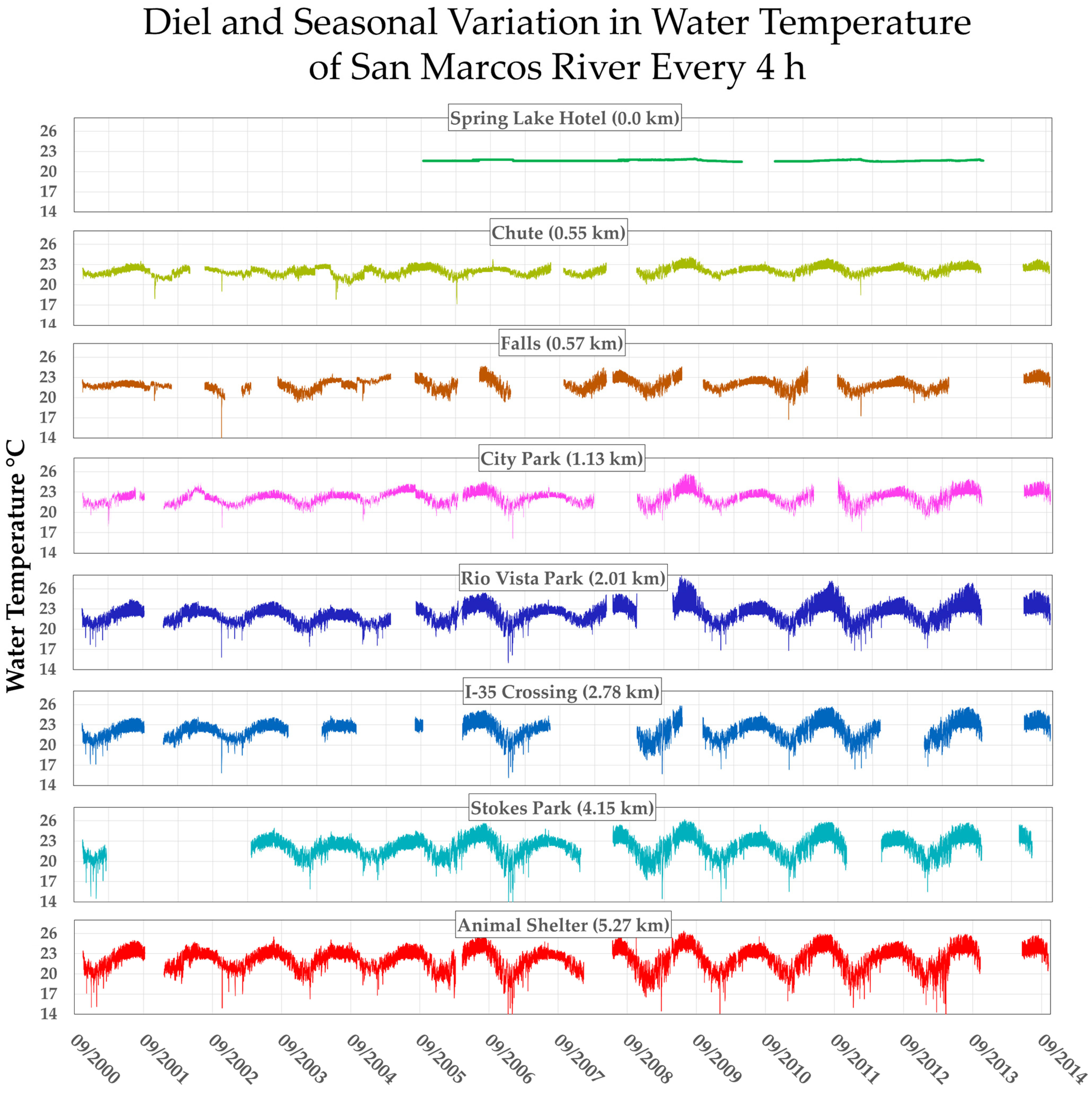
3.3.1. Overview of Contextual Data
3.3.2. Downstream Increase in Temperature Variance Is Inversely (But Tightly) Correlated with Huffmanela huffmani Abundance Measures in Wild-Caught SMR Fish
3.3.3. Downstream Depression of Huffmanela Abundance Probably Caused by Progressively Increasing Thermal Instability: Experimental Corroboration
3.4. Q4: Are There Corroborating Data Consistent with the Two Cenobiontic Host/Parasite Systems Having Been Separated for Thousands of Amphipod Generations?
3.4.1. River Flow Patterns
3.4.2. COI Genetics of Crenobiontic Hyalellids
3.4.3. Reproductive Isolation of the Crenobiontic Hyalellids
3.5. Q5: Are There Corroborating Data Consistent with the Two Crenobiontic Hyalellids Having Coevolved with Their Local Huffmanela Parasites Prior to the Repatriation of Hyalella cf. azteca to Both Springs?
