Halogens in Seaweeds: Biological and Environmental Significance
Abstract
1. General Introduction to Algae
2. Halogens
2.1. Definition, Discovery and History
2.1.1. Fluorine
2.1.2. Chlorine
2.1.3. Bromine
2.1.4. Iodine
2.1.5. Astatine
2.1.6. Tennessine
2.2. The Global Cycles of Halogens
2.3. Inorganic Biochemistry of the Halogens
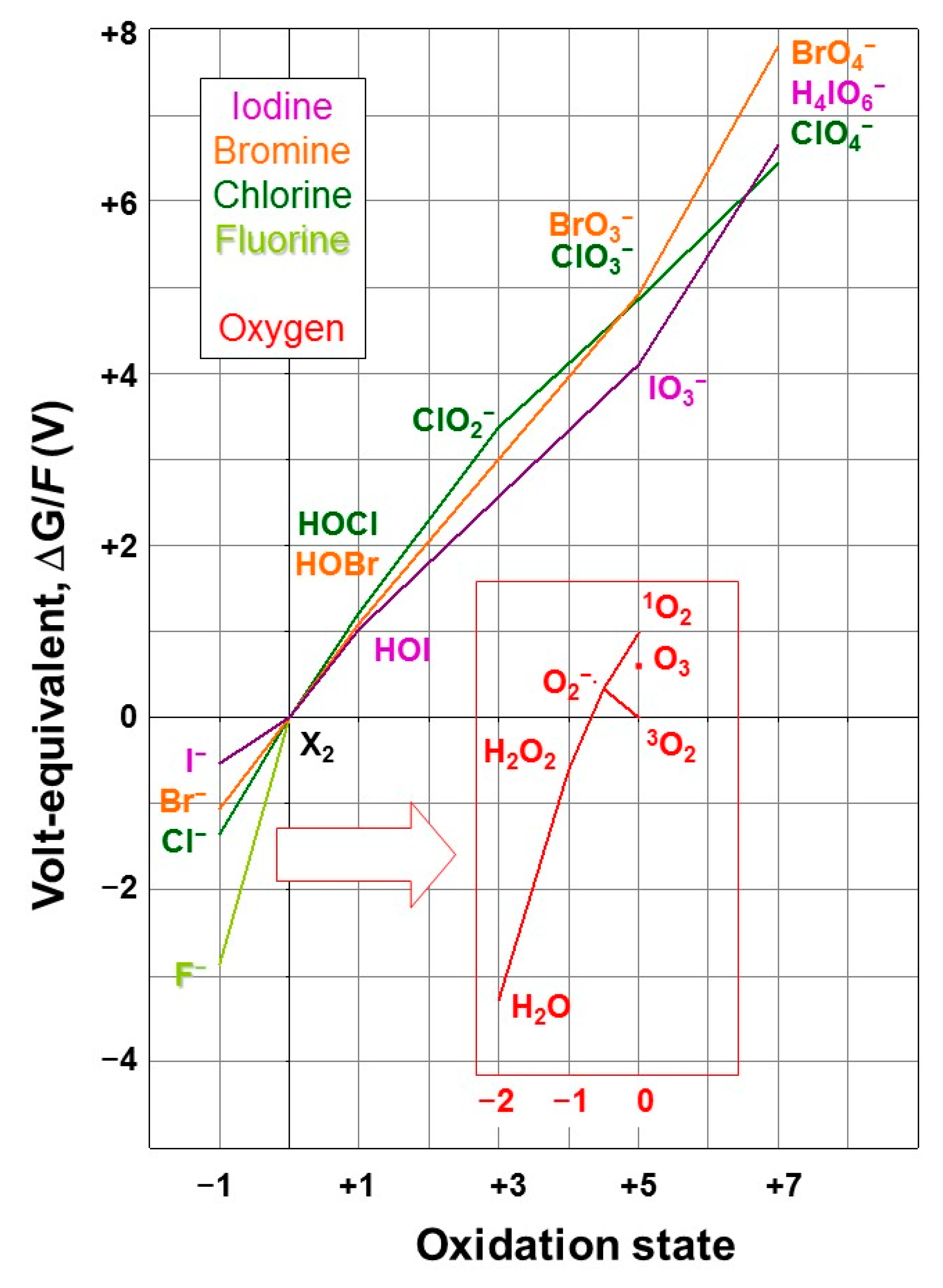
2.3.1. Halogen Oxidation States
2.3.2. Enzymatic Incorporation of Halide into Halocarbons
2.3.3. Halogen Oxyanions as Electron Acceptors
2.3.4. Dissimilatory and Assimilatory Organohalide Degradation
2.3.5. Halide Binding to Proteins and Nucleophilic Halogenation
2.4. Significance of Halogens
2.4.1. Environment
2.4.2. Human Physiology and Medicine
2.4.3. Dietary Iodine and Other Halogens in Seaweeds
3. Algae and Iodine Speciation in the Ocean
4. Algal Halogen Accumulation, Metabolism, Biochemistry
5. Algal Halogenated Natural Products
6. Vanadium Haloperoxidases
7. Algal Halogen Sources to the Atmosphere
8. Seaweeds as Bioindicators for Radioactive Iodine
9. General Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Carr, M.E.; Friedrichs, M.A.M.; Schmeltz, M.; Aita, M.N.; Antoine, D.; Arrigo, K.R.; Asanuma, I.; Aumont, O.; Barber, R.; Behrenfeld, M.; et al. A comparison of global estimates of marine primary production from ocean color. Deep. Sea Res. Part II Top. Stud. Oceanogr. 2006, 53, 741–770. [Google Scholar] [CrossRef]
- Huang, Y.; Nicholson, D.; Huang, B.; Cassar, N. Global Estimates of Marine Gross Primary Production Based on Machine Learning Upscaling of Field Observations. Glob. Biogeochem. Cycles 2021, 35, e2020GB006718. [Google Scholar] [CrossRef]
- Keller, M.D.; Bellows, W.K.; Guillard, R.R.L. Dimethyl sulfide production in marine phytoplankton. ACS Symp. Ser. 1989, 393, 167–182. [Google Scholar]
- Cabrita, M.T.; Vale, C.; Rauter, A.P. Halogenated compounds from marine algae. Mar. Drugs 2010, 8, 2301–2317. [Google Scholar] [CrossRef]
- Brodie, J.; Zuccarello, G.C. Systematics of the species rich algae: Red algal classification, phylogeny and speciation. In Reconstructing the Tree of Life: Taxonomy and Systematics of Species Rich Taxa; Hodkinson, T.R., Parnell, J.A.N., Eds.; CRC Press: Boca Raton, FL, USA, 2007; pp. 324–334. [Google Scholar]
- Guiry, M.D.; Guiry, G.M. AlgaeBase; World-Wide Electronic Publication; National University of Ireland: Galway, Ireland, 2021; Available online: http://www.algaebase.org (accessed on 1 December 2021).
- Van den Hoek, C.; Mann, D.G.; Jahns, H.M. Algae; Cambridge University Press: Cambridge, MA, USA, 1997. [Google Scholar]
- Moon-van der Staay, S.Y.; De Wachter, R.; Vaulot, D. Oceanic 18S rDNA sequences from picoplankton reveal unsuspected eukaryotic diversity. Nature 2001, 409, 607–610. [Google Scholar] [CrossRef]
- Baldauf, S.L. The deep roots of eukaryotes. Science 2003, 300, 1703–1706. [Google Scholar] [CrossRef]
- Baldauf, S.L. An overview of the phylogeny and diversity of eukaryotes. J. Syst. Evol. 2008, 46, 263–273. [Google Scholar]
- Krause-Jensen, D.; Lavery, P.; Serrano, O.; Marba, N.; Masque, P.; Duarte, C.M. Sequestration of macroalgal carbon: The elephant in the Blue Carbon room. Biol. Lett. 2018, 14, 20180236. [Google Scholar] [CrossRef]
- Hoegh-Guldberg, O.; Northrop, E.; Lubchenco, J. The ocean is key to achieving climate and societal goals. Science 2019, 365, 1372. [Google Scholar] [CrossRef]
- Holbrook, S.J.; Carr, M.H.; Schmitt, R.J.; Coyer, J.A. Effect of giant kelp on local abundance of reef fishes—The importance of ontogenic resource requirements. Bull. Mar. Sci. 1990, 47, 104–114. [Google Scholar]
- Kitada, S.; Nakajima, K.; Hamasaki, K.; Shishidou, H.; Waples, R.S.; Kishino, H. Rigorous monitoring of a large-scale marine stock enhancement program demonstrates the need for comprehensive management of fisheries and nursery habitat. Sci. Rep. 2019, 9, 5290. [Google Scholar] [CrossRef]
- Teagle, H.; Hawkins, S.J.; Moore, P.J.; Smale, D.A. The role of kelp species as biogenic habitat formers in coastal marine ecosystems. J. Exp. Mar. Biol. Ecol. 2017, 492, 81–98. [Google Scholar] [CrossRef]
- Hind, K.R.; Starko, S.; Burt, J.M.; Lemay, M.A.; Salomon, A.K.; Martone, P.T. Trophic control of cryptic coralline algal diversity. Proc. Natl. Acad. Sci. USA 2019, 116, 15080–15085. [Google Scholar] [CrossRef]
- Hurd, C.; Harrison, P.; Bischof, K.; Lobban, C. Seaweed Ecology and Physiology, 2nd ed.; Cambridge University Press: Cambridge, MA, USA, 2014. [Google Scholar]
- Knoll, A.H. The Multiple Origins of Complex Multicellularity. Annu. Rev. Earth Planet. Sci. 2011, 39, 217–239. [Google Scholar] [CrossRef]
- Cock, J.M.; Godfroy, O.; Macaisne, N.; Peters, A.F.; Coelho, S.M. Evolution and regulation of complex life cycles: A brown algal perspective. Curr. Opin Plant Biol. 2014, 17, 1–6. [Google Scholar] [CrossRef]
- Küpper, F.C. Iodine in Seaweed: Two Centuries of Research; Springer: Berlin, Germany, 2015; pp. 591–596. [Google Scholar]
- Küpper, F.C.; Feiters, M.C.; Olofsson, B.; Kaiho, T.; Yanagida, S.; Zimmermann, M.B.; Carpenter, L.J.; Lu, Z.; Jonsson, M.; Kloo, L. Purple fumes: The importance of iodine. Sci. Sch. 2013, 27, 45–53. [Google Scholar]
- Küpper, F.C.; Carpenter, L.J.; McFiggans, G.B.; Palmer, C.J.; Waite, T.J.; Boneberg, E.M.; Woitsch, S.; Weiller, M.; Abela, R.; Grolimund, D.; et al. Iodide accumulation provides kelp with an inorganic antioxidant impacting atmospheric chemistry. Proc. Natl. Acad. Sci. USA 2008, 105, 6954–6958. [Google Scholar] [CrossRef]
- Küpper, F.C.; Carrano, C.J. Key aspects of the iodine metabolism in brown algae: A brief critical review. Metallomics 2019, 11, 756–764. [Google Scholar] [CrossRef]
- Küpper, F.C.; Feiters, M.C.; Olofsson, B.; Kaiho, T.; Yanagida, S.; Zimmermann, M.B.; Carpenter, L.J.; Luther, G.W., III; Lu, Z.; Jonsson, M.; et al. Commemorating two centuries of iodine research: An interdisciplinary overview of current research. Angew. Chem.-Int. Ed. 2011, 50, 11598–11620. [Google Scholar] [CrossRef]
- Küpper, F.C.; Kroneck, P.M.H. Iodine Bioinorganic Chemistry: Physiology, Structures, and Mechanisms. In Iodine Chemistry and Applications; Kaiho, T., Ed.; John Wiley & Sons Inc.: Hoboken, NJ, USA, 2015; pp. 557–589. [Google Scholar]
- Pröschold, T.; Leliaert, F. Systematics of the Green Algae: Conflict of Classic and Modern Approaches. Syst. Assoc. Spec. Vol. 2007, 75, 123. [Google Scholar]
- Thomas, D.N. Seaweeds; The Natural History Museum: London, UK, 2002. [Google Scholar]
- Downs, A.J.; Adams, C.J. The Chemistry of Chlorine, Bromine, Iodine and Astatine: Pergamon Texts in Inorganic Chemistry; Elsevier: Amsterdam, The Netherlands, 2017; Volume 7. [Google Scholar]
- Rao, C.K.; Singbal, S. Seasonal Variations in Halides in Marine Brown Algae from PORBANDAR and Okha Coasts (NW Coast of India); CSIR–NIScPR: Delhi, India, 1995; Volume 24, pp. 137–141. [Google Scholar]
- Truesdale, V.W.; Luther, G.W.; Canosamas, C. Molecular-Iodine Reduction In Seawater—An improved rate-equation considering organic compounds. Mar. Chem. 1995, 48, 143–150. [Google Scholar] [CrossRef]
- Rao, C.K.; Indusekhar, V. Distribution of Certain Cations and Anions in Seaweeds and Seawater of Saurashtra Coast and Their Geochemical Significance; CSIR–NIScPR: Delhi, India, 1989. [Google Scholar]
- Pekov, I.V.; Lykova, I.S.; Bryzgalov, I.A.; Ksenofontov, D.A.; Zyryanova, L.A.; Litvinov, N.D. Uniquely high-grade iodide mineralization in the oxidation zone of the Rubtsovskoe base-metal deposit, Northwest Altai, Russia. Geol. Ore Depos. 2011, 53, 683–698. [Google Scholar] [CrossRef]
- Gołębiowska, B.; Pieczka, A.; Rzepa, G.; Matyszkiewicz, J.; Krajewski, M. Iodargyrite from Zalas (Cracow area, Poland) as an indicator of Oligocene–Miocene aridity in Central Europe. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2010, 296, 130–137. [Google Scholar] [CrossRef]
- Reich, M.; Palacios, C.; Alvear, M.; Cameron, E.; Leybourne, M.; Deditius, A. Iodine-rich waters involved in supergene enrichment of the Mantos de la Luna argentiferous copper deposit, Atacama Desert, Chile. Miner. Depos. 2009, 44, 719–722. [Google Scholar] [CrossRef]
- Millsteed, P.W. Marshite-miersite solid solution and iodargyrite from Broken Hill, New South Wales, Australia. Mineral. Mag. 1998, 62, 471–475. [Google Scholar] [CrossRef]
- Lodders, K. Solar system abundances and condensation temperatures of the elements. Astrophys. J. 2003, 591, 1220. [Google Scholar] [CrossRef]
- Davy, H. XXXI. Some experiments and observations on the substances produced in different chemical processes on fluor spar. Phil. Trans. R. Soc. 1814, 104, 74–93. [Google Scholar]
- Wisniak, J. The history of fluorine—from discovery to commodity. Indian J. Chem. Technol. 2002, 9, 363–372. [Google Scholar]
- Banks, R. Isolation of flourine by Moissan: Setting the scene. J. Fluor. Chem. 1986, 33, 3–26. [Google Scholar] [CrossRef]
- TH, Y.T. Relationship between Natural Water Quality and Health; United Nations Educational, Scientific and Cultural Organization: Paris, Frence, 1983. [Google Scholar]
- Kendrick, M.A. Halogens in seawater, marine sediments and the altered oceanic lithosphere. In The Role of Halogens in Terrestrial and Extraterrestrial Geochemical Processes; Springer: Cham, Switzerland, 2018; pp. 591–648. [Google Scholar]
- Weast, R.C.; Lide, D.; Astle, M.; Beyer, W. Handbook of Chemistry and Physics. –1989–1990; CRC Press: Boca Raton, FL, USA, 1989. [Google Scholar]
- Coley, N. Animal chemists and urinary stone. Ambix 1971, 18, 69–93. [Google Scholar] [CrossRef]
- Wisniak, J. The History of Bromine from Discovery to Commodity; NISCAIR-CSIR: New Delhi, India, 2002; pp. 263–271. [Google Scholar]
- Gay-Lussac, J.-L. Sur la combinaison de l’iode avec l’oxygène. Ann. Chim. 1813, 88, 319–321. [Google Scholar]
- Gay-Lussac, L.-J. Sur un nouvel acide formé avec la substance découverte par M. Courtois. Ann. Chim. 1813, 88, 311–318. [Google Scholar]
- Chance, R.J.; Tinel, L.; Sherwen, T.; Baker, A.R.; Bell, T.; Brindle, J.; Campos, M.L.A.M.; Croot, P.; Ducklow, H.; Peng, H.; et al. Global sea-surface iodide observations, 1967–2018. Sci. Data 2019, 6, 286. [Google Scholar] [CrossRef]
- Venturi, S.; Donati, F.M.; Venturi, A.; Venturi, M. Environmental iodine deficiency: A challenge to the evolution of terrestrial life? Thyroid 2000, 10, 727–729. [Google Scholar] [CrossRef] [PubMed]
- Oganessian, Y.T.; Abdullin, F.S.; Bailey, P.D.; Benker, D.E.; Bennett, M.E.; Dmitriev, S.N.; Ezold, J.; Hamilton, J.H.; Henderson, R.A.; Itkis, M.G.; et al. Synthesis of a New Element with Atomic NumberZ=117. Phys. Rev. Lett. 2010, 104, 142502. [Google Scholar] [CrossRef] [PubMed]
- Wong, G.T. The marine geochemistry of iodine. Rev. Aquat. Sci. 1991, 4, 45–73. [Google Scholar]
- Moyers, J.L.; Duce, R.A. Gaseous and particulate iodine in the marine atmosphere. J. Geophys. Res. Earth Surf. 1972, 77, 5229–5238. [Google Scholar] [CrossRef]
- Jones, C.E.; Hornsby, K.E.; Sommariva, R.; Dunk, R.M.; Mc Figgans, G.; Von Glasow, R.; Carpenter, L.J. Quantifying the contribution of marine organic gases to atmospheric iodine. Geophys. Res. Lett. 2010, 37, L18804. [Google Scholar] [CrossRef]
- Hoffmann, T.; O’Dowd, C.D.; Seinfeld, J.H. Iodine oxide homogeneous nucleation: An explanation for coastal new particle production. Geophys. Res. Lett. 2001, 28, 1949–1952. [Google Scholar] [CrossRef]
- Saiz-Lopez, A.; Shillito, J.A.; Coe, H.; Plane, J.M.C. Measurements and modelling of I2, IO, OIO, BrO and NO3 in the mid-latitude marine boundary layer. Atmos. Chem. Phys. 2003, 6, 1513–1528. [Google Scholar] [CrossRef]
- He, X.-C.; Tham, Y.J.; Dada, L.; Wang, M.; Finkenzeller, H.; Stolzenburg, D.; Iyer, S.; Simon, M.; Kürten, A.; Shen, J.; et al. Role of iodine oxoacids in atmospheric aerosol nucleation. Science 2021, 371, 589–595. [Google Scholar] [CrossRef] [PubMed]
- Martín, J.C.G.; Lewis, T.R.; Blitz, M.A.; Plane, J.M.C.; Kumar, M.; Francisco, J.S.; Saiz-Lopez, A. A gas-to-particle conversion mechanism helps to explain atmospheric particle formation through clustering of iodine oxides. Nat. Commun. 2020, 11, 4521. [Google Scholar] [CrossRef] [PubMed]
- Whitehead, D. The distribution and transformations of iodine in the environment. Environ. Int. 1984, 10, 321–339. [Google Scholar] [CrossRef]
