Carbon-Source-Dependent Toxicity of Carbon Dots: An Environmental Evaluation Using Brine shrimp
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Production of C-Dots
2.3. Characterization of C-Dots
2.4. Toxicity Test
2.5. Statistical Analysis
3. Results and Discussion
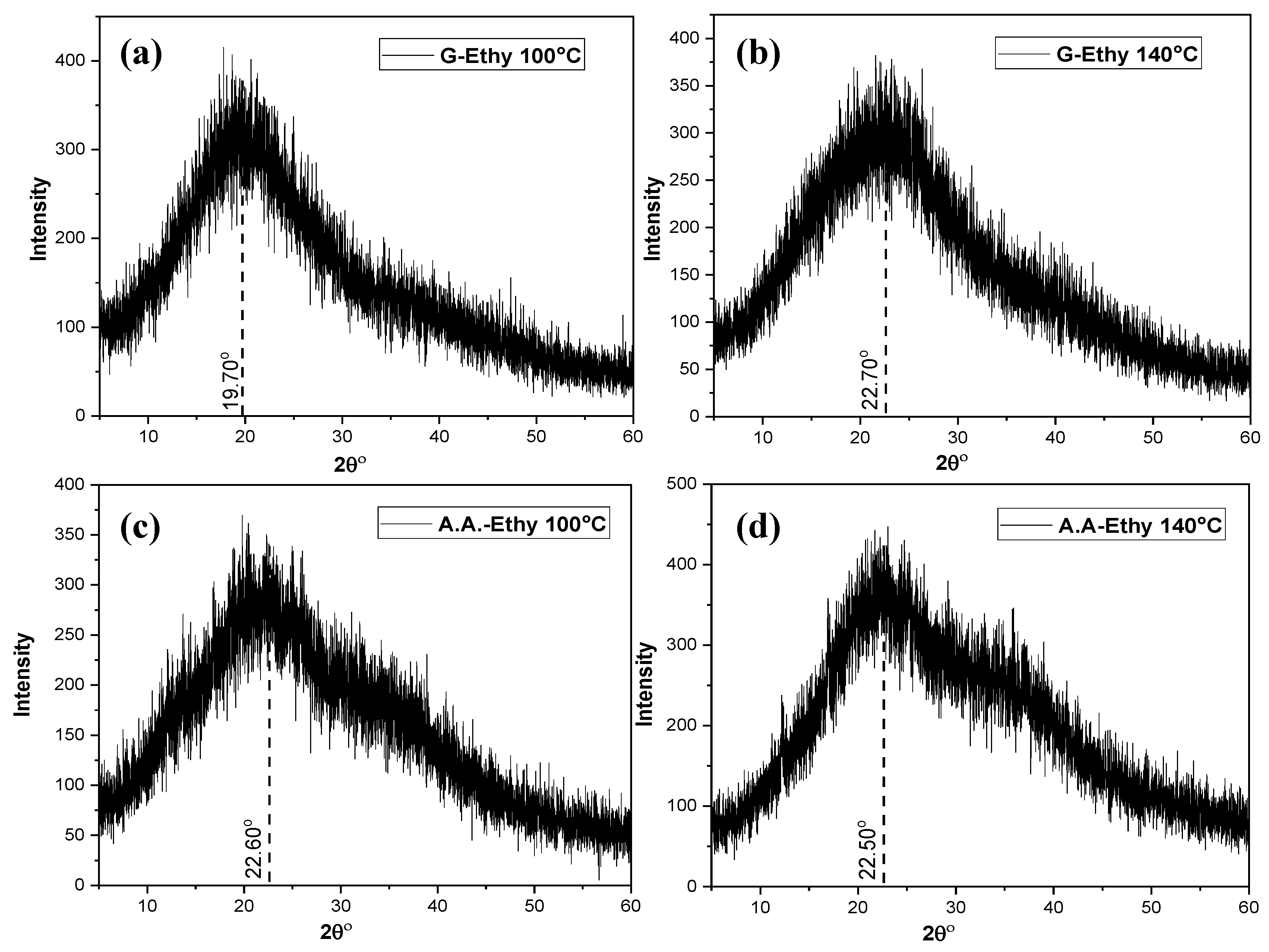
3.1. Morphological, Compositional, and Optical Characterization
3.2. Toxicity Test in Brine shrimp
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Elugoke, S.E.; Uwaya, G.E.; Quadri, T.W.; Ebenso, E.E. Carbon Quantum Dots: Basics, Properties, and Fundamentals. In ACS Symposium Series; Berdimurodov, E., Verma, D.K., Guo, L., Eds.; American Chemical Society: Washington, DC, USA, 2024; Volume 1465, pp. 3–42. ISBN 978-0-8412-9699-2. [Google Scholar]
- Abid, N.; Khan, A.M.; Shujait, S.; Chaudhary, K.; Ikram, M.; Imran, M.; Haider, J.; Khan, M.; Khan, Q.; Maqbool, M. Synthesis of Nanomaterials Using Various Top-down and Bottom-up Approaches, Influencing Factors, Advantages, and Disadvantages: A Review. Adv. Colloid Interface Sci. 2022, 300, 102597. [Google Scholar] [CrossRef]
- Sun, Y.-P. Carbon Dots: Exploring Carbon at Zero-Dimension; Springer International Publishing: Cham, Switzerland, 2020; ISBN 978-3-030-41183-1. [Google Scholar]
- De Medeiros, T.V.; Manioudakis, J.; Noun, F.; Macairan, J.-R.; Victoria, F.; Naccache, R. Microwave-Assisted Synthesis of Carbon Dots and Their Applications. J. Mater. Chem. C 2019, 7, 7175–7195. [Google Scholar] [CrossRef]
- Deng, J.; Hu, J.; Zhao, J.; An, N.; Liang, K.; Wang, Q.; Zhang, Z.; Wu, R.; Zhang, F. Eco Friendly Synthesis of Fluorescent Carbon Dots for the Sensitive Detection of Ferric Ions and Cell Imaging. Arab. J. Chem. 2021, 14, 103195. [Google Scholar] [CrossRef]
- Maruthupandi, M.; Varatharajan, P.; Shameem Banu, I.B.; Hafiz Mamat, M.; Vasimalai, N. White Light Emitting Diode and Anti-Counterfeiting Applications of Microwave Assisted Synthesized Green Fluorescent Carbon Dots Derived from Waste Curry Leaves. Results Opt. 2022, 8, 100249. [Google Scholar] [CrossRef]
- Balakrishnan, T.; Ang, W.L.; Mahmoudi, E.; Mohammad, A.W.; Sambudi, N.S. Formation Mechanism and Application Potential of Carbon Dots Synthesized from Palm Kernel Shell via Microwave Assisted Method. Carbon Resour. Convers. 2022, 5, 150–166. [Google Scholar] [CrossRef]