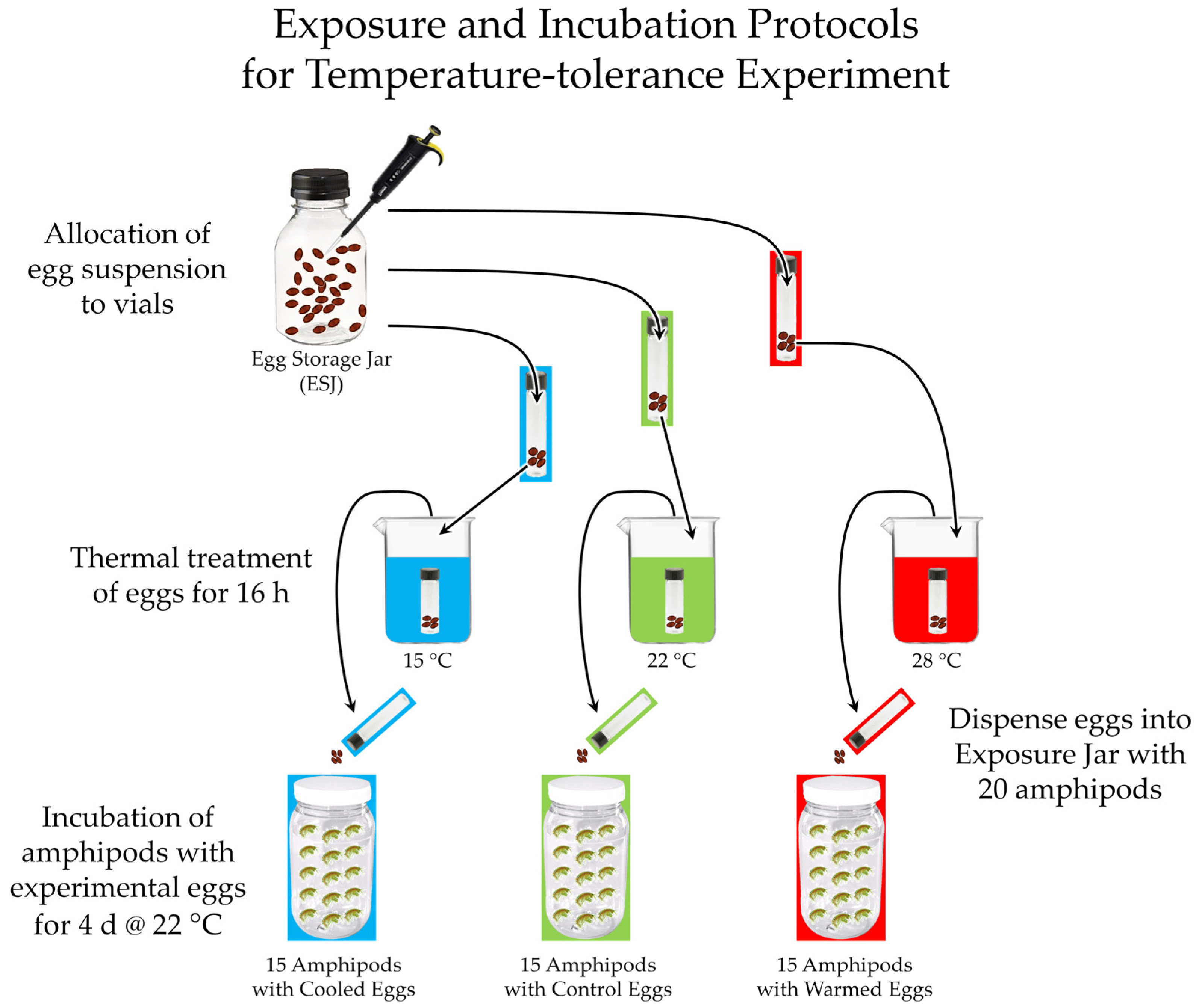
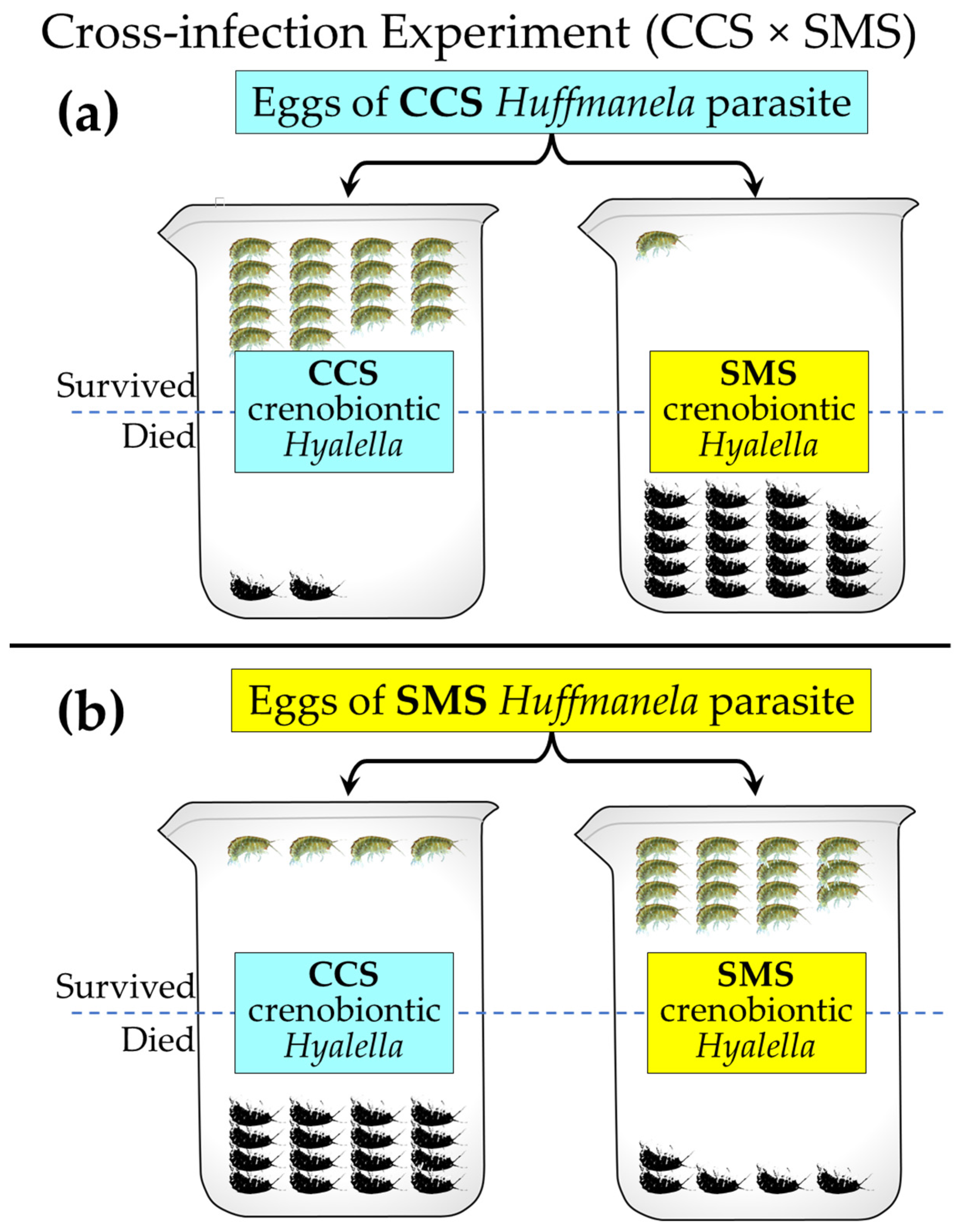
3.5.1. The Two Crenobiontic Host/Parasite Systems: Exposing Both Crenobiontic Hyalellids to Heavy Doses of Local vs. Exotic Huffmanela Eggs
3.5.2. Comparison of One Crenobiontic Amphipod/Huffmanela System with the Local Repatriated Amphipod/Huffmanela System: Differences in Chronic Reactions to the Parasite
4. Conclusions
4.1. Paleoecological Inference
4.2. A Proposed New Subcategory for Evolutionary Refugia
- The starting point is a diverse community of obligately aquatic plants and mostly invertebrate animals that are widely distributed among interconnected streams during a period of high regional precipitation associated with extensive glaciation just poleward of the region.