- Moreda-Piñeiro, A.; Romarís-Hortas, V.; Bermejo-Barrera, P. A review on iodine speciation for environmental, biological and nutrition fields. J. Anal. At. Spectrom. 2011, 26, 2107–2152. [Google Scholar] [CrossRef]
- Bowley, H.E. Iodine Dynamics in the Terrestrial Environment. Ph.D. Thesis, University of Nottingham, Nottingham, UK, 2013. [Google Scholar]
- Saunders, R.W.; Kumar, R.; Macdonald, S.M.; Plane, J.M.C. Insights into the Photochemical Transformation of Iodine in Aqueous Systems: Humic Acid Photosensitized Reduction of Iodate. Environ. Sci. Technol. 2012, 46, 11854–11861. [Google Scholar] [CrossRef]
- Fuge, R.; Johnson, C.C. Iodine and human health, the role of environmental geochemistry and diet, a review. Appl. Geochem. 2015, 63, 282–302. [Google Scholar] [CrossRef]
- Von Glasow, R.; Hughes, C. Biogeochemical Cycles: Bromine. Encycl. Atmos. Sci. 2014, 42, 194–200. [Google Scholar]
- Saiz-Lopez, A.; von Glasow, R. Reactive halogen chemistry in the troposphere. Chem. Soc. Rev. 2012, 41, 6448–6472. [Google Scholar] [CrossRef]
- Öberg, G. The natural chlorine cycle—Fitting the scattered pieces. Appl. Microbiol. Biotechnol. 2002, 58, 565–581. [Google Scholar] [CrossRef]
- Graedel, T.E.; Keene, W.C. The Budget and Cycle of Earth’s Natural Chlorine. Pure Appl. Chem. 1996, 68, 1689–1697. [Google Scholar] [CrossRef]
- Schlesinger, W.H.; Klein, E.M.; Vengosh, A. Global Biogeochemical Cycle of Fluorine. Glob. Biogeochem. Cycles 2020, 34, e2020GB006722. [Google Scholar] [CrossRef]
- Barnum, T.P.; Cheng, Y.; Hill, K.A.; Lucas, L.N.; Carlson, H.K.; Coates, J.D. Identification of a parasitic symbiosis between respiratory metabolisms in the biogeochemical chlorine cycle. ISME J. 2020, 14, 1194–1206. [Google Scholar] [CrossRef] [PubMed]
- Eychenne, R.; Chérel, M.; Haddad, F.; Guérard, F.; Gestin, J.-F. Overview of the Most Promising Radionuclides for Targeted Alpha Therapy: The “Hopeful Eight”. Pharmaceutics 2021, 13, 906. [Google Scholar] [CrossRef] [PubMed]
- Fraústo da Silva, J.J.R.; Williams, R.J.P. The Biological Chemistry of the Elements—The Inorganic Chemistry of Life; Clarendon Press: Oxford, UK, 1991. [Google Scholar]
- Youngblut, M.D.; Wang, O.; Barnum, T.P.; Coates, J.D. (Per)chlorate in Biology on Earth and Beyond. Annu. Rev. Microbiol. 2016, 70, 435–457. [Google Scholar] [CrossRef]
- Ingraham, L.L.; Meyer, D.L. Biochemistry of Dioxygen. In Biochemistry of the Elements; Frieden, E., Ed.; Plenum Press: New York, NY, USA, 1985; Volume 4. [Google Scholar]
- Villafañe, F. Where Is Ozone in the Frost Diagram? J. Chem. Educ. 2009, 86, 432. [Google Scholar] [CrossRef][Green Version]
- Lu, Z.; Jenkyns, H.; Rickaby, R.E. Iodine to calcium ratios in marine carbonate as a paleo-redox proxy during oceanic anoxic events. Geology 2010, 38, 1107–1110. [Google Scholar] [CrossRef]
- Lu, W.; Ridgwell, A.; Thomas, E.; Hardisty, D.S.; Luo, G.; Algeo, T.J.; Saltzman, M.R.; Gill, B.C.; Shen, Y.; Ling, H.-F.; et al. Late inception of a resiliently oxygenated upper ocean. Science 2018, 361, 174–177. [Google Scholar] [CrossRef]
- Lu, Z.; Lu, W.; Rickaby, R.E.M.; Thomas, E. Earth History of Oxygen and the iprOxy; Cambridge University Press: Cambridge, UK, 2020. [Google Scholar]
- Gkotsi, D.S.; Dhaliwal, J.; McLachlan, M.M.; Mulholand, K.R.; Goss, R.J. Halogenases: Powerful tools for biocatalysis (mechanisms applications and scope). Curr. Opin. Chem. Biol. 2018, 43, 119–126. [Google Scholar] [CrossRef]
- Ludewig, H.; Molyneux, S.; Ferrinho, S.; Guo, K.; Lynch, R.; Gkotsi, D.S.; Goss, R.J. Halogenases: Structures and functions. Curr. Opin. Struct. Biol. 2020, 65, 51–60. [Google Scholar] [CrossRef]
- Shaw, P.D.; Hager, L.P. Biological Chlorination. IV. Peroxidative Nature of Enzymatic Chlorination1. J. Am. Chem. Soc. 1959, 81, 6527–6528. [Google Scholar]
- Sundaramoorthy, M.; Terner, J.; Poulos, T.L. The crystal structure of chloroperoxidase: A heme peroxidase–cytochrome P450 functional hybrid. Structure 1995, 3, 1367–1378. [Google Scholar] [CrossRef]
- Loughran, N.B.; O’Connor, B.; Ó’Fágáin, C.; O’Connell, M.J. The phylogeny of the mammalian heme peroxidases and the evolution of their diverse functions. BMC Evol. Biol. 2008, 8, 101. [Google Scholar] [CrossRef] [PubMed]
- de Boer, E.; van Kooyk, Y.; Tromp, M.; Plat, H.; Wever, R. Bromoperoxidase from Ascophyllum nodosum: A novel class of enzymes containing vanadium as a prosthetic group? Biochim. Biophys. Acta (BBA) Protein Struct. Mol. Enzym. 1986, 869, 48–53. [Google Scholar] [CrossRef]
- Leblanc, C.; Vilter, H.; Fournier, J.-B.; Delage, L.; Potin, P.; Rebuffet, E.; Michel, G.; Solari, P.; Feiters, M.; Czjzek, M. Vanadium haloperoxidases: From the discovery 30 years ago to X-ray crystallographic and V K-edge absorption spectroscopic studies. Co-ord. Chem. Rev. 2015, 301, 134–146. [Google Scholar] [CrossRef]
- Held, A.M.; Halko, D.J.; Hurst, J.K. Mechanisms of chlorine oxidation of hydrogen peroxide. J. Am. Chem. Soc. 1978, 100, 5732–5740. [Google Scholar] [CrossRef]
- Kanofsky, J.R.; Hoogland, H.; Wever, R.; Weiss, S.J. Singlet oxygen production by human eosinophils. J. Biol. Chem. 1988, 263, 9692–9696. [Google Scholar] [CrossRef]
- Everett, R.R.; Butler, A. Bromide-assisted hydrogen peroxide disproportionation catalyzed by vanadium bromoperoxidase: Absence of direct catalase activity and implications for the catalytic mechanism. Inorg. Chem. 1989, 28, 393–395. [Google Scholar] [CrossRef]
- Wiesner, W.; van Pée, K.H.; Lingens, F. Purification and characterization of a novel bacterial non-heme chloroperoxidase from Pseudomonas pyrrocinia. J. Biol. Chem. 1988, 263, 13725–13732. [Google Scholar] [CrossRef]
- Hecht, H.; Sobek, H.; Haag, T.; Pfeifer, O.; Van Pée, K.-H.; Van Pée, K.-H. The metal-ion-free oxidoreductase from Streptomyces aureofaciens has an α/β hydrolase fold. Nat. Genet. 1994, 1, 532–537. [Google Scholar] [CrossRef]
- China, H.; Okada, Y.; Ogino, H. Production mechanism of active species on the oxidative bromination following perhydrolase activity. J. Phys. Org. Chem. 2016, 29, 84–91. [Google Scholar] [CrossRef]
- Kirk, O.; Conrad, L.S. Metal-Free Haloperoxidases: Fact or Artifact? Angew. Chem. Int. Ed. Engl. 1999, 38, 977–979. [Google Scholar] [CrossRef]
- Schofield, R.M.S.; Bailey, J.; Coon, J.J.; Devaraj, A.; Garrett, R.W.; Goggans, M.S.; Hebner, M.G.; Lee, B.S.; Lee, D.; Lovern, N.; et al. The homogenous alternative to biomineralization: Zn- and Mn-rich materials enable sharp organismal “tools” that reduce force requirements. Sci. Rep. 2021, 11, 17481. [Google Scholar] [CrossRef] [PubMed]
- Schofield, R.M.S.; Niedbala, J.C.; Nesson, M.H.; Tao, Y.; Shokes, J.E.; Scott, R.A.; Latimer, M.J. Br-rich tips of calcified crab claws are less hard but more fracture resistant: A comparison of mineralized and heavy-element biological materials. J. Struct. Biol. 2009, 166, 272–287. [Google Scholar] [CrossRef] [PubMed]
- Birkedal, H.; Khan, R.K.; Slack, N.; Broomell, C.; Lichtenegger, H.C.; Zok, F.; Stucky, G.D.; Waite, J.H. Halogenated Veneers: Protein Cross-Linking and Halogenation in the Jaws of Nereis, a Marine Polychaete Worm. ChemBioChem 2006, 7, 1392–1399. [Google Scholar] [CrossRef]
- Park, H.B.; Lam, Y.C.; Gaffney, J.P.; Weaver, J.C.; Krivoshik, S.R.; Hamchand, R.; Pieribone, V.; Gruber, D.F.; Crawford, J.M. Bright Green Biofluorescence in Sharks Derives from Bromo-Kynurenine Metabolism. iScience 2019, 19, 1291–1336. [Google Scholar] [CrossRef]
- Arakawa, Y.; Akiyama, Y.; Furukawa, H.; Suda, W.; Amachi, S. Growth Stimulation of Iodide-Oxidizing α-Proteobacteria in Iodide-Rich Environments. Microb. Ecol. 2012, 63, 522–531. [Google Scholar] [CrossRef]
- Suzuki, M.; Eda, Y.; Ohsawa, S.; Kanesaki, Y.; Yoshikawa, H.; Tanaka, K.; Muramatsu, Y.; Yoshikawa, J.; Sato, I.; Fujii, T.; et al. Iodide Oxidation by a Novel Multicopper Oxidase from the Alphaproteobacterium Strain Q-1. Appl. Environ. Microbiol. 2012, 78, 3941–3949. [Google Scholar] [CrossRef]
- Amachi, S.; Muramatsu, Y.; Akiyama, Y.; Miyazaki, K.; Yoshiki, S.; Hanada, S.; Kamagata, Y.; Ban-Nai, T.; Shinoyama, H.; Fujii, T. Isolation of Iodide-Oxidizing Bacteria from Iodide-Rich Natural Gas Brines and Seawaters. Microb. Ecol. 2005, 49, 547–557. [Google Scholar] [CrossRef]
- Gozlan, R.S.; Margalith, P. Iodide Oxidation byPseudomonas iodooxidans. J. Appl. Bacteriol. 1974, 37, 493–499. [Google Scholar] [CrossRef]
- Li, H.-P.; Daniel, B.; Creeley, D.; Grandbois, R.; Zhang, S.; Xu, C.; Ho, Y.-F.; Schwehr, K.A.; Kaplan, D.I.; Santschi, P.H.; et al. Superoxide Production by a Manganese-Oxidizing Bacterium Facilitates Iodide Oxidation. Appl. Environ. Microbiol. 2014, 80, 2693–2699. [Google Scholar] [CrossRef]
- Dairi, T.; Nakano, T.; Aisaka, K.; Katsumata, R.; Hasegawa, M. Cloning and Nucleotide Sequence of the Gene Responsible for Chlorination of Tetracycline. Biosci. Biotechnol. Biochem. 1995, 59, 1099–1106. [Google Scholar] [CrossRef] [PubMed]
- Keller, S.; Wage, T.; Hohaus, K.; Hölzer, M.; Eichhorn, E.; Van Pée, K.-H. Purification and Partial Characterization of Tryptophan 7-Halogenase (PrnA) from Pseudomonas fluorescens. Angew. Chem. Int. Ed. 2000, 39, 2300–2302. [Google Scholar] [CrossRef]
- Mori, S.; Pang, A.H.; Thamban Chandrika, N.; Garneau-Tsodikova, S.; Tsodikov, O.V. Unusual substrate and halide versatility of phenolic halogenase PltM. Nat. Commun. 2019, 10, 1255. [Google Scholar] [CrossRef] [PubMed]
- Podzelinska, K.; Latimer, R.; Bhattacharya, A.; Vining, L.C.; Zechel, D.L.; Jia, Z. Chloramphenicol biosynthesis: The structure of CmlS, a flavin-dependent halogenase showing a covalent flavin-aspartate bond. J. Mol. Biol. 2010, 397, 316–331. [Google Scholar] [CrossRef]
- Fisher, B.F.; Snodgrass, H.M.; Jones, K.A.; Andorfer, M.C.; Lewis, J.C. Site-Selective C–H Halogenation Using Flavin-Dependent Halogenases Identified via Family-Wide Activity Profiling. ACS Central Sci. 2019, 5, 1844–1856. [Google Scholar] [CrossRef]
- Gkotsi, D.S.; Ludewig, H.; Sharma, S.V.; Connolly, J.A.; Dhaliwal, J.; Wang, Y.; Unsworth, W.P.; Taylor, R.J.K.; McLachlan, M.M.W.; Shanahan, S.; et al. A marine viral halogenase that iodinates diverse substrates. Nat. Chem. 2019, 11, 1091–1097. [Google Scholar] [CrossRef]
- Vaillancourt, F.H.; Yin, J.; Walsh, C.T. SyrB2 in syringomycin E biosynthesis is a nonheme FeII α-ketoglutarate- and O2-dependent halogenase. Proc. Natl. Acad. Sci. USA 2005, 102, 10111–10116. [Google Scholar] [CrossRef]
- Blasiak, L.C.; Vaillancourt, F.H.; Walsh, C.T.; Drennan, C.L. Crystal structure of the non-haem iron halogenase SyrB2 in syringomycin biosynthesis. Nature 2006, 440, 368–371. [Google Scholar] [CrossRef]
- Galonić, D.P.; Barr, E.W.; Walsh, C.T.; Bollinger, J.M.; Krebs, C. Two interconverting Fe(IV) intermediates in aliphatic chlorination by the halogenase CytC3. Nat. Chem. Biol. 2007, 3, 113–116. [Google Scholar] [CrossRef]
- Wong, S.D.; Srnec, M.; Matthews, M.L.; Liu, L.V.; Kwak, Y.; Park, K.; Iii, C.B.B.; Alp, E.E.; Zhao, J.; Yoda, Y.; et al. Elucidation of the Fe(iv)=O intermediate in the catalytic cycle of the halogenase SyrB2. Nature 2013, 499, 320–323. [Google Scholar] [CrossRef]
- Mitchell, A.J.; Zhu, Q.; Maggiolo, A.O.; Ananth, N.R.; Hillwig, M.L.; Liu, X.; Boal, A.K. Structural basis for halogenation by iron- and 2-oxo-glutarate-dependent enzyme WelO5. Nat. Chem. Biol. 2016, 12, 636–640. [Google Scholar] [CrossRef] [PubMed]
- Hillwig, M.L.; Zhu, Q.; Ittiamornkul, K.; Liu, X. Discovery of a Promiscuous Non-Heme Iron Halogenase in Ambiguine Alkaloid Biogenesis: Implication for an Evolvable Enzyme Family for Late-Stage Halogenation of Aliphatic Carbons in Small Molecules. Angew. Chem. Int. Ed. Engl. 2016, 55, 5780–5784. [Google Scholar] [CrossRef] [PubMed]
- Hillwig, M.; Liu, X. A new family of iron-dependent halogenases acts on freestanding substrates. Nat. Chem. Biol. 2014, 10, 921–923. [Google Scholar] [CrossRef] [PubMed]
- DuBois, J.L.; Ojha, S. Production of Dioxygen in the Dark: Dismutases of Oxyanions. Met. Ions Life Sci. 2014, 15, 45–87. [Google Scholar]
- Kengen, S.W.M.; Rikken, G.B.; Hagen, W.R.; van Ginkel, C.G.; Stams, A. Purification and Characterization of (Per)Chlorate Reductase from the Chlorate-Respiring Strain GR-1. J. Bacteriol. 1999, 181, 6706–6711. [Google Scholar] [CrossRef]