- Kumar, P.; Dua, S.; Kaur, R.; Kumar, M.; Bhatt, G. A Review on Advancements in Carbon Quantum Dots and Their Application in Photovoltaics. RSC Adv. 2022, 12, 4714–4759. [Google Scholar] [CrossRef]
- Manzoor, S.; Dar, A.H.; Dash, K.K.; Pandey, V.K.; Srivastava, S.; Bashir, I.; Khan, S.A. Carbon Dots Applications for Development of Sustainable Technologies for Food Safety: A Comprehensive Review. Appl. Food Res. 2023, 3, 100263. [Google Scholar] [CrossRef]
- Redondo-Fernandez, G.; Cigales Canga, J.; Soldado, A.; Ruiz Encinar, J.; Costa-Fernandez, J.M. Functionalized Heteroatom-Doped Carbon Dots for Biomedical Applications: A Review. Anal. Chim. Acta 2023, 1284, 341874. [Google Scholar] [CrossRef]
- Tukhliyivich, B.E.; Verma, D.K. (Eds.) Carbon Dots in Biology: Synthesis, Properties, Biological and Pharmaceutical Applications, 1st ed.; De Gruyter Stem; De Gruyter: Boston, MA, USA, 2023; ISBN 978-3-11-079992-7. [Google Scholar]
- Singh, R.P.; Khan, R.; Singh, J.; Singh, K.R. (Eds.) Carbon Dots: Next-Generation Materials for Biomedical Applications; IOP Publishing: Bristol, UK, 2022; ISBN 978-0-7503-4641-2. [Google Scholar]
- Sengar, P.; Chauhan, K.; Hirata, G.A. Progress on Carbon Dots and Hydroxyapatite Based Biocompatible Luminescent Nanomaterials for Cancer Theranostics. Transl. Oncol. 2022, 24, 101482. [Google Scholar] [CrossRef] [PubMed]
- Costa, R.S.; De Castro, M.O.; Da Silva, G.H.; Delite, F.D.S.; Strauss, M.; Ferreira, O.P.; Martinez, D.S.T.; Viana, B.C. Carbon-Dots from Babassu Coconut (Orbignya Speciosa) Biomass: Synthesis, Characterization, and Toxicity to Daphnia Magna. Carbon Trends 2021, 5, 100133. [Google Scholar] [CrossRef]
- Liu, Y.-Y.; Li, J.-M.; Ji, R.; Zhang, H.; Zhang, W.; Miao, A.-J. Bioaccumulation Determines the Toxicity of Carbon Dots to Two Marine Dinoflagellates. Chemosphere 2023, 321, 138155. [Google Scholar] [CrossRef]
- Tiple, A.D.; Badwaik, V.J.; Padwad, S.V.; Chaudhary, R.G.; Singh, N.B. A Review on Nanotoxicology: Aquatic Environment and Biological System. Mater. Today Proc. 2020, 29, 1246–1250. [Google Scholar] [CrossRef]
- Yan, J.; Hou, S.; Yu, Y.; Qiao, Y.; Xiao, T.; Mei, Y.; Zhang, Z.; Wang, B.; Huang, C.-C.; Lin, C.-H.; et al. The Effect of Surface Charge on the Cytotoxicity and Uptake of Carbon Quantum Dots in Human Umbilical Cord Derived Mesenchymal Stem Cells. Colloids Surf. B Biointerfaces 2018, 171, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Namdee, K.; Khongkow, M.; Boonthod, S.; Boonrungsiman, S.; Jarussophon, S.; Pongwan, P.; Yata, T.; Saengkrit, N. Cell-Based Assay for Characterizing Cell Adhesion Properties of Active Targeted Nanoparticles under Static and Flow Condition Using an Integrated Flow Chamber. J. Drug Deliv. Sci. Technol. 2018, 45, 296–302. [Google Scholar] [CrossRef]