- The community becomes challenged by region-wide gradually reduced precipitation as the glacial margins retreat poleward.
- Evapotranspiration begins to exceed precipitation and many streams and springs that were formerly perennial become progressively more intermittent, and the distribution of the obligate aquatic community becomes progressively more fragmented.
- Many smaller springs stop flowing and lotic habitats become restricted to isolated flows fed by a few perennially rheocrenic springs, and perennial high-order rivers draining remote regions.
- The largest rheocrenic springs have spring runs extending from the springhead but disappear into ground and air, never connecting to other surface waters; others shrink back to limnocrenes in which the water remains thermally constant a meter or so deep where there is movement of groundwater.
- This condition is maintained for thousands of generations of the obligate aquatic invertebrates, which are now members of an isolated biocenosis continually bathed in a thermally constant environment with a stable bottom undisturbed by spates.
- The surviving clades eventually lose (to drift or reassignment) genetic loci for heat shock proteins that once allowed the organisms to adapt to varying temperatures, but also become extremely efficient competitors under these rigidly constrained physicochemical conditions.
- The drought breaks, and dependable precipitation in excess of evapotranspiration returns to the region.
- Spring-streams lengthen, eventually forming permanent connections with surface-fed streams, but crenobionts that survived through the drought in the springs can no longer survive through the temperature swings of progressively more ambient waters downstream.
- Sister clades that are relatives of the crenobionts, but which had retreated to other regions with dependable precipitation during the drought, now return and intermingle with the crenobionts.
- If the crenobionts are now reproductively isolated from the repatriated sister clades, a longitudinal density gradient is established, with the repatriated sister clades diminishing in density toward the spring head, and the crenobiontic clades diminishing downstream with physicochemical instability.
- The obligate aquatic community at the springhead now consists of a highly diverse mixture of surviving, but still endemic crenobionts and repatriated sister clades.
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
Appendix A. Review of Terminology Related to Refugia, Relicts, and Endemics
Appendix A.1. Terms Related to Endemics
Appendix A.2. Terms Related to Relicts
Appendix A.3. Terms Related to “Refugia”
Appendix A.3.1. Misapplications of “Refug-”
Appendix A.3.2. Problems with “Evolutionary Refugia”
Appendix A.3.3. “Refugia” with “Glacial” Modifiers
Appendix A.3.4. Recent Attempts to Rein in Refugial Misuses
Appendix A.3.5. The Need New Refugial Categories
Appendix A.4. Consequences of Terminological Ambiguity and Recommended Solutions
References
- Wallace, A.R. The Malay Archipelago; The Land of the Orang-Utan and the Bird of Paradise; A Narrative of Travel with Studies of Man and Nature; Macmillan & Co.: London, UK, 1869; Volumes 1 and 2. [Google Scholar]
- Huxley, T.H. On the classification and distribution of the Alectoromorphae and Heteromorphae. Proc. Zool. Soc. Lond. 1868, 1868, 294–319. [Google Scholar]
- Cracraft, J. Historical biogeography and earth history: Perspectives for a future synthesis. Ann. Mo. Bot. Gard. 1975, 62, 227–250. [Google Scholar] [CrossRef]
- Cantonati, M.; Füreder, L.; Gerecke, R.; Jüttner, I.; Eileen, J.C. Crenic habitats, hotspots for freshwater biodiversity conservation: Toward an understanding of their ecology. Freshw. Sci. 2012, 31, 463–480. [Google Scholar] [CrossRef]
- Hubbs, C.; Kuehne, R.A.; Ball, J.C. The fishes of the upper Guadalupe River, Texas. Tex. J. Sci. 1953, 5, 216–244. [Google Scholar]
- George, P.G.; Mace, R.E.; Petrossian, R. Aquifers of Texas; Report 380; Texas Water Development Board: Austin, TX, USA, 2011; p. viii+172.
- Jones, I.C. The northern segment of the Edwards (Balcones Fault Zone) Aquifer. In The Edwards Aquifer: The Past, Present, and Future of a Vital Water Resource; Sharp, J.M., Jr., Green, R.T., Schindel, G.M., Eds.; The Geological Society of America: Boulder, CO, USA, 2019; Volume mwr215, pp. 119–130. [Google Scholar]
- Craig, C.A.; Kollaus, K.A.; Behen, K.P.K.; Bonner, T.H. Relationships among spring flow, habitats, and fishes within evolutionary refugia of the Edwards Plateau. Ecosphere 2016, 7, 1–13. [Google Scholar] [CrossRef]
- Gibson, J.R.; Harden, S.J.; Fries, J.N. Survey and distribution of invertebrates from selected springs of the Edwards Aquifer in Comal and Hays Counties, Texas. Southwest. Nat. 2008, 53, 74–84. [Google Scholar] [CrossRef]
- Culver, D.C.; Pipan, T.; Schneider, K. Vicariance, dispersal and scale in the aquatic subterranean fauna of karst regions. Freshw. Biol. 2009, 54, 918–929. [Google Scholar] [CrossRef]
- Kauffman, E.G. Paleobiogeography and evolutionary response dynamic in the Cretaceous Western Interior Seaway of North America. In Jurassic-Cretaceous Biochronology and Paleogeography of North America; Westermann, G.E., Ed.; Geological Association of Canada: St. John’s, NL, Canada, 1984; Special Paper 27; Volume 27, pp. 273–306. [Google Scholar]
- Schindel, G.M. Genesis of the Edwards (Balcones Fault Zone) Aquifer. In The Edwards Aquifer: The Past, Present, and Future of a Vital Water Resource; The Geological Society of America: Boulder, CO, USA, 2019; pp. 9–18. [Google Scholar]
- Laubach, S.E.; Jackson, M.L.W. Origin of arches in the northwestern Gulf of Mexico basin. Geology 1990, 18, 595–598. [Google Scholar] [CrossRef]
- Barker, R.A.; Bush, P.W.; Baker, E.T. Geologic History and Hydrogeologic Setting of the Edwards-Trinity Aquifer System, West-Central Texas; US Department of the Interior, US Geological Survey: Reston, VA, USA, 1994.