- Sun, S.Q.; Chen, S.L. How does Mo-dependent perchlorate reductase work in the decomposition of oxyanions? Dalton Trans. 2019, 48, 5683–5691. [Google Scholar] [CrossRef]
- Amachi, S.; Kawaguchi, N.; Muramatsu, Y.; Tsuchiya, S.; Watanabe, Y.; Shinoyama, H.; Fujii, T. Dissimilatory Iodate Reduction by Marine Pseudomonas sp. Strain SCT. Appl. Environ. Microbiol. 2007, 73, 5725–5730. [Google Scholar] [CrossRef]
- Tamai, N.; Ishii, T.; Sato, Y.; Fujiya, H.; Muramatsu, Y.; Okabe, N.; Amachi, S. Bromate Reduction by Rhodococcus sp. Br-6 in the Presence of Multiple Redox Mediators. Environ. Sci. Technol. 2016, 50, 10527–10534. [Google Scholar] [CrossRef]
- Maymó-Gatell, X.; Tandoi, V.; Gossett, J.M.; Zinder, S.H. Characterization of an H2-utilizing enrichment culture that reductively dechlorinates tetrachloroethene to vinyl chloride and ethene in the absence of methanogenesis and acetogenesis. Appl. Environ. Microbiol. 1995, 61, 3928–3933. [Google Scholar] [CrossRef]
- Maymó-Gatell, X.; Chien, Y.-T.; Gossett, J.M.; Zinder, S.H. Isolation of a Bacterium That Reductively Dechlorinates Tetrachloroethene to Ethene. Science 1997, 276, 1568–1571. [Google Scholar] [CrossRef]
- Schubert, T.; Adrian, L.; Sawers, R.G.; Diekert, G. Organohalide respiratory chains: Composition, topology and key enzymes. FEMS Microbiol. Ecol. 2018, 94, fiy035. [Google Scholar] [CrossRef] [PubMed]
- Bommer, M.; Kunze, C.; Fesseler, J.; Schubert, T.; Diekert, G.; Dobbek, H. Structural basis for organohalide respiration. Science 2014, 346, 455–458. [Google Scholar] [CrossRef] [PubMed]
- Dolfing, J. Energetic Considerations in Organohalide Respiration. In Organohalide-Respiring Bacteria; Adrian, L., Löffler, F.E., Eds.; Springer: Berlin/Heidelberg, Germany, 2016; pp. 31–48. [Google Scholar]
- Yang, Y.; Sanford, R.; Yan, J.; Chen, G.; Cápiro, N.L.; Li, X.; Löffler, F.E. Roles of Organohalide-Respiring Dehalococcoidia in Carbon Cycling. mSystems 2020, 5, e00757-19. [Google Scholar] [CrossRef]
- Atashgahi, S.; Häggblom, M.M.; Smidt, H. Organohalide respiration in pristine environments: Implications for the natural halogen cycle. Environ. Microbiol. 2018, 20, 934–948. [Google Scholar] [CrossRef] [PubMed]
- Chekan, J.R.; Lee, G.Y.; El Gamal, A.; Purdy, T.N.; Houk, K.N.; Moore, B.S. Bacterial Tetrabromopyrrole Debrominase Shares a Reductive Dehalogenation Strategy with Human Thyroid Deiodinase. Biochemistry 2019, 58, 5329–5338. [Google Scholar] [CrossRef]
- Temme, H.R.; Carlson, A.; Novak, P.J. Presence, Diversity, and Enrichment of Respiratory Reductive Dehalogenase and Non-respiratory Hydrolytic and Oxidative Dehalogenase Genes in Terrestrial Environments. Front. Microbiol. 2019, 10, 1258. [Google Scholar] [CrossRef] [PubMed]
- Fetzner, S. Bacterial dehalogenation. Appl. Microbiol. Biotechnol. 1998, 50, 633–657. [Google Scholar] [CrossRef]
- Ang, T.-F.; Maiangwa, J.; Salleh, A.B.; Normi, Y.M.; Leow, T.C. Dehalogenases: From Improved Performance to Potential Microbial Dehalogenation Applications. Molecules 2018, 23, 1100. [Google Scholar] [CrossRef]
- Cavina, L.; Van Der Born, D.; Klaren, P.H.M.; Feiters, M.C.; Boerman, O.C.; Rutjes, F.P.J.T. Design of Radioiodinated Pharmaceuticals: Structural Features Affecting Metabolic Stability towards in Vivo Deiodination. Eur. J. Org. Chem. 2017, 2017, 3387–3414. [Google Scholar] [CrossRef]
- Macdonald, T.L.; Anders, M.W. Chemical Mechanisms of Halocarbon Metabolism. CRC Crit. Rev. Toxicol. 1983, 11, 85–120. [Google Scholar] [CrossRef]
- Cnubben, N.H.; Vervoort, J.; Boersma, M.G.; Rietjens, I.M. The effect of varying halogen substituent patterns on the cytochrome-P450 catalyzed dehalogenation of 4-halogenated anilines to 4-aminophenol metabolites. Biochem. Pharmacol. 1995, 49, 1235–1248. [Google Scholar] [CrossRef]
- Sinsheimer, J.E.; Wang, T.; Röder, S.; Shum, Y.Y. Mechanisms for biodehalogenation of iodocompounds. Biochem. Biophys. Res. Commun. 1978, 83, 281–286. [Google Scholar] [CrossRef]
- Zhang, C.; Kenny, J.R.; Le, H.; Deese, A.; Ford, K.A.; Lightning, L.K.; Fan, P.W.; Driscoll, J.P.; Halladay, J.S.; Hop, C.E.; et al. Novel mechanism for dehalogenation and glutathione conjugation of dihalogenated anilines in human liver microsomes: Evidence for ipso glutathione addition. Chem. Res. Toxicol. 2011, 24, 1668–1677. [Google Scholar] [CrossRef] [PubMed]
- Messerschmidt, A.; Prade, L.; Wever, R. Implications for the Catalytic Mechanism of the Vanadium-Containing Enzyme Chloroperoxidase from the Fungus Curvularia inaequalis by X-Ray Structures of the Native and Peroxide Form. Biol. Chem. 1997, 378, 309–315. [Google Scholar] [CrossRef]
- Dutzler, R.; Campbell, E.B.; MacKinnon, R. Gating the Selectivity Filter in ClC Chloride Channels. Science 2003, 300, 108–112. [Google Scholar] [CrossRef] [PubMed]
- Skitchenko, R.K.; Usoltsev, D.; Uspenskaya, M.; Kajava, A.V.; Guskov, A. Census of halide-binding sites in protein structures. Bioinformatics 2020, 36, 3064–3071. [Google Scholar] [CrossRef] [PubMed]
- Chang, T.M.; Berden, G.; Oomens, J.; Williams, E.R. Halide anion binding to Gly(3), Ala(3) and Leu(3). Int. J. Mass Spectrom. 2015, 377, 440–447. [Google Scholar] [CrossRef]
- O’Hagan, D.; Schaffrath, C.; Cobb, S.L.; Hamilton, J.T.; Murphy, C.D. Biosynthesis of an organofluorine molecule—A fluorinase enzyme has been discovered that catalyses carbon-fluorine bond formation. Nature 2002, 416, 279. [Google Scholar]
- Cadicamo, C.D.; Courtieu, J.; Deng, H.; Meddour, A.; O’Hagan, D. Enzymatic fluorination in Streptomyces cattleya takes place with an inversion of configuration consistent with an SN2 reaction mechanism. Chembiochem 2004, 5, 685–690. [Google Scholar] [CrossRef]
- Healy, E.F. The effect of desolvation on nucleophilic halogenase activity. Comput. Theor. Chem. 2011, 964, 91–93. [Google Scholar] [CrossRef]
- Vincent, M.A.; Hillier, I.H. The solvated fluoride anion can be a good nucleophile. Chem. Commun. 2005, 5902–5903. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Robinson, D.A.; McEwan, A.R.; O’Hagan, D.; Naismith, J.H. Mechanism of Enzymatic Fluorination in Streptomyces cattleya. J. Am. Chem. Soc. 2007, 129, 14597–14604. [Google Scholar] [CrossRef] [PubMed]
- Dong, C.; Huang, F.; Deng, H.; Schaffrath, C.; Spencer, J.B.; O’Hagan, D.; Naismith, J.H. Crystal structure and mechanism of a bacterial fluorinating enzyme. Nature 2004, 427, 561–565. [Google Scholar] [CrossRef] [PubMed]
- Sooklal, S.A.; Mpangase, P.T.; Tomescu, M.-S.; Aron, S.; Hazelhurst, S.; Archer, R.H.; Rumbold, K. Functional characterisation of the transcriptome from leaf tissue of the fluoroacetate-producing plant, Dichapetalum cymosum, in response to mechanical wounding. Sci. Rep. 2020, 10, 20539. [Google Scholar] [CrossRef]
- Eustaquio, A.; Pojer, F.; Noel, J.P.; Moore, B.S. Discovery and characterization of a marine bacterial SAM-dependent chlorinase. Nat. Chem. Biol. 2008, 4, 69–74. [Google Scholar] [CrossRef]
- Beer, L.L.; Moore, B.S. Biosynthetic Convergence of Salinosporamides A and B in the Marine Actinomycete Salinispora tropica. Org. Lett. 2007, 9, 845–848. [Google Scholar] [CrossRef]
- Wuosmaa, A.M.; Hager, L.P. Methyl Chloride Transferase: A Carbocation Route for Biosynthesis of Halometabolites. Science 1990, 249, 160–162. [Google Scholar] [CrossRef]
- Ohsawa, N.; Tsujita, M.; Morikawa, S.; Itoh, N. Purification and Characterization of a Monohalomethane-producing Enzyme S-adenosyl-L-methionine: Halide Ion Methyltransferase from a Marine Microalga, Pavlova pinguis. Biosci. Biotechnol. Biochem. 2001, 65, 2397–2404. [Google Scholar] [CrossRef]
- Schmidberger, J.W.; James, A.B.; Edwards, R.H.; Naismith, J.H.; O’Hagan, D. Halomethane Biosynthesis: Structure of a SAM-Dependent Halide Methyltransferase from Arabidopsis thaliana. Angew. Chem. Int. Ed. 2010, 49, 3646–3648. [Google Scholar] [CrossRef]
- Paul, C.; Pohnert, G. Production and role of volatile halogenated compounds from marine algae. Nat. Prod. Rep. 2011, 28, 186–195. [Google Scholar] [CrossRef]
- Küpper, F.C.; Miller, E.P.; Andrews, S.J.; Hughes, C.; Carpenter, L.; Meyer-Klaucke, W.; Toyama, C.; Muramatsu, Y.; Feiters, M.C.; Carrano, C.J. Emission of volatile halogenated compounds, speciation and localization of bromine and iodine in the brown algal genome model Ectocarpus siliculosus. JBIC J. Biol. Inorg. Chem. 2018, 23, 1119–1128. [Google Scholar] [CrossRef] [PubMed]
- Amachi, S. Microbial Contribution to Global Iodine Cycling: Volatilization, Accumulation, Reduction, Oxidation, and Sorption of Iodine. Microbes Environ. 2008, 23, 269–276. [Google Scholar] [CrossRef] [PubMed]
- WMO. Scientific Assessment of Ozone Depletion: 1994; WMO: Geneva, Switzerland, 1995. [Google Scholar]
- WMO. Scientific Assessment of Ozone Depletion: 1998; WMO: Geneva, Switzerland, 1999. [Google Scholar]
- Cicerone, R.J.; Walters, S.; Liu, S.C. Non-linear response of stratospheric ozone column to chlorine injections. J. Geophys. Res. -Ocean. 1983, 88, 3647–3661. [Google Scholar] [CrossRef]
- Farman, J.C.; Gardiner, B.G.; Shanklin, J.D. Large losses of total ozone in Antarctica reveal seasonal ClOx/NOx interaction. Nature 1985, 315, 207–210. [Google Scholar] [CrossRef]
- Solomon, S. The discovery of the Antarctic ozone hole. Nature 2019, 575, 46–47. [Google Scholar] [CrossRef]
- Barrie, L.A.; Bottenheim, J.W.; Schnell, R.; Crutzen, P.J.; Rasmussen, R.A. Ozone destruction and photochemical reactions at polar sunrise in the lower Arctic atmosphere. Nature 1988, 334, 138–141. [Google Scholar] [CrossRef]
- Crutzen, P.J. The influence of nitrogen oxides on the atmospheric ozone content. Q. J. R. Meteorol. Soc. 1970, 96, 320–325. [Google Scholar] [CrossRef]
- Molina, M.J.; Rowland, F.S. Stratospheric sink for chlorofluoromethanes: Chlorine atom-catalysed destruction of ozone. Nat. 1974, 249, 810–812. [Google Scholar] [CrossRef]
- McNeill, V.F. Obituary Mario Molina (1943–2020). Nature 2020, 587, 193. [Google Scholar] [CrossRef]
- Prather, M.J.; Blake, D.R.F. Sherwood Rowland (1927–2012). Nature 2012, 484, 168. [Google Scholar] [CrossRef]
- Prather, M.J.; McElroy, M.B.; Wofsy, S.C. Reductions in ozone at high concentrations of stratospheric halogens. Nature 1984, 312, 227–231. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.G.; Brune, W.H.; Proffitt, M.H. Ozone destruction by chlorine radicals within the Antarctic vortex: The spatial and temporal evolution of ClO-O3anticorrelation based on in situ ER-2 data. J. Geophys. Res. Earth Surf. 1989, 94, 11465–11479. [Google Scholar] [CrossRef]
- Gareau, B.J. A critical review of the successful CFC phase-out versus the delayed methyl bromide phase-out in the Montreal Protocol. Int. Environ. Agreem. -Politics Law Econ. 2010, 10, 209–231. [Google Scholar] [CrossRef]
- Jovanovic, D.; Lukinovic, M.; Vitosevic, Z. Environment and health—Thirty years of successful implementation of the Montreal protocol. Srpski Arhiv Za Celokupno Lekarstvo 2019, 147, 492–496. [Google Scholar] [CrossRef]
- Chipperfield, M.P.; Bekki, S.; Dhomse, S.; Harris, N.R.P.; Hassler, B.; Hossaini, R.; Steinbrecht, W.; Thiéblemont, R.; Weber, M. Detecting recovery of the stratospheric ozone layer. Nature 2017, 549, 211–218. [Google Scholar] [CrossRef]
- Weatherhead, E.C.; Andersen, S. The search for signs of recovery of the ozone layer. Nature 2006, 441, 39–45. [Google Scholar] [CrossRef]
- Koenig, T.K.; Baidar, S.; Campuzano-Jost, P.; Cuevas, C.A.; Dix, B.; Fernandez, R.P.; Guo, H.; Hall, S.R.; Kinnison, D.; Nault, B.A.; et al. Quantitative detection of iodine in the stratosphere. Proc. Natl. Acad. Sci. USA 2020, 117, 1860–1866. [Google Scholar] [CrossRef]
- Simpson, W.R.; Brown, S.S.; Saiz-Lopez, A.; Thornton, J.A.; von Glasow, R. Tropospheric Halogen Chemistry: Sources, Cycling, and Impacts. Chem. Rev. 2015, 115, 4035–4062. [Google Scholar] [CrossRef]
- Sherwen, T.; Evans, M.J.; Carpenter, L.J.; Andrews, S.J.; Lidster, R.T.; Dix, B.; Koenig, T.K.; Sinreich, R.; Ortega, I.; Volkamer, R.; et al. Iodine’s impact on tropospheric oxidants: A global model study in GEOS-Chem. Atmos. Chem. Phys. 2016, 16, 1161–1186. [Google Scholar] [CrossRef]