- Yamindago, A.; Lee, N.; Yum, S.; Woo, S. A Simple and Rapid Method for Toxicity Evaluation of Zinc Oxide Nanoparticle (ZnO NPs) in Benthic Animal Hydra Magnipapillata. MethodsX 2019, 6, 150–155. [Google Scholar] [CrossRef]
- Rajabi, S.; Ramazani, A.; Hamidi, M.; Naji, T. Artemia Salina as a Model Organism in Toxicity Assessment of Nanoparticles. DARU J. Pharm. Sci. 2015, 23, 20. [Google Scholar] [CrossRef] [PubMed]
- Hong, D.; Ma, Y.; Song, S.; Li, N.; Wang, J. Design, Synthesis, Brine shrimp Lethality and Cytotoxicity of Some Novel 17a-Aza-D-Homo-Androster-17-One Derivatives. Nat. Prod. Res. 2021, 35, 3985–3991. [Google Scholar] [CrossRef]
- Banti, C.; Hadjikakou, S. Evaluation of Toxicity with Brine shrimp Assay. Bio-Protocol 2021, 11, e3895. [Google Scholar] [CrossRef]
- Das Pramanik, D.; Lei, S.; Kay, P.; Goycoolea, F.M. Investigating on the Toxicity and Bio-Magnification Potential of Synthetic Glitters on Artemia Salina. Mar. Pollut. Bull. 2023, 190, 114828. [Google Scholar] [CrossRef] [PubMed]
- Arulvasu, C.; Jennifer, S.M.; Prabhu, D.; Chandhirasekar, D. Toxicity Effect of Silver Nanoparticles in Brine shrimp Artemia. Sci. World J. 2014, 2014, 256919. [Google Scholar] [CrossRef]
- Soledad-Flores, O.; Bailón-Ruiz, S.J.; Román-Velázquez, F. Rapid Synthesis of Non-Toxic, Water-Stable Carbon Dots Using Microwave Irradiation. Micro 2024, 4, 659–669. [Google Scholar] [CrossRef]
- Mintz, K.J.; Bartoli, M.; Rovere, M.; Zhou, Y.; Hettiarachchi, S.D.; Paudyal, S.; Chen, J.; Domena, J.B.; Liyanage, P.Y.; Sampson, R.; et al. A Deep Investigation into the Structure of Carbon Dots. Carbon 2021, 173, 433–447. [Google Scholar] [CrossRef]
- Zuo, K.; Liu, W.; Liu, X.; Liu, X. Phosphorescence of Carbon Dot: The Intrinsic Mechanism and Recent Progress. Carbon Trends 2023, 12, 100278. [Google Scholar] [CrossRef]
- Thain, S. IR Spectroscopy and FTIR Spectroscopy: How an FTIR Spectrometer Works and FTIR Analysis. Technol. Netw. Anal. Sep. 2022. Available online: https://www.technologynetworks.com/analysis/articles/ir-spectroscopy-and-ftir-spectroscopy-how-an-ftir-spectrometer-works-and-ftir-analysis-363938 (accessed on 3 March 2026).