- Woodruff, C.M., Jr.; Abbott, P.L. Drainage-basin evolution and aquifer development in a karstic limestone terrain South-Central Texas, USA. Earth Surf. Process. Landf. 1979, 4, 319–334. [Google Scholar] [CrossRef]
- White, K.; Davidson, G.R.; Paquin, P. Hydrologic evolution of the Edwards Aquifer recharge zone (Balcones fault zone) as recorded in the DNA of eyeless Cicurina cave spiders, south-central Texas. Geology 2009, 37, 339–342. [Google Scholar] [CrossRef]
- Collins, E.W. Geologic Map of the New Braunfels, Texas, 30 × 60 Minute Quadrangle, Texas: Geologic Framework of an Urban-Growth Corridor along the Edwards Aquifer, South-Central Texas; Miscellaneous Map #39; The University of Texas at Austin: Austin, TX, USA, 2000. [Google Scholar]
- Hart, M.B.; Yancey, T.E.; Leighton, A.D.; Miller, B.; Liu, C.; Smart, C.W.; Twitchett, R.J. The Cretaceous-Paleogene boundary on the Brazos River, Texas: New stratigraphic sections and revised interpretations. GCAGS J. 2012, 1, 69–80. [Google Scholar]
- Yancey, T.E. Depositional trends in siliciclastic deposits of the Stone City transgressive systems tract, middle Eocene, Texas. Trans.-Gulf Coast Assoc. Geol. Soc. 1995, 45, 581–586. [Google Scholar]
- Haq, B.U.; Hardenbol, J.; Vail, P.R. Chronology of fluctuating sea levels since the Triassic. Science 1987, 235, 1156–1167. [Google Scholar] [CrossRef]
- Zachos, J.; Pagani, M.; Sloan, L.; Thomas, E.; Billups, K. Trends, rhythms, and aberrations in global climate 65 Ma to present. Science 2001, 292, 686–693. [Google Scholar] [CrossRef]
- Miller, K.G.; Fairbanks, R.G.; Mountain, G.S. Tertiary oxygen isotope synthesis, sea level history, and continental margin erosion. Paleoceanography 1987, 2, 1–19. [Google Scholar] [CrossRef]
- Kominz, M.A.; Browning, J.V.; Miller, K.G.; Sugarman, P.J.; Mizintseva, S.; Scotese, C.R. Late Cretaceous to Miocene sea-level estimates from the New Jersey and Delaware coastal plain coreholes: An error analysis. Basin Res. 2008, 20, 211–226. [Google Scholar] [CrossRef]
- Kominz, M.A.; Miller, K.G.; Browning, J.V. Long-term and short-term global Cenozoic sea-level estimates. Geology 1998, 26, 311–314. [Google Scholar] [CrossRef]
- Cooke, M.J.; Stern, L.A.; Banner, J.L.; Mack, L.E.; Stafford Jr, T.W.; Toomey III, R.S. Precise timing and rate of massive late Quaternary soil denudation. Geology 2003, 31, 853–856. [Google Scholar] [CrossRef]
- Toomey III, R.S.; Blum, M.D.; Valastro Jr, S. Late Quaternary climates and environments of the Edwards Plateau, Texas. Glob. Planet. Change 1993, 7, 299–320. [Google Scholar] [CrossRef]
- Blum, M.D.; Valastro, S. Response of the Pedernales River of central Texas to late Holocene climatic change. Ann. Assoc. Am. Geogr. 1989, 79, 435–456. [Google Scholar] [CrossRef]
- Nordt, L.C. Archaeological Geology of the Fort Hood Military Reservation, Fort Hood, Texas; 25; Texas A&M Univ College Station Archeological Research Lab: College Station, TX, USA, 1992. [Google Scholar]
- Nickels, D.L.; Bousman, C.B. Texas River Center Archaeology, Test Excavations at 41HY160, Hays County, Texas; Texas State University: San Marcos, TX, USA, 2010. [Google Scholar]
- Davis, M.B.; Shaw, R.G. Range shifts and adaptive responses to Quaternary climate change. Science 2001, 292, 673–679. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.I.; Banner, J.L.; Musgrove, M. Holocene climate variability in Texas, USA: An integration of existing paleoclimate data and modeling with a new, high-resolution speleothem record. Quat. Sci. Rev. 2015, 127, 155–173. [Google Scholar] [CrossRef]
- Booth, R.K.; Jackson, S.T.; Forman, S.L.; Kutzbach, J.E.; Bettis, E.A.; Kreigs, J.; Wright, D.K. A severe centennial-scale drought in midcontinental North America 4200 years ago and apparent global linkages. Holocene 2005, 15, 321–328. [Google Scholar] [CrossRef]
- Asmerom, Y.; Polyak, V.; Burns, S.; Rassmussen, J. Solar forcing of Holocene climate: New insights from a speleothem record, southwestern United States. Geology 2007, 35, 1–4. [Google Scholar] [CrossRef]
- Anderson, R.Y.; Allen, B.D.; Menking, K.M. Geomorphic expression of abrupt climate change in southwestern North America at the glacial termination. Quat. Res. 2002, 57, 371–381. [Google Scholar] [CrossRef]
- Mensing, S.A.; Benson, L.V.; Kashgarian, M.; Lund, S. A Holocene pollen record of persistent droughts from Pyramid Lake, Nevada, USA. Quat. Res. 2004, 62, 29–38. [Google Scholar] [CrossRef]
- Russ, J.; Loyd, D.H.; Boutton, T.W. A paleoclimate reconstruction for southwestern Texas using oxalate residue from lichen as a paleoclimate proxy. Quat. Int. 2000, 67, 29–36. [Google Scholar] [CrossRef]
- Hall, S.A.; Penner, W.L. Stable carbon isotopes, C3–C4 vegetation, and 12,800 years of climate change in central New Mexico, USA. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2013, 369, 272–281. [Google Scholar] [CrossRef]