- Sherwen, T.; Evans, M.J.; Sommariva, R.; Hollis, L.D.J.; Ball, S.M.; Monks, P.S.; Reed, C.; Carpenter, L.J.; Lee, J.D.; Forster, G.; et al. Effects of halogens on European air-quality. Faraday Discuss. 2017, 200, 75–100. [Google Scholar] [CrossRef]
- Saiz-Lopez, A.; Fernandez, R.P.; Ordóñez, C.; Kinnison, D.E.; Martín, J.C.G.; Lamarque, J.-F.; Tilmes, S. Iodine chemistry in the troposphere and its effect on ozone. Atmos. Chem. Phys. 2014, 14, 13119–13143. [Google Scholar] [CrossRef]
- Saiz-Lopez, A.; Plane, J.M.C.; Baker, A.R.; Carpenter, L.; von Glasow, R.; Martin, J.C.G.; McFiggans, G.; Saunders, R.W. Atmospheric Chemistry of Iodine. Chem. Rev. 2011, 112, 1773–1804. [Google Scholar] [CrossRef] [PubMed]
- Méndez-Díaz, J.D.; Shimabuku, K.K.; Ma, J.; Enumah, Z.O.; Pignatello, J.J.; Mitch, W.A.; Dodd, M.C. Sunlight-Driven Photochemical Halogenation of Dissolved Organic Matter in Seawater: A Natural Abiotic Source of Organobromine and Organoiodine. Environ. Sci. Technol. 2014, 48, 7418–7427. [Google Scholar] [CrossRef] [PubMed]
- Read, K.; Mahajan, A.; Carpenter, L.; Evans, M.J.; Faria, B.V.E.; Heard, D.; Hopkins, J.; Lee, J.D.; Moller, S.; Lewis, A.; et al. Extensive halogen-mediated ozone destruction over the tropical Atlantic Ocean. Nature 2008, 453, 1232–1235. [Google Scholar] [CrossRef]
- von Glasow, R.; von Kuhlmann, R.; Lawrence, M.G.; Platt, U.; Crutzen, P.J. Impact of reactive bromine chemistry in the troposphere. Atmos. Chem. Phys. 2004, 4, 2481–2497. [Google Scholar] [CrossRef]
- Bloss, W.J.; Lee, J.D.; Johnson, G.P.; Sommariva, R.; Heard, D.; Saiz-Lopez, A.; Plane, J.M.C.; McFiggans, G.; Coe, H.; Flynn, M.; et al. Impact of halogen monoxide chemistry upon boundary layer OH and HO2 concentrations at a coastal site. Geophys. Res. Lett. 2005, 32, L06814. [Google Scholar] [CrossRef]
- Sander, R.; Rudich, Y.; Von Glasow, R.; Crutzen, P.J. The role of BrNO3 in marine tropospheric chemistry: A model study. Geophys. Res. Lett. 1999, 26, 2857–2860. [Google Scholar] [CrossRef]
- Carpenter, L.; Macdonald, S.M.; Shaw, M.D.; Kumar, R.; Saunders, R.W.; Parthipan, R.; Wilson, J.; Plane, J. Atmospheric iodine levels influenced by sea surface emissions of inorganic iodine. Nat. Geosci. 2013, 6, 108–111. [Google Scholar] [CrossRef]
- Carpenter, L.J.; Chance, R.J.; Sherwen, T.; Adams, T.J.; Ball, S.M.; Evans, M.J.; Hepach, H.; Hollis, L.D.; Hughes, C.; Jickells, T.D.; et al. Marine iodine emissions in a changing world. Proc. R. Soc. A 2021, 477, 20200824. [Google Scholar] [CrossRef]
- Legrand, M.; McConnell, J.R.; Preunkert, S.; Arienzo, M.; Chellman, N.; Gleason, K.; Sherwen, T.; Evans, M.J.; Carpenter, L. Alpine ice evidence of a three-fold increase in atmospheric iodine deposition since 1950 in Europe due to increasing oceanic emissions. Proc. Natl. Acad. Sci. USA 2018, 115, 12136–12141. [Google Scholar] [CrossRef]
- Cuevas, C.A.; Maffezzoli, N.; Corella, J.P.; Spolaor, A.; Vallelonga, P.; Kjær, H.A.; Simonsen, M.F.; Winstrup, M.; Vinther, B.; Horvat, C.; et al. Rapid increase in atmospheric iodine levels in the North Atlantic since the mid-20th century. Nat. Commun. 2018, 9, 1452. [Google Scholar] [CrossRef] [PubMed]
- Keller, J.M.; Boynton, W.V.; Karunatillake, S.; Baker, V.R.; Dohm, J.M.; Evans, L.G.; Finch, M.J.; Hahn, B.C.; Hamara, D.K.; Janes, D.M.; et al. Equatorial and midlatitude distribution of chlorine measured by Mars Odyssey GRS. J. Geophys. Res. Earth Surf. 2006, 112. [Google Scholar] [CrossRef]
- Wyngaarden, J.B.; Wright, B.M.; Ways, P. The Effect of certain anions upon the accumulation and retention of iodide by the thyroid gland. Endocrinology 1952, 50, 537–549. [Google Scholar] [CrossRef] [PubMed]
- Davila, A.F.; Willson, D.; Coates, J.D.; McKay, C.P. Perchlorate on Mars: A chemical hazard and a resource for humans. Int. J. Astrobiol. 2013, 12, 321–325. [Google Scholar] [CrossRef]
- Channer, D.; de Ronde, C.; Spooner, E. The Cl−Br−I− composition of ∼3.23 Ga modified seawater: Implications for the geological evolution of ocean halide chemistry. Earth Planet. Sci. Lett. 1997, 150, 325–335. [Google Scholar] [CrossRef]
- Burgess, R.; Goldsmith, S.L.; Sumino, H.; Gilmour, J.D.; Marty, B.; Pujol, M.; Konhauser, K.O. Archean to Paleoproterozoic seawater halogen ratios recorded by fluid inclusions in chert and hydrothermal quartz. Am. Miner. 2020, 105, 1317–1325. [Google Scholar] [CrossRef]
- Lu, Z.; Hoogakker, B.; Hillenbrand, C.-D.; Zhou, X.; Thomas, E.; Gutchess, K.M.; Lu, W.; Jones, L.; Rickaby, R.E.M. Oxygen depletion recorded in upper waters of the glacial Southern Ocean. Nat. Commun. 2016, 7, 11146. [Google Scholar] [CrossRef]
- Pohl, A.; Lu, Z.; Lu, W.; Stockey, R.G.; Elrick, M.; Li, M.; André, D.; Shen, Y.; He, R.; Finnegan, S.; et al. Reorganization of ocean circulation and oxygenation during late Ordovician glaciation. Nat. Geosci. 2021, in press. [Google Scholar]
- Hoogakker, B.A.A.; Lu, Z.; Umling, N.; Jones, L.; Zhou, X.; Rickaby, R.E.M.; Thunell, R.; Cartapanis, O.; Galbraith, E. Glacial expansion of oxygen-depleted seawater in the eastern tropical Pacific. Nature 2018, 562, 410–413. [Google Scholar] [CrossRef]
- Thilly, C.H.; Vanderpas, J.B.; Bebe, N.; Ntambue, K.; Contempre, B.; Swennen, B.; Moreno-Reyes, R.; Bourdoux, P.; Delange, F. Iodine deficiency, other trace elements, and goitrogenic factors in the etiopathogeny of iodine deficiency disorders (IDD). Biol. Trace Element Res. 1992, 32, 229. [Google Scholar] [CrossRef]
- Chatin, A. Recherches sur l’iode des eaux douces; de la présence de ce corps dans les plantes at les animaux terrestes. C. R. Acad. Sci. 1851, 31, 280–283. [Google Scholar]
- Zimmermann, M.B. Research on Iodine Deficiency and Goiter in the 19th and Early 20th Centuries. J. Nutr. 2008, 138, 2060–2063. [Google Scholar] [CrossRef] [PubMed]
- Baumann, F. Ueber das normale Vorkommen von Jod im Thierkörper. Z. Phys. Chem. 1896, 21, 319–330. [Google Scholar] [CrossRef][Green Version]
- Roos, E. Ueber die Wirkung des Thyrojodins. Biol. Chem. 1897, 22, 18–61. [Google Scholar] [CrossRef][Green Version]
- Yarrington, C.D.; Pearce, E.N. Dietary Iodine in Pregnancy and Postpartum. Clin. Obstet. Gynecol. 2011, 54, 459–470. [Google Scholar] [CrossRef]
- Pal, G. Endocrine Physiology. In Textbook of Medical Physiology; Ahuja Publishing House: New Delhi, India, 2007; p. 346. [Google Scholar]
- Hetzel, B.; Dunn, J. The iodine deficiency disorders: Their nature and prevention. Annu. Rev. Nutr. 1989, 9, 21–38. [Google Scholar] [CrossRef]
- Porterfield, S.P.; White, B.A. Endocrine Physiology; Mosby: London, UK, 2007. [Google Scholar]
- Delange, F. The Disorders Induced by Iodine Deficiency. Thyroid 1994, 4, 107–128. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Shan, Z.; Teng, W. Effects of Increased Iodine Intake on Thyroid Disorders. Endocrinol. Metab. 2014, 29, 240–247. [Google Scholar] [CrossRef]
- Zimmermann, M.B.; Gizak, M.; Abbott, K.; Andersson, M.; Lazarus, J.H. Iodine deficiency in pregnant women in Europe. Lancet Diabetes Endocrinol. 2015, 3, 672–674. [Google Scholar] [CrossRef]
- Solomon, B.L.; Evaul, J.E.; Burman, K.D.; Wartofsky, L. Remission rates with antithyroid drug therapy: Continuing influence of iodine intake? Ann. Intern. Med. 1987, 107, 510–512. [Google Scholar] [CrossRef]
- Blasco, B.; Rios, J.; Cervilla, L.; Sánchez-Rodrigez, E.; Ruiz, J.; Romero, L. Iodine biofortification and antioxidant capacity of lettuce: Potential benefits for cultivation and human health. Ann. Appl. Biol. 2008, 152, 289–299. [Google Scholar] [CrossRef]
- Venturi, S.; Venturi, M. Iodide, thyroid and stomach carcinogenesis: Evolutionary story of a primitive antioxidant? Eur. J. Endocrinol. 1999, 140, 371–372. [Google Scholar] [CrossRef] [PubMed]
- Eskin, B.; Stadel, B. Dietary iodine and cancer risk. Lancet 1976, 308, 807–808. [Google Scholar] [CrossRef]
- Ghent, W.R.; Eskin, B.A.; Low, D.E.; Hill, L.P. Iodine replacement in fibrocystic disease of the breast. Can. J. Surg. 1993, 36, 453–460. [Google Scholar] [PubMed]
- Parkin, D.M.; Whelan, S.L.; Ferlay, J.; Teppo, L.; Thomas, D.B. Cancer Incidence in Five Continents. In Lyon: International Agency for Research on Cancer; World Health Organization Scientific Publications: Geneva, Switzerland, 1997; Volume 8, pp. 66–68. [Google Scholar]
- Funahashi, H.; Imai, T.; Tanaka, Y.; Tobinaga, J.; Wada, M.; Morita, T.; Yamada, F.; Tsukamura, K.; Oiwa, M.; Kikumori, T.; et al. Suppressive effect of iodine on DMBA-induced breast tumor growth in the rat. J. Surg. Oncol. 1996, 61, 209–213. [Google Scholar] [CrossRef]
- Carpena, X.; Vidossich, P.; Schroettner, K.; Calisto, B.M.; Banerjee, S.; Stampler, J.; Soudi, M.; Furtmüller, P.G.; Rovira, C.; Fita, I.; et al. Essential Role of Proximal Histidine-Asparagine Interaction in Mammalian Peroxidases. J. Biol. Chem. 2009, 284, 25929–25937. [Google Scholar] [CrossRef]
- Singh, A.K.; Singh, N.; Sharma, S.; Singh, S.B.; Kaur, P.; Bhushan, A.; Srinivasan, A.; Singh, T.P. Crystal Structure of Lactoperoxidase at 2. 4 Å Resolution. J. Mol. Biol. 2008, 376, 1060–1075. [Google Scholar] [CrossRef]
- Ruf, J.; Carayon, P. Structural and functional aspects of thyroid peroxidase. Arch. Biochem. Biophys. 2006, 445, 269–277. [Google Scholar] [CrossRef]
- McCall, A.S.; Cummings, C.F.; Bhave, G.; Vanacore, R.; Page-McCaw, A.; Hudson, B.G. Bromine Is an Essential Trace Element for Assembly of Collagen IV Scaffolds in Tissue Development and Architecture. Cell 2014, 157, 1380–1392. [Google Scholar] [CrossRef]
- Tin, A.; Nadkarni, G.; Evans, A.M.; Winkler, C.A.; Bottinger, E.; Rebholz, C.M.; Sarnak, M.J.; Inker, L.A.; Levey, A.S.; Lipkowitz, M.S.; et al. Serum 6-Bromotryptophan Levels Identified as a Risk Factor for CKD Progression. J. Am. Soc. Nephrol. 2018, 29, 1939–1947. [Google Scholar] [CrossRef]
- Mayeno, A.N.; Curran, A.J.; Roberts, R.L.; Foote, C.S. Eosinophils Preferentially Use Bromide to Generate Halogenating Agents. J. Biol. Chem. 1989, 264, 5660–5668. [Google Scholar] [CrossRef]
- Wu, W.; Chen, Y.; D’Avignon, A.; Hazen, S.L. 3-Bromotyrosine and 3,5-Dibromotyrosine Are Major Products of Protein Oxidation by Eosinophil Peroxidase: Potential Markers for Eosinophil-Dependent Tissue Injury in Vivo. Biochemistry 1999, 38, 3538–3548. [Google Scholar] [CrossRef] [PubMed]
- Sabir, M.; Tan, Y.Y.; Aris, A.; Mani, A.R. The role of endogenous bromotyrosine in health and disease. Free Radic. Res. 2019, 53, 1019–1034. [Google Scholar] [CrossRef] [PubMed]
- Thomas, S.R.; McTamney, P.M.; Adler, J.M.; LaRonde-LeBlanc, N.; Rokita, S.E. Crystal Structure of Iodotyrosine Deiodinase, a Novel Flavoprotein Responsible for Iodide Salvage in Thyroid Glands. J. Biol. Chem. 2009, 284, 19659–19667. [Google Scholar] [CrossRef]
- Bianco, A.C.; Larsen, P.R. Cellular and Structural Biology of the Deiodinases. Thyroid 2005, 15, 777–786. [Google Scholar] [CrossRef]
- Cavallo, G.; Metrangolo, P.; Milani, R.; Pilati, T.; Priimagi, A.; Resnati, G.; Terraneo, G. The Halogen Bond. Chem. Rev. 2016, 116, 2478–2601. [Google Scholar] [CrossRef]
- Auffinger, P.; Hays, F.A.; Westhof, E.; Ho, P.S. Halogen bonds in biological molecules. Proc. Natl. Acad. Sci. USA 2004, 101, 16789–16794. [Google Scholar] [CrossRef]
- Bayse, C.A.; Rafferty, E.R. Is Halogen Bonding the Basis for Iodothyronine Deiodinase Activity? Inorg. Chem. 2010, 49, 5365–5367. [Google Scholar] [CrossRef]
- World Health Organization. Iodine Deficiency in Europe: A Continuing Public Health Problem; WHO: Geneva, Switzerland, 2007. [Google Scholar]
- World Health Organization, Nutrition Unit. Recommended Iodine Levels in Salt and Guidelines for Monitoring Their Adequacy and Effectiveness; WHO: Geneva, Switzerland, 1996. [Google Scholar]
- Crawford, B.A.; Cowell, C.T.; Emder, P.J.; Learoyd, D.L.; Chua, E.L.; Sinn, J.; Jack, M.M. Iodine toxicity from soy milk and seaweed ingestion is associated with serious thyroid dysfunction. Med J. Aust. 2010, 193, 413–415. [Google Scholar] [CrossRef]
- Müssig, K.; Thamer, C.; Bares, R.; Lipp, H.P.; Häring, H.U.; Gallwitz, B. Iodine-induced thyrotoxicosis after ingestion of kelp-containing tea. J. Gen. Intern. Med. 2006, 21, C11–C14. [Google Scholar] [CrossRef]
- Nishiyama, S.; Mikeda, T.; Okada, T.; Nakamura, K.; Kotani, T.; Hishinuma, A. Transient Hypothyroidism or Persistent Hyperthyrotropinemia in Neonates Born to Mothers with Excessive Iodine Intake. Thyroid 2004, 14, 1077–1083. [Google Scholar] [CrossRef] [PubMed]