- Boruah, J.S.; Sankaranarayanan, K.; Chowdhury, D. Insight into Carbon Quantum Dot–Vesicles Interactions: Role of Functional Groups. RSC Adv. 2022, 12, 4382–4394. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Huang, H.; Cao, W.; Mao, B.; Liu, Y.; Kang, Z. Advances in Carbon Dots: From the Perspective of Traditional Quantum Dots. Mater. Chem. Front. 2020, 4, 1586–1613. [Google Scholar] [CrossRef]
- Kim, S.H.; Man, M.T.; Lee, J.W.; Park, K.-D.; Lee, H.S. Influence of Size and Shape Anisotropy on Optical Properties of CdSe Quantum Dots. Nanomaterials 2020, 10, 1589. [Google Scholar] [CrossRef]
- Ozyurt, D.; Kobaisi, M.A.; Hocking, R.K.; Fox, B. Properties, Synthesis, and Applications of Carbon Dots: A Review. Carbon Trends 2023, 12, 100276. [Google Scholar] [CrossRef]
- Gao, Q.; Qu, F.; Zheng, W.; Lin, H. A Simple Method to Synthesize Graphitic Mesoporous Carbon Materials with Different Structures. J. Porous Mater. 2013, 20, 983–988. [Google Scholar] [CrossRef]
- He, S.; Turnbull, M.J.; Nie, Y.; Sun, X.; Ding, Z. Band Structures of Blue Luminescent Nitrogen-Doped Graphene Quantum Dots by Synchrotron-Based XPS. Surf. Sci. 2018, 676, 51–55. [Google Scholar] [CrossRef]
- Cong, Y.; Wang, Y.; Zhang, M.; Jin, F.; Mu, J.; Li, Z.; Wang, J. Lethal, Behavioral, Growth and Developmental Toxicities of Alkyl-PAHs and Non-Alkyl PAHs to Early-Life Stage of Brine shrimp, Artemia Parthenogenetica. Ecotoxicol. Environ. Saf. 2021, 220, 112302. [Google Scholar] [CrossRef]
- Browne, R.A.; Sorgeloos, P.; Trotman, C.N.A. (Eds.) Artemia Biology; CRC Press: Boca Raton, FL, USA, 1991; ISBN 978-0-8493-6729-8. [Google Scholar]
- Ross, S.; Wu, R.-S.; Wei, S.-C.; Ross, G.M.; Chang, H.-T. The Analytical and Biomedical Applications of Carbon Dots and Their Future Theranostic Potential: A Review. J. Food Drug Anal. 2020, 28, 678–696. [Google Scholar] [CrossRef] [PubMed]
- Tapia-Salazar, M.; Diaz-Sosa, V.R.; Cárdenas-Chávez, D.L. Toxicological Effect and Enzymatic Disorder of Non-Studied Emerging Contaminants in Artemia Salina Model. Toxicol. Rep. 2022, 9, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.; Liang, W.; Meziani, M.J.; Sun, Y.-P.; Yang, L. Carbon Dots as Potent Antimicrobial Agents. Theranostics 2020, 10, 671–686. [Google Scholar] [CrossRef]