- Ellwood, B.B.; Gose, W.A. Heinrich H1 and 8200 yr B.P. climate events recorded in Hall’s Cave, Texas. Geology 2006, 34, 753–756. [Google Scholar] [CrossRef]
- Smith, G.I.; Street-Perrott, F.A. Pluvial lakes of the western United States. Late-Quat. Environ. U. S. 1983, 1, 190–212. [Google Scholar]
- Sylvia, D.A.; Galloway, W.E. Morphology and stratigraphy of the late Quaternary lower Brazos valley: Implications for paleo-climate, discharge and sediment delivery. Sediment. Geol. 2006, 190, 159–175. [Google Scholar] [CrossRef]
- Musgrove, M.; Banner, J.L.; Mack, L.E.; Combs, D.M.; James, E.W.; Cheng, H.; Edwards, R.L. Geochronology of late Pleistocene to Holocene speleothems from central Texas: Implications for regional paleoclimate. GSA Bull. 2001, 113, 1532–1543. [Google Scholar] [CrossRef]
- Holsinger, J.R.; Longley, G. The Subterranean Amphipod Crustacean Fauna of an Artesian Well in Texas; Smithsonian Institution Press: Washington, DC, USA, 1980; Volume 308, p. 62. [Google Scholar]
- Hutchins, B.T. The conservation status of Texas groundwater invertebrates. Biodivers. Conserv. 2018, 27, 475–501. [Google Scholar] [CrossRef]
- Worsham, M.L.D.; Julius, E.P.; Nice, C.C.; Diaz, P.H.; Huffman, D.G. Geographic isolation facilitates the evolution of reproductive isolation and morphological divergence. Ecol. Evol. 2017, 7, 10278–10288. [Google Scholar] [CrossRef] [PubMed]
- Worsham, M.L.D.; Huffman, D.G.; Moravec, F.; Gibson, J.R. The life cycle of Huffmanela huffmani Moravec, 1987 (Nematoda: Trichosomoididae), an endemic marine-relict parasite of Centrarchidae from a Central Texas spring. Folia Parasitol. 2016, 63, 1. [Google Scholar] [CrossRef] [PubMed]
- Moravec, F. Revision of Capillariid Nematodes (Subfamily Capillariinae) Parasitic in Fishes. Studie ČSAV No. 3; Academia: Praha, Czech Republic, 1987; p. 144. [Google Scholar]
- Cox, M.K. The Distribution and Life Cycle of Huffmanela huffmani (Nematoda: Trichosomoididae). Master’s Thesis, Southwest Texas State University, San Marcos, TX, USA, 1998. [Google Scholar]
- Worsham, M.L.D. Huffmanela huffmani: Life Cycle, Natural History, and Biogeography. Master’s Thesis, Texas State University-San Marcos, San Marcos, TX, USA, 2015. [Google Scholar]
- Bond, A.T. Investigations into Huffmanela (Nematoda: Trichosomoididae): New Populations, Life Cycles, and Eggshell Ultrastructure. Master’s Thesis, Texas State University, San Marcos, TX, USA, 2020. [Google Scholar]
- Stevenson, M.M.; Peden, A.E. Description and ecology of Hyalella texana n. sp.(Crustacea: Amphipoda) from the Edwards Plateau of Texas. Am. Midl. Nat. 1973, 89, 426–436. [Google Scholar] [CrossRef]
- Bullard, S.A.; Moravec, F.; Ksepka, S.P.; Warren, M.B.; Dutton, H.R.; Huffman, D.G.; Yanong, R.P.E. Huffmanela cf. huffmani (Nematoda: Trichosomoididae) infecting swim bladder, peritoneum, and gonad of variable platyfish, Xiphophorus variatus (Cyprinodontiformes: Poeciliidae) and eastern mosquitofish, Gambusia holbrooki (Poeciliidae) in Florida; taxonomy, phylogenetic analysis, and pathological changes. Parasitol. Res. 2022; in press. [Google Scholar]
- Longley, G. The subterranean aquatic ecosystem of the Balcones Fault Zone Edwards Aquifer in Texas-threats from overpumping. In Proceedings of the First International Conference on Ground Water Ecology, Tampa, FL, USA, 26–27 April 1992; pp. 26–29. [Google Scholar]
- Ashcroft, M.B. Identifying refugia from climate change. J. Biogeogr. 2010, 37, 1407–1413. [Google Scholar] [CrossRef]
- Bennett, K.; Provan, J. What do we mean by ‘refugia’? Quat. Sci. Rev. 2008, 27, 2449–2455. [Google Scholar] [CrossRef]
- Cantonati, M.; Fensham, R.J.; Stevens, L.E.; Gerecke, R.; Glazier, D.S.; Goldscheider, N.; Knight, R.L.; Richardson, J.S.; Springer, A.E.; Tockner, K. Urgent plea for global protection of springs. Conserv. Biol. 2020, 35, 378–382. [Google Scholar] [CrossRef]
- Myers, A.A.; De Grave, S. Endemism: Origins and implications. Vie Et Milieu 2000, 50, 195–204. [Google Scholar]
- Harold, A.S.; Mooi, R.D. Areas of endemism: Definition and recognition criteria. Syst. Biol. 1994, 43, 261–266. [Google Scholar] [CrossRef]
- Heenan, P.B.; Millar, T.R.; Smissen, R.D.; McGlone, M.S.; Wilton, A.D. Phylogenetic measures of neo-and palaeo-endemism in the indigenous vascular flora of the New Zealand archipelago. Aust. Syst. Bot. 2017, 30, 124–133. [Google Scholar] [CrossRef]
- Keppel, G.; Van Niel, K.P.; Wardell-Johnson, G.W.; Yates, C.J.; Byrne, M.; Mucina, L.; Schut, A.G.T.; Hopper, S.D.; Franklin, S.E. Refugia: Identifying and understanding safe havens for biodiversity under climate change. Glob. Ecol. Biogeogr. 2012, 21, 393–404. [Google Scholar] [CrossRef]