- Müssig, K. Iodine-induced toxic effects due to seaweed consumption. Compr. Handb. Iodine 2009, 897–908. [Google Scholar]
- Palmieri, N.; Forleo, M.B. The potential of edible seaweed within the western diet. A segmentation of Italian consumers. Int. J. Gastron. Food Sci. 2020, 20, 100202. [Google Scholar] [CrossRef]
- Teas, J.; Pino, S.; Critchley, A.; Braverman, L.E. Variability of iodine content in common commercially available edible seaweeds. Thyroid 2004, 14, 836–841. [Google Scholar] [CrossRef]
- Lüning, K.; Mortensen, L. European aquaculture of sugar kelp (Saccharina latissima) for food industries: Iodine content and epiphytic animals as major problems. Bot. Mar. 2015, 58, 449–455. [Google Scholar] [CrossRef]
- Nitschke, U.; Stengel, D.B. Quantification of iodine loss in edible Irish seaweeds during processing. J. Appl. Phycol. 2016, 28, 3527–3533. [Google Scholar] [CrossRef]
- Nagataki, S. The Average of Dietary Iodine Intake due to the Ingestion of Seaweeds is 1.2 mg/day in Japan. Thyroid 2008, 18, 667–668. [Google Scholar] [CrossRef]
- Al-Adilah, H.; Peters, A.F.; Al-Bader, D.; Raab, A.; Akhdhar, A.; Feldmann, J.; Küpper, F.C. Iodine and fluorine concentrations in seaweeds of the Arabian Gulf identified by morphology and DNA barcodes. Bot. Mar. 2020, 63, 509–519. [Google Scholar] [CrossRef]
- Saenko, G.N.; Kravtsova, Y.Y.; Ivanenko, V.V.; Sheludko, S.I. Concentration of iodine and bromine by plants in the seas of Japan and Okhotsk. Mar. Biol. 1978, 47, 243–250. [Google Scholar] [CrossRef]
- Pavelka, S.; Vobecký, M.; Babicky, A. Halogen speciation in the rat thyroid: Simultaneous determination of bromine and iodine by short-term INAA. J. Radioanal. Nucl. Chem. Artic. 2008, 278, 575–579. [Google Scholar] [CrossRef]
- Romarís–Hortas, V.; García-Sartal, C.; Barciela-Alonso, M.D.C.; Domínguez-González, R.; Moreda-Piñeiro, A.; Bermejo-Barrera, P. Bioavailability study using an in-vitro method of iodine and bromine in edible seaweed. Food Chem. 2011, 124, 1747–1752. [Google Scholar] [CrossRef]
- Buzalaf, M.A.R.; Pessan, J.P.; Honorio, H.M.; Ten Cate, J.M. Mechanisms of action of fluoride for caries control. Monogr. Oral. Sci. 2011, 22, 97–114. [Google Scholar] [PubMed]
- Jenkins, G.N. The mechanism of action of fluoride in reducing caries incidence. Int. Dent. J. 1967, 17, 552. [Google Scholar]
- Voynar, A. Biological Role of Microelements in Human and Animal Organism; Vysshaya Shokola: Moscow, Russia, 1960. [Google Scholar]
- Krishnamachari, K.A. Skeletal fluorosis in humans: A review of recent progress in the understanding of the disease. Prog. Food Nutr. Sci. 1986, 10, 279–314. [Google Scholar] [PubMed]
- Li, S.; Smith, K.D.; Davis, J.H.; Gordon, P.B.; Breaker, R.R.; Strobel, S.A. Eukaryotic resistance to fluoride toxicity mediated by a widespread family of fluoride export proteins. Proc. Natl. Acad. Sci. USA 2013, 110, 19018–19023. [Google Scholar] [CrossRef]
- Yoo, J.I.; Seppälä, S.; O’Malley, M.A. Engineered fluoride sensitivity enables biocontainment and selection of genetically-modified yeasts. Nat. Commun. 2020, 11, 5459. [Google Scholar] [CrossRef]
- Wood, J.M.; Kennedy, F.S.; Wolfe, R.S. Reaction of multihalogenated hydrocarbons with free and bound reduced vitamin B12. Biochemistry 1968, 7, 1707–1713. [Google Scholar] [CrossRef]
- Carpenter, L.J.; Malin, G.; Liss, P.S.; Küpper, F.C. Novel biogenic iodine-containing trihalomethanes and other short-lived halocarbons in the coastal east Atlantic. Glob. Biogeochem. Cycles 2000, 14, 1191–1204. [Google Scholar] [CrossRef]
- Paul, N.A.; De Nys, R.; Steinberg, P. Chemical defence against bacteria in the red alga Asparagopsis armata: Linking structure with function. Mar. Ecol. Prog. Ser. 2006, 306, 87–101. [Google Scholar] [CrossRef]
- Küpper, F.C.; Leblanc, C.; Meyer-Klaucke, W.; Potin, P.; Feiters, M.C. Different speciation for bromine in brown and red algae, revealed by in vivo X-ray absorption spectroscopic studies. J. Phycol. 2014, 50, 652–664. [Google Scholar] [CrossRef]
- McAllister, T.A.; Cheng, K.-J.; Okine, E.K.; Mathison, G.W. Dietary, environmental and microbiological aspects of methane production in ruminants. Can. J. Anim. Sci. 1996, 76, 231–243. [Google Scholar] [CrossRef]
- IPCC. Climate Change 2007: The Physical Science Basis. In Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change; Solomon, S., Qin, D., Manning, M., Chen, Z., Marquis, M., Averyt, K.B., Tignor, M., Miller, H.L., Eds.; Cambridge University Press: Cambridge, UK, 2007; p. 1009. [Google Scholar]
- Roque, B.M.; Salwen, J.K.; Kinley, R.; Kebreab, E. Inclusion of Asparagopsis armata in lactating dairy cows’ diet reduces enteric methane emission by over 50 percent. J. Clean. Prod. 2019, 234, 132–138. [Google Scholar] [CrossRef]
- Roque, B.M.; Venegas, M.; Kinley, R.D.; de Nys, R.; Duarte, T.L.; Yang, X.; Kebreab, E. Red seaweed (Asparagopsis taxiformis) supplementation reduces enteric methane by over 80 percent in beef steers. PLoS ONE 2021, 16, e0247820. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Norman, H.C.; Kinley, R.D.; Laurence, M.; Wilmot, M.; Bender, H.; de Nys, R.; Tomkins, N. Asparagopsis taxiformis decreases enteric methane production from sheep. Anim. Prod. Sci. 2018, 58, 681–688. [Google Scholar] [CrossRef]
- Roque, B.M.; Brooke, C.G.; Ladau, J.; Polley, T.; Marsh, L.J.; Najafi, N.; Pandey, P.; Singh, L.; Kinley, R.; Salwen, J.K.; et al. Effect of the macroalgae Asparagopsis taxiformis on methane production and rumen microbiome assemblage. Anim. Microbiome 2019, 1, 3. [Google Scholar] [CrossRef]
- Machado, L.; Magnusson, M.; Paul, N.A.; de Nys, R.; Tomkins, N. Effects of Marine and Freshwater Macroalgae on In Vitro Total Gas and Methane Production. PLoS ONE 2014, 9, e85289. [Google Scholar] [CrossRef]
- Machado, L.; Magnusson, M.; Paul, N.; Kinley, R.D.; de Nys, R.; Tomkins, N. Dose-response effects of Asparagopsis taxiformis and Oedogonium sp. on in vitro fermentation and methane production. J. Appl. Phycol. 2015, 28, 1443–1452. [Google Scholar] [CrossRef]
- Machado, L.; Magnusson, M.; Paul, N.A.; Kinley, R.; De Nys, R.; Tomkins, N. Identification of bioactives from the red seaweed Asparagopsis taxiformis that promote antimethanogenic activity in vitro. J. Appl. Phycol. 2016, 28, 3117–3126. [Google Scholar] [CrossRef]
- Machado, L.; Tomkins, N.; Magnusson, M.; Midgley, D.J.; de Nys, R.; Rosewarne, C.P. In Vitro Response of Rumen Microbiota to the Antimethanogenic Red Macroalga Asparagopsis taxiformis. Microb. Ecol. 2017, 75, 811–818. [Google Scholar] [CrossRef]
- Zhu, P.; Li, D.; Yang, Q.; Su, P.; Wang, H.; Heimann, K.; Zhang, W. Commercial cultivation, industrial application, and potential halocarbon biosynthesis pathway of Asparagopsis sp. Algal Res. 2021, 56, 102319. [Google Scholar] [CrossRef]
- Küpper, F.C.; Schweigert, N.; Gall, E.A.; Legendre, J.-M.; Vilter, H.; Kloareg, B. Iodine uptake in Laminariales involves extracellular, haloperoxidase-mediated oxidation of iodide. Planta 1998, 207, 163–171. [Google Scholar] [CrossRef]
- Truesdale, V.W. The biogeochemical effect of seaweeds upon close-to natural concentrations of dissolved iodate and iodide in seawater—Preliminary study with Laminaria digitata and Fucus serratus. Estuarine, Coast. Shelf Sci. 2008, 78, 155–165. [Google Scholar] [CrossRef]
- Carrano, M.W.; Yarimizu, K.; Gonzales, J.L.; Cruz-López, R.; Edwards, M.S.; Tymon, T.M.; Küpper, F.C.; Carrano, C.J. The influence of marine algae on iodine speciation in the coastal ocean. ALGAE 2020, 35, 167–176. [Google Scholar] [CrossRef]
- Tymon, T.M.; Miller, E.P.; Gonzales, J.L.; Raab, A.; Küpper, F.C.; Carrano, C.J. Some aspects of the iodine metabolism of the giant kelp Macrocystis pyrifera (Phaeophyceae). J. Inorg. Biochem. 2017, 177, 82–88. [Google Scholar] [CrossRef]
- Chance, R.; Baker, A.R.; Küpper, F.C.; Hughes, C.; Kloareg, B.; Malin, G. Release and transformations of inorganic iodine by marine macroalgae. Estuar. Coast. Shelf Sci. 2009, 82, 406–414. [Google Scholar] [CrossRef]
- Gonzales, J.; Tymon, T.; Küpper, F.C.; Edwards, M.S.; Carrano, C.J. The potential role of kelp forests on iodine speciation in coastal seawater. PLoS ONE 2017, 12, e0180755. [Google Scholar]
- Carrano, M.W.; Carrano, C.J.; Edwards, M.S.; Al-Adilah, H.; Fontana, Y.; Sayer, M.D.; Katsaros, C.; Raab, A.; Feldmann, J.; Küpper, F.C. Laminaria kelps impact iodine speciation chemistry in coastal seawater. Estuar. Coast. Shelf Sci. 2021, 262, 107531. [Google Scholar] [CrossRef]
- Bartsch, I.; Wiencke, C.; Bischof, K.; Buchholz, C.; Buck, B.H.; Eggert, A.; Feuerpfeil, P.; Hanelt, D.; Jacobsen, S.; Karez, R.; et al. The genus Laminaria sensu lato: Recent insights and developments. Eur. J. Phycol. 2008, 43, 1–86. [Google Scholar] [CrossRef]
- Leblanc, C.; Colin, C.; Cosse, A.; Delage, L.; La Barre, S.; Morin, P.; Fiévet, B.; Voiseux, C.; Ambroise, Y.; Verhaeghe, E.; et al. Iodine transfers in the coastal marine environment: The key role of brown algae and of their vanadium-dependent haloperoxidases. Biochimie 2006, 88, 1773–1785. [Google Scholar] [CrossRef]
- Gall, E.A.; Küpper, F.C.; Kloareg, B. A survey of iodine content in Laminaria digitata. Bot. Mar. 2004, 47, 30–37. [Google Scholar] [CrossRef]
- Eschle Ueber den Jodgehalt einiger Algenarten. Z. Physiol. Chem. 1897, 23, 30–37. [CrossRef][Green Version]
- Golenkin, M. Algologische Notizen; 1. Das Vorkommen von freiem Iod bei Bonnemaisonia asparagoides. Bull. Société Impériale Nat. Moscou 1894, 8, 257–270. [Google Scholar]
- Sauvageau, C. Sur quelques algues floridées renfermant de l’iode à l’état libre. Bull. Stn. Biol. Arcachon 1925, 22, 3–43. [Google Scholar]
- Kylin, H. Über das Vorkommen von Jodiden, Bromiden und Jodidoxydasen bei Meeresalgen. Hoppe-Seyler’s Z. Physiol. Chem. 1929, 186, 50–84. [Google Scholar] [CrossRef]
- Dangeard, P. Sur le dégagement de l’iode chez les algues marines. Comptes Rendus Hebd. Séances Académie Sci. 1928, 186, 892–894. [Google Scholar]
- Tong, W.; Chaikoff, I.L. Metabolism of 131I by the marine alga, Nereocystis luetkeana. J. Biol. Chem. 1955, 215, 473–484. [Google Scholar] [CrossRef]
- Baily, N.A.; Kelly, S. Iodine Exchange in Ascophyllum. Biol. Bull. 1955, 109, 13–20. [Google Scholar] [CrossRef]
- Shaw, T. The mechanism of iodine accumulation by the brown sea weed Laminaria digitata. The uptake of 131I. Proc. Roy. Soc. Lond. B 1959, 150, 356–371. [Google Scholar]
- Shaw, T.I. The mechanism of iodine accumulation by the brown sea weed Laminaria digitata. II. Respiration and iodide uptake. Proc. Roy. Soc. Lond. B 1960, 152, 109–117. [Google Scholar]
- Huang, R.-J.; Thorenz, U.R.; Kundel, M.; Venables, D.S.; Ceburnis, D.; Ho, K.F.; Chen, J.; Vogel, A.L.; Küpper, F.C.; Smyth, P.P.A.; et al. The seaweeds Fucus vesiculosus and Ascophyllum nodosum are significant contributors to coastal iodine emissions. Atmospheric Chem. Phys. 2013, 13, 5255–5264. [Google Scholar] [CrossRef]
- Verhaeghe, E.F.; Fraysse, A.; Guerquin-Kern, J.-L.; Wu, T.-D.; Devès, G.; Mioskowski, C.; Leblanc, C.; Ortega, R.; Ambroise, Y.; Potin, P. Microchemical imaging of iodine distribution in the brown alga Laminaria digitata suggests a new mechanism for its accumulation. JBIC J. Biol. Inorg. Chem. 2007, 13, 257–269. [Google Scholar] [CrossRef] [PubMed]
- Kapanna, A.N.; Sitakara Rao, V. Iodine content of marine algae from Gujarat coast. J. Sci. Ind. Res. B Phys. Sci. 1962, 21, 559–560. [Google Scholar]
- Palmer, C.J.; Anders, T.L.; Carpenter, L.J.; Küpper, F.C.; McFiggans, G.B. Iodine and Halocarbon Response of Laminaria digitata to Oxidative Stress and Links to Atmospheric New Particle Production. Environ. Chem. 2005, 2, 282–290. [Google Scholar] [CrossRef]
- Küpper, F.C.; Carpenter, L.J.; Leblanc, C.; Toyama, C.; Uchida, Y.; Maskrey, B.H.; Robinson, J.; Verhaeghe, E.F.; Malin, G.; Luther, G.W., III; et al. In vivo Speciation studies and antioxidant properties of bromine in Laminaria digitata reinforce the significance of iodine accumulation for kelps. J. Exp. Bot. 2013, 64, 2653–2664. [Google Scholar] [CrossRef] [PubMed]