- Mavroidi, B.; Kaminari, A.; Sakellis, E.; Sideratou, Z.; Tsiourvas, D. Carbon Dots–Biomembrane Interactions and Their Implications for Cellular Drug Delivery. Pharmaceuticals 2023, 16, 833. [Google Scholar] [CrossRef] [PubMed]
- Chiorcea-Paquim, A.-M. 8-Oxoguanine and 8-Oxodeoxyguanosine Biomarkers of Oxidative DNA Damage: A Review on HPLC–ECD Determination. Molecules 2022, 27, 1620. [Google Scholar] [CrossRef]
- Martínez-Orgado, J.; Martínez-Vega, M.; Silva, L.; Romero, A.; De Hoz-Rivera, M.; Villa, M.; Del Pozo, A. Protein Carbonylation as a Biomarker of Oxidative Stress and a Therapeutic Target in Neonatal Brain Damage. Antioxidants 2023, 12, 1839. [Google Scholar] [CrossRef]
- Malmborg, V.; Elam, D.A.; Di Battista, V.; Rissler, J.; Clausen, P.A.; Vogel, U.; Wohlleben, W.; Jacobsen, N.R. Toxicity of Carbon Nanomaterials: A Model to Predict ROS Production from Easily Measurable Surface Characteristics. Carbon 2025, 234, 119997. [Google Scholar] [CrossRef]
- Romulo, A.; Suryoprabowo, S.; Setiarto, R.H.B.; Guo, Y. Carbon Dots as Multifunctional Nanomaterials: A Review on Antimicrobial Activities and Fluorescence-Based Microbial Detection. Molecules 2025, 30, 3969. [Google Scholar] [CrossRef]
- Lin, X.; Chen, T. A Review of in Vivo Toxicity of Quantum Dots in Animal Models. Int. J. Nanomed. 2023, 18, 8143–8168. [Google Scholar] [CrossRef]
- Tvrda, E. NBT Test. In Oxidants, Antioxidants and Impact of the Oxidative Status in Male Reproduction; Elsevier: Amsterdam, The Netherlands, 2019; pp. 195–205. ISBN 978-0-12-812501-4. [Google Scholar]
- Njus, D.; Kelley, P.M.; Tu, Y.-J.; Schlegel, H.B. Ascorbic Acid: The Chemistry Underlying Its Antioxidant Properties. Free Radic. Biol. Med. 2020, 159, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Sharma, N.; Sharma, A.; Lee, H.-J. The Antioxidant Properties of Green Carbon Dots: A Review. Environ. Chem. Lett. 2025, 23, 1061–1109. [Google Scholar] [CrossRef]
- Abbasi, R.; Shineh, G.; Mobaraki, M.; Doughty, S.; Tayebi, L. Structural Parameters of Nanoparticles Affecting Their Toxicity for Biomedical Applications: A Review. J. Nanopart Res. 2023, 25, 43. [Google Scholar] [CrossRef]
- Wang, H.; Sun, S.; Zhao, Y.; Wang, P.; Zhou, Y.; Sun, H.; Yang, J.; Cheng, K.; Li, S.; Lin, H. Carbon Dots with Integrated Photothermal Antibacterial and Heat-Enhanced Antioxidant Properties for Diabetic Wound Healing. Small 2024, 20, 2403160. [Google Scholar] [CrossRef]