- Davis, J.; Pavlova, A.; Thompson, R.; Sunnucks, P. Evolutionary refugia and ecological refuges: Key concepts for conserving Australian arid zone freshwater biodiversity under climate change. Glob. Change Biol. 2013, 19, 1970–1984. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, J.; Hochachka, P.W. Functional significance of isoenzymes in thermal acclimatization. Acetylcholinesterase from trout brain . Biochem. J. 1970, 116, 883–887. [Google Scholar] [PubMed]
- Somero, G.N. Thermal physiology and vertical zonation of intertidal animals: Optima, limits, and costs of living. Integr. Comp. Biol. 2002, 42, 780–789. [Google Scholar] [CrossRef] [PubMed]
- Tellier, F.; Tapia, J.; Faugeron, S.; Destombe, C.; Valero, M. The Lessonia nigrescens species complex (Laminariales, Phaeophyceae) shows strict parapatry and complete reproductive isolation in a secondary contact zone. J. Phycol. 2011, 47, 894–903. [Google Scholar] [CrossRef]
- Adams, N.E.; Inoue, K.; Seidel, R.A.; Lang, B.K.; Berg, D.J. Isolation drives increased diversification rates in freshwater amphipods. Mol. Phylogenetics Evol. 2018, 127, 746–757. [Google Scholar] [CrossRef]
- Krejca, J.K. Stygobite Phylogenetics as a tool for Determining Aquifer Evolution. Doctoral Dissertation, University of Texas, Austin, TX, USA, 2005. [Google Scholar]
- Abbott, P.L. On the hydrology of the Edwards Limestone, south-central Texas. J. Hydrol. 1975, 24, 251–269. [Google Scholar] [CrossRef]
- Mejía-Madrid, H.H.; Vázquez-Domínguez, E.; Pérez-Ponce de León, G. Phylogeography and freshwater basins in central Mexico: Recent history as revealed by the fish parasite Rhabdochona lichtenfelsi (Nematoda). J. Biogeogr. 2007, 34, 787–801. [Google Scholar] [CrossRef]
- Martínez-Aquino, A.; Ceccarelli, F.S.; Eguiarte, L.E.; Vázquez-Domínguez, E.; de León, G.P.-P. Do the historical biogeography and evolutionary history of the digenean Margotrema spp. across central Mexico mirror those of their freshwater fish hosts (Goodeinae)? PLoS ONE 2014, 9, e101700. [Google Scholar]
- Lumme, J.; Mäkinen, H.; Ermolenko, A.V.; Gregg, J.L.; Ziętara, M.S. Displaced phylogeographic signals from Gyrodactylus arcuatus, a parasite of the three-spined stickleback Gasterosteus aculeatus, suggest freshwater glacial refugia in Europe. Int. J. Parasitol. 2016, 46, 545–554. [Google Scholar] [CrossRef] [PubMed]
- Brune, G.M. Springs of Texas, Volume 1; Branch-Smith: Fort Worth, TX, USA, 1981. [Google Scholar]
- Musgrove, M.; Crow, C.L. Origin and Characteristics of Discharge at San Marcos Springs Based on Hydrologic and Geochemical Data (2008-10), Bexar, Comal, and Hays Counties, Texas; 141133454X; US Department of the Interior, US Geological Survey: Reston, VA, USA, 2012; p. viii+94.
- Schenck, J.R.; Whiteside, B.G. Distribution, habitat preference and population size estimate of Etheostoma fonticola. Copeia 1976, 1976, 697–703. [Google Scholar] [CrossRef]
- Jordan, D.S.; Gilbert, C.H. List of fishes collected in Arkansas, Indian Territory, and Texas, in September, 1884, with notes and descriptions. Proc. U. S. Natl. Mus. 1886, 1886, 1–25. [Google Scholar] [CrossRef]
- USFWS. National Wild Fish Health Survey Results for the Comal River, TX (CHN 15-16); FWS/R2/FR-SFHU/780; San Marcos Aquatic Resources Center (USFWS): San Marcos, TX, USA, 2015; p. 1.
- Michel, G.D. The Biology of Capillaria sp. (Nematoda: Capillariidae) from Swim Bladders of Sunfishes of the Upper San Marcos River. Master’s Thesis, Southwest Texas State University, San Marcos, TX, USA, 1984. [Google Scholar]
- O’Docharty, E.M. Studies on the life cycle of Huffmanela huffmani (Nematoda: Trichosomoididae). Master’s Thesis, Texas State University-San Marcos, San Marcos, TX, USA, 2007. [Google Scholar]
- Cox, M.K.; Huffman, D.G.; Moravec, F. Observations on the distribution and biology of Huffmanela huffmani (Nematoda: Trichosomoididae). Folia Parasitol. 2004, 51, 50–54. [Google Scholar] [CrossRef]
- EAA. The EAA Act: A Success Story. Available online: https://www.edwardsaquifer.org/business-center/legislation-rules/the-eaa-act-a-success-story/ (accessed on 25 February 2020).
- Hitchcock, A.S. New species and new names of grasses from Texas. J. Wash. Acad. Sci. 1933, 23, 449–456. [Google Scholar]
- Horne, F.; Kahn, A. Phylogeny of North American wild rice, a theory. Southwest. Nat. 1997, 42, 423–434. [Google Scholar]
- Hardy, T. (Chief Science Officer, Meadows Center for Water & the Environment, San Marcos, TX, USA) (Meadows Professor of Environmental Flows, Texas State University, San Marcos, TX, USA). Personal communication regarding ecological requirements of Texas wild rice, 2021.
- Heard, T.C. (Aquatic Biologist, Meadows Center for Water & the Environment, San Marcos, TX, USA). Personal communication regarding ecological requirements of Texas wild rice, 2021.