- Leri, A.; Mayer, L.M.; Thornton, K.R.; Northrup, P.; Dunigan, M.R.; Ness, K.J.; Gellis, A.B. A marine sink for chlorine in natural organic matter. Nat. Geosci. 2015, 8, 620–624. [Google Scholar] [CrossRef]
- Theiler, R.; Cook, J.C.; Hager, L.P.; Siuda, J.F. Halohydrocarbon Synthesis by Bromoperoxidase. Science 1978, 202, 1094–1096. [Google Scholar] [CrossRef]
- Beissner, R.S.; Guilford, W.J.; Coates, R.M.; Hager, L.P. Synthesis of brominated heptanones and bromoform by a bromoperoxidase of marine origin. Biochemistry 1981, 20, 3724–3731. [Google Scholar] [CrossRef]
- Leri, A.C.; Dunigan, M.R.; Wenrich, R.L.; Ravel, B. Particulate organohalogens in edible brown seaweeds. Food Chem. 2018, 272, 126–132. [Google Scholar] [CrossRef]
- Leri, A.; Marcus, M.A.; Myneni, S.C. X-ray spectromicroscopic investigation of natural organochlorine distribution in weathering plant material. Geochim. Cosmochim. Acta 2007, 71, 5834–5846. [Google Scholar] [CrossRef]
- Reina, R.G.; Leri, A.C.; Myneni, S.C.B. Cl K-edge X-ray Ssectroscopic investigation of enzymatic formation of organochlorines in weathering plant material. Environ. Sci. Technol. 2004, 38, 783–789. [Google Scholar] [CrossRef]
- Leri, A.C.; Hakala, A.; Marcus, M.A.; Lanzirotti, A.; Reddy, C.M.; Myneni, S.C.B. Natural organobromine in marine sediments: New evidence of biogeochemical Br cycling. Glob. Biogeochem. Cycles 2010, 24. [Google Scholar] [CrossRef]
- Küpper, F.C.; Müller, D.G.; Peters, A.F.; Kloareg, B.; Potin, P. Oligoalginate recognition and oxidative burst play a key role in natural and induced resistance of sporophytes of laminariales. J. Chem. Ecol. 2002, 28, 2057–2081. [Google Scholar] [CrossRef] [PubMed]
- Potin, P. Oxidative Burst and Related Responses in Biotic Interactions of Algae. In Algal Chemical Ecology; Springer: Berlin/Heidelberg, Germany, 2008; pp. 245–271. [Google Scholar]
- de Oliveira, L.S.; Tschoeke, D.A.; Magalhães Lopes, A.C.R.; Sudatti, D.B.; Meirelles, P.M.; Thompson, C.C.; Pereira, R.C.; Thompson, F.L. Molecular mechanisms for microbe recognition and defense by the red seaweed Laurencia dendroidea. Msphere 2017, 2, e00094-17. [Google Scholar] [CrossRef] [PubMed]
- Weinberger, F.; Hoppe, H.G.; Friedlander, M. Bacterial induction and inhibition of a fast necrotic response in Gracilaria conferta (Rhodophyta). J. Appl. Phycol. 1997, 9, 277–285. [Google Scholar] [CrossRef]
- Weinberger, F.; Friedlander, M.; Hoppe, H.-G. Oligoagars elicit a Physiological Response in Gracilaria conferta (Rhodophyta). J. Phycol. 1999, 35, 747–755. [Google Scholar] [CrossRef]
- Weinberger, F.; Friedlander, M. Endogenous and exogenous elicitors of a hypersensitive response in Gracilaria conferta (Rhodophyta). J. Appl. Phycol. 2000, 12, 139–145. [Google Scholar] [CrossRef]
- Weinberger, F.; Friedlander, M. Response of Gracilaria conferta (Rhodophyta) to oligoagars results in defense against agar-degrading epiphytes. J. Phycol. 2000, 36, 1079–1086. [Google Scholar] [CrossRef]
- Bouarab, K.; Potin, P.; Weinberger, F.; Correa, J.; Kloareg, B. The Chondrus crispus-Acrochaete operculata host-pathogen association, a novel model in glycobiology and applied phycopathology. J. Appl. Phycol. 2001, 13, 185–193. [Google Scholar] [CrossRef]
- Weinberger, F.; Richard, C.; Kloareg, B.; Kashman, Y.; Hoppe, H.-G.; Friedlander, M. Structure-activity relationships of oligoagar elicitors toward Gracilaria conferta (Rhodophyta). J. Phycol. 2001, 37, 418–426. [Google Scholar] [CrossRef]
- Weinberger, F.; Pohnert, G.; Kloareg, B.; Potin, P. A signal released by an enclophytic attacker acts as a substrate for a rapid defensive reaction of the red alga Chondrus crispus. Chembiochem 2002, 3, 1260–1263. [Google Scholar] [CrossRef]
- Weinberger, F.; Leonardi, P.; Miravalles, A.; Correa, J.A.; Lion, U.; Kloareg, B.; Potin, P. Dissection of two distinct defense-related responses to agar oligosaccharides in Gracilaria chilensis (Rhodophyta) and Gracilaria conferta (Rhodophyta). J. Phycol. 2005, 41, 863–873. [Google Scholar] [CrossRef]
- Küpper, F.C.; Kloareg, B.; Guern, J.; Potin, P. Oligoguluronates Elicit an Oxidative Burst in the Brown Algal Kelp Laminaria digitata. Plant Physiol. 2001, 125, 278–291. [Google Scholar] [CrossRef] [PubMed]
- Küpper, F.C.; Gaquerel, E.; Boneberg, E.-M.; Morath, S.; Salaün, J.-P.; Potin, P. Early events in the perception of lipopolysaccharides in the brown alga Laminaria digitata include an oxidative burst and activation of fatty acid oxidation cascades. J. Exp. Bot. 2006, 57, 1991–1999. [Google Scholar] [CrossRef] [PubMed]
- Zambounis, A.; Gaquerel, E.; Strittmatter, M.; Salaun, J.-P.; Potin, P.; Küpper, F.C. Prostaglandin A2 triggers a strong oxidative burst in Laminaria: A novel defense inducer in brown algae? ALGAE 2012, 27, 21–32. [Google Scholar] [CrossRef]
- Küpper, F.C.; Gaquerel, E.; Cosse, A.; Adas, F.; Peters, A.F.; Müller, D.G.; Kloareg, B.; Salaün, J.-P.; Potin, P. Free Fatty Acids and Methyl Jasmonate Trigger Defense Reactions in Laminaria digitata. Plant Cell Physiol. 2009, 50, 789–800. [Google Scholar] [CrossRef]
- Collén, J.; Ekdahl, A.; Abrahamsson, K.; Pedersén, M. The involvement of hydrogen peroxide in the production of volatile halogenated compounds by Meristiella gelidium. Phytochemistry 1994, 36, 1197–1202. [Google Scholar] [CrossRef]
- Pedersen, M.; Collen, J.; Abrahamsson, K.; Ekdahl, A. Production of halocarbons from seaweeds: An oxidative stress reaction? Sci. Mar. 1996, 60, 257–263. [Google Scholar]
- Weinberger, F.; Coquempot, B.; Forner, S.; Morin, P.; Kloareg, B.; Potin, P. Different regulation of haloperoxidation during agar oligosaccharide-activated defence mechanisms in two related red algae, Gracilaria sp. and Gracilaria chilensis. J. Exp. Bot. 2007, 58, 4365–4372. [Google Scholar] [CrossRef]
- Küpper, F.C.; Maier, I.; Müller, D.G.; Goer, S.L.D.; Guillou, L. Phylogenetic affinities of two eukaryotic pathogens of marine macroalgae, Eurychasma dicksonii (Wright) Magnus and Chytridium polysiphoniae Cohn. Cryptogam. Algol. 2006, 27, 165–184. [Google Scholar]
- Sekimoto, S.; Beakes, G.W.; Gachon, C.M.; Müller, D.G.; Küpper, F.C.; Honda, D. The Development, Ultrastructural Cytology, and Molecular Phylogeny of the Basal Oomycete Eurychasma dicksonii, Infecting the Filamentous Phaeophyte Algae Ectocarpus siliculosus and Pylaiella littoralis. Protist 2008, 159, 299–318. [Google Scholar] [CrossRef]
- Strittmatter, M.; Grenville-Briggs, L.J.; Breithut, L.; Van West, P.; Gachon, C.M.; Küpper, F.C. Infection of the brown alga Ectocarpus siliculosus by the oomycete Eurychasma dicksonii induces oxidative stress and halogen metabolism. Plant Cell Environ. 2016, 39, 259–271. [Google Scholar] [CrossRef] [PubMed]
- Wever, R.; Tromp, M.G.M.; Krenn, B.E.; Marjani, A.; Van Tol, M. Brominating activity of the seaweed Ascophyllum nodosum: Impact on the biosphere. Environ. Sci. Technol. 1991, 25, 446–449. [Google Scholar] [CrossRef]
- Scott, R. Observations on the Iodo-Amino-Acids of Marine Algae using Iodine-131. Nature 1954, 173, 1098–1099. [Google Scholar] [CrossRef]
- Klemperer, H.G. The accumulation of iodide by Fucus ceranoides. Biochem. J. 1957, 67, 381–390. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Manley, S.L. Micronutrient uptake and translocation by Macrocystis pyrifera (Phaeophyta). J. Phycol. 1984, 20, 192–201. [Google Scholar] [CrossRef]
- Manley, S.L.; Dastoor, M.N. Methyl halide (CH3X) production from the giant kelp, Macrocystis, and estimates of global CH3X production by kelp. Limnol. Oceanogr. 1987, 32, 709–715. [Google Scholar] [CrossRef]
- Manley, S.L.; Dastoor, M.N. Methyl iodide (CH3I) production by kelp and associated microbes. Mar. Biol. 1988, 98, 477–482. [Google Scholar] [CrossRef]
- Manley, S.L.; Goodwin, K.; North, W.J. Laboratory production of bromoform, methylene bromide, and methyl-iodide by macroalgae and distribution in nearshore Southern California waters. Limnol. Oceanogr. 1992, 37, 1652–1659. [Google Scholar] [CrossRef]
- Konotchick, T.; Dupont, C.L.; Valas, R.E.; Badger, J.H.; Allen, A.E. Transcriptomic analysis of metabolic function in the giant kelp, Macrocystis pyrifera, across depth and season. New Phytol. 2013, 198, 398–407. [Google Scholar] [CrossRef]
- Goodwin, K.D.; North, W.J.; Lidstrom, M.E. Production of bromoform and dibromomethane by Giant Kelp: Factors affecting release and comparison to anthropogenic bromine sources. Limnol. Oceanogr. 1997, 42, 1725–1734. [Google Scholar] [CrossRef]
- Stutz, J.; Jobson, B.T.; Sumner, A.L. Impact of Reactive Halogen Species on the Air Quality in California Coastal Areas; Final Report: CRC A-62-1/2 & ARB #05-307; CRC Press: Boca Raton, FL, USA, 2006. [Google Scholar]
- Finley, B.D.; Saltzman, E.S. Observations of Cl2, Br2, and I2 in coastal marine air. J. Geophys. Res. Atmos. 2008, 113. [Google Scholar] [CrossRef]
- Butler, A.; Sandy, M. Mechanistic considerations of halogenating enzymes. Nature 2009, 460, 848–854. [Google Scholar] [CrossRef]
- Graham, M.H.; Vasquez, J.A.; Buschmann, A.H. Global ecology of the giant kelp Macrocystis: From ecotypes to ecosystems. In Oceanography and Marine Biology; Gibson, R.N., Atkinson, R.J.A., Gordon, J.D.M., Eds.; Crc Press-Taylor & Francis Group: Boca Raton, FL, USA, 2007; Volume 45, pp. 39–88. [Google Scholar]
- Camargo, J.A. Fluoride toxicity to aquatic organisms: A review. Chemosphere 2002, 50, 251–264. [Google Scholar] [CrossRef]
- Tromp, M.; Van, T.T.; Wever, R. Reactivation of vanadium bromoperoxidase; inhibition by metallofluoric compounds. Biochim. Biophys. Acta (BBA) Protein Struct. Mol. Enzym. 1991, 1079, 53–56. [Google Scholar] [CrossRef]
- Ma, L.; Li, Y.; Meng, L.; Deng, H.; Li, Y.; Zhang, Q.; Diao, A. Biological fluorination from the sea: Discovery of a SAM-dependent nucleophilic fluorinating enzyme from the marine-derived bacterium Streptomyces xinghaiensis NRRL B24674. RSC Adv. 2016, 6, 27047–27051. [Google Scholar] [CrossRef]
- Huang, S.; Ma, L.; Tong, M.H.; Yu, Y.; O’Hagan, D.; Deng, H. Fluoroacetate biosynthesis from the marine-derived bacterium Streptomyces xinghaiensis NRRL B-24674. Org. Biomol. Chem. 2014, 12, 4828–4831. [Google Scholar] [CrossRef] [PubMed]
- Carter-Franklin, J.N.; Butler, A. Vanadium Bromoperoxidase-Catalyzed Biosynthesis of Halogenated Marine Natural Products. J. Am. Chem. Soc. 2004, 126, 15060–15066. [Google Scholar] [CrossRef]
- Young, E.G.; Langille, W.M. The occurrence of inorganic elements in marine algae of the Atlantic provinces of Canada. Can. J. Bot. 1958, 36, 301–310. [Google Scholar] [CrossRef]
- Mohamed, W.E.-D.; Hamad, M.T.M.H.; Kamel, M.Z. Application of statistical response surface methodology for optimization of fluoride removal efficiency by Padina sp. alga. Water Environ. Res. 2020, 92, 1080–1088. [Google Scholar] [CrossRef]
- Babu, A.N.; Reddy, D.S.; Kumar, G.S.; Ravindhranath, K.; Mohan, G.V.K. Sequential synergetic sorption analysis of Gracilaria Rhodophyta biochar toward aluminum and fluoride: A statistical optimization approach. Water Environ. Res. 2019, 92, 880–898. [Google Scholar] [CrossRef]
- Dembitsky, V.M.; Srebnik, M. Natural halogenated fatty acids: Their analogues and derivatives. Prog. Lipid Res. 2002, 41, 315–367. [Google Scholar] [CrossRef]
- Todd, J.S.; Proteau, P.J.; Gerwick, W.H. Egregiachlorides A-C: New chlorinated oxylipins from the marine brown alga Egregia menziesii. Tetrahedron Lett. 1993, 34, 7689–7692. [Google Scholar] [CrossRef]
- Kousaka, K.; Ogi, N.; Akazawa, Y.; Fujieda, M.; Yamamoto, Y.; Takada, Y.; Kimura, J. Novel Oxylipin Metabolites from the Brown Alga Eisenia bicyclis. J. Nat. Prod. 2003, 66, 1318–1323. [Google Scholar] [CrossRef] [PubMed]
- Jesus, A.; Correia-Da-Silva, M.; Afonso, C.; Pinto, M.; Cidade, H. Isolation and Potential Biological Applications of Haloaryl Secondary Metabolites from Macroalgae. Mar. Drugs 2019, 17, 73. [Google Scholar] [CrossRef]
- Harper, M.K. Introduction to the Chemical Ecology of Marine Natural Products. In Marine Chemical Ecology; CRC Press: Boca Raton, FL, USA, 2001. [Google Scholar]
- Dembitsky, V.M. Biogenic Iodine and Iodine-Containing Metabolites. Nat. Prod. Commun. 2006, 1, 1934578X0600100210. [Google Scholar] [CrossRef]