- Wolfram, J.; Zhu, M.; Yang, Y.; Shen, J.; Gentile, E.; Paolino, D.; Fresta, M.; Nie, G.; Chen, C.; Shen, H.; et al. Safety of Nanoparticles in Medicine. Curr. Drug Targets 2015, 16, 1671–1681. [Google Scholar] [CrossRef]
- Yang, C.; Yang, J.; Lu, A.; Gong, J.; Yang, Y.; Lin, X.; Li, M.; Xu, H. Nanoparticles in Ocular Applications and Their Potential Toxicity. Front. Mol. Biosci. 2022, 9, 931759. [Google Scholar] [CrossRef] [PubMed]
- Truskewycz, A.; Yin, H.; Halberg, N.; Lai, D.T.H.; Ball, A.S.; Truong, V.K.; Rybicka, A.M.; Cole, I. Carbon Dot Therapeutic Platforms: Administration, Distribution, Metabolism, Excretion, Toxicity, and Therapeutic Potential. Small 2022, 18, 2106342. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.; Wang, Y.; Sui, X.; Xie, T.; Zhang, J.; Huang, Y.; Men, Y.; Zhang, P.; Chen, J. Renal-Clearable Biomass-Derived Carbon Dots with Red Fluorescence for Masked Cryptic Kidney Injury Imaging. ACS Appl. Bio Mater. 2025, 8, 1148–1156. [Google Scholar] [CrossRef]
- Tegafaw, T.; Mulugeta, E.; Zhao, D.; Liu, Y.; Chen, X.; Baek, A.; Kim, J.; Chang, Y.; Lee, G.H. Surface Modification, Toxicity, and Applications of Carbon Dots to Cancer Theranosis: A Review. Nanomaterials 2025, 15, 781. [Google Scholar] [CrossRef]
- Wang, X.; Liang, D.; Wang, Y.; Peijnenburg, W.J.G.M.; Monikh, F.A.; Zhao, X.; Dong, Z.; Fan, W. A Critical Review on the Biological Impact of Natural Organic Matter on Nanomaterials in the Aquatic Environment. Carbon Res. 2022, 1, 13. [Google Scholar] [CrossRef]







Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Soledad-Flores, O.V.; Bailón-Ruiz, S.J. Carbon-Source-Dependent Toxicity of Carbon Dots: An Environmental Evaluation Using Brine shrimp. Foundations 2026, 6, 11. https://doi.org/10.3390/foundations6010011
Soledad-Flores OV, Bailón-Ruiz SJ. Carbon-Source-Dependent Toxicity of Carbon Dots: An Environmental Evaluation Using Brine shrimp. Foundations. 2026; 6(1):11. https://doi.org/10.3390/foundations6010011
Chicago/Turabian StyleSoledad-Flores, Olga V., and Sonia J. Bailón-Ruiz. 2026. "Carbon-Source-Dependent Toxicity of Carbon Dots: An Environmental Evaluation Using Brine shrimp" Foundations 6, no. 1: 11. https://doi.org/10.3390/foundations6010011
APA StyleSoledad-Flores, O. V., & Bailón-Ruiz, S. J. (2026). Carbon-Source-Dependent Toxicity of Carbon Dots: An Environmental Evaluation Using Brine shrimp. Foundations, 6(1), 11. https://doi.org/10.3390/foundations6010011