- Bush, A.O.; Lafferty, K.D.; Lotz, J.M.; Shostak, A.W. Parasitology meets ecology on its own terms: Margolis et al. revisited. J. Parasitol. 1997, 83, 575–583. [Google Scholar] [CrossRef]
- Groeger, A.W.; Brown, P.F.; Tietjen, T.E.; Kelsey, T.C. Water quality of the San Marcos River. Tex. J. Sci. 1997, 49, 16. [Google Scholar]
- Saunders, K.S.; Mayes, K.B.; Jurgensen, T.A.; Trungale, J.T.; Kleinsasser, L.J.; Aziz, K.; Fields, J.R.; Moss, R.E. An Evaluation of Spring Flows to Support the Upper San Marcos River Spring Ecosystem, Hays County, Texas; Resource Protection Division, Texas Parks and Wildlife Department: Austin, TX, USA, 2001.
- Hannan, H.H.; Dorris, T.C. Succession of a macrophyte community in a constant temperature river. Limnol. Oceanogr. 1970, 15, 442–453. [Google Scholar] [CrossRef]
- Nickels, D.L.; Bousman, C.B. Archaeological testing at San Marcos Springs (41HY160) for the Texas Rivers Center, Hays County, Texas. Index Tex. Archaeol. Open Access Gray Lit. Lone Star State 2010, 2010, 8. [Google Scholar] [CrossRef]
- Social_Science_Statistics. Spearman’s Rho Correlation Coefficient Calculator. Available online: https://www.socscistatistics.com/tests/pearson/default2.aspx (accessed on 4 March 2020).
- Logan, C.A.; Buckley, B.A. Transcriptomic responses to environmental temperature in eurythermal and stenothermal fishes. J. Exp. Biol. 2015, 218, 1915–1924. [Google Scholar] [CrossRef] [PubMed]
- Peck, S.B. Climatic change and the evolution of cave invertebrates in the Grand Canyon, Arizona; J. Caves Karst Stud. 1980, 42, 53–60. [Google Scholar]
- Chown, S.L.; Jumbam, K.R.; Sørensen, J.G.; Terblanche, J.S. Phenotypic variance, plasticity and heritability estimates of critical thermal limits depend on methodological context. Funct. Ecol. 2009, 23, 133–140. [Google Scholar] [CrossRef]
- Mishler, B.D.; Knerr, N.; González-Orozco, C.E.; Thornhill, A.H.; Laffan, S.W.; Miller, J.T. Phylogenetic measures of biodiversity and neo-and paleo-endemism in Australian Acacia. Nat. Commun. 2014, 5, 4473. [Google Scholar] [CrossRef] [PubMed]
- Fattorini, S. Endemism in historical biogeography and conservation biology: Concepts and implications. Biogeogr. J. Integr. Biogeogr. 2017, 32, 47–75. [Google Scholar] [CrossRef]
- Darwin, C.R. The Origin of Species by Means of Natural Selection, or the Preservation of Favoured Races in the Struggle for Life. London: John Murray. 6th Edition, with Additions and Corrections, 6th ed.; William Clowes and Sons: London, UK, 1872; p. xxii+460. [Google Scholar]
- Platnick, N.I.; Nelson, G. Composite areas in vicariance biogeography. Syst. Zool. 1984, 33, 328–335. [Google Scholar] [CrossRef]
- Grandcolas, P.; Nattier, R.; Trewick, S. Relict species: A relict concept? Trends Ecol. Evol. 2014, 29, 655–663. [Google Scholar] [CrossRef]
- Keppel, G.; Wardell-Johnson, G.W. Refugia: Keys to climate change management. Glob. Change Biol. 2012, 18, 2389–2391. [Google Scholar] [CrossRef]
- Robson, B.J.; Chester, E.T.; Mitchell, B.D.; Matthews, T.G. Disturbance and the role of refuges in mediterranean climate streams. Hydrobiologia 2013, 719, 77–91. [Google Scholar] [CrossRef]
- Burk, R.A.; Kennedy, J.H. Invertebrate communities of groundwater-dependent refugia with varying hydrology and riparian cover during a supraseasonal drought. J. Freshw. Ecol. 2013, 28, 251–270. [Google Scholar] [CrossRef]
- EAHCP. Habitat Conservation Plan 2018—Annual Report; Edwards Aquifer Authority: San Antonio, TX, USA, 2019. [Google Scholar]