- Wang, B.-G.; Gloer, J.B.; Ji, N.-Y.; Zhao, J.-C. Halogenated Organic Molecules of Rhodomelaceae Origin: Chemistry and Biology. Chem. Rev. 2013, 113, 3632–3685. [Google Scholar] [CrossRef]
- Gribble, G.W. Naturally Occurring Organohalogen Compounds—A Survey. J. Nat. Prod. 1992, 55, 1353–1395. [Google Scholar] [CrossRef]
- Gribble, G.W. Natural Organohalogens: Many More Than You Think! J. Chem. Educ. 1994, 71, 907–911. [Google Scholar] [CrossRef]
- Gribble, G.W. The diversity of naturally produced organohalogens. Chemosphere 2003, 52, 289–297. [Google Scholar] [CrossRef]
- Gribble, G.W. Natural organohalogens: A new frontier for medicinal agents? J. Chem. Educ. 2004, 81, 1441–1449. [Google Scholar] [CrossRef]
- Gribble, G.W. A recent survey of naturally occurring organohalogen compounds. Environ. Chem. 2015, 12, 396–405. [Google Scholar] [CrossRef]
- Gribble, G.W. Biological Activity of Recently Discovered Halogenated Marine Natural Products. Mar. Drugs 2015, 13, 4044–4136. [Google Scholar] [CrossRef] [PubMed]
- Cardozo, K.H.M.; Guaratini, T.; Barros, M.; Falcão, V.R.; Tonon, A.P.; Lopes, N.; Campos, S.; Torres, M.; Souza, A.D.O.; Colepicolo, P.; et al. Metabolites from algae with economical impact. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2007, 146, 60–78. [Google Scholar] [CrossRef] [PubMed]
- Blunt, J.W.; Carroll, A.R.; Copp, B.R.; Davis, R.A.; Keyzers, R.A.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2009, 26, 170–244. [Google Scholar] [CrossRef] [PubMed]
- Agatsuma, Y.; Seki, T.; Kurata, K.; Taniguchi, K. Instantaneous effect of dibromomethane on metamorphosis of larvae of the sea urchins Strongylocentrotus nudus and Strongylocentrotus intermedius. Aquaculture 2006, 251, 549–557. [Google Scholar] [CrossRef]
- Wright, J.T.; De Nys, R.; Poore, A.G.B.; Steinberg, P.D. Chemical defense in a marine alga: Heritability and the potential for selection by herbivores. Ecology 2004, 85, 2946–2959. [Google Scholar] [CrossRef]
- Steinberg, P.D.; De Nys, R. Chemical mediation of colonisation of seaweed surfaces. J. Phycol. 2002, 38, 621–629. [Google Scholar] [CrossRef]
- Dworjanyn, S.A.; De Nys, R.; Steinberg, P.D. Chemically mediated antifouling in the red alga Delisea pulchra. Mar. Ecol. Prog. Ser. 2006, 318, 153–163. [Google Scholar] [CrossRef][Green Version]
- Lane, A.; Stout, E.P.; Lin, A.-S.; Prudhomme, J.; Le Roch, K.; Fairchild, C.R.; Franzblau, S.; Hay, M.; Aalbersberg, W.; Kubanek, J. Antimalarial Bromophycolides J−Q from the Fijian Red Alga Callophycus serratus. J. Org. Chem. 2009, 74, 2736–2742. [Google Scholar] [CrossRef]
- Lane, A.L.; Nyadong, L.; Galhena, A.S.; Shearer, T.L.; Stout, E.P.; Parry, R.M.; Kwasnik, M.; Wang, M.D.; Hay, M.E.; Fernandez, F.M.; et al. Desorption electrospray ionization mass spectrometry reveals surface-mediated antifungal chemical defense of a tropical seaweed. Proc. Natl. Acad. Sci. USA 2009, 106, 7314–7319. [Google Scholar] [CrossRef]
- Paradas, W.C.; Salgado, L.T.; Sudatti, D.B.; Crapez, M.A.; Fujii, M.T.; Coutinho, R.; Pereira, R.C.; Filho, G.M.A. Induction of halogenated vesicle transport in cells of the red seaweed Laurencia obtusa. Biofouling 2010, 26, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Dworjanyn, S.A.; De Nys, R.; Steinberg, P.D. Localisation and surface quantification of secondary metabolites in the red alga Delisea pulchra. Mar. Biol. 1999, 133, 727–736. [Google Scholar] [CrossRef]
- Young, D.N.; Howard, B.M.; Fenical, W. Subcellular localization of brominated secondary metabolites in the red alga Laurencia snyderae. J. Phycol. 1980, 16, 182–185. [Google Scholar] [CrossRef]
- Paul, N.A.; Cole, L.; De Nys, R.; Steinberg, P.D. Ultrastructure of the gland cells of the red alga Asparagopsis armata (Bonnemaisoniaceae). J. Phycol. 2006, 42, 637–645. [Google Scholar] [CrossRef]
- Murphy, C.; Moore, R.M.; White, R.L. Peroxidases from marine microalgae. J. Appl. Phycol. 2000, 12, 507–513. [Google Scholar] [CrossRef]
- Winter, J.; Moore, B.S. Exploring the Chemistry and Biology of Vanadium-dependent Haloperoxidases. J. Biol. Chem. 2009, 284, 18577–18581. [Google Scholar] [CrossRef]
- Wever, R.; Hemrika, W. Vanadium haloperoxidases. In Handbook of Metalloproteins; Messerschmidt, A., Huber, R., Poulos, T., Wieghardt, K., Eds.; John Wiley & Sons: Chichester, UK, 2001; pp. 1417–1428. [Google Scholar]
- Butler, A.; Walker, J.V. Marine haloperoxidases. Chem. Rev. 1993, 93, 1937–1944. [Google Scholar] [CrossRef]
- Wever, R. Structure and function of vanadium haloperoxidases. In Vanadium—Biochemical and Molecular Biological Approaches; Michibata, H., Ed.; Springer: Berlin/Heidelberg, Germany, 2012; pp. 95–126. [Google Scholar]
- Blasiak, L.C.; Drennan, C.L. Structural Perspective on Enzymatic Halogenation. Acc. Chem. Res. 2008, 42, 147–155. [Google Scholar] [CrossRef]
- van Pe, K.H.; Dong, C.; Flecks, S.; Naismith, J.; Patallo, E.P.; Wage, T. Biological halogenation has moved far beyond haloperoxidases. In Advances in Applied Microbiology; Elsevier Academic Press Inc.: San Diego, CA, USA, 2006; Volume 59, pp. 127–157. [Google Scholar]
- Vilter, H. Vanadium-dependent haloperoxidases. In Vanadium and Its Role in Life; Sigel, H., Sigel, A., Eds.; Marcel Dekker: New York, NY, USA, 1995; pp. 325–362. [Google Scholar]
- Butler, A. Mechanistic considerations of the vanadium haloperoxidases. Co-ord. Chem. Rev. 1999, 187, 17–35. [Google Scholar] [CrossRef]
- Butler, A. Vanadium haloperoxidases. Curr. Opin. Chem. Biol. 1998, 2, 279–285. [Google Scholar] [CrossRef]
- Carter-Franklin, J.N.; Parrish, J.D.; Tschirret-Guth, R.A.; Little, A.R.D.; Butler, A. Vanadium Haloperoxidase-Catalyzed Bromination and Cyclization of Terpenes. J. Am. Chem. Soc. 2003, 125, 3688–3689. [Google Scholar] [CrossRef] [PubMed]
- Butler, A.; Carter-Franklin, J.N. The role of vanadium bromoperoxidase in the biosynthesis of halogenated marine natural products. Nat. Prod. Rep. 2004, 21, 180–188. [Google Scholar] [CrossRef]
- Messerschmidt, A.; Wever, R. X-ray structure of a vanadium-containing enzyme: Chloroperoxidase from the fungus Curvularia inaequalis. Proc. Natl. Acad. Sci. USA 1996, 93, 392–396. [Google Scholar] [CrossRef] [PubMed]
- Colin, C.; Leblanc, C.; Wagner, E.; Delage, L.; Leize-Wagner, E.; Van Dorsselaer, A.; Kloareg, B.; Potin, P. The Brown Algal Kelp Laminaria digitata Features Distinct Bromoperoxidase and Iodoperoxidase Activities. J. Biol. Chem. 2003, 278, 23545–23552. [Google Scholar] [CrossRef]
- Vilter, H. Peroxidases from Phaeophyceae IV. Fractionation and Location of Peroxidase Isoenzymes in Ascophyllum nodosum (L.) Le Jol. Bot. Mar. 1983, 26, 451–455. [Google Scholar] [CrossRef]
- Vilter, H. Peroxidases from phaeophyceae: A vanadium(V)-dependent peroxidase from Ascophyllum nodosum. Phytochemistry 1984, 23, 1387–1390. [Google Scholar] [CrossRef]
- Weyand, M.; Hecht, H.-J.; Kieß, M.; Liaud, M.-F.; Vilter, H.; Schomburg, D. X-ray structure determination of a vanadium-dependent haloperoxidase from Ascophyllum nodosum at 2.0 Å resolution. J. Mol. Biol. 1999, 293, 595–611. [Google Scholar] [CrossRef]
- Feiters, M.C.; Leblanc, C.; Küpper, F.C.; Meyer-Klaucke, W.; Michel, G.; Potin, P. Bromine is an Endogenous Component of a Vanadium Bromoperoxidase. J. Am. Chem. Soc. 2005, 127, 15340–15341. [Google Scholar] [CrossRef]
- Itoh, N.; Izumi, Y.; Yamada, H. Characterization of nonheme type bromoperoxidase in Corallina pilulifera. J. Biol. Chem. 1986, 261, 5194–5200. [Google Scholar] [CrossRef]
- Itoh, N.; Izumi, Y.; Yamada, H. Purification of bromoperoxidase from Corallina pilulifera. Biochem. Biophys. Res. Commun. 1985, 131, 428–435. [Google Scholar] [CrossRef]
- Itoh, N.; Izumi, Y.; Yamada, H. Characterization of nonheme iron and reaction mechanism of bromoperoxidase in Corallina pilulifera. J. Biol. Chem. 1987, 262, 11982–11987. [Google Scholar] [CrossRef]
- Wever, R.; Plat, H.; de Boer, E. Isolation procedure and some properties of the bromoperoxidase from the seaweed Ascophyllum nodosum. Biochim. Biophys. Acta (BBA) Protein Struct. Mol. Enzym. 1985, 830, 181–186. [Google Scholar] [CrossRef]
- Sheffield, D.; Harry, T.; Smith, A.; Rogers, L. Purification and characterization of the vanadium bromoperoxidase from the macroalga Corallina officinalis. Phytochemistry 1992, 32, 21–26. [Google Scholar] [CrossRef]
- Shimonishi, M.; Kuwamoto, S.; Inoue, H.; Wever, R.; Ohshiro, T.; Izumi, Y.; Tanabe, T. Cloning and expression of the gene for a vanadium-dependent bromoperoxidase from a marine macro-alga, Corallina pilulifera. FEBS Lett. 1998, 428, 105–110. [Google Scholar] [CrossRef]
- Vreeland, V.; Ng, K.L.; Epstein, L. cDNA sequence and active recombinant vanadium bromoperoxidase from Fucus embryos. Mol. Biol. Cell 1998, 9, 180A. [Google Scholar]
- Ohshiro, T.; Nakano, S.; Takahashi, Y.; Suzuki, M.; Izumi, Y. Occurrence of bromoperoxidase in the marine green macro-alga, Ulvella lens, and emission of volatile brominated methane by the enzyme. Phytochemistry 1999, 52, 1211–1215. [Google Scholar] [CrossRef]
- Isupov, M.; Dalby, A.; Brindley, A.A.; Izumi, Y.; Tanabe, T.; Murshudov, G.N.; Littlechild, J.A. Crystal structure of dodecameric vanadium-dependent bromoperoxidase from the red algae Corallina officinalis. J. Mol. Biol. 2000, 299, 1035–1049. [Google Scholar] [CrossRef]
- Suthiphongchai, T.; Boonsiri, P.; Panijpan, B. Vanadium-dependent bromoperoxidases from Gracilaria algae. J. Appl. Phycol. 2007, 20, 271–278. [Google Scholar] [CrossRef]
- Manley, S.L.; Barbero, P.E. Physiological constraints on bromoform (CHBr3) production by Ulva lactuca (Chlorophyta). Limnol. Oceanogr. 2001, 46, 1392–1399. [Google Scholar] [CrossRef]
- Littlechild, J.; Rodriguez, E.G.; Isupov, M. Vanadium containing bromoperoxidase—Insights into the enzymatic mechanism using X-ray crystallography. J. Inorg. Biochem. 2009, 103, 617–621. [Google Scholar] [CrossRef]
- Fournier, J.-B.; Rebuffet, E.; Delage, L.; Grijol, R.; Meslet-Cladière, L.; Rzonca, J.; Potin, P.; Michel, G.; Czjzek, M.; Leblanc, C. The Vanadium Iodoperoxidase from the Marine Flavobacteriaceae Species Zobellia galactanivorans Reveals Novel Molecular and Evolutionary Features of Halide Specificity in the Vanadium Haloperoxidase Enzyme Family. Appl. Environ. Microbiol. 2014, 80, 7561–7573. [Google Scholar] [CrossRef] [PubMed]
- Gribble, G.W. Naturally occuring organohalogen compounds—A comprehensive survery. In Progress in the Chemistry of Organic Natural Products; Springer: Cham, Switzerland, 1996; pp. 1–423. [Google Scholar]
- Ballschmiter, K. Pattern and sources of naturally produced organohalogens in the marine environment: Biogenic formation of organohalogens. Chemosphere 2003, 52, 313–324. [Google Scholar] [CrossRef]
- Rasmussen, R.A.; Khalil, M.A.K.; Gunawardena, R.; Hoyt, S.D. Atmospheric methyl iodide (CH3I). J. Geophys. Res. Ocean. 1982, 87, 3086–3090. [Google Scholar] [CrossRef]
- Class, T.; Ballschmiter, K. Chemistry of organic traces in air. J. Atmos. Chem. 1988, 6, 35–46. [Google Scholar] [CrossRef]
- Itoh, N. Volatile halogenated compounds from marine algae; Their formation mechanisms and geochemical aspects. Recent Res. Devel. Phytochem. 1997, 1, 309–327. [Google Scholar]
- Butler, J.H. Better budgets for methyl halides? Nature 2000, 403, 260–261. [Google Scholar] [CrossRef]
- de Jong, E.; Field, J.A. Sulfur tuft and turkey tail: Biosynthesis and Biodegradation of Organohalogens by Basidiomycetes. Annu. Rev. Microbiol. 1997, 51, 375–414. [Google Scholar] [CrossRef]
- Rezanka, T.; Spižek, J. Griseofulvin and other biologically active halogen containing compounds from fungi. Elsevier 2005, 32, 471–547. [Google Scholar]
- Gribble, G.W. Recently Discovered Naturally Occurring Heterocyclic Organohalogen Compounds. Heterocycles 2012, 84, 157–207. [Google Scholar] [CrossRef]
- Lovelock, J.E. Natural halocarbons in the air and in the sea. Nature 1975, 256, 193–194. [Google Scholar] [CrossRef]
- Scarratt, M.; Moore, R. Production of methyl chloride and methyl bromide in laboratory cultures of marine phytoplankton. Mar. Chem. 1996, 54, 263–272. [Google Scholar] [CrossRef]
- Richter, U.; Wallace, D.W.R. Production of methyl iodide in the tropical Atlantic Ocean. Geophys. Res. Lett. 2004, 31. [Google Scholar] [CrossRef]
- Karlsson, A.; Auer, N.; Schulz-Bull, D.; Abrahamsson, K. Cyanobacterial blooms in the Baltic—A source of halocarbons. Mar. Chem. 2008, 110, 129–139. [Google Scholar] [CrossRef]
- Blunt, J.W.; Copp, B.R.; Munro, M.H.; Northcote, P.T.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2006, 23, 26–78. [Google Scholar] [CrossRef] [PubMed]
- Erickson, K.L. Constituents of Laurencia. Mar. Nat. Prod. Chem. Biol. Perspect. 1983, 5, 131–257. [Google Scholar]
- Roche, J.; Yagi, Y. Sur la fixation de l’iode radioactif par les algues et sur les constituants iodés des Laminaires. Comptes Rendus Société Biol. Paris 1952, 146, 642–645. [Google Scholar]
- Kelly, S.; Baily, N.A. The uptake of radioactive iodine by Ascophyllum. Biol. Bull. 1951, 100, 188–190. [Google Scholar] [CrossRef]
- Kelly, S. Respiration and iodine uptake in Ascophyllum. Biol. Bull. 1953, 104, 138–145. [Google Scholar] [CrossRef]
- Nightingale, P.; Malin, G.; Liss, P.S. Production of chloroform and other low molecular-weight halocarbons by some species of macroalgae. Limnol. Oceanogr. 1995, 40, 680–689. [Google Scholar] [CrossRef]
- Lim, Y.-K.; Phang, S.-M.; Rahman, N.A.; Sturges, W.T.; Malin, G. Halocarbon emissions from marine phytoplankton and climate change. Int. J. Environ. Sci. Technol. 2017, 14, 1355–1370. [Google Scholar] [CrossRef]
- Laturnus, F.; Giese, B.; Wiencke, C.; Adams, F.C. Low-molecular-weight organoiodine and organobromine compounds released by polar macroalgae—The influence of abiotic factors. Anal. Bioanal. Chem. 2000, 368, 297–302. [Google Scholar] [CrossRef] [PubMed]
- Berg, W.W.; Heidt, L.E.; Pollock, W.; Sperry, P.D.; Cicerone, R.J.; Gladney, E.S. Brominated organic species in the Arctic atmosphere. Geophys. Res. Lett. 1984, 11, 429–432. [Google Scholar] [CrossRef]
- Fogelqvist, E. Carbon tetrachloride, tetrachloroethylene, 1, 1, 1-trichloroethane and bromoform in Arctic seawater. J. Geophys. Res. Ocean. 1985, 90, 9181–9193. [Google Scholar] [CrossRef]
- Moore, R.M.; Zafiriou, O.C. Photochemical production of methyl iodide in seawater. J. Geophys. Res. Earth Surf. 1994, 99, 16415–16420. [Google Scholar] [CrossRef]
- Brownell, D.K.; Moore, R.M.; Cullen, J.J. Production of methyl halides by Prochlorococcus and Synechococcus. Glob. Biogeochem. Cycles 2010, 24. [Google Scholar] [CrossRef]
- Williams, J.; Gros, V.; Atlas, E.; Maciejczyk, K.; Batsaikhan, A.; Schöler, H.F.; Forster, C.; Quack, B.; Yassaa, N.; Sander, R.; et al. Possible evidence for a connection between methyl iodide emissions and Saharan dust. J. Geophys. Res. Earth Surf. 2007, 112. [Google Scholar] [CrossRef]
- Sturges, W.T.; Cota, G.F.; Buckley, P.T. Vertical profiles of bromoform in snow, sea ice, and seawater in the Canadian Arctic. J. Geophys. Res. Ocean. 1997, 102, 25073–25083. [Google Scholar] [CrossRef]
- Keng, F.S.-L.; Phang, S.-M.; Rahman, N.A.; Leedham, E.C.; Hughes, C.; Robinson, A.D.; Harris, N.R.P.; Pyle, J.A.; Sturges, W.T. Volatile halocarbon emissions by three tropical brown seaweeds under different irradiances. J. Appl. Phycol. 2013, 25, 1377–1386. [Google Scholar] [CrossRef]
- Mtolera, M.S.; Collén, J.; Pedersén, M.; Ekdahl, A.; Abrahamsson, K.; Semesi, A.K. Stress-induced production of volatile halogenated organic compounds in Eucheuma denticulatum (Rhodophyta) caused by elevated pH and high light intensities. Eur. J. Phycol. 1996, 31, 89–95. [Google Scholar] [CrossRef]
- Collén, J.; Porcel, B.; Carré, W.; Ball, S.G.; Chaparro, C.; Tonon, T.; Barbeyron, T.; Michel, G.; Noel, B.; Valentin, K.; et al. Genome structure and metabolic features in the red seaweed Chondrus crispus shed light on evolution of the Archaeplastida. Proc. Natl. Acad. Sci. USA 2013, 110, 5247–5252. [Google Scholar] [CrossRef]
- Berg, W.W.; Sperry, P.D.; Rahn, K.A.; Gladney, E.S. Atmospheric bromine in the Arctic. J. Geophys. Res. Ocean. 1983, 88, 6719–6736. [Google Scholar] [CrossRef]
- Fan, S.-M.; Jacob, D.J. Surface ozone depletion in Arctic spring sustained by bromine reactions on aerosols. Nature 1992, 359, 522. [Google Scholar] [CrossRef]
- Scarratt, M.G.; Moore, R.M. Production of chlorinated hydrocarbons and methyl iodide by the red microalga Porphyridium purpureum. Limnol. Oceanogr. 1999, 44, 703–707. [Google Scholar] [CrossRef]
- Hughes, C.; Malin, G.; Nightingale, P.; Liss, P.S. The effect of light stress on the release of volatile iodocarbons by three species of marine microalgae. Limnol. Oceanogr. 2006, 51, 2849–2854. [Google Scholar] [CrossRef]
- Hughes, C.; Sun, S. Light and brominating activity in two species of marine diatom. Mar. Chem. 2016, 181, 1–9. [Google Scholar] [CrossRef]
- Mithoo-Singh, P.K.; Keng, F.S.-L.; Phang, S.-M.; Elvidge, E.C.L.; Sturges, W.T.; Malin, G.; Rahman, N.A. Halocarbon emissions by selected tropical seaweeds: Species-specific and compound-specific responses under changing pH. PeerJ 2017, 5, e2918. [Google Scholar] [CrossRef] [PubMed]
- Abrahamsson, K.; Choo, K.-S.; Pedersén, M.; Johansson, G.; Snoeijs, P. Effects of temperature on the production of hydrogen peroxide and volatile halocarbons by brackish-water algae. Phytochemistry 2003, 64, 725–734. [Google Scholar] [CrossRef]
- Mehrtens, G.; Laturnus, F. Halogenating activity in an Arctic population of brown macroalga Laminaria saccharina (L.) Lamour. Polar Res. 1997, 16, 19–26. [Google Scholar]
- Laturnus, F.; Adams, F.C.; Wiencke, C. Methyl halides from Antarctic macroalgae. Geophys. Res. Lett. 1998, 25, 773–776. [Google Scholar] [CrossRef]
- Atkinson, H.M.; Huang, R.-J.; Chance, R.; Roscoe, H.K.; Hughes, C.; Davison, B.; Schönhardt, A.; Mahajan, A.S.; Saiz-Lopez, A.; Hoffmann, T.; et al. Iodine emissions from the sea ice of the Weddell Sea. Atmos. Chem. Phys. 2012, 12, 11229–11244. [Google Scholar] [CrossRef]
- O’Dowd, C.D.; Jimenez, J.L.; Bahreini, R.; Flagan, R.C.; Seinfeld, J.H.; Hämeri, K.; Pirjola, L.; Kulmala, M.; Jennings, S.G.; Hoffmann, T. Marine aerosol formation from biogenic iodine emissions. Nature 2002, 417, 632–636. [Google Scholar] [CrossRef]
- Kerkweg, A.; Jöckel, P.; Warwick, N.; Gebhardt, S.; Brenninkmeijer, C.A.M.; Lelieveld, J. Consistent simulation of bromine chemistry from the marine boundary layer to the stratosphere—Part 2: Bromocarbons. Atmos. Chem. Phys. 2008, 8, 5919–5939. [Google Scholar] [CrossRef]
- Liang, Q.; Stolarski, R.S.; Kawa, S.R.; Nielsen, J.E.; Douglass, A.R.; Rodriguez, J.M.; Blake, D.R.; Atlas, E.L.; Ott, L.E. Finding the missing stratospheric Br-y: A global modeling study of CHBr3 and CH2Br2. Atmos. Chem. Phys. 2010, 10, 2269–2286. [Google Scholar] [CrossRef]
- Ordóñez, C.; Lamarque, J.-F.; Tilmes, S.; Kinnison, D.E.; Atlas, E.L.; Blake, D.R.; Santos, G.S.; Brasseur, G.; Saiz-Lopez, A. Bromine and iodine chemistry in a global chemistry-climate model: Description and evaluation of very short-lived oceanic sources. Atmos. Chem. Phys. 2012, 12, 1423–1447. [Google Scholar] [CrossRef]
- Warwick, N.J.; Pyle, J.A.; Carver, G.D.; Yang, X.; Savage, N.H.; O’Connor, F.M.; Cox, R.A. Global modeling of biogenic bromocarbons. J. Geophys. Res. -Atmos. 2006, 111, D24305. [Google Scholar] [CrossRef]
- Butler, J.H.; King, D.B.; Lobert, J.M.; Montzka, S.; Yvon-Lewis, S.; Hall, B.D.; Warwick, N.; Mondeel, D.J.; Aydin, M.; Elkins, J.W. Oceanic distributions and emissions of short-lived halocarbons. Glob. Biogeochem. Cycles 2007, 21. [Google Scholar] [CrossRef]
- Carpenter, L.J.; Liss, P.S. On temperate sources of bromoform and other reactive organic bromine gases. J. Geophys. Res. Earth Surf. 2000, 105, 20539–20547. [Google Scholar] [CrossRef]
- Quack, B.; Wallace, D.W.R. Air-sea flux of bromoform: Controls, rates, and implications. Glob. Biogeochem. Cycles 2003, 17. [Google Scholar] [CrossRef]
- Yokouchi, Y.; Hasebe, F.; Fujiwara, M.; Takashima, H.; Shiotani, M.; Nishi, N.; Kanaya, Y.; Hashimoto, S.; Fraser, P.; Toom-Sauntry, D.; et al. Correlations and emission ratios among bromoform, dibromochloromethane, and dibromomethane in the atmosphere. J. Geophys. Res. Earth Surf. 2005, 110. [Google Scholar] [CrossRef]
- Carpenter, L.J. Iodine in the Marine Boundary Layer. Chem. Rev. 2003, 103, 4953–4962. [Google Scholar] [CrossRef]
- Yokouchi, Y.; Osada, K.; Wada, M.; Hasebe, F.; Agama, M.; Murakami, R.; Mukai, H.; Nojiri, Y.; Inuzuka, Y.; Toom-Sauntry, D.; et al. Global distribution and seasonal concentration change of methyl iodide in the atmosphere. J. Geophys. Res. Earth Surf. 2008, 113, D18311. [Google Scholar] [CrossRef]
- Bell, N.; Hsu, L.; Jacob, D.J.; Schultz, M.; Blake, D.R.; Butler, J.H.; King, D.B.; Lobert, J.M.; Maier-Reimer, E. Methyl iodide: Atmospheric budget and use as a tracer of marine convection in global models. J. Geophys. Res. Earth Surf. 2002, 107, e4340. [Google Scholar] [CrossRef]
- Menard, H.W.; Smith, S.M. Hypsometry of ocean basin provinces. J. Geophys. Res. Earth Surf. 1966, 71, 4305–4325. [Google Scholar] [CrossRef]
- Ziska, F.; Quack, B.; Abrahamsson, K.; Archer, S.D.; Atlas, E.; Bell, T.; Butler, J.H.; Carpenter, L.J.; Jones, C.E.; Harris, N.R.P.; et al. Global sea-to-air flux climatology estimates of bromoform, dibromomethane and methyl iodide. Atmos. Chem. Phys. 2013, 13, 8915–8934. [Google Scholar] [CrossRef]
- McFiggans, G.; Coe, H.; Burgess, R.; Allan, J.; Cubison, M.; Alfarra, M.R.; Saunders, R.; Saiz-Lopez, A.; Plane, J.M.C.; Wevill, D.; et al. Direct evidence for coastal iodine particles from Laminaria macroalgae—Linkage to emissions of molecular iodine. Atmospheric Chem. Phys. 2004, 4, 701–713. [Google Scholar] [CrossRef]
- Saiz-Lopez, A.; Plane, J.M.C.; McFiggans, G.; Williams, P.I.; Ball, S.M.; Bitter, M.; Jones, R.L.; Hongwei, C.; Hoffmann, T. Modelling molecular iodine emissions in a coastal marine environment: The link to new particle formation. Atmos. Chem. Phys. 2006, 6, 883–895. [Google Scholar] [CrossRef]
- Hou, X.; Dahlgaard, H.; Nielsen, S. Iodine-129 Time Series in Danish, Norwegian and Northwest Greenland Coast and the Baltic Sea by Seaweed. Estuar. Coast. Shelf Sci. 2000, 51, 571–584. [Google Scholar] [CrossRef]
- Hou, X.; Hansen, V.; Aldahan, A.; Possnert, G.; Lind, O.C.; Lujaniene, G. A review on speciation of iodine-129 in the environmental and biological samples. Anal. Chim. Acta 2009, 632, 181–196. [Google Scholar] [CrossRef]
- Morita, T.; Niwa, K.; Fujimoto, K.; Kasai, H.; Yamada, H.; Nishiutch, K.; Sakamoto, T.; Godo, W.; Taino, S.; Hayashi, Y.; et al. Detection and activity of iodine-131 in brown algae collected in the Japanese coastal areas. Sci. Total Environ. 2010, 408, 3443–3447. [Google Scholar] [CrossRef]
- Manley, S.L.; Lowe, C.G. Canopy-Forming Kelps as California’s Coastal Dosimeter: 131I from Damaged Japanese Reactor Measured in Macrocystis pyrifera. Environ. Sci. Technol. 2012, 46, 3731–3736. [Google Scholar] [CrossRef]
- Lebeau, D.; Leroy, N.; Doizi, D.; Wu, T.-D.; Guerquin-Kern, J.-L.; Perrin, L.; Ortega, R.; Voiseux, C.; Fournier, J.-B.; Potin, P.; et al. Mass spectrometry—Based imaging techniques for iodine-127 and iodine-129 detection and localization in the brown alga Laminaria digitata. J. Environ. Radioact. 2021, 231, 106552. [Google Scholar] [CrossRef]
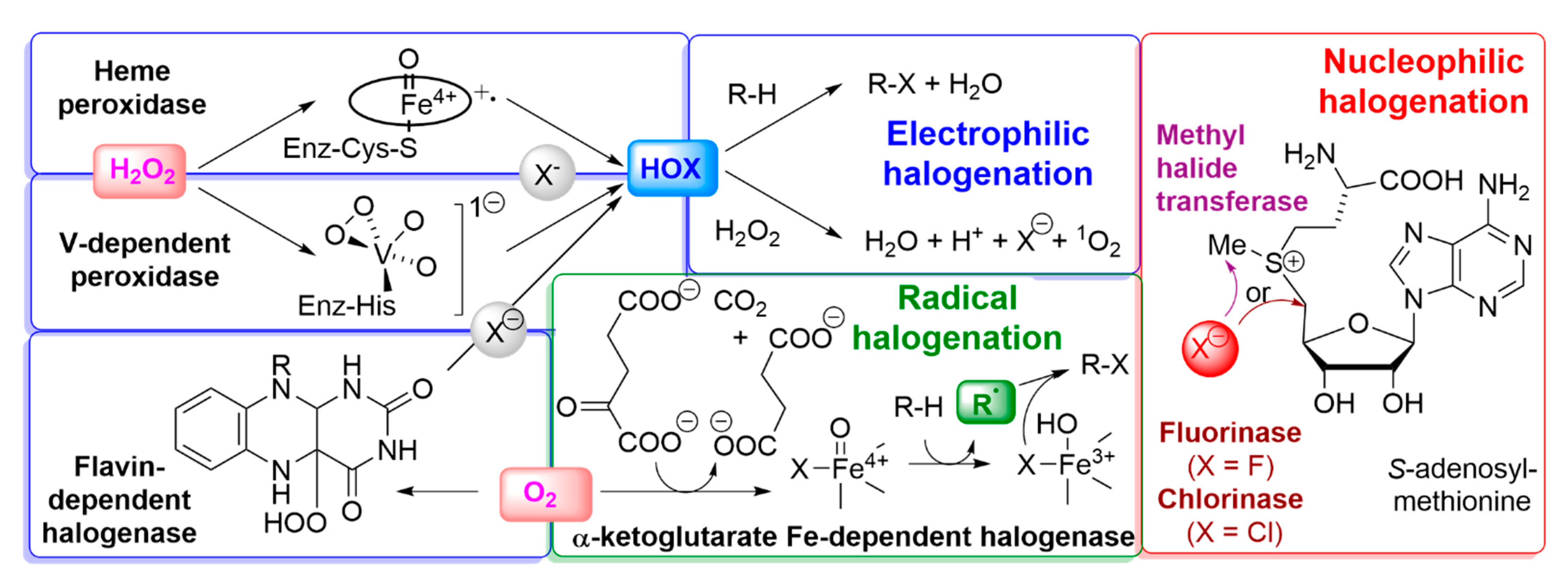
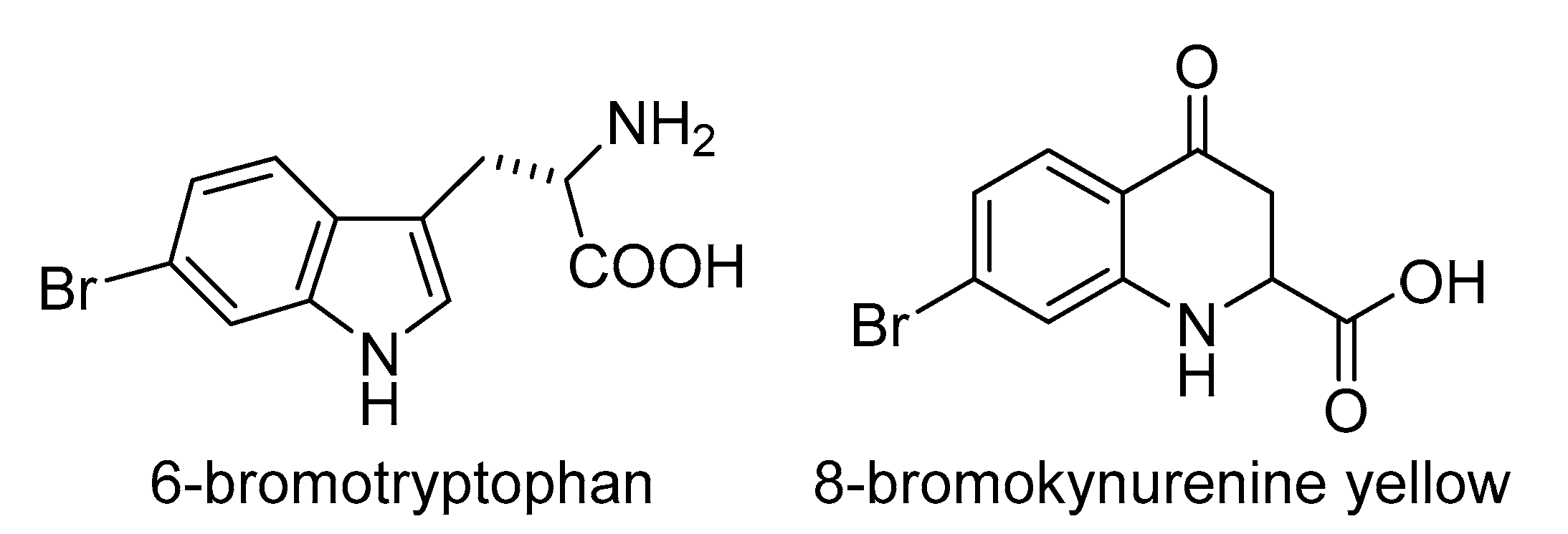

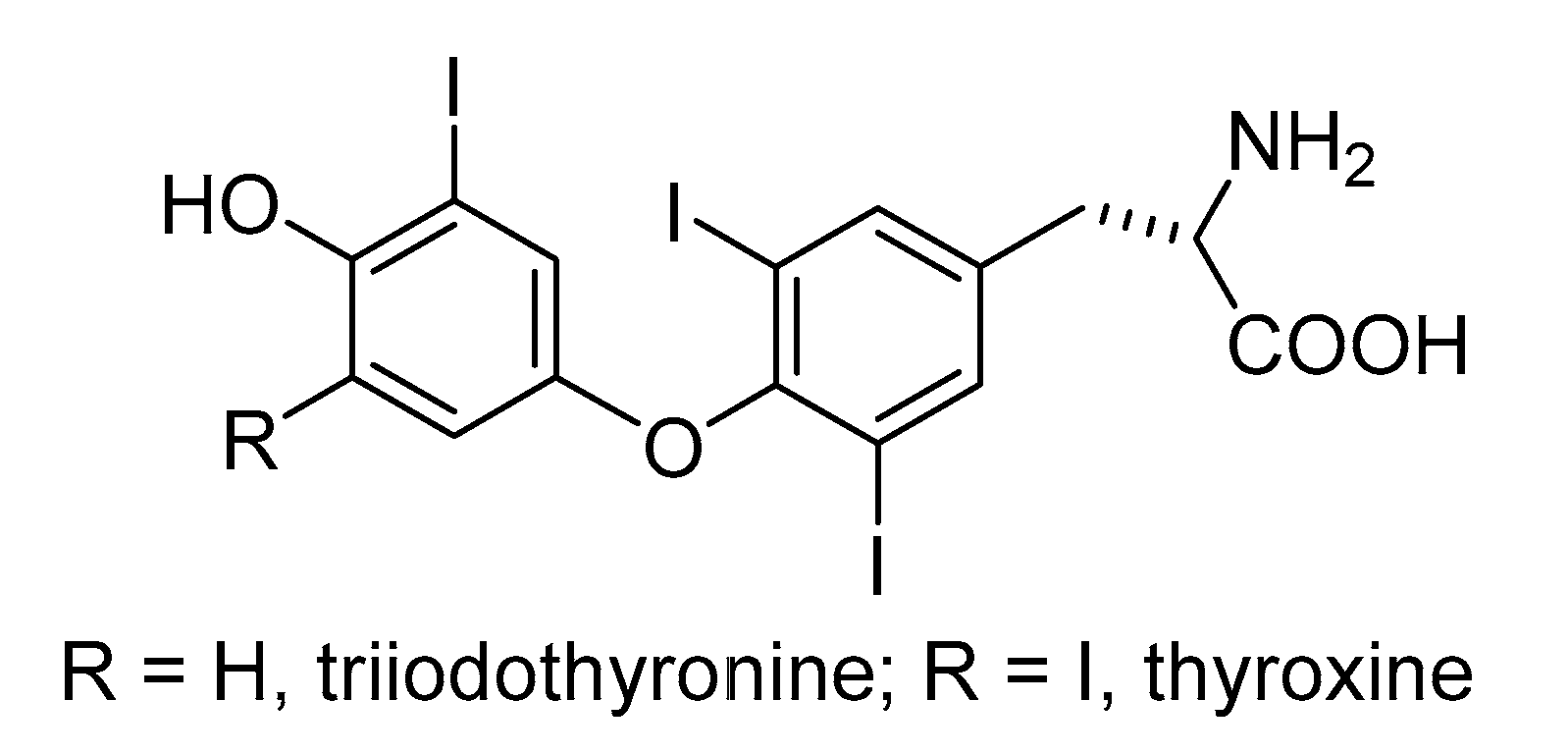

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Adilah, H.; Feiters, M.C.; Carpenter, L.J.; Kumari, P.; Carrano, C.J.; Al-Bader, D.; Küpper, F.C. Halogens in Seaweeds: Biological and Environmental Significance. Phycology 2022, 2, 132-171. https://doi.org/10.3390/phycology2010009
Al-Adilah H, Feiters MC, Carpenter LJ, Kumari P, Carrano CJ, Al-Bader D, Küpper FC. Halogens in Seaweeds: Biological and Environmental Significance. Phycology. 2022; 2(1):132-171. https://doi.org/10.3390/phycology2010009
Chicago/Turabian StyleAl-Adilah, Hanan, Martin C. Feiters, Lucy J. Carpenter, Puja Kumari, Carl J. Carrano, Dhia Al-Bader, and Frithjof C. Küpper. 2022. "Halogens in Seaweeds: Biological and Environmental Significance" Phycology 2, no. 1: 132-171. https://doi.org/10.3390/phycology2010009
APA StyleAl-Adilah, H., Feiters, M. C., Carpenter, L. J., Kumari, P., Carrano, C. J., Al-Bader, D., & Küpper, F. C. (2022). Halogens in Seaweeds: Biological and Environmental Significance. Phycology, 2(1), 132-171. https://doi.org/10.3390/phycology2010009