- Van Wyk, J.A. Refugia-overlooked as perhaps the most potent factor concerning the development of anthelmintic resistance. Onderstepoort J. Vet. Res. 2001, 68, 55–67. [Google Scholar] [PubMed]
- Deuel, N.R.; Conner, L.M.; Miller, K.V.; Chamberlain, M.J.; Cherry, M.J.; Tannenbaum, L.V. Habitat selection and diurnal refugia of gray foxes in southwestern Georgia, USA. PLoS ONE 2017, 12, e0186402. [Google Scholar] [CrossRef] [PubMed]
- Stewart, J.R.; Lister, A.M.; Barnes, I.; Dalén, L. Refugia revisited: Individualistic responses of species in space and time. Proc. R. Soc. B Biol. Sci. 2010, 277, 661–671. [Google Scholar] [CrossRef]



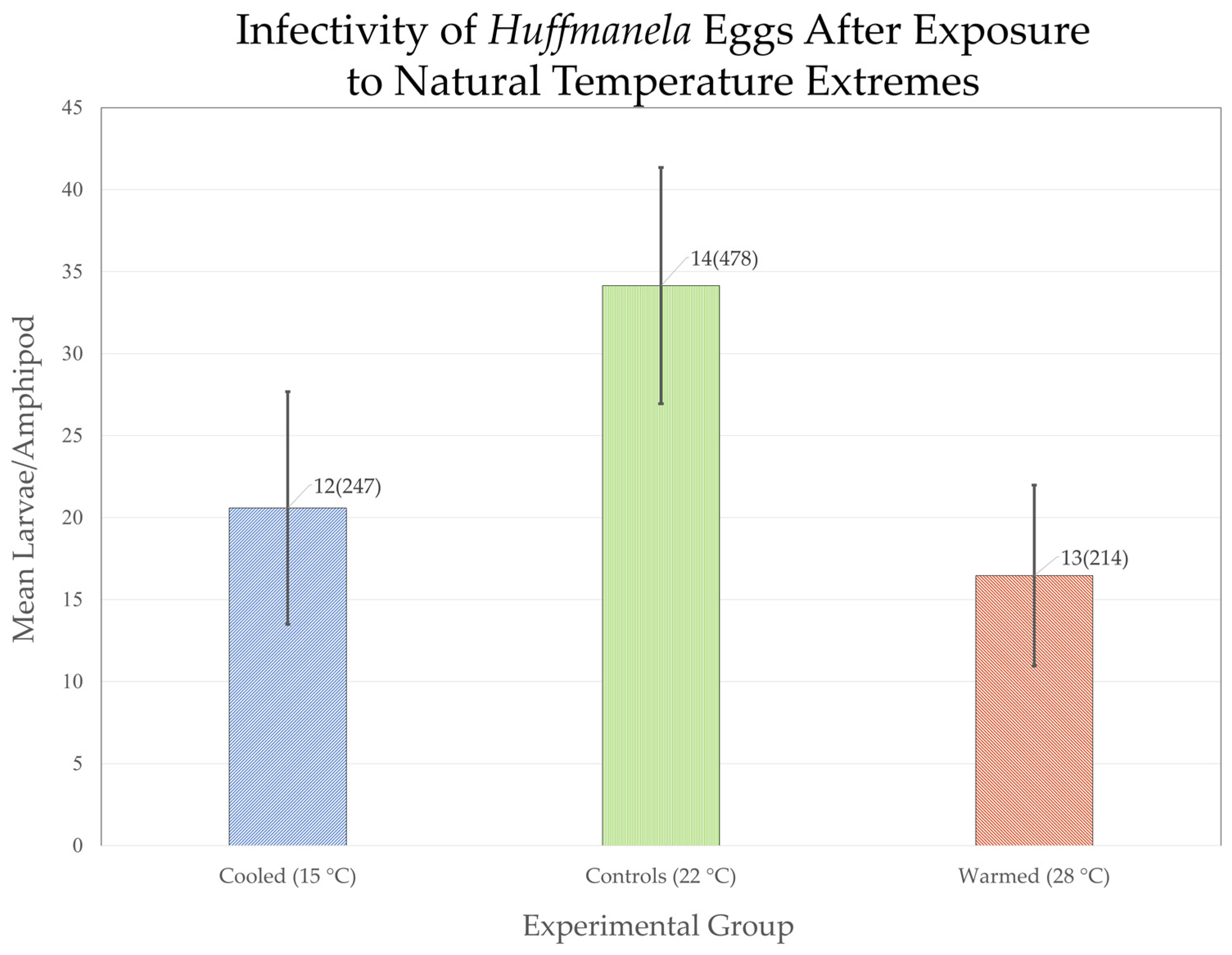
| Experimental Group | Days 1–7: Incubation | Days 8–14: Treatment | Days 15–21: Incubation | Day 22: Challenge | Day 27: Evaluation |
|---|---|---|---|---|---|
| Desiccated | Evaporating w/o replenishment | Dry all week | Rehydrated andreplenished | Six lab-reared amphipods added | All 6 amphipods negative for larvae |
| Control | Evaporating with replenishment | Continued replenishment | Continued replenishment | Six lab-reared amphipods added | Many live larvae in all 6 amphipods |
| Egg-Density Rating | Rating Criteria for Each Field |
|---|---|
| 5 | Transmitted light entirely blocked by multiple layers of eggs |
| 4 | More than 50% of field blocked by eggs |
| 3 | Between 25% and 50% of field blocked by eggs |
| 2 | Up to 24% of field blocked by eggs |
| 1 | Only a few spots of eggs observed in field |
| 0 | No eggs detected in field |
| Huffmanela Egg Clade | |||
|---|---|---|---|
| CCS | SMS | ||
| Crenobiontic Hyalellaclade | CCS | 20 | 20 |
| SMS | 20 | 20 | |
| # Died (of 20) | Null Expectation | Test Statistics | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Huffmanela Egg Source | Huffmanela Egg Source | ||||||||
| CCS | SMS | CCS | SMS | Φ = | −0.75 | ||||
| Hyalella Source | CCS | 18 | 4 | 22 | CCS | 11 | 11 | χ2 (Yates) = | 18.24 |
| SMS | 1 | 15 | 16 | SMS | 8 | 8 | df = | 1 | |
| 19 | 19 | 38 | p < | 0.0001 | |||||
| SMS H. huffmani Larvae in Presumed-Naïve Hyalella cf. azteca | SMS H. huffmani Larvae in Presumed Co-Evolved SMS Hyalella sp. | |
|---|---|---|
| Larval viability | >6 w | ≲2 w |
| Evidence of rejection | none | Immobility → calcification |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Worsham, M.L.D.; Bond, A.; Gibson, J.R.; Huffman, D.G. Biogeography of Selected Spring Endemics in Texas Interglacial-Drought Refugia with Unexpected Insights from a Spring-Dependent Nematode Parasite. Hydrobiology 2023, 2, 97-133. https://doi.org/10.3390/hydrobiology2010007
Worsham MLD, Bond A, Gibson JR, Huffman DG. Biogeography of Selected Spring Endemics in Texas Interglacial-Drought Refugia with Unexpected Insights from a Spring-Dependent Nematode Parasite. Hydrobiology. 2023; 2(1):97-133. https://doi.org/10.3390/hydrobiology2010007
Chicago/Turabian StyleWorsham, Mclean L. D., Alan Bond, James Randy Gibson, and David G. Huffman. 2023. "Biogeography of Selected Spring Endemics in Texas Interglacial-Drought Refugia with Unexpected Insights from a Spring-Dependent Nematode Parasite" Hydrobiology 2, no. 1: 97-133. https://doi.org/10.3390/hydrobiology2010007
APA StyleWorsham, M. L. D., Bond, A., Gibson, J. R., & Huffman, D. G. (2023). Biogeography of Selected Spring Endemics in Texas Interglacial-Drought Refugia with Unexpected Insights from a Spring-Dependent Nematode Parasite. Hydrobiology, 2(1), 97-133. https://doi.org/10.3390/hydrobiology2010007






